Abstract
Cardiac tumors are a very rare condition. Mostly, they are benign tumors (75%), with myxomas being the most frequent. The remaining 25% are malignant; either primary malignant sarcoma or secondary metastases. Given the small number of cases reported and the lack of prospective and randomized clinical trials, the level of evidence for the optimal multimodal treatment of primary cardiac sarcomas is very low and the optimal imaging diagnostic workup is not well established. In particular, 18F-FDG-PET/CT is not yet included in routine diagnosis of cardiac masses. Here, we report four illustrative clinical cases and a review of the literature on the current available data on the role of 18F-fluorodeoxyglucose PET/CT imaging in cardiac tumors.
Introduction
Cardiac tumors are a very rare disease. Thus, optimal diagnosis and treatment management are not well established, to date. Here, we report an overview on current available data in literature on these tumors, focusing on the role of imaging, with particular reference to 18-fluorine-fluorodeoxyglucose positron emission tomography/computed tomography (PET/CT).
Epidemiology of cardiac tumors
Cardiac tumors account only for a small fraction of all cardiac masses, mostly represented by pseudotumors like thrombi, vegetations, abscesses, aneurysms.
The estimated frequency of cardiac tumors ranges from 0.0017% to 0.33%, mostly represented by benign tumors (75%), with cardiac myxomas being the most common, accounting for nearly half of them. 1
Primary malignant cases account for 25% of all cardiac tumors, while secondary cardiac tumors, either by metastatic spread or by direct invasion, are far more common (40–50 times more frequent).
Autopsy series reviews found a 2.9–8.4% frequency of metastatic tumors involving the heart.2,3 Cardiac metastases originate mainly from lung, breast, kidney and cutaneous melanoma.
Among the primary malignant tumors of the heart, the most frequent are sarcomatous in nature. All types of sarcomas may be observed in the heart with a predominance of rhabdomyosarcoma in childhood and angiosarcomas or undifferentiated sarcomas in adulthood. They affect mostly patients in the fourth decade of life and survival remains extremely poor: in older retrospective case series, regardless of the type of treatment, most patients died within 12–16.5 months after initial diagnosis. 4 In more recent series, survival reaches 38.8 months but only for patients who underwent complete resection in referral centers. 5
Treatment of cardiac sarcomas
Given the small number of cases reported and the lack of randomized clinical trials, the level of evidence for the optimal multimodal management of primary cardiac sarcomas is low. There is no standardized evidence-based approach which is then often extrapolated from that of extracardiac soft tissue sarcomas with the inevitable limits due to the cardiac location itself.
Complete surgical resection is rarely achievable in practice, given the aggressive and infiltrative nature of these tumors (only 13.3% of cases were resectable in the largest series of primary cardiac sarcomas reported by the French Sarcoma Group in a multi-institutional retrospective study). 5
Moreover, it is easy to understand how dose-limiting problems are related to radiotherapy. The value of preoperative or postoperative radiotherapy (an effective adjuvant tool for sarcomas arising at other sites) is very limited in the treatment of cardiac sarcomas considering that the heart is usually more sensitive than the tumor to radiation injury, and that the ventricular wall cannot be treated with a radical radiation dose without substantial risk of cardiomyopathy and chronic pericarditis.
Generally, surgical resection represents the cornerstone of therapy in primary localized cardiac sarcoma. Only surgical resection with pathologically negative margins (R0 resection) is associated with a prospect of prolonged survival. However, despite complete surgical resection, tumor recurrence and metastases occur early, mostly within 6–12 months, and frequently. A high rate of both relapse and distant metastases within the follow-up period is reported in a series of malignant cardiac tumors seen over 15 years (45.5% and 72.2%, respectively). 6
Neoadjuvant or adjuvant chemotherapy may be administered in order to downsize the tumor and facilitate R0 resection or to reduce risk of relapse and distant metastases. However, the rarity of these tumors hinders generation of reliable evidence in favor of or against neoadjuvant and adjuvant chemotherapy in cardiac sarcoma. In the literature, the optimal treatment approach regarding postoperative adjuvant therapy remains unclear: some studies found that the adjuvant approach failed to modify the natural course of the disease,7,8 while others have demonstrated a better survival for patients whose surgery was followed by adjuvant chemotherapy.6,9
Overall, anthracyclines (in particular, doxorubicin) and nitrogen mustard alkylating agents (in particular, ifosfamide), which represent the preferred chemotherapeutic choice to treat sarcomas originating from other sites, are the most common chemotherapy agents used also for cardiac sarcomas. However, the typical cardiotoxicity of anthracyclines should be kept in mind when adjuvant treatment is proposed to a patient.
In brief, given the limited and conflicting results of adjuvant treatment, this approach may be considered for a patient–physician shared decision making in a subset of selected fit and high-risk patients and, in any case, a strict follow up must be planned, even after apparently completed treatment.
In case of unresectable disease or presence of metastases, palliative chemotherapy should be offered, although in some cases, palliative surgical debulking may also be proposed, with the aim of relieving rapidly progressing symptoms.
As reported so far, cardiac sarcomas show a high degree of complexity: due to their location and dismal prognosis, a multidisciplinary approach is crucial from the very beginning of patient management. Because of their rarity, diagnosis, imaging techniques, surgical, radiotherapy or chemotherapy treatments and follow-up strategy still pose a serious challenge for physicians.
Imaging techniques
With regard to noninvasive diagnostic modalities, echocardiography and magnetic resonance imaging (MRI) have always represented the two most used and sensitive techniques to detect and attempt to characterize suspected cardiac masses.
Echocardiography, especially the transesophageal route, is usually the initial imaging modality. It may show the tumor, its extent and its hemodynamic consequences. Cardiac MRI provides further information about morphology, location and extent of the mass. In case of suspected cardiac malignancies, CT scan is also useful to assess extracardiac extent and distant metastasis, if any.
In particular, the multiplanar assessment of anatomy, tissue composition, and functional impact afforded by MRI, allows accurate confirmation of the presence of a space-occupying lesion, localization and assessment of the extent of involvement, evaluation of the functional impact of the lesion, as well as tissue characterization. Such information is important not only for diagnosis but also for prognosis determination and therapy planning. As such, cardiac MRI has rapidly evolved as the reference standard technique for early assessment and differentiation between a non-neoplastic mass and a tumor mass, be it benign or malignant.
A quick diagnosis facilitates the initiation of a proper treatment, which may in turn improve outcome of patients affected by these rare and aggressive neoplasms. Therefore, a high level of suspicion is required, and, over the years, efforts have been made to define criteria for malignancy of cardiac masses. Findings suggestive of a malignant cardiac tumor at MRI include: right atrial location; involvement of more than one cardiac chamber, size > 5 cm, hemorrhagic pericardial effusion, broad base of attachment, extension into the mediastinum or great vessels and a ‘moderate,’ ‘strong,’ or heterogeneous delayed enhancement pattern. 10
18 F-fluorodeoxyglucose positron emission tomography/computed tomography
Scattered case reports in literature describe how integrated PET/CT with FDG imaging can incidentally show cardiac neoplasia, particularly metastatic involvement,11–17 or may be used in case of suspected aggressive cardiac malignancy to strengthen the diagnosis and detect possible occult distant disease.18–26
However, unlike many other solid tumors, FDG PET does not have an established role in the routine evaluation of cardiac tumors, which is probably due both to their low frequency and to the physiologically high uptake of FDG in the myocardium, although with interindividual and intraindividual variability. 27
In the normal myocardium, metabolism is primarily oxidative and uses various substrates, including glucose, free fatty acids and lactate. The choice of substrate depends on its availability and the physiological and pathologic condition of the myocardium. The European Association of Nuclear Medicine (EANM) guidelines suggest that, in case of evaluation of a lesion in the heart or very close to the myocardium, dietary recommendations can be helpful. 28 Several reports indicate that a low-carbohydrate diet, associated with prolonged fasting, is adequate for shifting myocardium metabolism toward fatty acid consumption. The EANM 2015 guidelines recommend a low-carbohydrate diet for 24 h prior to the PET/CT study or, at least, a low-carbohydrate meal before starting the 6 h fasting period preceding the study.
In order to standardize the high-fat/low-carbohydrate meal, a very recent report by the Society of Nuclear Medicine and Molecular Imaging-American Society of Nuclear Cardiology (SNMMI-ASNC) Expert Consensus indicates in detail the foods permitted and prohibited before PET scanning. 29
Two larger studies have been conducted to assess the diagnostic value of 18F-FDG PET (integrated with CT or MRI imaging) in the diagnostic algorithm and in the noninvasive preoperative determination of malignancy and metastatic spread of cardiac tumors, with the ultimate aim of stratifying patients and selecting and monitoring therapies.30,31
In the study by Rahbar and Colleagues, 18F-FDG PET/CT scans (whole body imaging with low-dose CT) of 24 consecutive patients with newly diagnosed cardiac tumors were retrospectively analyzed. 30 The patients underwent standard oncological preparation for PET examination (with a fasting period of at least 6 h). As a result, background myocardial and blood pool uptake corresponded to a mean standardized uptake value (SUV) of 2.1 ± 0.6 and 1.6 ± 0.4, respectively. The maximum standardized uptake values (SUVmax) of the tumors were measured and compared between patients affected by benign and malignant cardiac tumors (both cardiac primaries or metastases). The study showed that mean SUVmax was 2.8 ± 0.6 in benign cardiac tumors and significantly higher both in malignant primary and in secondary cardiac tumors (8.0 ± 2.1 and 10.8 ± 4.9, respectively), (p < 0.001), assessing an SUVmax cutoff with high sensitivity at 3.5.
In the study by Nensa and Colleagues, 20 patients were prospectively assessed using integrated cardiac 18F-FDG PET/MRI to evaluate whether this integrated imaging provided significant benefit over 18F-FDG PET or MRI alone. 31 In order to optimally suppress physiologic glucose uptake in the myocardium, patients were adequately prepared with a high-fat, low-carbohydrate diet for a period of 24 h before the examination, and 50 IU/kg of unfractionated heparin was intravenously administered 15 min before 18F-FDG injection. Blood glucose levels at the time of tracer injection were less than 120 mg/dl. As a result, the mean SUV in the blood pool was 1.3 ± 0.3. The study showed that the mean SUVmax in all nonmalignant cases (benign tumors, thrombi, and scar tissue) was 2.3 ± 1.2, whereas the mean SUVmax in primary and secondary malignant tumors was significantly higher at 13.2 ± 6.2 (p < 0.0004). Despite the obvious bias of statistical power related to the poor sample size, in this study, the combination of 18F-FDG PET and MRI yielded 100% sensitivity and specificity. In fact, there were only two cases (both with an SUVmax of 5.2) that could not be differentiated using 18F-FDG PET alone, then unequivocally identified as malignancy and patch tissue by MRI, respectively. In another case, MR imaging misclassified scar tissue as local relapse of cardiac angiosarcoma, but a low SUVmax of 2.2 made malignant relapse unlikely.
Illustrative cases on the role of 18F-fluorodeoxyglucose positron emission tomography/computed tomography in cardiac tumors
Herein we report four illustrative clinical cases of patients affected by cardiac tumors, referred to our institution. 18F-FDG PET/CT imaging was performed during the patients’ oncologic examination, proving to be a very useful tool in detecting neoplasms and guiding physicians’ choices. The study protocol was approved by the ethics committee of Sant’Orsola Hospital-Bologna, Italy (approval no. 164/2017/O/Oss) as part of a large retrospective analysis of patients with rare tumors. All patients provided written informed consent for inclusion in the study (Figures 1–4).
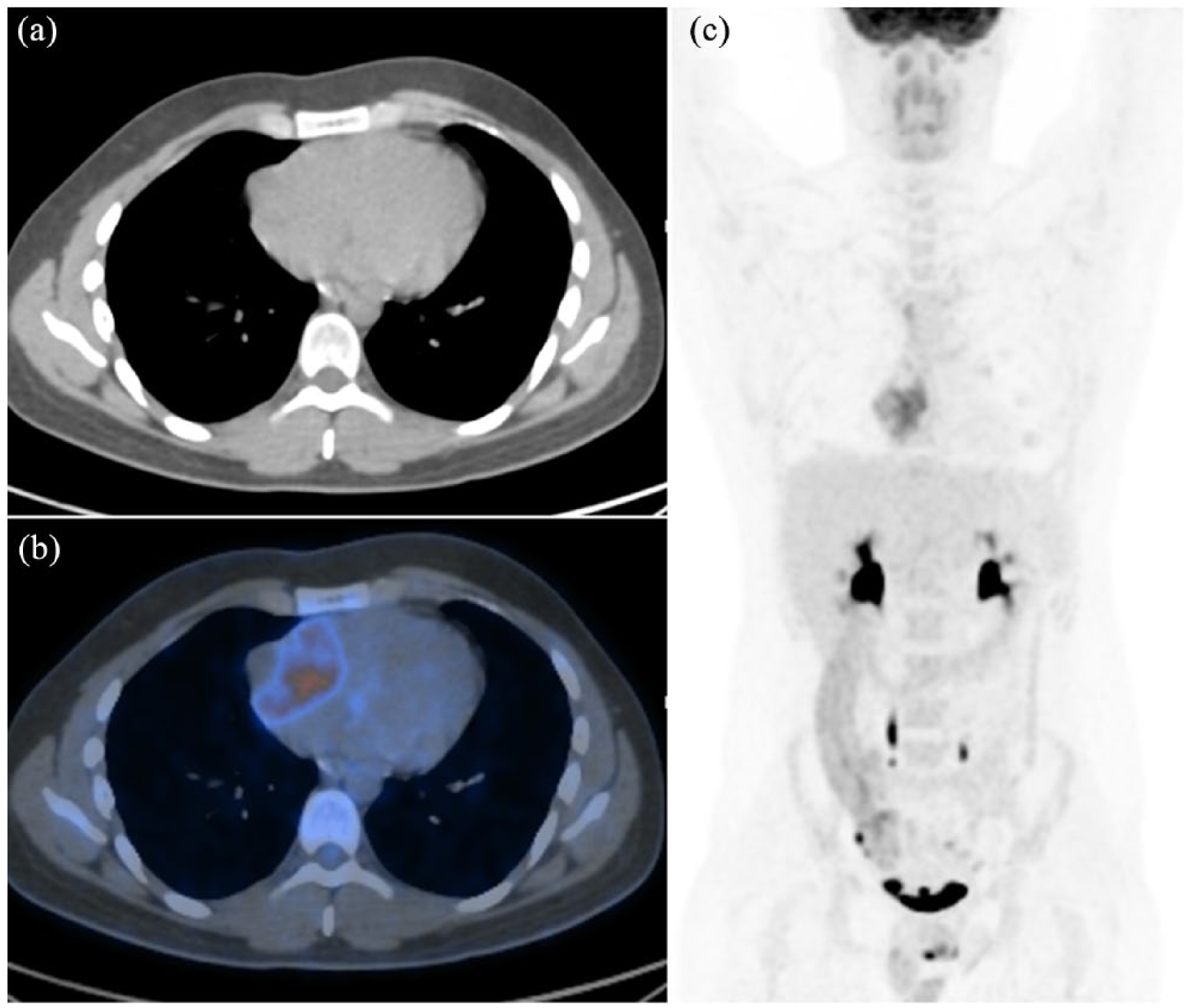
18F-fluorodeoxyglucose positron emission tomography/computed tomography images.

18F-fluorodeoxyglucose positron emission tomography/computed tomography images of a 74-year-old man with abnormal uptake in the right ventricule.

18F-fluorodeoxyglucose positron emission tomography/computed tomography images of a 38-year-old man with abnormal uptake (SUVmax = 8.7) in the right atrium.

18F-fluorodeoxyglucose positron emission tomography/computed tomography images.
Conclusion
Cardiac tumors are very uncommon. Given their rarity and complexity, optimal diagnosis and treatment management are still unknown.
Within the noninvasive diagnostic modalities to characterize suspected cardiac masses, echocardiography and MRI have represented the two most used and sensitive techniques.
18F-FDG PET has not an established role in the imaging workup of cardiac tumors, while it may have a substantial meaning in the diagnostic algorithm and in the noninvasive preoperative determination of malignancy and metastatic spread of cardiac tumors.
Although based on small case series, the few available studies, as well as our illustrative cases reported, suggest that PET imaging can add useful information to CT or MRI alone, improving the diagnostic workup in patients with cardiac masses both in the preoperative and post-treatment setting, enabling definition of surgical indication and early identification of local or distant recurrences that are key elements in the overall treatment strategy.
In brief, by reviewing available literature and based on our reported experience, PET/CT, unlike other instrumental investigations, through the functional study of the heart, allows: (a) obtaining information about the malignant potential of doubtful masses, based on SUVmax evaluation; (b) estimation of disease stage, also revealing potential myocardial involvement and pericardial spread; (c) evaluation of potential postoperative residual disease (in order to avoid false positives, the examination should be performed after an appropriate time period of 3/4 weeks from surgery); (d) performing early evaluation of response to chemotherapy, especially in light of the recent introduction of target therapies for some types of these tumors.
Of course, for patients that are going to be scanned with PET/CT with FDG to assess the myocardium, a specific and accurate preparation (high-fat/low-carbohydrate diet plus prolonged fasting) is mandatory. Future studies are eagerly awaited to better define and validate this role on larger series.
