Abstract
Background:
Intensive triplet chemotherapy/bevacizumab significantly increased metastatic colorectal cancer (MCRC) outcome. This phase II study investigated the safety/activity of FIr-C/FOx-C triplet/cetuximab (CET) in first-line RAS wild-type and the prediction of individual limiting toxicity syndromes (LTS) by pharmacogenomic biomarkers.
Methods:
A Simon two-step design was used: p0 70%, p1 85%, power 80%, α5%, β20%; projected objective response rate (ORR) I step 14/19. FIr-C/FOx-C: 5-fluorouracil (5-FU) 12h-timed flat infusion 900 mg/m2 d1–2, 8–9, 15–16, 22–23; irinotecan (CPT-11) 160 mg/m2 d1 and 15, oxaliplatin 80 mg/m2 d8 and 22; CET 400mg/m2 then 250 mg/m2 d1, 8, 15, 22; every 28 days. Toxicity, and individual LTS were evaluated, compared by a Chi-square test; and activity/efficacy by log-rank. 5-FU/CPT-11 pharmacogenomic biomarkers, 5-FU degradation rate (5-FUDR), single nucleotide polymorphisms (SNPs) ABCB1, CYP3A4, DYPD, UGT1A1 were evaluated in patients with LTS and at a recommended dose.
Results:
A total of 29 patients <75 years, with a primary/intermediate Cumulative Index Rating Scale were enrolled; the median age was 59 years; there were 7 young-elderly (yE; 24%). Recommended CPT-11/5-FU doses were 120/750 mg/m2. In the intent-to-treat analysis, the ORR was 58.6%. The primary endpoint was met in patients who received the planned three treatment cycles: the objective response (OR) was 14/18 (78%). At a median follow up of 18 months, progression-free survival (PFS) was 12, and overall survival (OS) was 23 months. At the recommended doses (received dose intensity >80%), grade 3–4 toxicities were: diarrhea 23%, asthenia 15%, vomiting 8%, hypertransaminasemy 8%; LTS 19 (65.5%), with 83% in yE patients. LTS prevalently multiple (ms) versus single site were 59% versus 7% (p = 0.006). The prevalence of reduced FUDR was 56%, SNPs CYP3A4 22%, UGT1A1 71%, and of >2 positive pharmacogenomics biomarkers was 78%, prevalently reported in patients who developed gastrointestinal LTS.
Conclusions:
FIr-C/FOx-C is highly active and tolerable at recommended doses in non-elderly RAS wild-type MCRC patients. LTS provided an evaluation of the toxicity burden in individual patients. Reduced FUDR, CYP3A4, and UGT1A1 SNPs may predict individual LTS-ms in patients at risk of limiting gastrointestinal toxicity.
Trial registration:
The trial was registered at Osservatorio Nazionale sulla Sperimentazione Clinica dei Medicinali (OsSC) Agenzia Italiana del Farmaco (AIFA) Numero EudraCT 2009-016793-32.
Keywords
Introduction
Therapeutic strategy of metastatic colorectal cancer (MCRC) is planned by predictive/prognostic biomarkers and the patients’ fitness for selected targeted drugs, antivascular endothelial growth factor (anti-VEGF) or anti-epidermal growth factor receptor (anti-EGFR), associated with doublet or more intensive triplet chemotherapy, properly weighing the expected clinical outcome with toxicity in individual patients. 1
A major concern of adding more drugs in an intensive chemotherapy regimen is schedule design, to obtain the active and tolerable dose intensity (DI) for each drug. We previously showed that 12-hour (10 p.m. to 10 a.m.) timed flat infusion (TFI) 5-fluorouracil (5-FU) could be safely associated with irinotecan (CPT-11) at a high 5-FU/DI without leucovorin. We thus, designed FIr/FOx triplet chemotherapy consisting of weekly TFI/5-FU and alternating CPT-11 and oxaliplatin (OXP), 2 and developed a FIr-B/FOx schedule adding bevacizumab (BEV), reaching an objective response rate (ORR) of 82%, progression-free survival (PFS) of 12 months, and overall survival (OS) of 28 months, consistent with those reported by the FOLFOXIRI/BEV schedule, not significantly different in the KRAS exon 2 wild-type and mutant.3–6 In the phase III TRIBE trial, FOLFOXIRI/BEV significantly increased clinical outcomes over the doublet plus BEV, and reached a median OS of 37.1 months in the RAS wild-type. 7
The anti-EGFR drugs, cetuximab (CET) or panitumumab (PAN), added to doublet chemotherapy in EGFR-overexpressing and KRAS exon 2 wild-type MCRC reported an ORR of 39–68%, a PFS of 7.2–10.6 months, and an OS of 19.9–26.1 months. 1 In the RAS wild-type, in a recent study, CET or PAN added to FOLFOX or FOLFIRI reached a median PFS of 10.1–13 months, and an OS of 26–41.3 months.8–13 Anti-EGFR addition to triplet chemotherapy was evaluated in phase I and II trials reaching an ORR of 79–80.9%, a PFS of 9.5–14 months, and an OS of 24.7–37 months, in molecularly unselected patients; in the KRAS exon 2 wild-type, 60–83.3%, 9.9–16 months, 30.3 to not reached; in RAS wild-type, 67.8–89%, 11.2–11.3 months, not reached, respectively.14–19 The range of reported individual grade 3–4 toxicities of diarrhea (25–53%), asthenia (9.5–31.7%), mucositis (6–14%), neutropenia (23.3–48%), and febrile neutropenia (3–6%), represented the main concern for intensive regimens when adding anti-EGFR to triplet chemotherapy. Pharmacogenomic biomarkers, specifically 5-FU degradation rate (5-FUDR) and single nucleotide polymorphisms (SNPs) of different genes, such as ABCB1, CYP3A4, DYPD, UGT1A1 encoding for enzymes involved in 5-FU and CPT11 metabolism have been evaluated to predict gastrointestinal toxicity in individual patients. 20
The present phase II study evaluated a first-line FIr-C/FOx-C schedule, adding CET to FIr/FOx triplet chemotherapy, in RAS wild-type MCRC, to assess the activity and safety at recommended doses. 5-FUDR and ABCB1, CYP3A4, DYPD, UGT1A1 SNPs were detected and related to gastrointestinal-limiting toxicity in individual patients to evaluate their predictive relevance.
Materials and methods
Patient eligibility
Patients were eligible if they had histologically confirmed diagnosis of measurable MCRC; KRAS exon 2 wild-type, then KRAS/NRAS wild-type tumors; age 18–75 years, specifically <65 years (non-elderly), ⩾65 and <75 (young-elderly, yE); World Health Organization performance status (PS) ⩽2; adequate hematological, renal, hepatic functions; life expectancy >3 months. The ineligibility criteria were: pregnancy and breast-feeding; uncontrolled severe diseases; cardiovascular disease (uncontrolled hypertension, arrhythmia, ischemic cardiac diseases in the last year); thromboembolic disease, coagulopathy, pre-existing bleeding diatheses; surgery within the previous 28 days; previous adjuvant chemotherapy or radiotherapy completed <6 months before.
The Cumulative Index Rating Scale (CIRS) was used to evaluate comorbidity status, and only patients with primary and intermediate CIRS stage were enrolled. 21 The primary CIRS stage consisted of Independent Instrumental Activity of Daily Living (IADL), and absent or mild grade comorbidities; intermediate CIRS stage, dependent or independent IADL, and <3 mild or moderate grade comorbidities. Patients with secondary CIRS stage, consisting of ⩾3 comorbidities or a severe comorbidity, with or without dependent IADL, were not enrolled.
Treatment was approved by Agenzia Italiana del Farmaco for administration in-label for MCRC treatment in Italian public hospitals, and published in Gazzetta Ufficiale Repubblica Italiana (N.1, 2 Gennaio 2009). The study was approved by the local ethical committee (Comitato Etico, Azienda Sanitaria Locale n.4 L’Aquila, Regione Abruzzo, Italia) and conducted in accordance with the Declaration of Helsinki. All patients provided written informed consent for inclusion in the present study, regarding the proposed treatment and specifying the addition of exploratory pharmacogenomic analysis to better address individual toxicity.
Schedule
This was a single-arm, single-center phase II study evaluating the activity and safety of 5-FU, CPT-11, OXP, CET (FIr-C/FOx-C) as a first-line treatment of RAS wild-type MCRC.
The FIr-C/FOx-C schedule consisted of 5-FU associated with alternating CPT-11/CET or OXP/CET according to the following weekly schedule: TFI/5-FU (Fluorouracil Teva; Teva Italia, Milan, Italy) 900 mg/m2/day, over 12 hours (from 10:00 p.m. to 10:00 a.m.), on days 1–2, 8–9, 15–16, and 22–23; CPT-11 (Campto; Pfizer, Latina, Italy) 160 mg/m2, as a 90-min intravenous infusion in NaCl 0.9% 250 ml, on days 1 and 15; cetuximab (Erbitux; Merck, Darmstadt, Germany), at a loading dose of 400 mg/m2, followed by 250 mg/m2, intravenous infusion in NaCl 0.9% 600 ml over 120 min at first time, then 300 ml over 60 min, on days 1, 8, 15, and 22; OXP (Eloxatin; Sanofi-Aventis, Milan, Italy) as a 2-hour intravenous infusion in dextrose 5% 250 ml, 80 mg/m2, on days 8 and 22. Cycles were repeated every 4 weeks. 5-FU was administered by portable pump (CADD Plus, SEVIT) using a venous access device. In subsequent dose-finding steps, 5-FU and CPT-11 were reduced at recommended doses 750 mg/m2 and 120 mg/m2, respectively, due to limiting diarrhea.
Biomarkers and pharmacogenomic analyses
The KRAS/NRAS genotype was evaluated on paraffin-embedded tissue blocks from the primary tumor or metastatic site by direct sequencing. KRAS/NRAS exon 2–4 sequence reactions were performed from polymerase chain reaction (PCR)-amplified tumor DNA using a Big Dye V3.1 Terminator Kit, electrophoresis in ABI Prism 3130xl Genetic Analyzer, and analyzed using GeneMapper Analysis Software, version 4.0 (Applied Biosystems, Foster City, CA, USA), as described elsewhere.5,6
Exploratory pharmacogenomic analyses consisted of: 5-FUDR defining reduced metabolizers if degradation rate was <1.2 ng/ml/106 cells/min; and the detection of seven SNPs of four genes involved in 5-FU and CPT-11 metabolism, ABCB1, C3435T and C1236T; CYP3A4, 1B and 53; DYPD1, IVS14+1 and A166G; UGT1A1, 28. First, they were evaluated on-treatment in patients with limiting toxicity syndrome (LTS) at a first dose level; then, they were evaluated before starting treatment at a recommended dose level.
Study design
The primary endpoint was ORR, evaluated according to the Response Evaluation Criteria in Solid Tumors 22 ; activity evaluation was planned after three cycles of administered treatment. Secondary endpoints were toxicity, PFS, OS, analyzed using the Kaplan–Meier method. 23 PFS was defined as the length of time between the beginning of treatment and disease progression or death (resulting from any cause) or last contact; OS was between beginning of treatment and death or last contact. A log-rank test was used to compare PFS and OS. 24 Patients were classified according to involved metastatic sites, liver-limited (L-L) and other/multiple metastatic sites (O/MM). 25 L-L patients were evaluated at baseline and every three cycles of treatment by a multidisciplinary team, consisting of a medical oncologist, liver surgeon and radiologist, to dynamically evaluate resectability according to previously reported categories. 25 Clinical evaluation of the response was made by computerized tomography scan; positron emission tomography was added based on the investigators’ assessment; and objective responses were confirmed 3 months later. Follow up was scheduled every 3 months up to disease progression or death. Toxicity was registered according to National Cancer Institute Common Toxicity Criteria (NCI-CTC, version 3.0). Limiting toxicity (LT) was defined as grade 3–4 non-hematological toxicity (mainly represented by diarrhea, mucositis, neurotoxicity, hand–foot syndrome, asthenia, skin rash), grade 4 hematologic toxicity, febrile neutropenia, or any toxicity determining >2 weeks treatment delay.
LTS, consisting of at least one LT associated or not with other limiting or grade 2 toxicities, were evaluated,3,26,27 and classified as: LTS single site (LTS-ss), if characterized only by the LT; LTS multiple sites (LTS-ms), if characterized by ⩾2 LTs or an LT associated with other, at least grade 2, non-limiting toxicities. A Chi-square test was used to compare the rates of LTS-ms and LTS-ss. 28
Statistical design
This phase II study was planned according to the two-step Simon’s design 29 : assuming an ORR of 70%, as a minimal interesting activity, 14 objective responses among the first 19 enrolled patients were necessary for the first step; to verify the alternative hypothesis of an ORR of 85%, 46 objective responses among 59 total patients enrolled were necessary; power (1 − β) was 80%; and the error probability, α, was 5%. The p0 was determined as the activity of triplet combinations plus BEV in overall MCRC3,4; p1 as projected ORR using the present intensive combination, increasing activity ⩾15% in genetically selected patients.
Alterations of 5-FUDR and ABCB1, CYP3A4, DYPD, UGT1A1 SNPs were evaluated and compared with LTS occurrence.
Results
Patient demographics
From January 2010 to June 2016, 29 consecutive, unselected patients, (16 KRAS exon 2 wild-type and 13 RAS wild-type), were enrolled. From April 2014, patients were prospectively tested for KRAS/NRAS mutational status, and KRAS/NRAS wild-type were eligible. RAS mutational analyses were performed in 25 primary tumors and four metastases (2 local recurrences, 1 lung, and 1 liver). Retrospective analysis of KRAS/NRAS genotype among 12 previously enrolled patients, revealed NRAS exon 2 and exon 3 mutant genotype in 2 patients (1 not evaluable for activity at a PFS of 21 months, and 1 with stable disease at a PFS of 8 months). Table 1 describes the patient features: male/female ratio (20/9); median age, 59 years; yE (7, 24%); PS 0–1/2 (26/3); CIRS primary/intermediate (10/19); metastatic disease metachronous (9, 31%), synchronous (20, 69%); liver metastasis (19, 65.5%); metastatic site single (15, 52%), multiple (14, 48%); L-L (7, 24%).
Patient features.

c. i., continuous infusion; CIRS, Cumulative Illness Rating Scale; WHO, World Health Organization.
Activity and efficacy
Among all 29 enrolled patients evaluable in the intent-to-treat (ITT) analysis (Table 2), the ORR was 58.6% [α 0.05, confidence interval (CI) ± 18]): 14 partial responses (48.3%), 3 complete responses (10.3%), 6 stable diseases (20.7%), no progressive disease.
Activity and efficacy data.
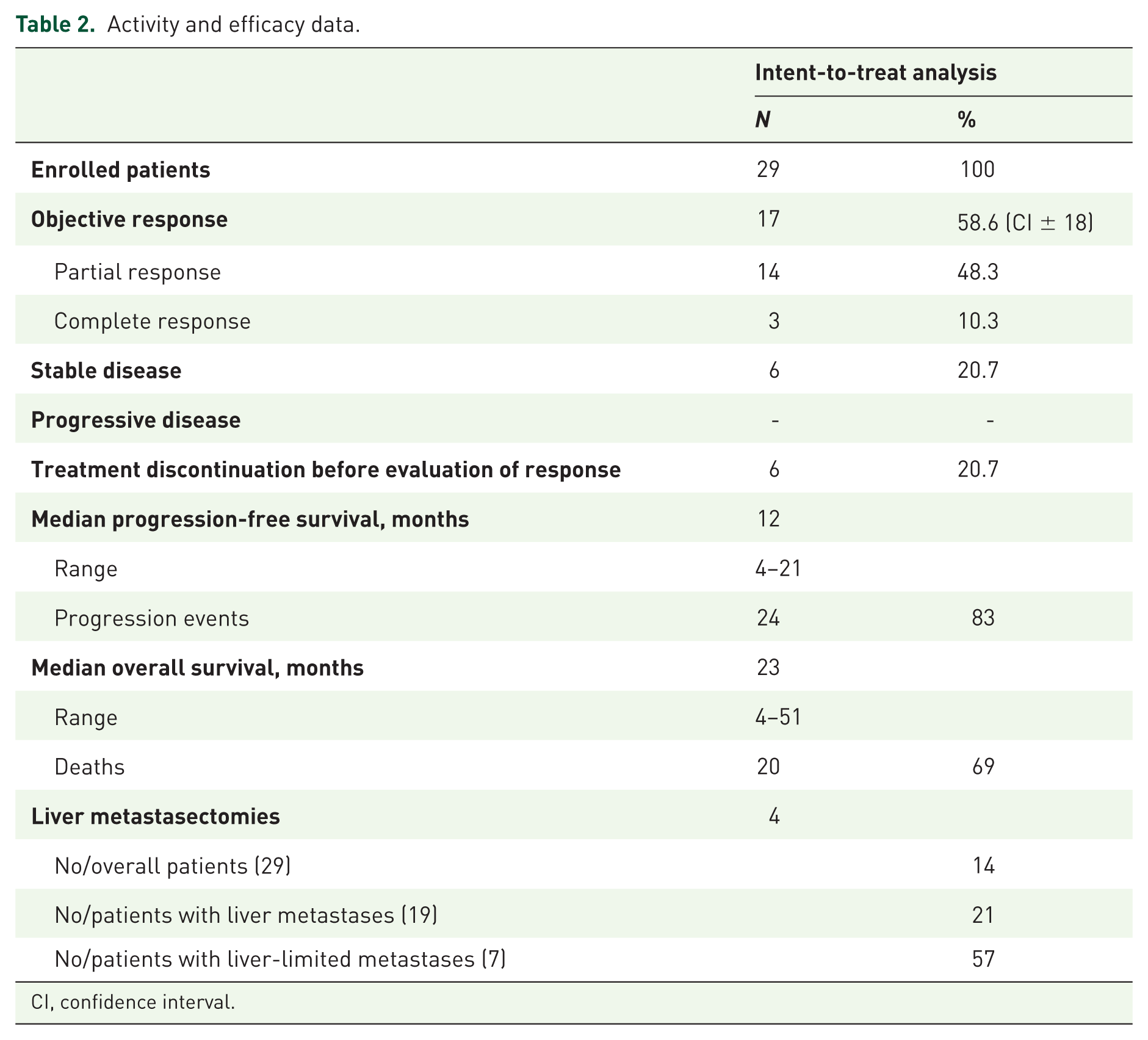
CI, confidence interval.
A total of six patients (20.7%) discontinued treatment due to LTS. Among the 23 patients who received at least two cycles of treatment, the ORR was 74% (α 0.05, CI ± 18). The expected 14 objective responses of the first step of Simon’s two-step design were obtained in 18 patients who received the planned three cycles of treatment (5 patients were evaluated after two cycles for secondary resection of metastases), with an ORR 78% (α 0.05, CI ± 20). The disease control rate was 100%. Among responsive patients, the median deepness of response was 50% (20–100%).
At a median follow up of 18 months (Supplementary Figure S1), the median PFS was 12 months (4–21): 24 events occurred, and five patients (17%) were progression-free. The median OS was 23 months (4–51): 20 events occurred, with nine patients (31%) alive. Among RAS wild-type, the median PFS was 12 months (4 to 20+), and the median OS was 23 months (4–51). Among the 23 patients who received at least two cycles of treatment, the median PFS was 11 months (5–20), and the median OS was 23 months (5+ to 48+). Among the 13 patients treated at the recommended doses, the ORR was 80% (α 0.05, CI ± 26). Liver metastasectomies were performed in 4 patients (14%); 4 out of 19 with liver metastases (21%); 4 out 7 with L-L (57%). The resection of metastatic lymph nodes and lung metastasis after the clinical complete response of liver metastases was performed in two patients. Among six yE patients, the median PFS was 12 months (5–14), and the median OS was15 months (9–48). The OS was significantly worse in yE patients compared with non-elderly (p = 0.045).
Dose-finding and toxicity
The projected FIr-C/FOx-C schedule was first evaluated in seven enrolled patients and 34 administered cycles [Supplementary Table S1(a)]; dose reductions were implemented due to prevalent LTS characterized by grade 3–4 diarrhea (four patients, 57%), also associated with limiting stomatitis/mucositis, skin rash, and neutropenia (1 patient, 14%). Thrombosis and cardiac ischemia were also observed in one patient (11%). Then, nine patients received CPT-11 at 140 mg/m2 and 5-FU at 800 mg/m2/day for 36 administered cycles [Supplementary Table S1(b)]; dose reductions were implemented due to prevalent LTS characterized by grade 3–4 diarrhea in six patients (66%), also associated with limiting asthenia in four patients (44%), hypokalemia in two patients (22%), neutropenia in three patients (33%), and grade 4 febrile neutropenia in one patient (11%). In the third dose-finding step, 13 patients received CPT-11 at 120 mg/m2 and 5-FU at 750 mg/m2/day and 49 cycles were administered (Table 3). Cumulative grade 3–4 toxicities, by patients, were: diarrhea (three patients, 23%), asthenia (two, 15%), and vomiting and hypertransaminasemy (one, 8%). Cumulative grade 2 toxicities were prevalently characterized by: diarrhea (eight patients, 61.5%), asthenia (four, 31%), and skin rash (three, 23%).
Cumulative toxicity: second amendment, recommended doses.

NCI–CTC, National Cancer Institute Common Toxicity Criteria.
Among the 29 enrolled patients and 119 administered cycles, cumulative grade 3–4 toxicities by patients were [Supplementary Table S1(c)]: diarrhea (13, 44%), asthenia (6, 21%), neutropenia (4, 13%), and vomiting (3, 10%). Cumulative grade 2 toxicities were prevalently characterized by: diarrhea (13, 45%), asthenia (10, 34%), vomiting (9, 31%), skin rash (6, 21%), and anorexia (6, 21%).
Overall, 14 out of 29 patients (48%) discontinued FIr-C/FOx-C treatment due to LT: diarrhea (10), asthenia (1), cardiac ischemia (1), thrombosis (1), and oxaliplatin infusional hypersensitivity reaction (1). LTS were observed in 19 patients [65.5%; Table 4; Supplementary Table 4(a)], yE (5 out of 6, 83%): LTS-ss (2, 7%), LTS-ms (17, 59%). LTSs were significantly represented by LTS-ms compared with LTS-ss (Chi-square, 7.703, p = 0.006). LTS-ms was characterized by LT associated with other, at least grade 2, non-limiting toxicities were detected in 10 patients (34%); ⩾2 LTs in 7 (24%). The two LTS-ss were characterized by: grade 2 neutropenia with >2 weeks treatment delay; and an oxaliplatin infusional hypersensitivity reaction. The seven LTS-ms, with double LTs, were characterized by grade 3–4 diarrhea and grade 3 asthenia associated with other toxicities, and grade 3 skin rash and grade 3 stomatitis/mucositis associated with grade 2 asthenia and diarrhea. The 10 LTS-ms, characterized by LT associated with other, at least grade 2, non-limiting toxicities, were prevalently characterized by grade 3–2 diarrhea associated with grade 2–3 asthenia (Supplementary Table S2).
Limiting toxicity syndromes: overall and in young-elderly patients.

LT, limiting toxicity; LTS, limiting toxicity syndrome.
Dose intensity
The median number of administered cycles was 3 (range 1–12). The median received DI (rDI) per cycle, at recommended doses of 5-FU at 750 mg/m2/day and CPT-11 at 120 mg/m2, were (Table 5): 5-FU 1280 (214.45–1800) mg/m2/w, 85.3%; CPT-11 56 (20–80), 93.3%; OXP 32 (10–40), 80%; and cetuximab 200 (58–287.5), 80%. The median rDI per patient was: 5-FU 1238 (457–1700) mg/m2/w, 82.5%; CPT-11 54 (20–80), 90%; OXP 30 (10–40), 75%; and cetuximab 206 (79–268.75), 82%. In yE, the median rDIs per cycle were: 5-FU 1066 (400–1400) mg/m2/w, 71%; CPT-11 45.5 (26–70), 76%; OXP 26.5 (0–40), 66%; and cetuximab 178.5 (58–250), 71%.
Dose intensity.

5-FU, 5-fluorouracil; CET, cetuximab; CPT-11, irinotecan; DI, dose-intensity; OXP, oxaliplatin.
Pharmacogenomic analyses
5-FUDR and ABCB1, CYP3A4, DYPD, UGT1A1 SNPs were evaluated and compared with LTS occurrence in 14 treated patients (48.3%; Table 6): after the treatment at the first dose level in the 4 who showed a LTS characterized by limiting diarrhea, out of 6 LTS; before starting treatment at the second and third recommended dose level, 10 out of 22 (45.5%), of whom 5 showed LTS and 5 did not. Overall, 9 patients out of 19 who showed LTS were evaluated (47.4%). Prevalence of pharmacogenomic alterations in 14 patients, and in 9 with or 5 without LTSs were: reduced 5-FUDR, 6/14 (43%), 5/9 (56%), 1/5 (20%); ABCB1, 10/14 (71%), 7/9 (78%), 3/5 (60%); CYP3A4, 2/14 (14%), all with LTS, 2/9 (22%); DYPD, 2 out of 13 (15%), all in 5 without LTS (40%); UGT1A1, 6 out of 12 evaluable (50%), 5 out of 7 (71%) and 1 out of 5 (20%). Overall, 9 out of 14 evaluated patients (65%) showed ⩾2 pharmacogenomic alterations, including a reduced 5-FUDR, CYP3A4 or UGT1A1 SNPs (range 1–3), 7 out of 9 with LTS (78%), all 6 with limiting diarrhea, and 2 out of 5 without LTS (40%). Pharmacogenomic data in the 14 individual patients (Supplementary Table S3) show that in the cohort of 4 patients with limiting diarrhea: 3 had reduced 5-FUDR and UGT1A1 SNPs (75%), 2 had CYP3A4+ (50%); all 4 showed ⩾2 pharmacogenomic alterations (range 2–4). In the cohort of 10 patients analyzed before starting treatment at third dose level (76.9%): 5 showed LTS, two characterized by limiting diarrhea, associated with UGT1A1 SNPs with reduced 5-FUDR in 1 patient; 5 did not show LTS.
Pharmacogenomic analyses: prevalence according to LTS.

FUDR, 5-fluorouracil degradation rate; LTS, limiting toxicity syndrome.
Discussion
Preliminary phase II data of CET addition to triplet chemotherapy according to the FIr-C/FOx-C schedule reported an ORR of 58.6% in the ITT analysis, due to a 20.7% discontinuation rate related to LT occurring before the first treatment evaluation. Overall, 14 out of 29 patients (48%) discontinued FIr-C/FOx-C treatment due to LT, mostly represented by diarrhea. The ORR among the 23 patients who received at least two cycles of treatment was 74%, with a PFS of 12 months, confirming that such an intensive regimen may contribute to increased efficacy, also by raising the resection rate of liver metastases, as a first-line treatment of RAS wild-type MCRC. Thus, the present study confirmed the need of proper selection of fit patients and evaluation of toxicity.14–19
Different schedules of CET addition to triplet chemotherapy, chrono-IFLO, ERBIRINOX, FOLFOXIRI have been proposed, and reported a high activity (ORR 70–83%), and liver resection rate >60%.14–17 A phase II trial in 37 KRAS/NRAS/HRAS/BRAF wild-type patients evaluated PAN addiction to modified FOLFOXIRI, with an ORR of 60–89%, R0 resection rate of 35%, median PFS of 11.3 and 13.3 months, respectively.18,19 The high discontinuation rate of started treatment (48%) due to LT, mainly characterized by diarrhea associated with asthenia, confirmed that individual patient toxicity was the main limitation to the use of intensive schedules associating triplet chemotherapy and anti-EGFR-targeted agents in clinical practice. BEV addiction to FOLFOXIRI in a randomized phase III TRIBE trial reported increased clinical outcomes also in wild-type KRAS/NRAS/BRAF patients. 7
To more properly assess feasibility and individual toxicity, we proposed the FIr-C/FOx-C schedule, associating weekly, nightly-TFI/FU without folinic acid, and weekly alternating CPT-11/CET or OXP/CET, designed from the previously reported FIr-B/FOx schedule associating triplet chemotherapy with the anti-VEGF bevacizumab, that may achieve a preferable toxicity profile without neutropenia, high activity and clinical outcome consistent with those reported in phase III TRIBE trial (ORR 82%, PFS 12 months, and OS 28 months).3,4,30 The recommended doses of TFI/5-FU of 750 mg/m2/day and CPT-11 of 120 mg/m2 were obtained by progressive dose-finding, with a median rDI ⩾ 80% and cumulative grade 3–4 limiting toxicities of diarrhea (23%), asthenia (15%), vomiting, and hypertransaminasemy, without neutropenia, as previously reported with the FIr-B/FOx schedule (10%), while the FOLFOXIRI schedule added to BEV or CET was frequently characterized by limiting neutropenia (50%) and febrile neutropenia (8.8%).4,18 In Phase II studies associating CET with triplet chemotherapy in an every-2-weeks schedule, according to chrono-IFLO, ERBIRINOX, and FOLFOXIRI regimens, or PAN to modified FOLFOXIRI, prevalent limiting grade 3–4 were more consistent, respectively: diarrhea (35–53%); neutropenia (13–48%); and asthenia (12–31.7%).14–19 Thus, the FIr-C/FOx-C schedule may be more tolerable than other reported schedules, even if we confirm that intensive regimens adding CET to triplet chemotherapy frequently require proper clinical management of toxicity and treatment modulations due to moderate/severe toxicities.
To this aim, as previously proposed, we added the evaluation of individual LTS, in order to more properly evaluate the clinical relevance of toxicity in individual patients.3,26,27 Overall, the FIr-C/FOx-C schedule determined 65.5% of individual LTS, significantly more represented by LTS-ms (59%) than LTS-ss (p = 0.006), mostly characterized by LT associated with other at least grade 2 non-limiting toxicities (34%) or ⩾2 LTs (24%), prevalently diarrhea and asthenia associated with other toxicities, or skin rash and stomatitis/mucositis associated with asthenia and diarrhea. LTS were previously observed in 44% of MCRC patients treated with FIr-B/FOx, and equally involving single or multiple sites.3,26 In yE patients, the median rDI at recommended doses was <80%, and individual LTS were 83%, mostly LTS-ms, suggested elderly age >65 years as an exclusion criterion for intensive regimens associating triplet chemotherapy and anti-EGFR. With limitations of a small number of enrolled patients, the present preliminary phase II data showed that FIr-C/FOx-C may be a feasible schedule in clinical practice to gain expected rDI ⩾ 80% and high activity, weighed by an acceptable toxicity profile, except for elderly MCRC patients. Further prospective studies will confirm the feasibility and safety of the present schedule. To this aim, we proposed to add LTS as an indicator, providing careful evaluation of the cumulative toxicity burden in individual patients, also reflecting the addictive grade of specific toxicities determined by different drugs sharing common toxicity profiles, and inter-patient variability.
SNPs in the dihydropyrimidine dehydrogenase (DPD) gene (DPYD) and UGT1A1 influence fluoropyrimidines and CPT-11 adverse events, and justify inter-patient variability in the safety profile. 20 In two phase II trials of the triplet capecitabine, OXP, CPT-11 (COI regimen) plus BEV or CET, prevalent grade 3–4 toxicities were, respectively: diarrhea (19 and 46%), neutropenia (3 and 7%), and asthenia (0 and 7%). 31 An independent significant association with severe toxicity and treatment modifications was found for DPYD c.496A > G (p = 0.022) and c.1896 T > C (p = 0.027), trend for UGT1A1*28 (p = 0.054). 31 The UGT1A1*28 allele lead to decreased glucuronidation of the metabolite SN-38 and an increased risk of severe CPT-11-induced neutropenia.20,31,32 To further relate the occurrence of individual toxicity, specific companion analysis of pharmacogenomic biomarkers of FU and CPT-11 toxicity, including 5-FUDR, and ABCB1, CYP3A4, DYPD, UGT1A1 SNPs was performed and related with LTS occurrence in 14 individual MCRC patients (48.3%). The prevalence of pharmacogenomic alterations was 5-FUDR (43%), SNPs of ABCB1 (71%), CYP3A4 (14%), DYPD (15%), UGT1A1 (50%), in the range of those reported.20,32 With limitations of only 14 patients evaluated (48.3%), and particularly for the relationship between pharmacogenomic biomarkers and occurrence of LTS or not, the present exploratory analysis showed that: reduced FUDR was observed in 56% patients with LTS and only in one without LTS (20%); CYP3A4 SNPs were detected in two patients with LTS (22%) while they were not reported in five without LTS; UGT1A1 variants were prevalently detected in patients with LTS (71%), and only in one (20%) without LTS. ABCB1 SNPs were not differently represented according to LTS, and DYPD1 SNPs were all detected in patients without LTS (40%). Most MCRC patients (65%), specifically those who developed gastrointestinal LTS (78%), showed >1 pharmacogenomic alteration, including reduced FUDR, CYP3A4 or UGT1A1 SNPs (range 1–3). The present exploratory data of pharmacogenomic biomarkers compared with LTS occurrence show that reduced 5-FUDR, and CYP3A4 and UGT1A1 SNPs may predict the occurrence of individual LTS, particularly at recommended dose, and specifically gastrointestinal LTS. Absence of alterations of these biomarkers may justify increased tolerability of triplet chemotherapy associated with anti-EGFR at the recommended doses. 5-FUDR, and CYP3A4, UGT1A1 SNPs may help the selection of patients fit for intensive regimens adding anti-EGFR to triplet chemotherapy, and require further validation in prospective studies. To this aim, the present study can represent a proof of concept concerning the effective integration of toxicity evaluation including LTS with companion pharmacogenomic analysis to more properly evaluate a selection of patients fit for an intensive regimen such as FIr-C/FOx-C, that should be prospectively evaluated as a model in clinical practice.
Conclusions
FIr-C/FOx-C intensive triplet chemotherapy plus CET in RAS wild-type MCRC may be a highly active and tolerable regimen at recommended doses in non-elderly patients, to be verified in the wider patient population.
Individual LTS could represent a parameter providing careful evaluation of the toxicity burden in individual patients and inter-patient variability. Integrated evaluation of LTS with pharmacogenomics biomarkers suggests predictive relevance of 5-FUDR, and CYP3A4, UGT1A1 SNPs for a priori identification of individual patients at risk of limiting gastrointestinal toxicity. Companion pharmacogenomic analyses need to be prospectively validated in clinical trials.
Supplemental Material
846421_supplementary_mater – Supplemental material for Intensive first-line FIr-C/FOx-C association of triplet chemotherapy plus cetuximab in RAS wild-type metastatic colorectal cancer patients: preliminary phase II data and prediction of individual limiting toxicity syndromes by pharmacogenomic biomarkers
Supplemental material, 846421_supplementary_mater for Intensive first-line FIr-C/FOx-C association of triplet chemotherapy plus cetuximab in RAS wild-type metastatic colorectal cancer patients: preliminary phase II data and prediction of individual limiting toxicity syndromes by pharmacogenomic biomarkers by Gemma Bruera, Silvia Massacese, Francesco Pepe, Umberto Malapelle, Antonella Dal Mas, Eugenio Ciacco, Giuseppe Calvisi, Giancarlo Troncone, Maurizio Simmaco and Enrico Ricevuto in Therapeutic Advances in Medical Oncology
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
