Abstract
Chemoradiotherapy (CRT) is extensively used prior to surgery for rectal cancer to provide significantly better local control, but the radiotherapy (RT), as the other component of CRT, has been subject to less interest than the drug component in recent years. With considerable developments in RT, the use of advanced techniques, such as intensity-modulated radiotherapy (IMRT) in rectal cancer, is garnering more attention nowadays. The radiation dose can be better conformed to the target volumes with possibilities for synchronous integrated boost without increased complications in normal tissue. Hopefully, both local recurrence and toxicities can be further reduced. Although those seem to be of interest, many issues remain unresolved. There is no international consensus regarding the radiation schedule for preoperative RT for rectal cancer. Moreover, an enormous disparity exists regarding the RT delivery. With the advent of IMRT, variations will likely increase. Moreover, time to surgery is also quite variable, as it depends upon the indication for RT/CRT in the clinical practices. In this review, we discuss the options and problems related to both the dose–time fractionation schedule and time to surgery; furthermore, it addresses the research questions that need answering in the future.
Introduction
Surgery is the cornerstone of curative therapy for rectal cancer, and combined-modality treatment is the recommended adjuvant, or neoadjuvant therapy. Multimodality therapy is often used for tumor downstaging or downsizing, anal sphincter, or other organ preservation, as well as improvements in local control (LC) or even overall survival (OS). Preoperative chemoradiotherapy (CRT) has been shown comparable or superior to postoperative treatment in terms of various end points,1–3 and preoperative radiation dose and time interval are significant predictors of the pathological complete response (pCR) rate and downstaging. 4 However, different viewpoints exist regarding the optimal dose–time fractionation schedule of preoperative radiotherapy (RT) and time to surgery.
Conventionally, long-course RT (i.e. 1.8–2.0 Gy per day; total dose of 45–50.4 Gy), frequently combined with chemotherapy, has been the preferred approach for a majority of patients in most countries, particularly in the United States and in Southern Europe. Short-course RT (i.e. 5 Gy per day; total dose of 25 Gy) and surgery within the following week has been commonly used in Sweden and some other countries in Northern and Western Europe. Recently, short-course RT with delay to surgery has also been demonstrated a useful alternative to these two schedules. 5 In fact, treatment differences exist even across institutions within the same country. 6
Traditionally, preoperative RT has been delivered via three-dimensional conformal RT (3D-CRT) with three- or four-field techniques for rectal cancer. Nowadays, advanced techniques, such as intensity-modulated radiotherapy (IMRT), are widely and successfully used for prostate, head-and-neck, and other cancers.7–9 The theoretical rationale for using such highly conformal techniques is sound for rectal cancer; however, the potential clinical benefits remain debatable. 10 Thus, we performed searches on PubMed, EMBASE, and MEDLINE databases (2000 to May 2019) using the medical subject heading term ‘rectal cancer.’ Additional keywords included ‘preoperative,’ ‘radiotherapy,’ ‘chemoradiotherapy,’ ‘surgery,’ ‘dose,’ and ‘time interval.’ Furthermore, we reviewed reference lists from retrieved articles and textbooks to identify additional articles of interest. We discussed the options of both dose–time fractionation schedule and time to surgery using prior standards (i.e. 3D-CRT), and advanced technologies (i.e. IMRT). Given the growing concerns about precision medicine, understanding the patterns of new technology is particularly important.
The era of 3D-CRT
Local recurrence (LR) is a serious problem because it causes disabling symptoms and successful salvage of pelvic recurrence is rarely possible. Just 34 years ago, the LR risk was greatly reduced from 25% to 16% with the advent of postoperative RT, with anterior and posterior parallel opposed fields. 11 In 2004, the German Rectal Cancer Study Group demonstrated improved LC and reduced toxicity when CRT with a three- or four-field technique was delivered preoperatively instead of postoperatively, and LR at 5 years was further reduced from 13% to 6%. 12 In 2005, at a median follow up of 13 years, the Swedish rectal cancer trial eventually reported that LR was only 9% after short-course RT with immediate surgery. 13 Therefore, preoperative RT/CRT for rectal cancer was found to be beneficial for reducing LR rates.
Conventional as well as hypo- and hyperfractionated RT strategies and time intervals
During the last 2 decades, more modern trials have examined the most appropriate treatment schedule. Polish and Australian trials compared long-course CRT (28 × 1.8 Gy) and surgery 4–6 weeks later with short-course RT (5 × 5 Gy) and surgery within 7 days for cT3/T4 disease.14–16 No significant differences were observed in postoperative complications, LC, late toxicity, recurrence-free survival (RFS), disease-free survival (DFS), or OS; nevertheless, a significantly higher acute radiation toxicity was observed with long-course CRT. In 2017, the Stockholm III trial used three regimens: either short-course RT (5 × 5 Gy) with surgery within 1 week or after 4–8 weeks or 25 × 2 Gy with surgery after 4–8 weeks. 5 No significant differences in local and distant recurrences or in RFS and OS were reported among the three different RT regimens. Compared with short-course RT with immediate surgery, postoperative complications were significantly reduced by delaying surgery; however, acute radiation-induced toxicities were seen in ~7% of these patients after much delay. In addition to a hypofractionated RT regimen (5 × 5 Gy), a hypofractionated RT schedule (30 Gy in 10 once-daily fractions) was tested in China to minimize side effects without compromising therapeutic efficacy. 17 After a median follow up of 63.8 months, 5-year DFS and OS rates were 64.5% and 75.6% respectively. Moreover, grade ⩾3 acute toxicity rates was only 1.2%, and the total grade ⩾3 late RT toxicity rate was down to 2.7%. 18
In order to verify the hypothesis that hyperfractionated accelerated radiotherapy (HART) may provide a favorable long-term outcome compared to hypofractionated RT, the pelvis was irradiated twice daily, with a minimal interfraction interval of 6h, and a total dose of 42 Gy was administered in doses of 1.5 Gy per fraction. 19 Surgery was performed 1–2 weeks after RT. The results showed that the physical, emotional, and social functioning of long-term survivors were significantly better with HART; however, when compared with hypofractionated RT, there was no significant difference regarding LC and OS. In order to ensure that the overall treatment time was shorter than the proliferation delay Tk, set to 7 days, 20 RT was delivered with a single fraction of 2.5 Gy twice daily (⩾6 h intervals) to a total dose of 25 Gy. Surgery was performed the following week. 21 The clinical trial showed that LC was excellent in primarily resectable rectal cancer. Combined with S-1 as a radiosensitizer, this regimen of a 4-week delay in surgery also showed acceptable oncologic outcomes for T3 rectal cancer.22,23
There are many other dose fractionations of preoperative RT in addition to the above schedules. Figure 1(a, b) shows the most commonly used regimens.24–52 Here, a biologically effective dose (BED) was calculated according to a linear–quadratic (LQ) model of radiation effect. BED was evaluated at the isocenter. In this model, α/β ratio of 10 was adopted for tumor tissue.20,53,54 Most of the regimens had larger BEDs of ⩾30 Gy. Meanwhile, the dose curves steepened and became concentrated for d > 1 Gy after the overall treatment time (OTT) was considered. If BED3/BED10 is used to represent the risk/benefit ratio of preoperative RT, 3–4 Gy per fraction using once-daily RT regimen or 2 Gy per fraction using twice/thrice-daily RT regimen might be optimal [Figure 1(c, d)]. In most centers, RT fractionation schedules and time to surgery are based on their clinical practice experiences. Given the different combination schemes of dose fractionation and time to surgery, a goal interval of 6–8 weeks is the most commonly used value in clinical trials [Figure 2(a)].5,15,17,18,21,24,27,28,31,32,34–36,38–42,44,46–48,50,52,55–94 Moreover, because of factors such as acute radiation reaction, there are some discrepancies between the goal intervals and true intervals. 95 Despite limited samples, a linear correlation can be observed between them in Figure 2(b). In addition, because of the semi-Poisson distribution of the actual time interval, 95 the mean values of goal time gaps are usually smaller than the median true time intervals [Figure 2(b)].5,18,21,24,32,36,38,41,43,44,46,47,52,56,61,70,73,74,79,80,82,90,91,93,96–108 It seems that surgery is usually performed early for most patients within a planned schedule.

Linear–quadratic-model-based BEDs of most commonly used schedules of preoperative RT.
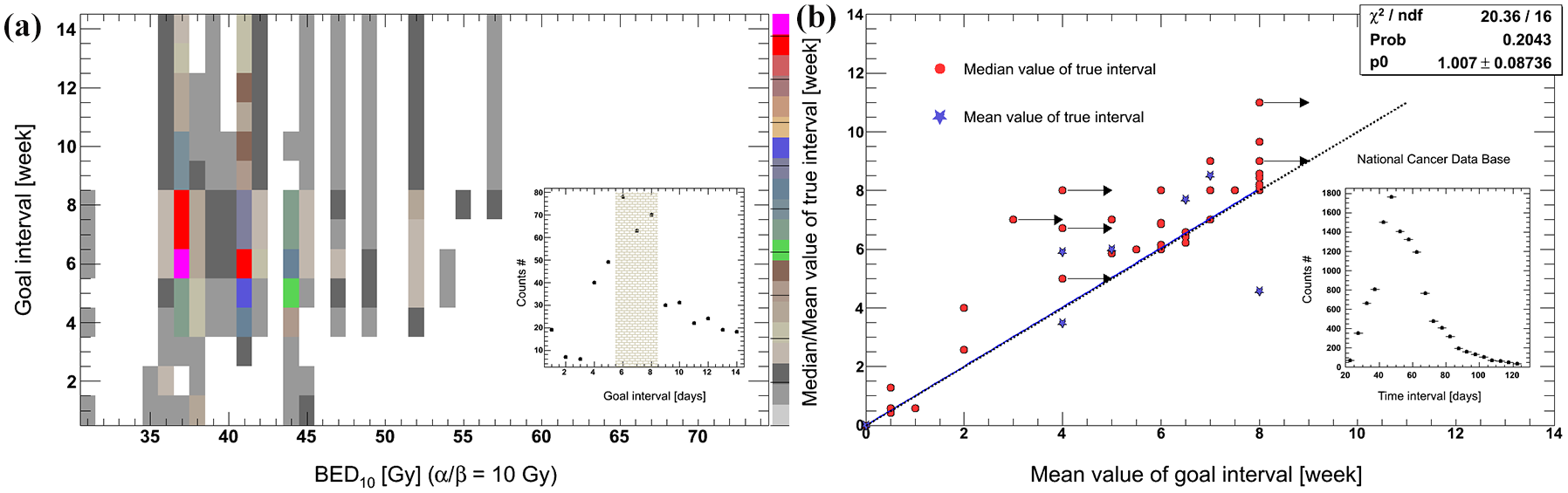
The most commonly used time intervals including goal and actual values.
Although the goals of preoperative RT/CRT are to minimize the recurrence risk, optimize survival, and avoid toxicity, different strategies have led to different outcomes. However, it is clinically relevant to wait for the highest degree of pathological response, as this helps to identify the optimal time to surgery and increases the chance of R0 resection. Moreover, patients might exhibit such favorable response that they become candidates for a watch-and-wait approach or local excision. 109 Additionally, patients with a pCR might have better DFS and OS.110,111 Therefore, an enhanced radiation response is necessary for a better pathologic response after preoperative RT/CRT.
Early endpoints: pathologic tumor response
Several parameters have been considered to quantify tumor response, such as T (tumor size) and N (number of nearby lymph nodes) status downstaging and pCR. In China, a pCR rate of 4.5% and a downstaging rate of 70.2% were achieved using the 30 Gy protocol and surgery after 2 weeks. 18 A high pCR rate of 11.8% was reported in the short-course RT-with-delay arm. 112 A Polish trial showed an even higher pCR of 16.1% using conventional long-course CRT and surgery 4–6 weeks later. 14 The tumor response could be further increased by the addition of specific chemotherapy regimens in preoperative setting. 71
Furthermore, a highly significant dose–response relationship was observed. For example, a trend toward increased pCR with higher doses was reported, with pCR being 15%, 23%, and 33% at 40 Gy, 46 Gy, and 50 Gy, respectively. 79 Figure 3(a) also shows that increasing RT doses were associated with tumor response, but the incremental rates were different because of additional chemotherapy, and RT techniques, etc.4,26,74,75,79,113–118 However, if OTT was considered using this LQ model, 20 the trends became very similar among some trials [Figure 3(b, c, d)]. Furthermore, improved response could be enhanced with intraoperative RT or with high-dose-rate γ-ray or contact X-ray brachytherapy boost.119–121
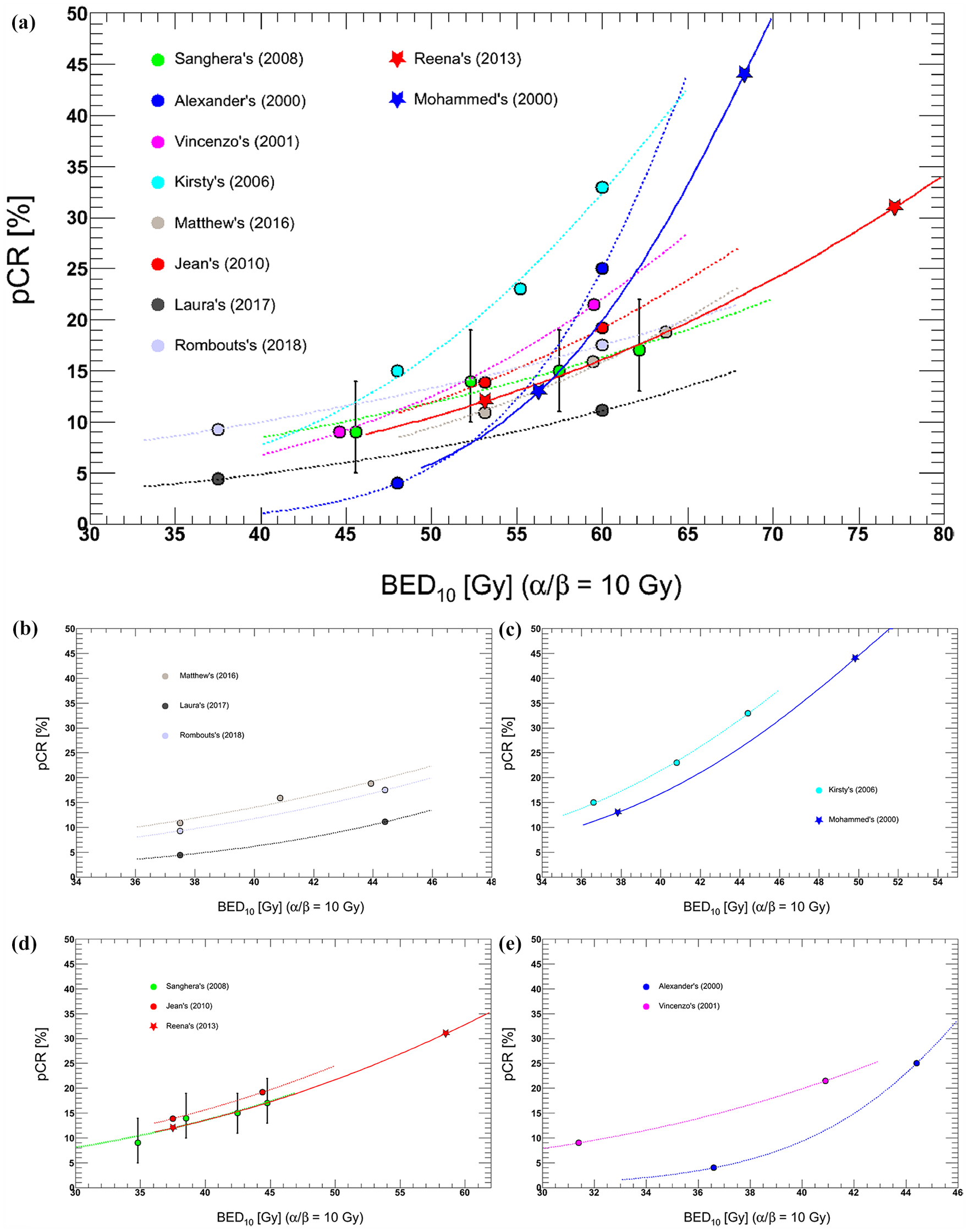
Comparison of reported dose–response relationships between BED10 and pCR.
Tumor regression takes time (median volume-halving time, 14 days). 122 Several studies have previously demonstrated improved pCR after long time intervals (Table 1, longer intervals might not increase pCR in particular cohorts).83,84,96,123–126 The Korean Radiation Oncology Group found that pCR steadily increased after 5–6 weeks, escalated over 10% after 6–7 weeks, and peaked at 9–10 weeks for locally advanced rectal cancer. The downstaging rate increased steadily until 6–7 weeks and declined afterward. 127 For patients with cT1-4N0-2M0-1, the highest pCR rates were observed at approximately 10–11 weeks from the end of long-course CRT. 128 A waiting time exceeding 11 weeks might be associated with higher morbidity and a more difficult surgical resection because of tissue fibrosis and friability. 129 After accounting for well-known confounders, such as comorbidities and tumor characteristics, an optimal threshold of 56 days (8 weeks) was determined after completion of neoadjuvant CRT for minimizing the risk of positive margins and maximizing pathologic downstaging. 95
Literature review.

p < 0.05.
5-FU, 5-fluorouracil; CAP, capecitabine; c.i., continuous infusion; CPT-11, irinotecan; DFS, disease-free survival; FL, 5-fluorouracil + leucovorin; FOLFOX6, oxaliplatin + leucovorin + 5-FU; IORT, intraoperative radiotherapy; LR, local recurrence; LV, leucovorin; mFOLFOX-6, modified FOLFOX-6; IC, irinotecan + capecitabine; N, number of nearby lymph nodes stage; NR, not reported; OS, overall survival; pCR, pathological complete response; RT, radiotherapy; T, tumor size stage; TME, total mesorectal excision; XELOX, oxaliplatin + capecitabine.
Long-term endpoints: LC and survival
After a median follow up of 11 years, the German CAO/ARO/AIO-94 trial reported that 10-year LR and OS rates were 7.1% and 59.6%, respectively, using 50.4 Gy in 28 fractions. 1 After a median follow up of 12 years, the TME trial finally reported that 10-year LR and OS rates were 5% and 48% respectively, with short-course RT. 143 The effect of RT on LC persisted, as well as the absence of a survival benefit. Nevertheless, it significantly improved survival in patients with a pCR, downstaging, or a negative circumferential margin.110,111,144,145
Early systematic reviews concluded that preoperative RT at a BED of >30 Gy reduced LR risk and improved OS,25,146 and that no significant difference was observed in outcomes for different time intervals between conventional neoadjuvant CRT and surgery. 147 Moreover, higher doses increased LR reduction. A linear dose–response effect of BED was seen on the risk reduction of LR, and an exponent correlation was detected between LR and BED (Figure 4).15,37,74,78,79,114,148,149 Each 1 Gy increase in BED would reduce LR rates by 1.36–1.72%; hence, it was proposed that a BED of approximately 68.8–73.5 Gy would be needed to achieve 100% LC. 29
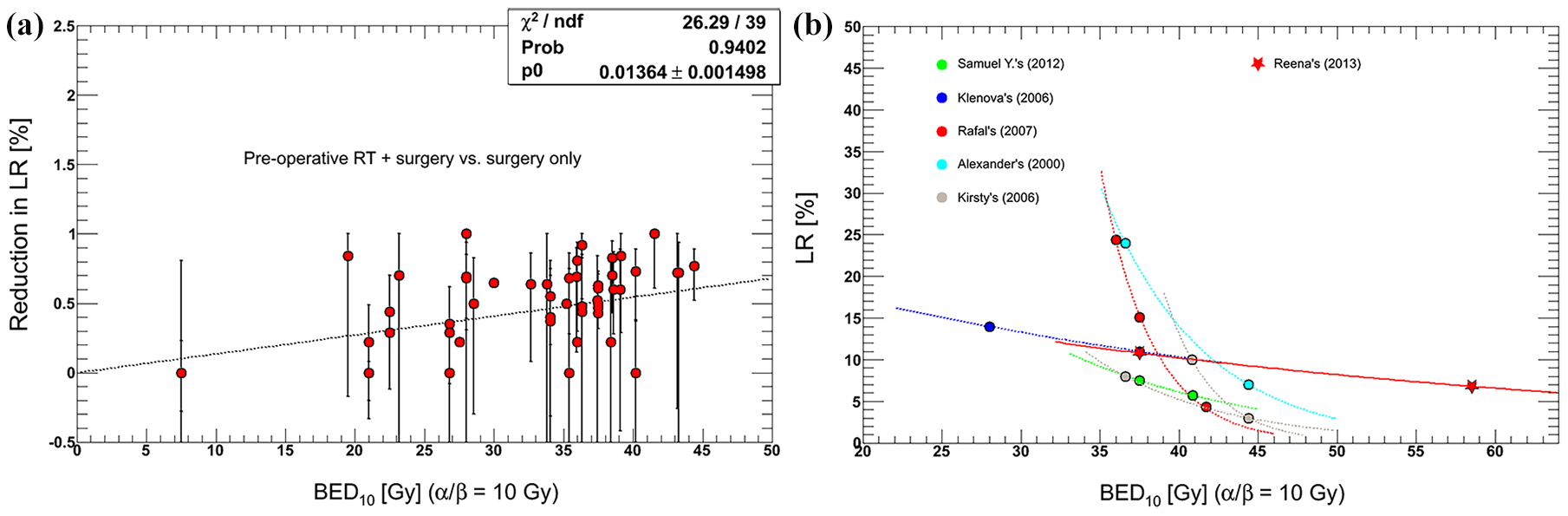
Comparison of reported dose–response relationships between BED10 and LR.
In contrast to the linear effect of BED on reduction in LR, the effects of BED on DFS and OS were not linear due to considerable heterogeneities. After dose escalation with three dose levels of 40 Gy, 46 Gy and 50 Gy, 2-year actuarial LR-free survival rates were 72%, 90%, and 89%, respectively; DFS rates were 62%, 84%, and 78%, respectively; OS rates were 72%, 94%, and 92%, respectively. A statistically significant increase in survival was seen with doses of ⩾46 Gy, but there was no difference in survival between doses of 46 Gy and 50 Gy. 79 However, after a long follow up of 11.9 years, patients with a concomitant dose boost (52.5 Gy) exhibited higher rates of 10-year OS than those for whom a conventional approach was used (45 Gy; 71.6% versus 62.4%). 100
As a radiation sensitizer, chemotherapy may augment RT. It may also sterilize circulating micrometastases and impede disease progression and distant organ involvement. 150 Prolongation of DFS and OS are ultimately expected. However, previous studies have demonstrated that a combination of preoperative RT and preoperative, with or without postoperative fluorouracil-based chemotherapy, would only further increase LC, without showing any significant differences in DFS and OS.58,151 However, it might benefit patients with a tumor 10–15 cm from the anal verge in terms of DFS. 152 Recently, the final results of the German CAO/ARO/AIO-94 trial showed that DFS at 3 years could be improved after adding oxaliplatin to fluorouracil-based neoadjuvant CRT for patients with cT3–4 rectal cancer. However, this trial had serious methodological shortcomings. 55 Although the benefits of chemotherapy on DFS are limited, as shown by the published data, current guidelines continue recommending a chemotherapy course because there is no sufficient evidence to conclude there is no absolute benefit of chemotherapy.153,154
Side effects: toxicities and complications
Preoperative CRT can induce serious side effects such as diarrhea, urinary tract infection, sexual dysfunction, and secondary malignancies.155–157 Meanwhile, toxicities and complications related to RT have also increased with the greater use of neoadjuvant CRT.158,159 The impact of RT on sexual, urinary, and anal functions has been documented in many previous trials, although surgery is likely to be the major factor.160–162 However, in 2019, a prospective study demonstrated that neoadjuvant CRT for lower rectal cancer did not affect postoperative urinary function, 163 treatments, the timing, and evaluation methods vary largely among these trails. Direct investigations of the effect of RT dose on the anorectal function have been reported recently; 164 a higher RT dose to anal sphincter complex tends to worsen the long-term anorectal function.
Furthermore, many trials demonstrated that there were no significant differences in severe late toxicity and quality of life between short-course RT with immediate surgery, and conventionally fractionated CRT with delayed surgery; however, CRT clearly increased the grade 3–4 acute toxicity.5,14,15 A recent retrospective analysis revealed that a dose boost did not increase the grade ⩾ 2 chronic toxicity after neoadjuvant CRT. 165 Interestingly, the Radiation Therapy Oncology Group (RTOG) trial 0012 compared hyperfractionated radiation (55–60 Gy) with once-daily radiation (50–55 Gy) and also found the similar acute and late toxicities. 57 Regarding the effect of timing intervals on toxicity, although the Stockholm III trial revealed that acute toxicity was only <1% after RT with immediate surgery compared with 5–7% after RT with delay, it is possible that these toxicities were obscured by early postoperative complications. 5
The addition of chemotherapy to preoperative RT has a sound radiobiological rationale,58,151 but will simultaneously increase grade III and IV acute toxicities.166,167 In a study, gastrointestinal (GI) toxicity was more frequently observed in the CRT group than in the RT-alone group (28.1% versus 12.9%, respectively); 168 a consequence of the increased toxicity was that the patients could not receive the full treatment or they experienced interruptions that could have a negative impact on outcomes. At present, no statistical difference was reported in late toxicity between the preoperative RT and CRT groups.
In summary, moderate RT dose escalation using the 3D-CRT technique and appropriate chemotherapy administration might be effective. The optimal time interval depends on clinical endpoints. There still remains a scope for the optimization of RT/CRT schedules. Although there is conflicting evidence because of various factors, strategies with the potential to improve outcomes, while reducing toxicities, are needed to guide future designs.
The era of IMRT
Early in 1993, MacFarlane and colleagues reported that total mesorectal excision (TME) instead of conventional surgery had led to substantial improvements in morbidity and survival. 169 It is hoped that improvements in RT techniques will further reduce LR and adverse events, and increase the survival. As an innovative technique, IMRT allows conformal dose distribution in the target while sparing the bladder and bowels. It is of critical importance for accurate target determination and strict dose–volume constraints. With the integration of image guidance and IMRT, both a more precise definition of target volume and accurate irradiation are allowed. Organ motion with changes in shape, size and position can be observed; a small target margin can be applied, consequently reducing potential toxicity. Using a synchronous integrated boost (SIB) technique, the dose per fraction can be further increased to the primary tumor while shortening the treatment time. However, adequate quality controls of procedures are always required. 155
Point: IMRT improves clinical endpoints
Multiple retrospective studies have shown that preoperative IMRT or volumetric-modulated arc therapy (VMAT, arc-based IMRT) is associated with a clinically significant reduction in GI or genitourinary (GU) toxicity, with or without improvement in LC compared with 3D-CRT.106,107,170–172 Furthermore, these modalities can potentially prevent delays in time to surgery and reduce hospitalizations, emergency department visits, and treatment breaks. 173 However, no significant differences were noted in tumor responses, DFS and OS. 107
Furthermore, several prospective studies have shown encouraging results. Preoperative IMRT with an SIB [46 + 55.2 (Gy) in 23 fractions] was explored. Surgery was performed 6 weeks later. 174 The grade ⩾ 3 late GI and GU toxicity was 9% and 4%, respectively; 5-year LC and OS were 97% and 68%, respectively. These values were in line with the results after preoperative CRT.12,151 In order to reach the best loco–regional control and to prevent systemic relapse, RT and chemotherapy are usually integrated. A Turkish study adopted hypofractionated RT (33 Gy/10 fractions), with concurrent oral capecitabine. Surgery was scheduled 6–8 weeks after the end of CRT; 11.5% of patients had pCR, and no grade 3–4 toxicity was observed. 175 Another phase II trial studied IMRT (47.5 Gy in 19/20 fractions) in combination with capecitabine and oxaliplatin (CAPOX). TME was scheduled 4–6 weeks after the CRT. A pCR was observed in 13% of patients, and major response in 48%, which seemed to translate into improved outcomes such as LC of 100%, DFS of 84%, and OS of 87%, after a median follow up of 55 months. 47
Moreover, preoperative IMRT with an SIB without dose escalation [41.8 + 46.2/48.4 (Gy) in 22 fractions], with concomitant capecitabine, was tested. Surgery was performed 6–8 weeks later. The rate of grade ⩾ 3 acute toxicity was 2.4%. A total of 25.5% patients achieved pCR, with 2-year LC, DFS, and OS rates of 100% for these patients. 92 If dose was escalated with an SIB [46 + 57.5 (Gy) in 23 fractions], and concomitant with capecitabine, surgery was planned around 8 weeks. A total of 30.6% of patients could achieve pCR with quite acceptable toxicity profiles. 44 At a median follow up of 38.2 months, the similar treatment schemes [45 + 55 (Gy) in 25 fractions, capecitabine, surgery 8 weeks later] resulted in 2-year DFS and OS of 90% and 90%, respectively, with a high pCR rate of 35%. 41
To obtain a better tumor response, elevating treatment dose has been considered a feasible method. Preoperative capecitabine and IMRT with an SIB [45 + 55 (Gy) in 25 fractions] were used and TME followed 6 weeks later. The crude pCR rate was up to 38%, with 50% achieving downstaging. 91 Recently, a near-total neoadjuvant approach was tested using multiagent chemotherapy, that is, sequential short-course IMRT (5 × 5 Gy) and FOLFOX (fluorouracil, leucovorin calcium, and oxaliplatin) followed by TME. A higher T downstaging of 75% and a superior 3-year DFS rate of 85% were observed compared with conventional neoadjuvant CRT (41% and 68%, respectively). 94
Counterpoint on IMRT and corresponding deliberation
Although preoperative IMRT has shown improved oncological outcomes, conflicting results are constantly being published. A retrospective study has demonstrated that IMRT was associated with worse R0 resection rates and sphincter preservation, without any differences in pathologic downstaging, unplanned readmission, or long-term OS. 176 A prospective phase II study used VMAT-SIB [45 + 57.5 (Gy) in 25 fractions] and two-drug chemotherapy CAPOX. Radical resection was performed 8 weeks after treatment. Although a very high tumor response was achieved, an acute toxicity rate of 44% was also recorded. 50 RTOG 0822 studied IMRT (25 × 1.8 Gy), followed by a boost (3 × 1.8 Gy) using 3D-CRT with concurrent CAPOX. Surgery was performed 4–8 weeks after CRT. A grade ⩾ 2 GI toxicity rate of 51.5% occurred preoperatively, which substantially exceeded the target rate of 28%. 177
The role of preoperative IMRT in rectal cancer remains to be determined at this juncture. Moreover, the addition of different chemotherapy and different treatment sequences confound it more. CRT-to-surgery interval also affects these clinical endpoints in the era of IMRT. 178 More trials with the prospective aim to further explore the efficiency of preoperative IMRT are expected.
Meanwhile, the limitations and potential difficulties inherent to IMRT, that is, dose inhomogeneity and integral dose, must be considered. Patient selection is of utmost importance. IMRT is also technically challenging, because the oncological outcomes are highly dependent on accurate target determination and dose–volume parameters. Careful quality assurance with regards to target delineation, image guidance, and plan optimization constraints is needed prior to treatment.
Using the SIB technique, two different doses per fraction are usually delivered in two different target regions, that is, a two-target approach. Neoadjuvant chemotherapy prior to preoperative RT/CRT gives us a chance to induce tumor regression, which allows the dose to the macroscopic postchemotherapy tumor to be increased by several additional Gy using the third targets. Additionally, a dynamic target could be generated within the frame of adaptive RT to accompany dose escalation. Also, it appears promising to harness functional imaging to guide dose to subvolumes of the target with a high tumor load and de-escalate dose to low-risk volumes.
In summary, trimodality therapy for rectal cancer inherently has uncertainties: treatment sequence, timing, and duration of the various modalities. Many treatment paradigms have been tested, such as surgery ± adjuvant RT/CRT, and preoperative RT/CRT/chemotherapy ± surgery ± CRT (Figure 5). However, until now, most fractionation schedules in preoperative RT have been empirical and based on the outcome of clinical trials. Fractionation schedules and the time interval are rather homogeneous across various institutions. Given patient selection and other treatment interventions, one cannot accurately assess whether and to what extent they influence clinical outcomes. If a radiobiological response model for fractionation is established on the basis of previous clinical studies, the controversy regarding dose fractionation schedules and time interval may disappear.

Treatment schemes for IMRT and CRT.
In addition, with the widespread standardization of surgery, diversification of drug, and precision of RT, the specific modality will be eliminated or used more sufficiently for a subset of patients, such as a ‘watch and wait’ strategy after preoperative RT/CRT, neoadjuvant chemotherapy only, and multidrug CRT. The priority for future research should be subgroups of patients who might receive relatively greater benefit from innovative treatment techniques. Moreover, with the development of technology and change in people’s understanding, the optimal regimen will also constantly change. These studies will be critical to further implementation of precision medicine through maximizing clinical outcomes, while minimizing associated toxicities.
New era: particle RT such as proton and heavy ions
Particle RT has recently garnered great attention. It can deliver radiation with a highly conformal dose distribution while maintaining minimal excess dose to normal tissues. Additionally, it is coupled with various biological advantages, especially for heavy-ion beam, such as a lack of oxygen effect and less cell cycle-related radiosensitivity. It enables treatment of diseases that are inaccessible with conventional RT, for example, postoperative recurrence of rectal cancer.
A recent report has shown that patients were treated with 73.6 GyE (physical dose multiplied by relative biological effectiveness) in 16 fractions using carbon ion. The 5-year LC rate was 88% and survival was 59%. 179 These figures are higher than those with photon RT. Moreover, particle RT might be further optimized by dose escalation or hypofractionation. Given a high rate of distant metastases in most studies, concurrent and adjuvant systemic therapies should also be investigated.
Supplemental Material
Supplementary_Figure – Supplemental material for Dose–time fractionation schedules of preoperative radiotherapy and timing to surgery for rectal cancer
Supplemental material, Supplementary_Figure for Dose–time fractionation schedules of preoperative radiotherapy and timing to surgery for rectal cancer by Fu Jin, Huanli Luo, Juan Zhou, Yongzhong Wu, Hao Sun, Hongliang Liu, Xiaodong Zheng and Ying Wang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Fu Jin and Huanli Luo contributed equally to this work. The authors thank the following colleagues for their assistance and advice in this study: Shi Li, Xia Tan, Xianfeng Liu, Xia Huang, Qicheng Li, Mingsong Zhong, Han Yang, Chao Li, Yanan He, Xiumei Tian, Da Qiu, Guanglei He, Li Yin, Guang Li, and Bo Li. The interim findings on relationships between biologically effective dose and pathological complete response, and local recurrence were presented at the ESMO as a poster. The poster’s abstract was published in Annals of Oncology (2019) 30 (suppl_9):ix30-ix41.10.1093/annonc/mdz421.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Natural Science Foundation of China under grant no. 11575038 and 11805025.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
