Abstract
Glioblastoma is the most common adult primary brain tumor that occurs in the central nervous system and is characterized by rapid growth and diffuse invasiveness with respect to the adjacent brain parenchyma, which renders surgical resection inefficient. Although it is a highly infiltrative tumor, it is rarely disseminated beyond the central nervous system, wherein extracranial metastasis is a unique but rare manifestation of this kind of tumor. It is very common for acquired immunodeficiency syndrome (AIDS) patients to be infected with the human immunodeficiency virus (HIV), which suggests that a possible association between HIV infection and tumor development exists. In this paper, we present a new case of a young patient’s HIV-associated glioblastoma, with glioblastoma metastasis within the T9 vertebral body and lymph nodes in the anterior neck tissue. Initially, the patient was diagnosed with a grade III plastic astrocytoma. The patient lived a normal life for a year while being treated with temozolomide, radiotherapy, and highly active antiretroviral therapy. However, the tumor quickly evolved into a glioblastoma. We believe that the drastic progression of the tumor from a grade III anaplastic astrocytoma to a metastatic glioblastoma is due to the HIV infection that the patient had acquired, which contributed to a weakened immune system, thus accelerating progression of the cancer.
Keywords
Background
Gliomas are tumors derived from the neoplastic transformation of glial cells and are the most common form of brain tumors. 1 The type of gliomas originating from glial cells and their precursors are also known as astrocytomas. 2 Further, astrocytic tumors are classified histologically and molecularly based on the World Health Organization’s criteria. They are known as grade III and IV astrocytomas or glioblastomas and are considered to be malignant. 1 The median survival period of patients with grade III anaplastic astrocytoma is 3 years. 2 Moreover, glioblastoma are the most malignant and aggressive brain tumors, with a poor prognosis.3,4 On the other hand, the median survival period of patients with glioblastoma is 14–16 months, which is 2 years less than of 10–15% of those patients with access to the gold standard treatment available nowadays. 5
In 1983, the human immunodeficiency virus (HIV), which causes acquired immune deficiency syndrome (AIDS), was discovered. The virus replicates in TCD4+ cells, resulting in a progressive decline in T cell immunity. 6 The replication of HIV may, among other things, contribute to chronic inflammation and lead to progression of AIDS, resulting in the eventual death of the patient. 6 More than half of patients with AIDS demonstrate some kind of neurological disorder, especially in the 10% of those diagnosed with brain tumors, including glioblastoma.7,8 Some reports, however, suggest that there is an increased possibility of glioblastoma occurring at a younger age in the HIV-infected population compared with the general healthy population. 9 Since the introduction of highly active antiretroviral therapy in 1998, a substantial decrease in HIV/AIDS-associated mortality has been reported. 10 However, when treated via highly active antiretroviral therapy, patients develop a chronic immune activation that contributes to the progression of cancers by stimulating the production of nitrogen species and reactive oxygen, ensuring cell proliferation, along with an enhanced secretion of pro-carcinogenic chemokines, cytokines, and related mechanisms.11,12 Moreover, the immune cell functions of the patients undergoing highly active antiretroviral therapy are not fully recovered and may become impaired, even after a year of effective therapy, a phenomenon that contributes to the formation of neoplasms. 13
In the current report, we present a new case of a young patient’s HIV-associated glioblastoma with glioblastoma metastasis at the T9 vertebral body and lymph nodes in the anterior neck tissue.
Case presentation
History and case evolution
A 32-year-old, seemingly healthy, man presented an acute syncope while practicing physical activities, followed by hemiplegia on the right side, right labial commissure deviation, and disorientation. At the emergency unit, his brain computed tomography scan showed a intraparenchymal hematoma in the left basal ganglia, measuring 3.8 × 3.1 × 2.8 cm 3 , with edema in a small area that induced a contralateral deviation in the midline structures. The patient was a former smoker who had stopped smoking for a month, and therefore, at that time, was likely to have suffered a hemorrhagic stroke. A cerebral arteriography further showed occlusion of the middle cerebral artery. During clinical evaluation at the hospital, laboratory tests showed that the patient was HIV1 positive. At the time, his CD4 count was 333 and his viral load was 7792 copies/ml without any associated co-morbidity (hepatitis B and C, cytomegalovirus, toxoplasmosis, and fluorescent treponemal antibody absorption test results were found to be negative). Further, the patient started highly active antiretroviral therapy during his hospitalization. Magnetic resonance imaging of his brain showed an expansive lesion in the periventricular region and in the internal capsule on the left side, with extension to the thalamus, inferior to the cerebral peduncle, along with the corona radiate and a semi-oval white center at the top left, measuring about 4.5 × 4.0 × 4.6 cm 3 . The tumor mass presented a heterogeneous signal intensity on T1 and T2, with a large amount of blood residue and hypo-intense signal on conducting susceptibility-weighted imaging, with heterogeneous and irregular enhanced contrast. Moreover, there was a central lesion area with a necrotic aspect, without the contrast being enhanced or diffusion restriction being encountered in the uppermost portion of the lesion. These findings suggested an associated neoplasm in the same area where the hemorrhagic changes were found (Figure 1).

Magnetic resonance imaging of the brain when the tumor was diagnosed. In (A) and (B), we can see the first magnetic resonance imaging that the patient underwent when he discovered the grade III anaplastic astrocytoma. In (C) and (D), we can view the perfusion magnetic resonance imaging with the surrounding edema and the peripheral contrast hypercapnia, indicating a malignant neoplasm. In (E) we can observe a hematoxylin and eosin staining of the first resection of the central nervous system lesion. Here, we can note the gemistocytic astrocyte’ proliferation, with mild-to-moderate pleomorphism. In (F) and (G), we can observe the first magnetic resonance imaging the patient underwent, which suggests the grade III anaplastic astrocytoma had already progressed to a glioblastoma.
The patient underwent a stereotactic biopsy of the supposed neoplasm 2 months after his stroke, when he was diagnosed with an anaplastic astrocytoma. The neoplasia could not be completely resected owing to its location. In addition to the administration of antiviral drugs for HIV, the patient started chemotherapy with temozolomide, and underwent five cycles of 30 Gy radiotherapy, leading a normal life for a year.
At 1 year and 7 months following disease onset, the patient developed chronic dorsalgia, which worsened after physical therapy. A magnetic resonance imaging of the thoracic spine indicated a pathological fracture of the 9th thoracic vertebral body, along with spinal cord compression. The patient underwent a surgical procedure in order to decompress his spinal cord with laminectomy and partial corpectomy, followed by pedicular screw stabilization. The biopsy of the ninth thoracic vertebral body mass led to the diagnosis of glioblastoma metastasis in this vertebral body. Moreover, a month prior to the spine surgery, a mass was noted in the patient’s anterior neck tissue, which was also analyzed via magnetic resonance imaging. Therefore, he was asked to undergo a lymph node biopsy, which also showed glioblastoma metastasis (Figure 2).

Magnetic resonance imaging of the thoracic spine and neck. In (A), we can see the magnetic resonance imaging of the thoracic spine, wherein a pathologic fracture of the ninth thoracic vertebral body was diagnosed with spinal cord compression. The biopsy of the T9 vertebral body mass diagnosed glioblastoma metastasis in this vertebral body. In (B), we can view the magnetic resonance imaging of the neck, wherein we can observe an augmentation in the anterior neck tissue; the lymph node biopsy, together with immunohistochemistry studies, also diagnosed with metastasis from the glioblastoma. In (C), we can note neoplastic gemistocytic proliferation infiltrating the skin. In (D), the neoplastic cells diffuse the glial fibrillary acidic protein positivity, whereas (E) shows Ki-67positive cells.
At 3 months after his first spine surgery, the patient reported acute anterior thoracic and rib pain, despite a series of spine radiotherapy sessions, and in addition to several chemotherapy cycles. The new thoracic spine magnetic resonance imaging showed a recurrence of spinal cord compression on the same site as before, with foramen stenosis resulting from the regrowth of the tumor mass in the vertebral body. Subsequently, the patient underwent a new palliative surgical procedure to decompress the spinal cord, along with the debridement of the spine canal, as a result of which we managed to isolate his cancer cells (Figure 3). At that time, a thoracic computed brain tomography scan showed multiple lytic lesions on the ribs and the scapula, whereas the thoracic magnetic resonance imaging showed extensive edema on the same sites, suggesting permeating lesions (secondary implants). Towards the end of his life, the patient presented metastatic masses in the cervical area (ribs, scapula, and neck) and in the spine, wherein an extracranial tumor emerged from the first biopsy path, all of which were compatible with the glioblastoma metastasis. After palliative spine surgery, the patient developed liver insufficiency as well as pneumonia, and was thus treated with antibiotics. After 2 weeks of palliative treatment, the patient died as a consequence of his disease.
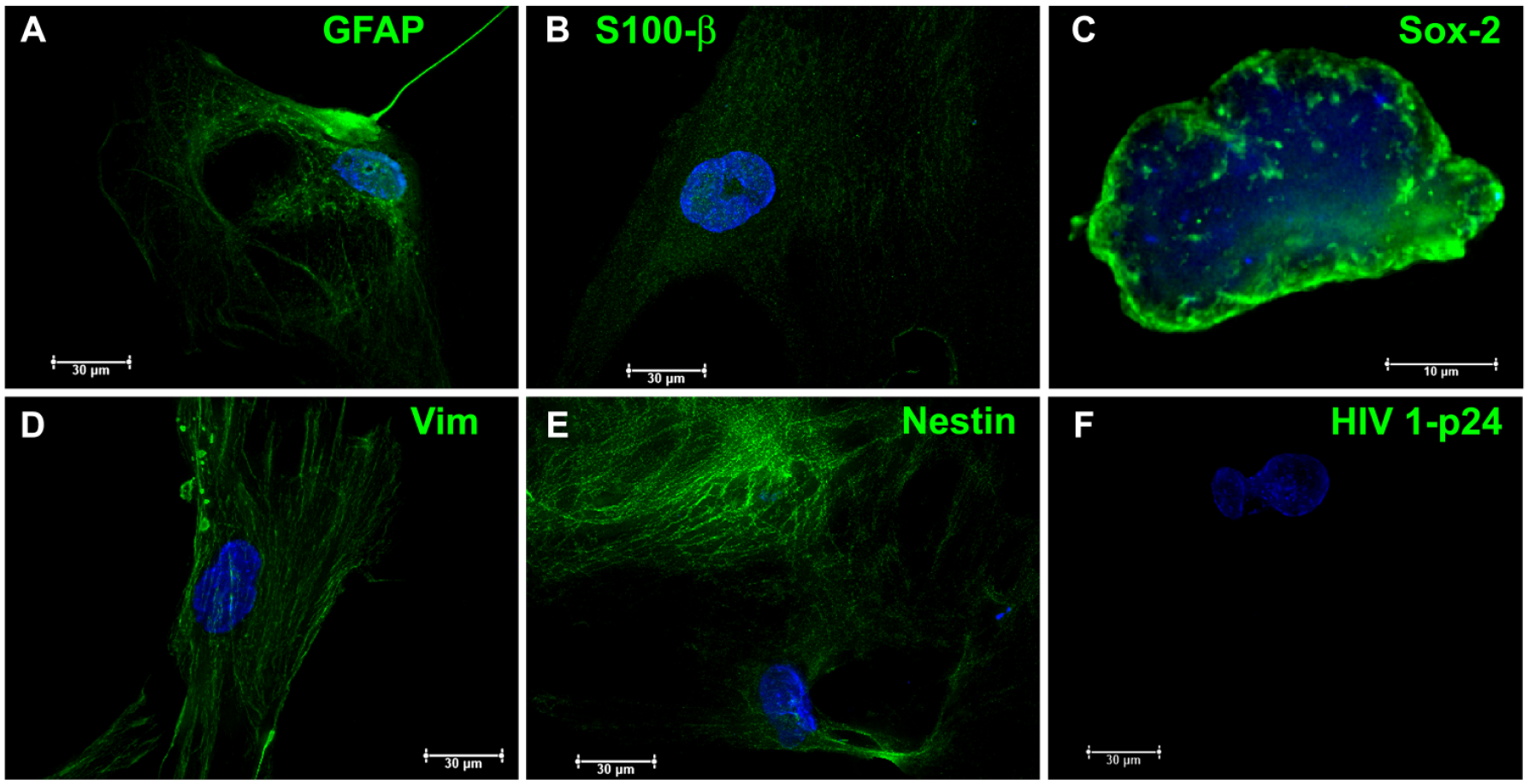
Characterization of glioblastoma cells isolated from the spinal cord metastasis. In (A) and (B), we can observe the met-GBM18 primary cells that were isolated from the patient in his last surgery for decompression, and stained for glial fibrillary acidic protein as well as S100β in order to confirm their glial origins. In (C), we can view a cell staining for SOX-2, which is a stem cell marker demonstrating that this cell line has properties identical to cancer stem cells. In (D), we can see a cell staining for vimentin, which demonstrates that this cell line possesses epithelial–mesenchymal transition properties that are closely related to its capacity to migrate and undergo metastasis. In (E), we can note a cell staining for nestin, demonstrating its neural origin. In (F), we cannot observe any staining for HIV, as expected.
Pathological description of the clinical case and methodology
Representative sections of the patient’s stereotactic biopsy were stained with hematoxylin and eosin; a small area with a diffuse and rich proliferation of gemistocytic astrocytes could be observed (Figure 1E). Endothelial proliferation was observed, after which single-labeled immunoperoxidase staining was performed using standard procedures. For this purpose, the following primary antibodies were used: monoclonal anti-glial fibrillary acidic protein (GFAP EP672Y, Cell Marque; 1:500), mouse monoclonal anti-IDH-1R132H/DIA-H09 (Optistain; 1:300), and monoclonal anti-Ki67 (SP6, Cell Marque; 1:5000). The immunohistochemistry showed diffuse positivity for the glial fibrillary acidic protein, was IDH-1R132H negative, and displayed a proliferation index of 8% (Ki–67) (Figure 2C–E). In addition, the diagnosis showed a grade III anaplastic astrocytoma (World Health Organization). The vertebral body and the resections of the lymph nodes depicted the same histology as that of diffuse gemistocytic astrocyte and cell pleomorphism. However, in these samples, we could still identify necrosis. Again, the diagnosis showed metastatic glioblastoma.
When the patient underwent a palliative spine surgery for decompression 2 years after disease onset, his metastatic glioblastoma (met-GBM18) cells were isolated, established, and characterized in our laboratory (Figure 3). This study was approved by the Clinical Research Ethics Committee of Instituto Estadual do Cérebro Paulo Niemeyer (CAAE:90670018.4.0000.8110). The cells were cultured in Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12, supplemented with 10% fetal bovine serum, and maintained at 37°C in an atmosphere containing 95% air and 5% CO2. For immunofluorescence, the met-GBM18 cells were cultured on coverslips in 24-well plates as described previously. 14 The cells were subsequently stained using standard procedures. In summary, the following primary antibodies were used: polyclonal rabbit anti-GFAP (Dako, Carpinteria, CA, USA; 1:200), polyclonal rabbit S100 (Dako; 1:400), anti-SOX-2 (Cell Signaling Technology, Danvers, MA, USA; 1:400), monoclonal mouse anti-Vimentin Clone V9 (Dako; 1:100), Anti-Nestin Antibody (Millipore, Bedford, MA, USA; 1:200), and anti-HIV-1 p24 monoclonal mouse (NIH AIDS reagent program, NIH, Bethesda, MD, USA; 1:1000). As a control for the nonspecific binding of secondary antibodies, primary antibodies were omitted. The secondary antibodies were goat anti-rabbit IgG or goat anti-mouse IgG, conjugated with Alexa Fluor 488 (1:750). The nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI). In all cases, no reactivity was observed when the primary antibody was absent. The cell preparations were mounted directly on the ProLong® Gold Antifade reagent and visualized in a DMi8 advanced fluorescence microscope (Leica Microsystems, Wetzlar, Germany).
Discussion
In this paper, we have reported the case of a 32-year-old patient who presented syncope while engaging in physical activities. Only after the patient had been hospitalized did post laboratory tests indicate that he was HIV1 positive. Owing to this condition, it was not clear whether the initial ischemia could have been caused by a neoplasm. After a biopsy, a grade III anaplastic astrocytoma was diagnosed, which was unfortunately considered inoperable due to its location. The patient started chemotherapy with temozolomide and underwent five cycles of 30 Gy radiotherapy, thereby leading a normal life for a year. However, the grade III anaplastic astrocytoma quickly evolved into a metastatic glioblastoma in the spinal cord and the anterior neck tissue. The patient’s general condition worsened rapidly after the glioblastoma diagnosis, and he died a year later.
HIV is a neurotrophic virus that usually targets macrophages and microglia within the central nervous system.15,16 It does not manifest itself in glial tumors, as per the analysis of immunohistochemistry. 17 This corroborates our findings, as we could not observe the same after conducting immunocytochemistry staining for HIV on the cells we isolated from the patient after his last surgery for spinal cord decompression. It is believed that the HIV virus has an indirect effect on glial cell transformation, facilitating either the activation of oncogenes or the inactivation of tumor suppressors. 18 Although HIV has already been demonstrated in astrocytes in vitro,15,19 it is also known that astrocytes cannot maintain HIV replication and gene expression owing to the limitations of the HIV life cycle. 20 The resistance of astrocytes to HIV-mediated cytotoxicity, in conjunction with the persistence of HIV in a host genome, helps in the transformation of astrocytes during infection. 15 Further, it is believed that astrocytes can serve as essential HIV repositories, and act as potential mediators of HIV-induced neuronal damage. 21
Moreover, the tumor microenvironment is fundamental to controlling the progression to, and development of, cancer. The immune system plays a pivotal role in this regard. Immune surveillance within the CNS is crucial to maintaining healthy brain functions. It is well known that HIV-positive patients possess an unhealthy immune system that cannot control tumorigenesis properly, thus causing a faster progression of cancer and/or a more frequent clinical presentation of tumors at a younger age. 9 It has also been demonstrated that HIV infection promotes the secretion of several cytokines, such as interleukin-1, interleukin-6, interleukin-8, and tumor necrosis alpha, all of which aid in the progression of the glioma. 15 The overexpression of transforming growth factor-beta by macrophages in the brains of HIV patients plays an important role in maintaining this immunosuppressive micro environment, in addition to helping with astrocytoma and glioblastoma progression. 15 We already know that favoring brain tissue does not express transforming growth factor-beta. 15 Some reports suggest that there is a link between the development of astrocytomas and/or glioblastomas and reduced immune surveillance.22,23 Furthermore, a previous study demonstrated a correlation of 40% between patients with advanced cancer and AIDS. 24 In addition, it was recently demonstrated that patients with HIV-glioma have a worse prognosis than glioblastoma patients without HIV. Indeed, HIV is also associated with an increased incidence of glioblastoma inpatients.9,25 Currently, cancer therapy is focused on immunotherapy based on immune checkpoints blockade, to favor the immune system and eventually help tumor regression. 26 A systematic review recently described that HIV-infected patients in an advanced-stage of cancer (melanoma or non-small cell lung cancer) tolerated immune checkpoint inhibitory therapy well, i.e. the presence of HIV infection appears not to affect the efficacy of immune checkpoint inhibitor therapy. However, the correlation between safety and efficacy of immune checkpoint inhibitor therapy and HIV load, CD4 cell count, tumor mutation burden, or programmed cell death-ligand 1 expression has not yet been evaluated. 27
In our research, we believe that our patient developed a glioblastoma from a grade III anaplastic astrocytoma rapidly because he was found to be HIV-positive; this was discovered only 2 days after being hospitalized, following a syncope that led to a hemiplegia on the right side of his body. As he was an ex-smoker who had stopped smoking for only a month, the first diagnostic hypothesis was stroke. At the time, his CD4 count was 333 and viral load was 7792 copies/mL without any associated co-morbidity. The patient was subsequently administered highly active antiretroviral therapy during his hospitalization. However, he died 2 years and 3 months after he experiencing the syncope. We believe his immune system became compromised due to his HIV infection, which drove the transformation of glial cells into a grade III anaplastic astrocytoma. Furthermore, we argue that HIV infection also contributed to the rapid evolution of the disease into a glioblastoma, thus also causing the patient to present different metastasic foci. It is already known that patients with high-grade gliomas after radiation and temozolomide treatment have a CD4 count reduction (below 300 cells/mm3). 28 However, the development of extracranial metastasis is rare. Moreover, immunocompetent patients rarely develop glioblastoma metastasis, being, therefore, considered marginal donors for organ transplant. On the other hand, studies have demonstrated that patients who received organs from those patients died of glioblastoma metastasis, probably due to an immunosuppressed immune system. 29 We believe that our patient’s immune system was probably as equally compromised as the immune system of a transplant patient, as he was taking a number of drugs such as temozolomide (in the first 1.5 years) and irinotecan, together with bevacizumab (in the last year), in addition to oxcarbazepine, clobazam, cotrimoxazole, and dexamethasone. Apart from these drugs, he was also being treated with radiotherapy and highly active antiretroviral therapy. This combination of drugs along with the treatments he received caused his immune system to collapse.
Footnotes
Acknowledgements
We would like to thank Professors Vivaldo Moura Neto and Leila Chimelli for their careful reading of the manuscript and all the support they have provided for our project. We are also grateful to Professor Rodrigo Martinez (in memoriam) for his help in searching the patient’s clinical history. We also wish to thank Aytel Marcelo Teixeira da Fonseca for all the support and information that helped us elucidate this case in detail.
Author contributions
The manuscript was conceptualized, written and edited by TCLSS. LFR evaluated and participated in the palliative surgical procedure to decompress the spinal cord. AHSC made the pathological description of the case and analyzed all biopsies. The work was supervised by TCLSS. All authors read and approved the final submitted version of the manuscript.
Availability of data and material
All data generated or analyzed during this study are included in this published article.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval
The submission of this case report was approved by the Clinical Research Ethics Committee of Instituto Estadual do Cérebro Paulo Niemeyer (CAAE:10611219.3.0000.8110).
Ethics approval for the isolation of patient cells was obtained from the Clinical Research Ethics Committee of Instituto Estadual do Cérebro Paulo Niemeyer (CAAE:90670018.4.0000.8110), as we have established a cell line for future studies.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research would not have been possible without the support extended by the National Institute for Translational Neuroscience (INNT), Ministry of Science and Technology, Brazil. We are also indebted to the Support and Evaluation of Graduate Education (CAPES) Foundation (Ministry of Education, Brazil), the National Council for Scientific and Technological Development (CNPq), the Foundation Carlos Chagas Filho Research Support of the State of Rio de Janeiro (FAPERJ), the Ary Frauzino Foundation for Cancer Research, and the Pro-Health Charitable Association of Social and Hospital Assistance and Hospital Psiquiátrico Espírita Mahatma Gandhi, Brazil (Pró-Saúde Associação Beneficente de Assistência Social e Hospitalar).
Informed consent
Written informed consent for publication of this case report and for the isolation of cells to establish a new cell line for future research was obtained from the patient’s legally authorized representative rather than from the patient himself. Although the patient was capable of understanding his actions and making a reasoned decision, he was unable to sign as he was disabled due to illness.
