Abstract
A network meta-analysis of the comparative effectiveness of neurokinin 1 (NK-1) inhibitors in the prophylaxis of highly emetogenic chemotherapy induced nausea and vomiting has been conducted. Eligible studies included randomized trials evaluating aprepitant, fosaprepitant, netupitant (NEPA), casopitant and rolapitant containing regimens in the setting of highly emetogenic chemotherapy. Primary outcomes of interest include complete response (CR) and rate of no significant nausea. After preclusion of ineligible studies, 19 studies were included in the final analysis. The majority of the regimens containing NK-1 inhibitors (including NEPA, aprepitant/palonosetron (palono)/dexamethasone (dexa), casopitant/granisetron (grani) or ondansetron (ondan)/dexa, aprepitant/ondan/dexa) are better than regimens not containing them (palono/dexa, ondan/dexa, grani/dexa) in terms of achieving a CR in the overall phase. Moreover, casopitant/grani or ondan/dexa and aprepitant/grani or ondan/dexa are better than rolapitant/ondan or grani/dexa in terms of CR achievement [odds ratio (OR) 1.62, 95% credible interval (CrI) 1.14–2.23, and OR 1.28, 95% CrI 1.01–1.59, respectively]. Taking into consideration the limitations of cross-trial comparisons, regimens containing neurokinin inhibitors are associated with higher CR rates than regimens not containing them. Moreover, casopitant and aprepitant regimens seem to be more effective than rolapitant regimens.
Introduction
Chemotherapy-induced nausea and vomiting (CINV) are still considered among the major hurdles of administering appropriate anticancer treatment in spite of all major advances in its understanding and its treatment [Aapro et al. 2015; Jordan et al. 2015].
According to the degree of emetogenicity, anticancer drugs can be classified into highly emetogenic, moderately emetogenic, low emetogenic and minimally emetogenic treatment [Roila et al. 2010]; however, some practical considerations face such classification including the very rapid rate of appearance and approval of newer anticancer agents that were not present at the time of the original 2004 Perugia consensus statement [Navari and Aapro, 2016]; another consideration deals with the very broad range of ‘the moderately emetogenic chemotherapy’ category that is from 30% to 90% risk of emesis which means that standardizing one mode of treatment for this group may result in overtreatment for regimens at the lower end of the range or undertreatment for regimens at the upper end of the range. A third issue is related to the fact that this classification was based mainly on the experience with intravenous chemotherapy agents; thus, with the increasing incorporation of oral anticancer agents in the management of patients with cancer, a different emetogenicity classification has to be applied for oral anticancer agents rather than just adopting the older classification [Kottschade et al. 2016].
The process of CINV itself may be classified temporally in a timetable fashion into early phase (between 0 and 24 h), delayed phase (between 24 and 120 h) and overall phase (between 0 and 120 h) [Van Den Brande et al. 2014; Zhou et al. 2015]. Such a temporal classification pattern has proved very beneficial in terms of evaluating and comparing different treatment options for CINV.
Following the rapid establishment of 5HT3 inhibitors as cornerstones of antiemetic treatment, the launching of neurokinin 1 (NK-1) inhibitors in the past decade has further boosted the strength of different antiemetic protocols employed [Hesketh, 2001]. Aprepitant was the first agent approved from this group, followed by fosaprepitant and then several other agents evaluated, including netupitant and rolapitant [Hesketh et al. 2003; Poli-Bigelli et al. 2003].
Objective of the meta-analysis
This network meta-analysis aims to provide a detailed comparative assessment of the efficacy of regimes containing one of the NK-1 inhibitors (aprepitant, fosaprepitant, rolapitant, casopitant, netupitant) in the prevention of CINV from highly emetogenic chemotherapy.
Methodology
Search strategy
A comprehensive search for literature published in English was performed in the following databases: Medline, Cochrane library and Google scholar in order to identify all relevant citations; the date of the last search was 5 September 2015. Meeting abstracts including ASCO (American Society of Clinical Oncology) were also checked. An additional hand search of references of primary original research was conducted for potential ‘cross references’. Citations with the following words in their titles or abstracts were examined: ‘rolapitant’ or ‘aprepitant’ or ‘casopitant’ or ‘fosaprepitant’ or ‘netupitant’, and ‘emesis’ or ‘vomiting’ or ‘nausea’. No protocol has previously been published for this meta-analysis.
Selection criteria
Inclusion criteria:
Clinical studies that evaluate antiemetic regimens based on any of the above agents in the prevention of CINV from highly emetogenic chemotherapy in adults.
Efficacy measures were reported.
Exclusion criteria:
Non-English language records were excluded.
Data extraction
Data were extracted by review authors. All eligible articles underwent initial assessment for relevance. The following data were extracted if available: authors, issuing year, treatment plan, number of patients, complete response (CR) defined as no emesis and no use of rescue drugs, rate of no significant nausea (nausea <25 mm on a visual analogue scale) and rate of no emesis.
Outcome measures
The outcome measures of interest were CR, rate of no significant nausea and rate of no emesis. The three outcome measures were principally evaluated in the overall phase (i.e. from 0 to 120 h after chemotherapy). The cardinal outcome determinants were outlined using descriptive statistics. This meta-analysis follows the guidelines provided by the PRISMA statement (Preferred Reporting Items for Systematic Reviews and Meta-Analyses report) [Moher et al. 2009]. Quality of the included studies was assessed through the use of Jadad score (Table 2) [Jadad et al. 1996].
Data analysis
The statistical analysis integrated both direct and indirect evidence to get estimates of the relative efficacy of each of the treatments evaluated across the different randomized controlled trials (RCTs). Data were assessed using a Bayesian meta-analyses approach [Lu and Ades, 2004; Caldwell et al. 2005], conducted using WinBUGS 1.4.3 software, and data entry was conducted using NetMETAXL software. Based on the distributions of relative treatment effects, the probability that a certain intervention was more efficacious than another was calculated based on ranking. Both fixed-effects and random-effects models were used. These models estimate odds ratio (ORs) for rates of both CR and rate of no significant nausea and their corresponding 95% credible intervals (CrIs) that capture the uncertainty. Random-effects analyses used vague priors for treatment effects. Sensitivity analyses were performed through reporting results for fixed-effects and random-effects models. A total of 1000 burn in runs and model runs were conducted. Simplified convergence testing was conducted from within the NetMETAXL software.
Ethical approval
This article does not contain any studies with human participants conducted by any of the authors.
Results
Search results
Figure 1 summarizes the PRISMA diagram for the study selection procedure; 699 results were obtained from the searches in Medline (n = 166 studies) and other databases (n = 533). Of these results, 165 were duplicates and 500 did not meet the eligibility criteria and were therefore excluded. Of the 34 possibly eligible studies after the initial screening, a full text search resulted in the removal of 15 studies. Hence, 19 studies were included in the final analysis; 12 phase III studies and 7 randomized phase II studies [Schnadig et al. 2014; De Wit et al. 2003; Hesketh et al. 2003; Poli-Bigelli et al. 2003; Schmoll et al. 2006; Herrington et al. 2008; Grunberg et al. 2009; Roila et al. 2009; Takahashi et al. 2010; Saito et al. 2013; Stiff et al. 2013; Wenzell et al. 2013; Gralla et al. 2014; Hu et al. 2014; Schmitt et al. 2014; Ando et al. 2015; Rapport et al. 2015a; Rapport et al. 2015b] (Table 1). Four studies evaluated rolapitant-based regimens, one study evaluated a netupitant/palonosetron (palono) (NEPA)-based regimen, three studies evaluated aprepitant/palono-based regimens, 10 studies evaluated aprepitant/ondansetron (ondan) or granisetron (grani)-based regimens (one of which is the NEPA randomized study), two studies evaluated casopitant-based regimens and two studies evaluated fosaprepitant-based regimens.
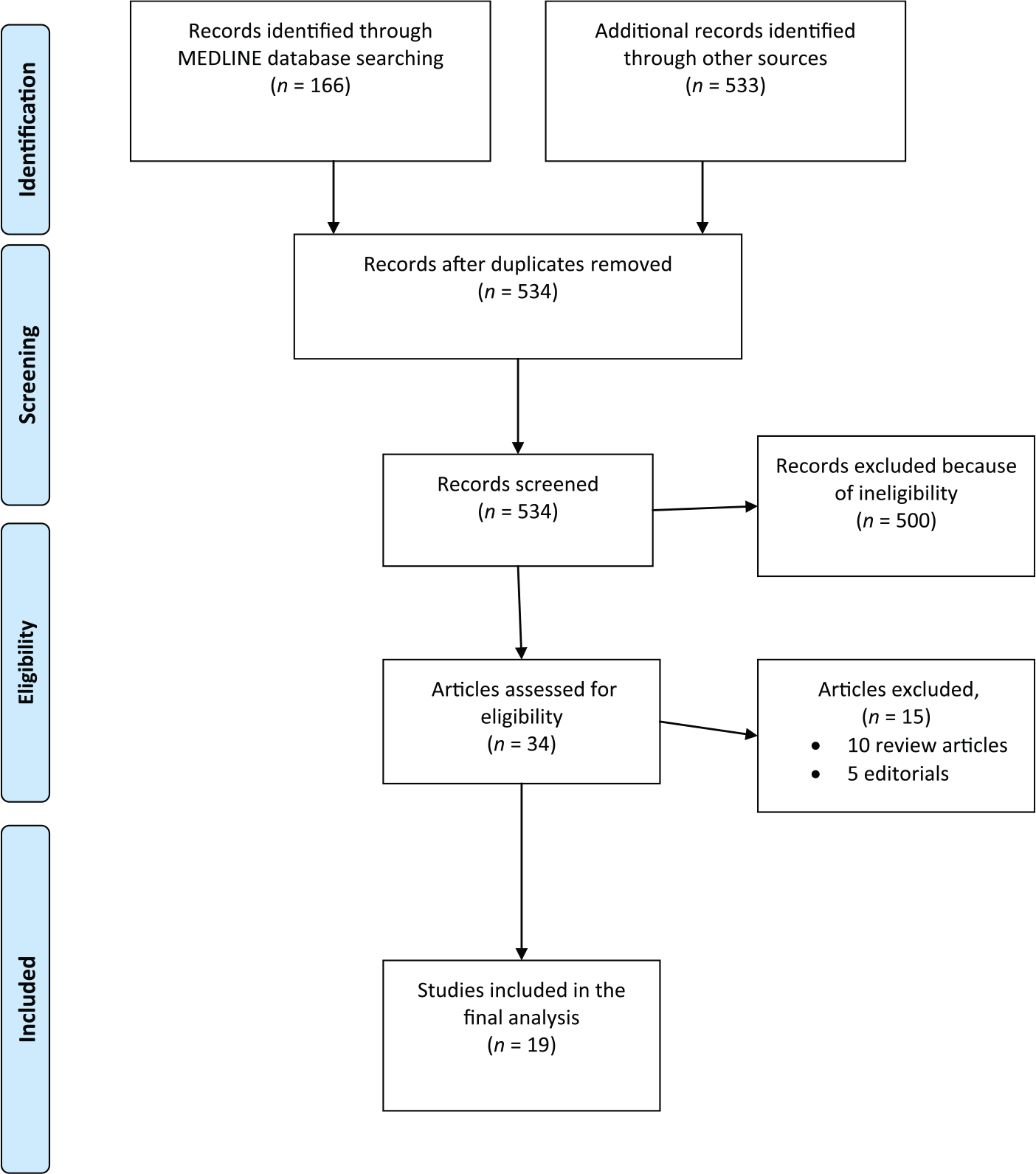
Flowchart of study selection procedure.
Efficacy outcomes in the included studies.

CR, complete response defined as no emesis and no use of rescue medication; NEPA, netupitant/palonosetron; N/R, not reported; RCT, randomized controlled trial; HEC: highly emetogenic chemotherapy.
Population characteristics
A total of 6788 patients were included in the analysis. All patients were enrolled in studies evaluating highly emetogenic chemotherapy and thus have adequate haematological, hepatic and renal functions. The baseline characteristics and the relevant outcomes in each trial are summarized in Table 1.
Quality of included studies
Table 2 summarizes the principal elements of the Jadad quality assessment for each of the included studies, including randomization, blinding and an account of all patients in addition to the overall score.
Jadad quality scale of the included studies.

Results of indirect comparison
The principal clinical outcomes evaluated in the indirect analysis were CR (overall phase) and no significant nausea (overall phase). An OR greater than one indicates improved outcome. A CrI around the point estimate is reported as a measure of uncertainty. A 95% CrI above 1.0 gives a 95% probability of improved outcome. All results were principally reported using a random-effects model with vague priors.
Indirect comparison for the overall phase of CR (from 0 to 120 h after chemotherapy)
These findings suggest that the majority of the regimens containing NK-1 inhibitors [including NEPA, aprepitant/palono/dexamethasone (dexa), casopitant/grani or ondan/dexa, aprepitant/ondan/dexa] are better than regimens not containing them (palono/dexa, ondan/dexa, grani/dexa) in terms of achieving a CR in the overall phase (Figure 2a–c).

(a) Forest plots of odds ratio (OR) of complete response (CR) associated with different neurokinin-based regimens. (b) League table of different neurokinin-based regimens in terms of CR achievement. (c) Rankogram of different neurokinin-based regimens in terms of CR achievement. CI, credible interval; dexa, dexamethasone; grani, granisetron; ondan, ondansetron; NEPA, netupitant/palonosetron; palono, palonosetron.
Other interdrug indirect comparisons among the neurokinin inhibitors themselves suggested that casopitant/grani or ondan/dexa and aprepitant/grani or ondan/dexa are better than rolapitant/ondan or grani/dexa in terms of CR achievement (OR 1.62, 95% CrI 1.14–2.23, and OR 1.28, 95% CrI 1.01–1.59, respectively).
None of the other interdrug indirect comparisons indicated statistically significant differences between the other neurokinin inhibitors and Table 2b (league table) provided a crude efficacy arrangement for neurokinin inhibitors in terms of ability to achieve CR rates.
Indirect comparison of the overall phase of rate of no significant nausea (from 0 to 120 h after chemotherapy)
None of the interdrug indirect comparisons indicated statistically significant differences between the evaluated regimens and the relevant league table provided a crude efficacy arrangement for neurokinin inhibitors in terms of their ability to achieve a higher rate of no significant nausea (Figure 3a–c).

(a) Forest plots of odds ratio (OR) of rate of no significant nausea associated with different neurokinin-based regimens. (b) League table of different neurokinin-based regimens in terms of rate of no significant nausea achievement. (c) Rankogram of different neurokinin-based regimens in terms of rate of no significant nausea achievement. CI, credible interval; dexa, dexamethasone; grani, granisetron; ondan, ondansetron; NEPA, netupitant/palonosetron; palono, palonosetron.
Discussion
To my knowledge, this is the most up to date meta-analysis to provide a comparative assessment of the efficacy of neurokinin inhibitor based regimens in the prophylaxis of highly emetogenic CINV. The indirect comparison from this analysis revealed that the majority of the regimens containing NK-1 inhibitors (including NEPA, aprepitant/palono/dexa, casopitant/grani or ondan/dexa, aprepitant/ondan/dexa) are better than regimens not containing them (palono/dexa, ondan/dexa, grani/dexa) in terms of achieving a CR in the overall phase. However, none of the other interdrug indirect comparisons revealed significant differences in terms of rates of no significant nausea.
Gastrointestinal toxicities (including nausea and vomiting) have been recorded for many anticancer therapies and they have always been a major cause of disturbed quality of life as well as lost treatment compliance [Schwartzberg, 2014].
The development of neurokinin inhibitors is considered to be the fruitful consequence of improved understanding of the biology of CINV and the role of different neuronal receptors in its occurrence. The two most important groups of receptors evaluated to date include 5HT3 receptors and NK-1 receptors [Rojas et al. 2014].
The approach to managing CINV has undergone revolutionary changes during the past two decades. Initially, the introduction of 5HT3 inhibitors (e.g. granisetron, dolasetron and ondansetron) in the 1990s changed the landscape of supportive care in patients receiving highly emetogenic chemotherapy. The first decade of the twenty-first century has come with another major breakthrough; that is, the introduction of aprepitant as the first representative of the group of NK-1 inhibitors [Hesketh et al. 2003; Poli-Bigelli et al. 2003; Schmoll et al. 2006]. Since then, a series of other NK-1 inhibitors have been introduced into clinical practice, including fosaprepitant (which is actually a water-soluble prodrug of aprepitant), casopitant (whose approval processes have been halted by the sponsoring company despite interesting phase III results), netupitant (which has been used in combination with palono and summarized as NEPA) and lastly rolapitant [Lasseter et al. 2007; Navari, 2007; Grunberg et al. 2009; Roila et al. 2009].
The above developments have been accompanied by another major achievement with the introduction of a second-generation 5HT3 inhibitor (palono) which has been shown to better older generation 5HT3 inhibitors (grani/ondan) in randomized controlled studies [Aapro et al. 2006; Saito et al. 2009]. The indirect comparison from this analysis has shown that NK-1 inhibitor based regimens not containing palono have a higher ability to achieve CR than the palono/dexa combination.
However, most of the randomized studies proving the superiority of NK-1 inhibitors have used a standard control arm comprising older generation 5HT3 inhibitors (grani or ondan) plus dexa. Thus although the superiority of these regimens against a grani or ondan/dexa combination was clear, the efficacy vis à vis palono/dexa or among the neurokinin inhibitors themselves was not clear for the majority of these antiemetic regimens. This has provided the principal rationale and motive to conduct this analysis in order to study the relative efficacy of these agents which can be investigated further by randomized controlled studies.
What is also interesting about these results is that despite the clear-cut superiority of NK-1 inhibitor regimens over older 5HT3 only regimens in controlling vomiting and achieving CR, the risk of significant nausea is not tackled to the same extent by these agents. This is an interesting area of future research and development in forthcoming antiemetic studies.
Other than the efficacy differences between NK-1 inhibitors discussed above, some other aspects need to be taken into consideration when choosing the appropriate NK-1 inhibitor to be used. For example, pharmacokinetic data have revealed interesting differences among these agents with regard to effect on the cytochrome P450 3A4 metabolizing pathway, with aprepitant and NEPA having a profound effect on this pathway while rolapitant does not [Poma et al. 2013]. This point has to be taken into consideration when prescribing a NK-1 inhibitor to a patient with cancer receiving other drugs potentially metabolized by this pathway. Other points of interest when evaluating these drugs is the minimal cardiac risk imposed by some of the older 5HT3 inhibitors and represented by prolonged QT interval in some of the published studies [George et al. 2010]. This is particularly relevant in older patients and those receiving concomitant medications that may further prolong this interval. This finding may be in favour of using palonosetron-based regimens because of the reduced risk of QT prolongation.
Among the other agents that have shown interesting antiemetic effects has been the atypical antipsychotic olanzapine which is equivalent to aprepitant in reducing emesis but is superior to aprepitant in reducing delayed nausea in a randomized phase III study [Navari et al. 2011].
The principal weakness of this meta-analysis is the presence of some degree of heterogeneity among included studies. Proper sensitivity analyses and meticulous review of all published data have been conducted to overcome this. Moreover, the network meta-analysis has some inherent limitations in performing indirect comparisons mainly due to selection biases.
Conclusion
This meta-analysis has demonstrated that compared with regimens not containing NK-1 inhibitors, the majority of the regimens containing NK-1 inhibitors are better in terms of achieving a CR in the overall phase. Bearing in mind the caveats of a cross-trial comparison, casopitant/grani or ondan/dexa seems to be a better combination than rolapitant/ondan or grani/dexa in terms of CR achievement. Moreover, none of the other inter-drug indirect comparisons revealed significant differences in terms of rates of no significant nausea.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
