Abstract
Background:
Many patients with solid tumours or nonmyeloid haematopoietic tumours develop symptomatic anaemia, which has a major impact on quality of life (QoL). The efficacy of erythropoiesis-stimulating agents (ESAs) in improving QoL and reducing blood transfusions has been widely demonstrated. Binocrit® (biosimilar epoetin alfa) is an ESA indicated in the European Union for treating chemotherapy-induced anaemia. The aim of this study was to investigate the effect of Binocrit® on haemoglobin (Hb) levels in anaemic cancer patients in Italian clinical practice.
Methods:
The ANEMONE study was a national, longitudinal, retrospective, multicentre observational study. Patients had to be 18 years or older, with a solid tumour or non-Hodgkin’s lymphoma, Hodgkin’s disease or multiple myeloma, receiving chemotherapy, and treated with Binocrit® to manage chemotherapy-induced anaemia. The primary outcomes were the proportion of patients with a Hb increase ⩾1 g/dl during the first 4 weeks and with a Hb increase ⩾2 g/dl during the first 12 weeks.
Results:
A total of 245 patients were enrolled and 215 patients were evaluable for statistical analysis. In the first 4 weeks, 49.3% of patients showed an increase in Hb of ⩾1 g/dl: 45.5% in patients with solid tumours and 52.1% in patients with haematological malignancies. In the first 12 weeks, 51.6% of patients showed an increase in Hb of ⩾2 g/dl (48.4% solid tumours, 54.2% haematological diseases). Treatment with Binocrit® was well tolerated.
Conclusions:
These results confirm the effectiveness and safety of Binocrit® for chemotherapy-induced anaemia in routine practice in patients with solid tumours, lymphoma and myeloma.
Keywords
Introduction
About 50% of patients with solid tumours or nonmyeloid haematopoietic tumours develop symptomatic anaemia during the course of their disease [Bokemeyer et al. 2004]. In cancer patients, anaemia may have multiple causes; it can result from cancer itself, concomitant diseases, older age, inflammation, malnutrition, low iron levels and treatment with chemotherapy [Skillings et al. 1995]. About two-thirds of cancer patients undergoing chemotherapy developed anaemia during treatment in the European Cancer Anaemia Survey (ECAS) [Ludwig et al. 2004].
Anaemia has a substantial impact on patients’ quality of life (QoL), and is largely responsible for the fatigue reported by many cancer patients. An increase in haemoglobin (Hb) level can produce a meaningful improvement in fatigue, with related improvement in physical, functional, emotional and overall well-being [Cella et al. 2004]. In cases of severe anaemia, red blood cell (RBC) transfusions may be needed [Ludwig et al. 2004]. These are effective, but inconvenient, in short supply and not without associated risks and cost [Abraham and Sun, 2012]. Furthermore, low levels of Hb before or during chemotherapy cycles may require dose reductions or delays in administration, resulting in a decrease in the overall treatment intensity.
Anaemia is defined as a Hb level <10 or 11 g/dl, with severe anaemia defined as a Hb level <8 g/dl [Bokemeyer et al. 2007]. Although considerable progress has been made in preventing or alleviating many of the common toxicities associated with cancer and its therapy, anaemia continues to be relatively undertreated [Loney et al. 2000]. The efficacy of erythropoiesis-stimulating agents (ESAs) in correcting anaemia in cancer patients has been widely demonstrated [Littlewood et al. 2003; Bohlius et al. 2006]. These substances improve QoL and reduce the need for transfusion. The administration of ESAs in cancer patients has been reported to be associated with an increased risk of thromboembolic events, reduced survival and reduced time to disease progression [Bennett et al. 2008]. However, in many studies ESAs had been used with target Hb levels significantly higher than those currently recommended by guidelines. According to current guidelines, ESAs should be used only in patients with chemotherapy-induced anemia, and treatment should be discontinued once the chemotherapy course is completed, and these agents should not be used when the aim of treatment is cure.
Recently, the UK National Institute for Health and Care Excellence (NICE) found ESA usage in cancer-induced anaemia to be cost-effective, largely due to the availability of lower cost biosimilar ESAs. Treatment guidelines and recommendations have been published regarding the safe use of ESAs [Bokemeyer et al. 2007; Rizzo et al. 2002]. Although the evidence for specifying the optimal strategy of monitoring iron and instituting iron repletion is not optimal, the majority of these guidelines also suggest the importance of using intravenous (IV) iron in combination with ESAs to ensure patients are iron replete [Bokemeyer et al. 2007; Rizzo et al. 2002].
The Anaemia Cancer Treatment (ACT) study assessed ESA treatment response rates in anaemic patients (Hb ⩽ 11 g/dl). It demonstrated that 65% of patients showed an increase in Hb of ⩾1 g/dl over 8–10 weeks after initiation, while a rise of Hb ⩾2 g/dl was observed in 34% of patients [Ludwig et al. 2009]. This study confirmed the importance of anaemia in real-life clinical practice and suggested that that the treatment response rates were lower than reported in randomized controlled trials. Observational studies are therefore important to assess the real-world effectiveness of these agents in the context of a rapidly evolving disease management framework.
Binocrit® (biosimilar epoetin alfa) is an ESA indicated for the treatment of anaemia as a result of chronic kidney disease or chemotherapy-induced anaemia [Binocrit® Summary of Product Characteristics (available at http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000725/WC500053680.pdf)]. However, data on the use of Binocrit® for the treatment of chemotherapy-induced anaemia in clinical practice are limited [Weigang-Köhler et al. 2009; Kerkhofs et al. 2012; Rodriguez Garzotto et al. 2014].
The aim of this retrospective study was to investigate the effect of Binocrit® on Hb levels in anaemic cancer patients in Italian clinical practice. Secondary aims were to describe: (i) the characteristics of the treated population; (ii) all adverse events (AEs) over the 12-week observational period; (iii) factors related to improvement of anaemia; and (iv) key factors considered by clinicians in the management of anaemia.
Materials and methods
Study design
The ANEMONE (Anaemia management in oNcohaEMatological patients treated with binOcrit: an italiaN retrospective obsErvational study) study was designed as a national, longitudinal, retrospective, multicentre observational study. It involved 23 Italian oncology or haematology centres. The study was approved by the Institutional Review Boards of all participating centres. Patients included in the study were those who started treatment with Binocrit® according to normal clinical practice before site activation, in order to manage chemotherapy-induced anaemia.
For each patient included, the decision to prescribe treatment with Binocrit® was taken by their doctor regardless of enrolment in the ANEMONE study. Data were retrospectively collected from clinical medical records over a 12-week observational period (± 1 week) after starting Binocrit® administration. Alive and reachable patients were consecutively enrolled at their normal scheduled visits, during which they signed the informed consent to the study and to retrospective data collection from existing clinical medical records. During the second half of the enrolment period, deceased/unreachable patients were also consecutively included in descending order of Binocrit® start date according to the list of all potentially eligible patients generated by each site at the time of activation. This study was retrospective and observational, and did not affect the relationship between clinicians and patients or standard patient follow-up patterns. Clinicians in the study remained free to decide on the treatment of their patients. The enrolment period lasted 10 months. Data were collected using an electronic data collection sheet, which automatically checked for possible data entry errors and inconsistencies. Site monitoring visits were performed at six participating centres, where all data were validated with source data.
Eligibility criteria
To be eligible for inclusion, patients (both males and females) had to be 18 years or older at initiation of Binocrit®, presenting with a solid tumour, non-Hodgkin’s lymphoma, Hodgkin’s disease or multiple myeloma. They were to be receiving chemotherapy (alone or in combination with other treatments). Treatment to manage chemotherapy-induced anaemia with Binocrit® had to have been started before site activation and data in medical records had to be available for the period from the start of treatment with Binocrit® for the following 12 weeks.
Patients were not eligible if they received treatments for their cancer or anaemia in the context of a clinical trial during the retrospective observational period, if they were in treatment only with radiotherapy or molecular targeted therapy, if they presented with acute leukaemia or a chronic myeloproliferative syndrome, if they presented with uncorrected haemolytic anaemia or anaemia due to a deficiency not related to cancer (e.g. deficiency of iron, vitamin B12 or folic acid), or if they received any other treatment for anaemia (e.g. other ESAs or transfusion) in the 28 days preceding the start of treatment with Binocrit®.
Study aims and sample size
The primary aim of the study was to describe the trend of Hb levels in a 12-week observational period in patients receiving chemotherapy for a solid tumour, lymphoma or myeloma and treated with Binocrit®.
In detail, the two primary outcomes considered for the evaluation of the primary objective were:
the proportion of patients who, during the first 4 weeks after the start of treatment with Binocrit®, showed an increase in Hb of ⩾1 g/dl;
the proportion of patients who, during the first 12 weeks after the start of treatment with Binocrit®, showed an increase in Hb of ⩾2 g/dl.
Secondary objectives of the study were to describe the treated population, to assess AEs occurring during the 12-week observational period, the evaluation of demographic and clinical factors potentially related to improvement of anaemia, and the description of the key factors considered by clinicians in the management of anaemia.
According to feasibility considerations based on clinicians’ opinions, a total of 300 patients were expected. Sample size was not based on the power to investigate a formal statistical hypothesis but the achievable estimate precision was evaluated based on literature data. In the ACT study [Ludwig et al. 2009], the proportion of patients who showed an increased level of Hb ⩾ 1 g/dl was 65.0%, with a median time to response of 4.7 weeks, while the proportion of patients who showed an increased level of Hb ⩾ 2 g/dl was 33.7%, with a median time to response of 6 weeks. Starting from these results, we assumed that the two proportions of interest at 4 and 12 weeks would be 32.5% and 33.7%, respectively. Thus, considering 32.5% as the expected value of the primary outcome, with a sample size of 300 patients (corresponding to 270 evaluable patients, assuming 10% of patients as not evaluable due to screening failures, missing data, etc.), the 95% confidence interval (CI) for the expected proportion would be 32.5% ± 5.6%. A smaller sample size would mean lower estimated precision, but the precision was acceptable (relative error ⩽ 20%), even for 200 evaluable patients.
Statistical analysis
Quantitative variables were described in terms of mean, standard deviation, median, 25th and 75th percentile [interquartile range (IQR)], minimum and maximum. Qualitative variables were described in terms of absolute and relative frequency, with bilateral 95% CI (where relevant).
For both primary outcomes, the last available value in the month preceding the first administration of Binocrit® was considered as the Hb level at the start of treatment.
The first outcome was calculated as the number of patients who, during the first 4 (+1) weeks after the start of treatment, showed at least one Hb level which exceeded the level at the start of treatment by ⩾1 g/dl in absence of blood transfusion or other ESA treatment during the 28 days preceding that value, divided by the number of evaluable patients with at least one Hb value in that period. If an increased level of Hb ⩾ 1 g/dl in the first 4 weeks occurred when the patient had undergone blood transfusion or other ESA treatment within 28 days before that increased value, the patient was still considered in the analysis but as a patient who did not show that increase.
The second outcome was calculated as the number of patients who, during the first 12 (+1) weeks after the start of treatment, showed at least one Hb level which exceeded the level at the start of treatment by at least 2 g/dl, with the same caveats as above. Moreover, the mean and standard deviation of Hb level at each time point was provided and tested by repeated-measure analysis of variance (ANOVA).
All AEs (serious related or unrelated, non-serious related or unrelated) arising during the 12-week observational period were described. Incidence of AEs was calculated for all events overall and by event type.
The association between demographic and clinical factors and the improvement of anaemia was studied by means of univariate logistic regression models. The dependent variable of these models was anaemia improvement (present versus absent), while the independent variables were each of the factors of interest (age, gender, Karnofsky performance status, baseline Hb level, tumour type).
For the description of key factors considered by clinicians in the management of anaemia, clinicians were asked to report the key criteria they take into account in management of anaemia and in initiating treatment with Binocrit® (for example maintenance of QoL, reduction of transfusions, expected improvement of Hb level and patient request). These were described in terms of frequency.
All the analyses were performed using SAS for Windows, release 9.2 (SAS Institute Inc., US).
Results
From June 2013 to March 2014, 245 patients were enrolled in the study. Of these, 30 were excluded due to violations of the eligibility criteria or unavailability of at least one Hb value during the Binocrit® monitoring period. Thus, 215 patients were evaluable for statistical analysis.
The main demographic and clinical characteristics of the patients are shown in Table 1. Median age was 68 years in females, and 71.5 years in males. Half of patients presented with at least one relevant comorbidity. Overall, 95 (44.2%) patients presented with a primary solid tumour, while 120 (55.8%) had lymphoma or myeloma. Among patients with primary solid tumours, the most frequent sites were lung, prostate and breast, and 26.3% of patients had received prior radiotherapy while 44.2% had received previous chemotherapy.
Baseline characteristics of evaluable patients.

Relevant comorbidities with frequency >5% are reported. Percentages are computed out of the number of evaluable patients (N = 215). For primary solid tumour site and type of haematological malignancy, percentages are computed out of the number of evaluable patients with solid tumour (N = 95) and with haematological malignancy (N = 120), respectively.
Hb, haemoglobin; IQR, interquartile range.
Among patients with haematological malignancies, non-Hodgkin’s lymphoma was most common. A total of 5% of haematological patients had received previous radiotherapy and 30.8% of patients had received previous chemotherapy.
Among all patients, the median Hb level at Binocrit® treatment initiation was 9.6 g/dl (IQR 8.9, 10.1 g/dl). During the 12-week observation period, 42 patients (19.5%) received iron supplementation: 21 (9.8%) received IV iron, and 27 (12.6%) received oral supplementation. Figure 1 shows iron administration at each time point.

Iron administration during the observation period.
Treatment with Binocrit®
Mean [± standard deviation (SD)] starting dose of Binocrit® was 35,673 ± 8449 IU/injection, and almost all patients (95.2%) started treatment with one injection per week (N = 104 patients with data available for the three time points). Almost half of patients (103, 47.9%) discontinued Binocrit® within 12 weeks; in 14.6% of these cases treatment was discontinued because chemotherapy was discontinued and in 75.7% because targeted values were attained (i.e. anaemia improvement was reached). In particular mean (SD) Hb values at treatment start were 9.34 (1.02) versus 9.83 (0.94) in patients who did not versus did discontinue because anaemia was improved (Student’s t-test p value = 0.0187).
Binocrit® was administered to half of the patients for at least 10 weeks during the observation period (range 1–13 weeks). No AEs leading to adjustment of dosage of Binocrit® or temporary suspension of treatment with the drug were reported. Initial Binocrit® dosage was increased during treatment in two patients, who continued the increased dosage until the end of the observation period.
Primary outcomes
Of 205 patients with at least one Hb value in the first 4 (+1) weeks after Binocrit® start (88 with solid tumours and 117 with haematological malignancies), 101 (49.3%) showed a ⩾1 g/dl increase in Hb in the first 4 weeks of treatment: 45.5% in patients with solid tumours, 52.1% in patients with haematological malignancies (Figure 2). Considering all evaluable patients (N = 215) in the first 12 weeks after treatment initiation, 72.6% of patients showed a ⩾1 g/dl increase in Hb (69.5% in patients with solid tumours, 75.0% in patients with lymphoma/myeloma) and 51.6% of patients showed a ⩾2 g/dl increase in Hb level (48.4% in patients with solid tumours, 54.2% in patients with haematological malignancies).
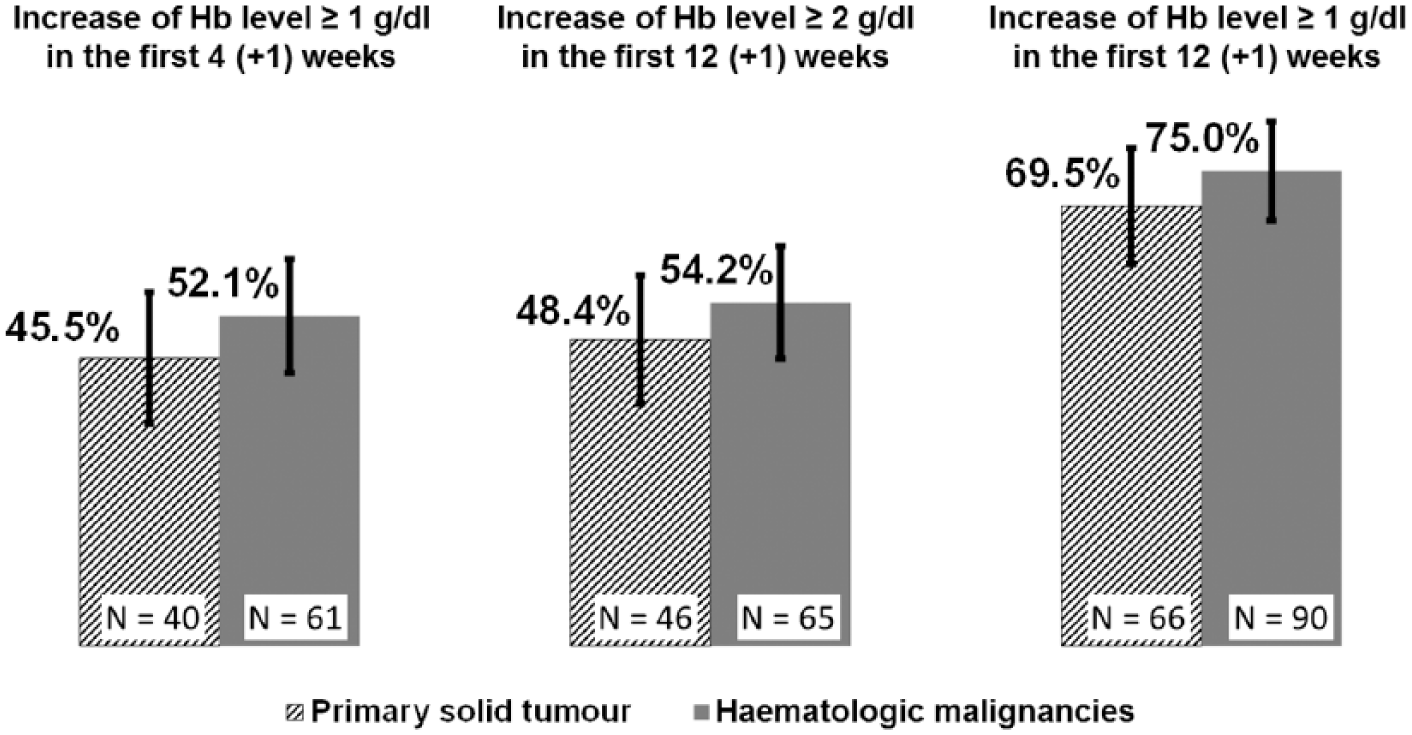
Increase of haemoglobin level during the observation period.
Considering the first 12 weeks after treatment initiation and only patients who did not discontinue Binocrit® (N = 112), 66.1% showed a ⩾1 g/dl increase in Hb level (62.5% in patients with solid tumours, 68.8% in patients with haematological malignancies), while 46.4% showed a ⩾2 g/dl increase in Hb level (39.6% in patients with solid tumours, 51.6% in patients with haematological malignancies). Considering only patients who did not discontinue Binocrit® or who discontinued it after the first 5 weeks (N = 150), 72.0% of patients showed a ⩾1 g/dl increase in Hb (66.7% in patients with solid tumours, 75.6% in patients with haematological malignancies), and 51.3% showed a ⩾2 g/dl increase in Hb (45.0% in patients with solid tumours, 55.6% in patients with haematological malignancies).
Among patients with available Hb values at each time point, a statistically significant increase in Hb levels in both patients with solid tumours and in patients with haematological malignancies was observed (Figure 3).

Haemoglobin level during the observation period.
In univariate logistic regression models, improvement in Hb level was not significantly associated with age, gender or Karnofsky performance status, tumour type (Table 2). The only variable that was significantly associated with improvement in Hb level was baseline Hb level: patients with higher baseline Hb level had a lower chance of obtaining an improvement (odds ratio 0.572, 95% CI 0.423–0.773).
Univariate logistic regression models for improvement of anaemia.

Note: The dependent variable was anaemia improvement (present versus absent), defined as either ⩾1 g/dl increased Hb level after 4 (+1) weeks from Binocrit® treatment start or ⩾2 g/dl increased Hb level after 12 (+1) weeks from Binocrit® treatment start.
CI, confidence interval; Hb, haemoglobin.
Among 21 patients who received IV iron administration, 16 (76.2%) experienced anaemia improvement; among the 194 patients who did not receive IV iron administration, 116 (59.8%) experienced anaemia improvement (not statistically significant). A total of 38 patients received RBC transfusions during the observation period: 23 (10.7%) by 4 weeks, and 25 (11.6%) by 12 weeks.
Adverse events
Overall, 29 (13.5%) patients experienced one or more AEs during the observation period, for a total of 63 AEs. The most commonly observed AEs were asthenia (nine events) and dyspnoea (six events), followed by oedema, diarrhoea, nausea and anorexia (three events each). Out of 63 events, 40 (63.5%) were mild, 15 (23.8%) were moderate and 8 (12.7%) were severe. Overall, four patients experienced one or more serious AEs (a total of six serious AEs): all these events were judged unrelated to Binocrit®. There were two patients that experienced adverse drug reactions: one superficial venous thrombosis and one deep venous thrombosis, both considered possibly related to Binocrit® administration. Antibody formation to biosimilar epoetin alpha was not specifically evaluated, in any case no patients developed pure red cell aplasia (PRCA) during this study.
Key factors taken into account by clinicians in the management of anaemia
Key considerations reported by clinicians in the management of anaemia are summarized in Table 3. The main reason taken into account by clinicians to treat chemotherapy-induced anaemia was maintenance of patients’ QoL (72.1%), followed by maintenance of chemotherapy dose (16.7%) and reduction of transfusions (6.5%). Key factors reported by clinicians in the decision to start Binocrit® included (i) the existence of hospital instructions (ESAS available in pharmacy) (38.1%) and (ii) maintenance of QoL (27.9%) and (iii) expected efficacy of the chosen treatment (15.8%).
Key factors in the management of anaemia as reported by clinicians.

Percentages are computed out of the number of evaluable patients (N = 215).
Discussion
In this retrospective, observational study, treatment with biosimilar epoetin alfa (Binocrit®) in a series of patients undergoing chemotherapy for solid tumours or lymphoma and myeloma was associated with a clinically relevant increase in Hb level (⩾1 g/dl) in the first 4 weeks after treatment initiation in nearly half (49%) of patients. This proportion was similar in the two groups: 45% in patients with primary solid tumours and 52% in patients with haematological disorders. Furthermore, in the first 12 weeks after treatment initiation, 73% of patients achieved a Hb increase of ⩾1 g/dl and 52% achieved an increase of ⩾2 g/dl. These proportions were also similar in patients with solid tumours and haematological malignancies.
These results are largely consistent with literature about the activity of ESAs. In the ACT study, an Hb increase of >1 g/dl was obtained in 65% of patients [Ludwig et al. 2009]. The ACT study was run before the 2008 ESA-class label change and United States Food and Drug Administration (FDA) black box warning, so one could postulate that treatment habits in Italy are likely to be more conservative during the period of our later study (i.e. less aggressive ESA treatment); this hypothesis requires further evaluation. Our secondary analysis of potential association between demographic and clinical variables and successful treatment in terms of Hb increase did not show a significant impact of sex, performance status and age. The only variable showing a statistically significant association with the chance of success in Hb increase was baseline Hb level, with a significantly greater chance of benefit from Binocrit® treatment in patients starting treatment with lower Hb values. This finding suggests that, even in a population of patients with a relatively low Hb value, with a high risk of QoL deterioration due to anaemia and a high risk of transfusion, treatment with Binocrit® is able to deliver a clinically meaningful benefit.
Our results show a relatively low use of iron supplementation in clinical practice, and this could have had a detrimental effect on the increase in Hb level obtained with Binocrit®. However, this is not the first report showing under-treatment of iron deficiency in patients with cancer and chemotherapy-induced anaemia. In a recent survey asking oncologists and haematologists from nine European countries about their last five patients treated for chemotherapy-induced anaemia, iron status was measured (by serum ferritin) in less than half (48%) of patients [Ludwig et al. 2014]. Despite the demonstration of iron deficiency in 42% of evaluated patients, iron supplementation was only used in 31% of the cases, with oral administration in the majority of cases. While there is little evidence that oral iron supplements increase response to ESAs, there is consistent evidence of a better response to ESAs when iron is administered IV in patients with absolute or functional iron deficiency [Aapro et al. 2008; Gafter-Gvili et al. 2014]. Optimal treatment of chemotherapy-induced anaemia should include an evaluation of iron status (transferrin saturation and ferritin) and optimal use of IV iron supplementation, when needed, along with ESAs, in order to obtain an effective increase in Hb levels and avoid blood transfusions.
In most countries, the sustainability of the health system has been seriously challenged in recent years. Although the cost of drugs is not the only factor associated with this problem, it is considered a leading driver of the phenomenon [Cornes, 2012]. A biosimilar drug is a successor to a biological medicine that has lost patent protection or exclusivity [Bennett et al. 2014]. Biosimilars are approved in highly-regulated markets around the world, following very stringently-defined regulatory pathways, after they have demonstrated similar safety, efficacy and quality to the reference product. Increase in the use of biosimilar drugs has been proposed as one way of controlling the escalating costs of healthcare. Using biosimilar ESAs for supportive cancer care could produce substantial cost savings, potentially increasing accessibility to primary anticancer treatment. A recent study simulated the budget impact of biosimilar ESAs in EU countries, and estimated the number of patients who could be treated with rituximab, bevacizumab or trastuzumab from the associated cost savings in a hypothetical panel of 100,000 patients [Abraham et al. 2014]. The simulation showed that, under fixed dosing, the savings from 100% conversion were €110,592,159, translating into an additional 9770 rituximab, 3912 bevacizumab, or 3713 trastuzumab treatments.
Another recent study evaluated the comparative cost efficiency of different ESAs in the management of chemotherapy-induced anaemia [Aapro et al. 2015]. The administration of biosimilar epoetin alfa was shown to be consistently cost-efficient with respect to treatment with originator epoetin alfa, epoetin beta and darbepoetin alfa (both once weekly and once every 3 weeks), under different dosing scenarios.
Along with the increasing adoption of biosimilar drugs in clinical practice, patient exposure to these products continues to increase. As of February 2014, the estimated exposure to Binocrit® was over 300,000 patient-years, with >5000 patients studied in clinical trials [Aapro, 2013]. No relevant differences in safety profiles between biosimilar and reference products have been demonstrated [Weigang-Köhler et al. 2009; Abraham and MacDonald, 2012]. From this point of view, our study confirmed the safety of Binocrit®, with a low incidence of AEs (13%) and only two adverse reactions considered related to Binocrit® administration.
A limitation of our study is that it was designed as a retrospective, observational analysis, which cannot produce the high level of evidence associated with randomized, controlled trials. On the other hand, its observational nature allowed confirmation of the effectiveness and safety of Binocrit® outside the controlled and selected conditions of randomized trials. Furthermore, the retrospective design ensured there was no interaction between study conduct and clinician’s behaviour. Thus, the ANEMONE study offers an overview of the use of Binocrit® to manage chemotherapy-induced anaemia in a considerable sample of patients from Italian clinical practice. Although participating sites did not represent a random sample of all Italian ones, an effort was made to represent the north, centre and south of Italy. The consecutive enrolment procedure and inclusion of deceased patients were performed to reduce the risk of selection bias.
In conclusion, the results of this retrospective, observational experience confirm the effectiveness and safety of biosimilar epoetin alfa (Binocrit®) in the treatment of anaemia in cancer patients undergoing chemotherapy in routine practice. Our results can be considered reassuring about the use of Binocrit® in this setting.
Footnotes
Acknowledgements
This study was sponsored by Sandoz S.p.A., Italy. Scientific and technical coordination, data management, and statistical support were provided by MediData.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Sandoz S.p.A., Italy.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
