Abstract
The use of targeted therapeutics known as poly(adenosine diphosphate–ribose) polymerase (PARP) inhibitors in the management of ovarian cancer is currently transforming clinical practice. The PARP inhibitor rucaparib is indicated in the UK, European Union and the United States for use in the treatment and maintenance settings for patients with relapsed ovarian cancer. Here, we discuss some of the real-world challenges and side effects that we have encountered while prescribing rucaparib, and we provide practical guidance on how the individual members of our multidisciplinary team (MDT), including a clinician, chemotherapy nurse practitioner, and clinical pharmacist, collaborate to manage these side effects. If recognized early, the side effects experienced by patients during rucaparib therapy, which include fatigue, nausea and vomiting, liver enzyme elevations, and anemia, can be easily managed. For example, providing patients with prophylactic antiemetics can help them avoid nausea, and early detection of decreases in hemoglobin levels allows for proactive interventions to alleviate anemia. The MDT should work together with the patient to identify potential side effects early and manage them effectively. The aim of this proactive approach is to maintain patients on rucaparib for optimal clinical benefit, while minimizing the potential negative impact of side effects on patient quality of life.
Keywords
Introduction
Ovarian cancer is the sixth leading cause of cancer-related deaths among women in the UK, with 4100 deaths annually. 1 Women with ovarian cancer initially undergo a combination of surgery and six cycles of platinum-doublet chemotherapy, although most patients with advanced disease will experience recurrence (Figure 1).2–6 Secondary debulking surgery and/or a second round of platinum-based chemotherapy may be considered in patients with platinum-sensitive ovarian cancer. 7 In the past, patients were closely followed after second-line chemotherapy until disease recurrence, and further relapses were treated with additional chemotherapy such as a platinum doublet (if platinum sensitive) or a single nonplatinum agent. However, response rates decrease with subsequent rounds of chemotherapy and eventually fatal chemotherapy resistance develops. 8

Overview of treatment pathway for patients with recurrent ovarian cancer eligible for PARPi therapy.
Recently, the treatment of ovarian cancer has been revolutionized by the introduction of poly(adenosine diphosphate–ribose) polymerase (PARP) inhibitors (rucaparib, olaparib, and niraparib). These drugs are approved in the UK, European Union, and the United States in the maintenance setting following a response to second or subsequent lines of platinum chemotherapy,9–14 with olaparib recently approved for maintenance treatment in the first-line setting for women with a BRCA mutation. 11 In the UK, European Union, and the United States, rucaparib is also approved in the treatment setting for patients with relapsed, BRCA-mutated, platinum-sensitive ovarian cancer;9,14 olaparib is approved in this setting in the United States (Figure 1). 13 In contrast to the treatment setting, in which therapy is initiated at the time of relapse, in the maintenance setting, patients with a complete or partial response to chemotherapy receive targeted therapy following the last cycle of chemotherapy. The goals of targeted maintenance treatment are to maintain the response achieved with chemotherapy, thereby extending clinically meaningful survival by delaying disease progression (and symptoms due to disease), and to prolong the period between treatment cycles, which allows patients to avoid the toxicities associated with chemotherapy that can negatively affect their quality of life.15,16
Here, we review the most frequent side effects observed during rucaparib therapy and provide practical guidance on the management of these side effects from the perspective of the multidisciplinary team (MDT) responsible for patients with ovarian cancer at Imperial College Healthcare National Health Service (NHS) Trust, London. The aim of proactive side effect management is to ensure that patients are able to remain on rucaparib therapy to derive optimal clinical benefit, while avoiding the negative impact of potential side effects on their quality of life.
Rucaparib: mode of action and efficacy in the maintenance and treatment settings
Rucaparib is a potent and selective, oral, small-molecule inhibitor of PARP1, PARP2, and PARP3.17–19 PARPs are crucial for the repair of single-strand breaks in DNA. Defects in the homologous recombination repair pathway, such as a BRCA1 or BRCA2 mutation, combined with inhibition of PARP proteins result in the accumulation of DNA damage and cell death through a mechanism known as synthetic lethality.20–22 Half of all high-grade serous tubo-ovarian cancers (including fallopian tube and primary peritoneal cancers) may have a homologous recombination repair deficiency at the time of diagnosis, with up to 18% of tumors harboring a germline BRCA mutation, 7% a somatic BRCA mutation, and 20% a mutation in or epigenetic silencing of another homologous recombination repair gene.23,24
Rucaparib is indicated as monotherapy for the maintenance treatment of adult patients with platinum-sensitive relapsed high-grade epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in response (complete or partial) to platinum-based chemotherapy. 9 Authorization for use in the maintenance setting was based on data from the randomized, phase III trial ARIEL3. 15 In ARIEL3, progression-free survival (PFS) was significantly longer with rucaparib than placebo in all primary analysis groups. 15 The median PFS in patients with a BRCA-mutant (i.e. deleterious germline or somatic BRCA mutation) carcinoma was 16.6 (95% confidence interval [CI]: 13.4, 22.9) months in the rucaparib group versus 5.4 (95% CI: 3.4, 6.7) months in the placebo group (hazard ratio [HR]: 0.23; 95% CI: 0.16, 0.34; p <0.0001). In patients who have a carcinoma harboring a homologous repair deficiency (i.e. BRCA mutation or BRCA wild type with high loss of heterozygosity [⩾16%]), the median PFS was 13.6 (95% CI: 10.9, 16.2) months versus 5.4 (95% CI: 5.1, 5.6) months, respectively (HR: 0.32; 95% CI: 0.24, 0.42; p <0.0001). In the intention-to-treat population, which included all patients regardless of BRCA mutation status, the median PFS was 10.8 (95% CI: 8.3, 11.4) months versus 5.4 (95% CI: 5.3, 5.5) months, respectively (HR: 0.36; 95% CI: 0.30, 0.45; p <0.0001). These results show that rucaparib significantly prolongs PFS when given in the maintenance setting to patients who have demonstrated a response to platinum-based chemotherapy. Recent data from ARIEL3 also show that rucaparib maintenance treatment did not adversely impact the possibility for patients to benefit from subsequent therapy, as time to disease progression on the subsequent line of therapy or death was significantly longer for patients who received maintenance rucaparib (21.1 [95% CI: 18.1, 23.7] months) than for those who received placebo (16.5 [95% CI: 14.6, 18.4] months; HR: 0.62; 95% CI: 0.48, 0.79; p = 0.0001). 25 Data on overall survival, an important endpoint of any oncology clinical trial, are not yet mature for ARIEL3. Given their demonstrated efficacy benefits,15,26,27 many clinicians have incorporated PARP inhibitors as standard of care in the maintenance setting.
In the treatment setting, rucaparib is indicated as monotherapy treatment for adult patients with platinum-sensitive, relapsed or progressive, BRCA-mutated (germline and/or somatic), high-grade epithelial ovarian, fallopian tube, or primary peritoneal cancer who have been treated with two or more prior lines of platinum-based chemotherapy and who are unable to tolerate further platinum-based chemotherapy. 9 The European Commission’s conditional marketing authorization for rucaparib in the treatment setting was based on an integrated efficacy analysis of 79 patients with platinum-sensitive ovarian cancer and a BRCA mutation participating in Study 10 (ClinicalTrials.gov identifier: NCT01482715) or ARIEL2 (ClinicalTrials.gov identifier: NCT01891344).9,28 A total of 51 of 79 patients (64.6%) achieved a best objective response of complete response (10.1%) or partial response (54.4%).9,28 Rucaparib treatment was associated with a median PFS of 10.9 (95% CI: 8.4, 12.8) months and median duration of response of 9.7 (95% CI: 7.4, 12.9) months.9,28
MDT and role in management of patients with ovarian cancer
In our hospital, the MDT responsible for the nonsurgical management of patients with ovarian cancer comprises clinicians, a chemotherapy nurse practitioner, a clinical oncology nurse specialist, and a clinical pharmacist (Figure 2).

Structure of the nonsurgical MDT for managing side effects that may occur during rucaparib therapy.
The clinician develops a treatment plan with the patient. This typically involves: discussing various options and treatment strategies and presenting clinical study data in a way that patients can easily understand so they are aware of the goals of treatment; obtaining a full medication history (including alternative medications); obtaining written patient consent; prescribing the first cycles of rucaparib; initiating rucaparib therapy; fully discussing the side effects that may occur during rucaparib therapy and providing information on how they can be managed; providing information on rucaparib dosing (i.e. when and how to take the tablets); and providing guidance on when to call the team for further advice.
The chemotherapy nurse practitioner and/or clinical nurse specialist provides day-to-day management of patients. At Imperial College Healthcare NHS Trust, the chemotherapy nurse practitioner manages patients from cycle 3 onwards. This involves evaluating patients’ fitness for treatment, including the assessment of blood test results, prescribing rucaparib, answering and triaging enquiries from the patient when they are feeling ill, providing advice on how to manage symptoms and side effects, and prescribing necessary supportive medications. Patients are given contact details of the chemotherapy unit and 24-h emergency hotline and are advised on who to contact in different situations. Each patient is also given the contact details of a clinical nurse specialist who supports them through their entire treatment journey.
The pharmacist screens and dispenses rucaparib and supportive medications. The pharmacist counsels patients on how to take rucaparib, makes sure the dosage instructions are clear, and may discuss concomitant medication and/or complementary therapies. Patients are advised to inform the MDT if there any changes to their medications. The pharmacist also acts as a source of information on medicines for the rest of the MDT, e.g. providing advice on drug–drug interactions. In addition, the pharmacist has a key role in ensuring the correct strength of tablets is available for each patient and that the medication stock is up to date. In some institutions, the pharmacist may provide patient counseling during the initial cycle of rucaparib therapy. Additional pharmacist-led clinics may be set up by pharmacists with an independent prescribing qualification.
Patients and/or their caregivers are closely involved in treatment decisions and should report any side effects or symptoms that occur during rucaparib therapy, in particular those that have a negative impact on their daily living and quality of life. We encourage the use of a diary to document symptoms, especially through the first month of treatment.
Initiating rucaparib
We typically start discussions about the possibility of maintenance treatment with a PARP inhibitor when a patient with recurrent, platinum-sensitive ovarian cancer initiates second-line chemotherapy. During these discussions, we emphasize that maintenance treatment would proceed only if the patient has a complete or partial response to chemotherapy. We explain that, unlike chemotherapy, maintenance treatment is not limited to a specific number of cycles and that patients will continue receiving therapy until disease progression. Patients are provided with objective information about the efficacy of PARP inhibitors in the maintenance setting, particularly in relation to their BRCA mutation status, to provide context for the anticipated magnitude of clinical benefits. We inform patients that rucaparib is an oral medication taken twice daily, and patients will be responsible for adhering to the dose regimen for the duration of therapy. Key differences between the safety profiles of chemotherapy and rucaparib are discussed, as certain side effects such as diarrhea and nausea may be more frequent during rucaparib therapy than with chemotherapy.
Rucaparib can also be prescribed as a third-line or later therapy for the treatment of women with a BRCA mutation with recurrent platinum-sensitive ovarian cancer (Figure 1). We typically discuss this option with patients when they experience a relapse following second-line or later chemotherapy.
After discussing all options, if the patient meets all appropriate criteria and elects to receive rucaparib in the treatment or maintenance setting, an application for rucaparib is made and full consent is gained from the patient. A meeting with the nonsurgical MDT is scheduled for the week the patient is due to begin therapy, and the team is updated regarding the treatment plan. Such plans outline the details of rucaparib therapy, as well as potential side effects that may occur and how they can be managed. At Imperial College Healthcare NHS Trust, our standard practice is to meet with patients after they have received 2 weeks of rucaparib to perform laboratory assessments (e.g. blood counts) and review any side effects they have had so that any tolerability concerns can be quickly addressed. If blood counts are stable, and there is no clinical indication for more frequent testing, we meet the patient again 2 weeks later and continue with regular follow ups throughout therapy (e.g. every 4 weeks for a year, then every 8 weeks thereafter). We explain that the starting dose of rucaparib is 600 mg twice a day, taken as two 300 mg tablets twice a day. If a dose reduction is required, we carefully explain to the patient what the new dose will be, how many tablets to take, and whether the tablet dosage will change.
Importance of BRCA mutation testing
At our institution, we discuss and, with appropriate consent, perform germline BRCA mutation testing as early as possible, often at the initial consultation with the medical oncology team. Germline testing is important, as the results can provide prognostic information regarding survival, impact treatment options, have implications for cancer risk in family members, and guide the implementation of appropriate risk mitigation measures for the development of other cancers.29,30 All clinicians are trained to discuss the BRCA mutation test, and patients are informed that if a germline mutation is detected, they will be referred to the genetics team for further counseling and that family members may also need to be tested. Consent forms are held in our patient electronic records; a copy is given to the patient and one is held with the regional genetics center. Knowledge of the BRCA mutation status forms part of the discussion about the potential magnitude of the efficacy benefit with rucaparib. 15 Somatic BRCA mutation testing is forming part of the standard of care for all patients in the future, which is particularly important as PARP inhibitor treatment moves into the first-line setting.31–34 In the future, screening for mutations in other homologous recombination repair genes that confer sensitivity to PARP inhibitors may also become part of clinical practice.
Potential side effects during rucaparib therapy
The most frequent side effects (occurring in >20% of treated patients) during rucaparib treatment or maintenance treatment include nausea, asthenia and/or fatigue, vomiting, anemia and/or decreased hemoglobin, abdominal pain, dysgeusia, increased serum alanine aminotransferase (ALT)/aspartate aminotransferase (AST), decreased appetite, diarrhea, thrombocytopenia and/or decreased platelet count, and creatinine elevations.9,15,35 The majority of these side effects are grade 1 or 2. The most frequently occurring grade ⩾3 side effects in both the treatment and maintenance settings include anemia and/or decreased hemoglobin, asthenia and/or fatigue, and increased serum ALT/AST.9,15,35 In clinical trials and our own experience, a number of additional side effects may occur in some patients, including constipation, photosensitivity, rash, headache, and dizziness (occurring in ≈10–35% of patients treated in clinical trials).
In our clinical experience, most side effects that occur during rucaparib therapy are manageable and unlikely to lead to drug discontinuation if appropriate guidance is followed. The most frequent side effects also tend to be transient, especially with appropriate management measures. In clinical trials, some side effects tended to occur early (within the first 2–3 weeks), including fatigue (median time to onset, 15 days), nausea (5 days), and dysgeusia (7 days), whereas others occurred later during rucaparib therapy (up to 2 months), such as anemia (median time to onset, 56 days) and thrombocytopenia (52 days). 36
Side effects that occur during rucaparib therapy should be proactively managed so that patients can continue rucaparib dosing to optimize clinical benefit. However, some side effects may need to be managed by rucaparib dose reduction and/or interruption if symptoms persist or are not adequately controlled by supportive measures. 9 Dose reductions are implemented in increments of 100 mg per day, from 600 mg twice daily to a minimum of 300 mg twice daily. 9 Side effects leading to dose reduction and/or interruption typically occur within the first month and include anemia, ALT/AST elevations, and fatigue/asthenia.15,37 The provision of proactive and effective supportive care by the MDT is crucial in reducing the need for dose modifications. Below, we provide practical guidance for the management of specific side effects that may occur during rucaparib therapy by the members of the MDT (see Table 1 for summary), based on published consensus guidelines and the clinical experience of the Imperial College Healthcare NHS Trust MDT.
Summary of practical guidance for managing side effects encountered during rucaparib therapy.

Consider dose reduction and/or interruption for any side effects not controlled by supportive measures; if rucaparib is interrupted for >14 days due to toxicity, it may need to be discontinued.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; GFR, glomerular filtration rate.
Practical management of nonhematologic side effects
Fatigue
The majority of patients experience grade 1 or 2 fatigue while receiving rucaparib (see Table 2 for grading). Fatigue is a frequent side effect of many anticancer therapies, and patients taking rucaparib may have fatigue related to chronic toxicity from prior chemotherapy. Fatigue may also be associated with underlying causes, such as anemia, uncontrolled pain, poor nutrition, hypothyroidism, emotional distress, and sleep disturbances, which are possible to optimize or treat. Iron, hematinics, magnesium, and vitamin D levels also should be checked for deficiencies. 39
CTCAE guidelines for grading of potential side effects which may occur during rucaparib therapy. a
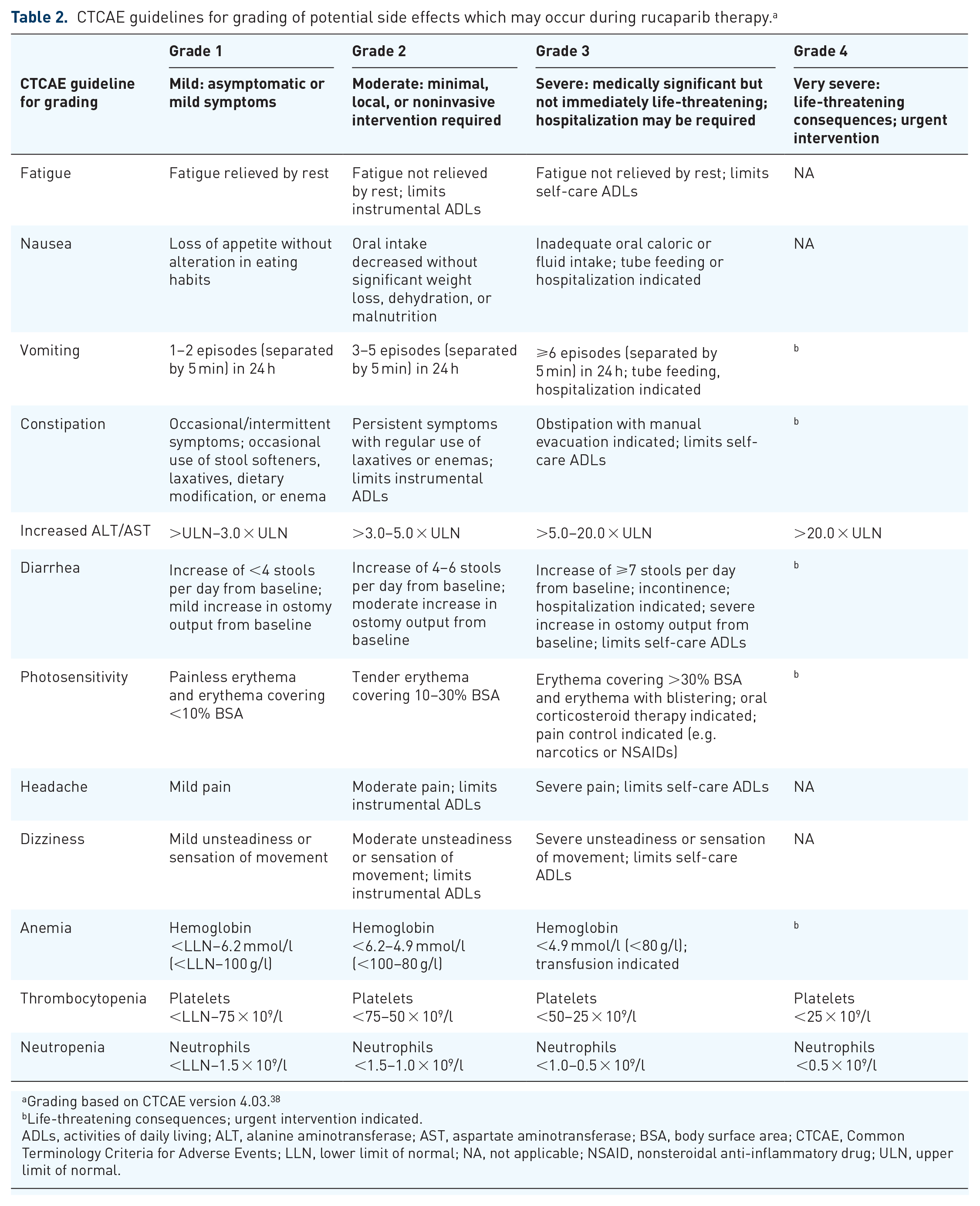
Grading based on CTCAE version 4.03. 38
Life-threatening consequences; urgent intervention indicated.
ADLs, activities of daily living; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BSA, body surface area; CTCAE, Common Terminology Criteria for Adverse Events; LLN, lower limit of normal; NA, not applicable; NSAID, nonsteroidal anti-inflammatory drug; ULN, upper limit of normal.
The approach to managing this side effect includes informing patients of expected patterns of fatigue and discussing ways to conserve energy that may allow them to remain active. For example, an appropriate and regular exercise regimen should be recommended. Since other possible reasons for fatigue might be psychological or attributable to poor nutrition or sleep, the clinician or nurse may need to make referrals for counseling, nutrition consultation, or sleep therapy. 40 For patients with grade 3 or 4 fatigue, more extensive investigations may be necessary to determine other possible contributing factors, such as concomitant medications, pain, or anemia; dose interruption could be considered. If fatigue persists despite the above measures and is affecting quality of life, a dose reduction may be necessary.
Nausea and vomiting
Patients frequently experience nausea during the first cycle of rucaparib therapy. We consider a prescription for prophylactic antiemetics the best way to manage this potential side effect. We advise patients to take regular antiemetics during the first few days of treatment and not to wait for nausea to occur. Ideally, antiemetics should be taken an hour and a half prior to rucaparib for maximal effect. If no nausea occurs, then the antiemetics can be reduced. The antiemetics domperidone and metoclopramide are recommended to not be used for more than 5 days. At Imperial College Healthcare NHS Trust, we also encourage patients to keep a food diary that can help to identify triggers of nausea and/or vomiting. A dose reduction and/or interruption may be necessary for grade 2 nausea (Table 2) not controlled by medications and/or supportive care. Grade 3 or 4 nausea and vomiting should be investigated for other causes, since this is not a typical side effect of rucaparib; other possible reasons could include concomitant medications or a partial or complete bowel obstruction.
ALT/AST elevations
ALT/AST elevations occur most frequently during the first few weeks of rucaparib treatment. Therefore, at Imperial College Healthcare NHS Trust, we monitor patients more closely in the first few weeks of therapy (at 2 weeks and monthly thereafter), unless significant elevations occur. In clinical trials, elevated ALT and AST levels observed during treatment with rucaparib were not accompanied by other signs of potential liver damage, such as elevations of bilirubin.15,35,37 After an initial increase, ALT and AST levels in these trials typically decreased to within the normal range and remained consistent for up to 2 years. For grade 2 increases (Table 2), treatment modification is not necessary. According to the summary of product characteristics (SmPC), for grade 3 increases, rucaparib can be continued at the same dose if bilirubin levels are less than the upper limit of normal and alkaline phosphatase levels are less than three times the upper limit of normal. 9 Weekly monitoring is recommended until ALT/AST levels return to near normal. 9 If ALT/AST levels do not decline within 2 weeks, rucaparib should be interrupted until they return to grade ⩽2; rucaparib can then be resumed at the same or at a reduced dose. 9 For grade 4 increases, treatment with rucaparib should be interrupted until ALT/AST levels return to grade ⩽2; rucaparib may then be resumed at a reduced dose. If a dose interruption is required for ⩾14 days, then we would consider discontinuing rucaparib.
Creatinine elevations
Electrolytes should be checked at the start of treatment and monthly thereafter. No starting dose modifications are required in patients with mild-to-moderate renal impairment. 9 There are currently no data in patients with severe renal impairment (creatinine clearance <30 ml/min). Elevations of creatinine (grade 1 or 2) were seen in 15–20% of patients in clinical trials in the first weeks of treatment, but levels stabilized over time without dose adjustments and remained within normal ranges for up to 2 years.15,35 We advise patients to keep well hydrated during treatment and warn them that changes to their renal function may occur, although no change in rucaparib dose is necessary. This laboratory abnormality may be due to the inhibition of the renal transporters MATE1 and MATE2-K, since some PARP inhibitors are known to affect these transporters in vitro,9,41,42 although the creatinine elevations may not be directly associated with renal toxicity. Notably, in a retrospective study of patients who received PARP inhibitors, discordance between estimated glomerular filtration rate (GFR) based on serum creatinine levels and calculated GFR obtained through a nuclear medicine scan was commonly observed (63% of matched assessments). 43 At Imperial College Healthcare NHS Trust, if there is a concern regarding creatinine elevations that may prevent a patient being treated with rucaparib (e.g. grade 3 or 4 rise in creatinine or a calculated creatinine clearance <30 ml/min), a nuclear medicine GFR scan may be performed to obtain a more accurate assessment of renal function, which could allow for the continuation of rucaparib treatment if no true dysfunction is identified despite rising creatinine. Other causes of renal dysfunction such as obstructions or use of other medications associated with renal toxicity, should be ruled out in patients with a significant rise in creatinine. Rucaparib should be interrupted in patients with grade 3 or 4 rises in creatinine; if true renal dysfunction is confirmed, dose reduction may be required once creatinine levels have improved. If alternative causes are ruled out and toxicity does not resolve to grade 1 or better within 14 days, rucaparib may need to be discontinued.
Diarrhea
Patients who develop diarrhea while taking rucaparib should first undergo the appropriate clinical assessments to exclude any underlying causes (e.g. infection, prior constipation, or medication-induced diarrhea). Preventing dehydration with oral hydration is necessary. Grade 2 or 3 diarrhea (Table 2) may be alleviated through dietary adjustments and/or antidiarrheal medication (e.g. loperamide or codeine). Antidiarrheal medications, intravenous fluids, and electrolyte replacement should be provided to patients with more severe diarrhea. Antibiotics may be needed if an infection is confirmed.
Constipation
Following debulking surgery (which may have included bowel resection) and chemotherapy, many women experience a change in bowel function, particularly constipation. This may be exacerbated by the use of rucaparib. Patients should be informed of self-care and prevention strategies for constipation, such as staying adequately hydrated, dietary modifications, and, when needed, use of osmotic or stimulant laxatives. For full rectum or fecal impaction, suppositories and/or enemas may be required (Table 1). 44 Concomitant medications, such as ondansetron and calcium supplements, may also contribute to constipation and should be considered when managing this side effect.
Photosensitivity and rash
Patients taking rucaparib may experience increased sensitivity to the sun and/or other sources of ultraviolet light (e.g. tanning beds and manicure setting lights). Before initiating rucaparib therapy, patients should be advised to avoid spending time in direct sunlight and to use sun protection while outdoors (e.g. wearing a hat and protective clothing and using a high sun protection factor sunscreen and lip balm). 9 Rash can usually be proactively managed with topical creams and emollients, adding a course of topical steroid cream if needed.
Headache and dizziness
Patients taking rucaparib may experience headache; however, this is a common complaint in the general population, and we would assess for any contributing causes. A full headache history should be taken, along with a neurological examination. If headache corresponds with initiation of rucaparib, further imaging is unlikely to be required but could be considered. Headache may be managed with simple analgesics; occasionally, dose reductions or interruptions are required.
Patients may experience dizziness while taking rucaparib. A full clinical assessment of dizziness is required to ascertain what the patient means by feeling dizzy; this can be done by asking questions, such as ‘Is the room spinning?’ and ‘Do you feel “lightheaded” on standing?’ Other symptoms, including hearing changes, nausea, and headache, should be investigated and categorized as persistent or intermittent. We assess for any contributing cause and consider referral to an ear, nose, and throat specialist or perform a hearing test, if required. Often, reassurance that this may be a side effect of the medication is all that is required. A dose reduction may be needed if the patient’s dizziness does not resolve and is affecting their quality of life.
Practical management of hematologic side effects
Prior to commencing rucaparib, the clinician should confirm that blood counts have recovered from previous chemotherapy to grade 1 or normal. The clinician and/or chemotherapy nurse practitioner should ensure that the blood count results are acceptable before prescribing rucaparib and may liaise with the clinical pharmacist if a dose reduction is required. Hematologic toxicities should be managed according to rucaparib’s SmPC and local treatment guidelines. In general, for grade 2 side effects (Table 2), supportive care (e.g. transfusions, erythropoiesis stimulating agents, and iron replacement) is recommended.39,45,46 Dose reduction and/or interruption should be considered for grade 3 or 4 hematologic side effects 9 and can be implemented by the clinician and/or chemotherapy nurse practitioner, although overall responsibility lies with the clinician. At Imperial College Healthcare NHS Trust, we consider discontinuing rucaparib treatment if it is interrupted for ⩾14 days without the improvement of a hematologic side effect to grade 1 or better. In addition, if hematologic toxicities do not resolve despite dose reduction and/or interruption, urgent hematology referral may be required to investigate for potential myelodysplastic syndrome/acute myeloid leukemia (MDS/AML). Investigations should include bone marrow sampling and cytogenetics. If MDS/AML is confirmed, rucaparib should be permanently discontinued. This potentially fatal side effect was observed in 1% of rucaparib-treated patients in ARIEL3. 15
Anemia
Anemia and/or decreased hemoglobin of any grade was reported in 42% of patients in clinical trials (median time to onset, 56 days), 36 with 17–22% of patients requiring a treatment interruption and/or dose reduction.15,28 Hemoglobin levels should be monitored at least every 28 days during rucaparib therapy. Blood transfusions may be required for patients who develop anemia, once other causes (e.g. iron, B12, or folate deficiencies) have been excluded. For grade 1 or 2 anemia, optimization of iron levels with hematinics (supplementary vitamin B12 and folates) should be attempted. 39 For recurrent episodes of anemia, dose reduction and/or interruption should be considered. However, in our experience, blood transfusions and the use of erythropoietic-stimulating agents can allow patients to continue on the same dose of rucaparib. If anemia recurs, dose reduction is required.
Thrombocytopenia
Across clinical trials of rucaparib, thrombocytopenia and/or decreased platelet count of any grade was reported in 26% of patients (median time to onset, 52 days), 36 with 13–18% of patients requiring a treatment interruption and/or dose reduction.15,28 Rucaparib can be continued if grade 1 thrombocytopenia occurs. Grade 2 thrombocytopenia requires close monitoring and potential dose reduction. If grade 3 or 4 thrombocytopenia (Table 2) occurs, rucaparib should be interrupted until the patient’s platelet count has recovered to near normal and then restarted at a reduced dose. For profound thrombocytopenia (platelet count <20 × 109/l), patients require regular blood checks (every few days) to closely monitor platelets until recovered. For a platelet count of <10 × 109/l, a platelet transfusion should be considered. Medications that can induce thrombocytopenia, such as heparin, are commonly co-administered in many cancer patients and so need to be reviewed and possibly withheld if significant thrombocytopenia occurs.
Neutropenia
Neutropenia and/or decreased neutrophil count of any grade was reported in 16% of patients in clinical trials (median time to onset, 67 days; data on file), with 7% of patients requiring a treatment interruption and/or dose reduction. 15 Rucaparib can be continued at the same dose if a patient experiences grade 1 or 2 neutropenia (Table 2). For grade 3 or 4 neutropenia, rucaparib may need to be interrupted until neutrophil cell counts return to grade 1 or 2. If persistent neutropenia occurs, a dose reduction is likely to be required. 47
Practical management of concomitant medications
When starting rucaparib, the clinician takes a full medication history and patients are advised to alert the MDT if they start any new medications so that these can be checked for interactions. In vitro data demonstrated that rucaparib is metabolized by the cytochrome P450 enzymes CYP2D6, CYP3A4, and CYP1A2. 9 Although CYP3A4 metabolism is slow, caution is required if a patient is taking a concomitant medication that is a strong CYP3A4 inhibitor (e.g. clarithromycin) or inducer (e.g. phenytoin) which might increase or decrease rucaparib levels, respectively. 9
Rucaparib is also a moderate inhibitor of CYP1A2 and a mild inhibitor of CYP2C9, CYP2C19, and CYP3A. 9 If a patient is taking a medication metabolized by these enzymes (e.g. tizanidine, warfarin, omeprazole, or midazolam, respectively) the clinician should implement appropriate clinical monitoring and consider dose adjustments as outlined in the SmPC. 9 Rucaparib is a substrate of P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP) and also inhibits BCRP. Caution is recommended when using strong inhibitors of P-gp (e.g. clarithromycin) or medications that are BCRP substrates (e.g. rosuvastatin). 9 Due to inhibition of the renal drug transporters MATE-1 and MATE-2K and hepatic drug transporters OCT1 and OCT2 by rucaparib, caution is advised when co-administering rucaparib with medications such as metformin, as this could increase renal elimination and decrease liver uptake of these products. 9
Impact on service and patients
Impact on service
The monitoring and management of side effects that may occur during rucaparib therapy have an impact on the oncology clinic due to a greater frequency of clinical patient reviews, more frequent blood tests, and a greater level of interaction between MDT members and the patient than during regular follow up. In an already stretched NHS, this increased burden must not be underestimated.
Side effects that require dose reductions may also lead to medication wastage if a patient needs a midcycle dose reduction requiring a different-strength tablet. The potential cost impact of this medication wastage can be minimized by maintaining good communication within the MDT.
Impact on patient quality of life beyond side effects of treatment
Patients may experience disruption of normal daily activities and incur additional travel-related costs due to increased clinic visits. One practical way to address these challenges is to provide the patient with blood test results by telephone and/or have local general practitioners perform blood tests to cut down on travel and waiting times. Communication by email is also an efficient way to inform patients of blood test results and whether they can pick up their medication. In addition, the psychological impact of living with a disease that cannot be cured can be further exacerbated by the side effects of therapy. Patients may wish to spend time away from hospital, and regular hospital visits required for a maintenance treatment is a constant reminder of their illness, which some patients wish to avoid. By developing approaches for proactive side-effect management that focus on early detection and timely intervention, the MDT can help alleviate this psychological burden and ensure patients maintain their quality of life (see patient perspective box).
Patient perspective.

Conclusion
Rucaparib has clinical benefits for patients with ovarian cancer in both the maintenance and treatment settings, such as extending PFS and reducing tumor burden. However, these benefits may be negatively impacted if side effects prevent a patient from remaining on rucaparib therapy or decrease the patient’s quality of life. The majority of side effects that occur during rucaparib therapy can be easily managed with early and proactive intervention by the clinical MDT responsible for patient care. Patients should be empowered to inform their treating team of side effects so that they can be quickly addressed and appropriate advice given.
We have provided practical guidance for the effective management of side effects encountered during rucaparib therapy, as implemented by the MDT at Imperial College Healthcare NHS Trust. We hope that this guidance will help other MDTs set up PARP inhibitor clinics. Although the role of the MDT members may vary in different hospitals (e.g. roles of clinical pharmacist and chemotherapy nurse practitioners as prescribers of rucaparib and supportive medications), the overall aim of delivering the best patient care and minimizing the impact of side effects that occur during rucaparib therapy will be the same. The MDT approach provides the supportive structure needed to maintain patients on rucaparib for optimal clinical benefit, while minimizing the potential negative impact of side effects on patient quality of life.
Footnotes
Author contributions
All authors contributed to the writing and review of the manuscript and approved the final version for submission. All authors agree to be accountable for the accuracy and integrity of the work.
Conflict of interest statement
Laura Tookman has served on an advisory board for AstraZeneca, has served as a consultant for Tesaro, and has received compensation for travel from Tesaro.
Jonathan Krell has served on advisory boards for Clovis Oncology, AstraZeneca, and Tesaro.
Baleseng Nkolobe has served on an advisory board for Clovis Oncology and has received honoraria and compensation for travel from Tesaro.
Laura Burley declares no competing interests.
Iain A. McNeish has served on advisory boards for Clovis Oncology, AstraZeneca, Takeda, and Tesaro and receives institutional funding from AstraZeneca.
Consent for publication
The patient perspective statement included in the article was obtained from the patient, who provided written consent for its inclusion and publication.
Data availability
Not applicable.
Ethics approval and consent to participate
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: medical writing and editorial support funded by Clovis Oncology, Inc., were provided by Stephen Mason, Nathan Yardley, and Shannon Davis of Ashfield Healthcare Communications (Middletown, CT, USA).
