Abstract
While initially effective, androgen deprivation therapy (ADT) is not curative, and nearly all men with advanced prostate cancer will eventually progress to the more resistant, and ultimately lethal form of the disease, so called castration-resistant prostate cancer (CRPC). The maintenance of androgens within the prostate cancer microenvironment likely represents one of the key mechanisms by which this transition from hormone-sensitive to CRPC occurs. This can be accomplished either through intratumoral androgen biosynthesis or the active transport of androgens and androgenic precursors into the tumor microenvironment. More recently, preclinical and clinical data supported therapeutic strategies that seek to target these two mechanisms, either through the use of drugs that impair androgen biosynthesis (e.g. inhibiting the steroidogenic enzymes CYP17 and AKR1C3 with abiraterone and indomethacin, respectively) or drugs that inhibit the SLCO transporters responsible for importing androgens (e.g. statins).
Keywords
Introduction
Even in its most resistant form, the vast majority of prostate cancers remain dependent on androgen receptor (AR) signaling [Schweizer and Yu, 2015]. Indeed, prostate cancer’s reliance on androgens has been recognized since the 1940s, and inhibiting AR signaling with androgen deprivation therapy (ADT) (i.e. surgical castration, luteinizing hormone-releasing hormone (LHRH) analogues) represent the first example of a ‘targeted’ cancer therapy [Huggins and Hodges, 2002]. Consequently, a number of adaptive changes occur whereby prostate cancer cells are able to survive in spite of low circulating androgen levels, many of which involve the AR [Robinson et al. 2015; Schweizer and Yu, 2015]. Tumor outgrowth while on ADT represents a key transition point from hormone-sensitive to a more aggressive, and ultimately lethal form of the disease: castration-resistant prostate cancer (CRPC).
While there are varied mechanisms by which AR signaling is able to persist in a castration-resistant state, the maintenance of intratumoral androgens at levels sufficient to drive tumor growth likely represent an important driver of the CRPC phenotype [Titus et al. 2005; Mostaghel et al. 2007]. Key sources of these persistent androgens are intratumoral steroidogenesis through overexpressing steroidogenic enzymes (e.g. AKR1C3, 3βHSD and CYP17), and the active transport of androgens into the tumor microenvironment [Montgomery et al. 2008; Cai et al. 2011; Sharifi, 2013; Liu et al. 2015]. Notably, the recognition that CRPC is largely driven by persistent AR signaling led to the development of the next generation AR-directed therapies abiraterone [a cytochrome P450 17 (CYP17) inhibitor] and enzalutamide (a potent AR antagonist), both capable of disrupting the A–ligand interaction (abiraterone through ligand depletion and enzalutamide through directly antagonizing the receptor) [De Bono et al. 2011; Scher et al. 2012; Ryan et al. 2013; Beer et al. 2014].
Abiraterone, a CYP17 inhibitor, was largely developed to target the production of persistent extragonadal (i.e. adrenal and intratumoral) androgens, and is United States Food and Drug Administration approved for men with metastatic CRPC on the basis of phase III data, indicating that it affords an overall survival benefit [De Bono et al. 2011; Ryan et al. 2013]. Perhaps not surprising, the androgenic precursor dehydroepiandrosterone sulfate (DHEAS) has been found to persist within the prostate following neoadjuvant treatment with abiraterone plus LHRH agonist therapy, which may in theory be sufficient to drive continued tumor growth [Tamae et al. 2015]. Steroid transporters have been implicated as mediators of persistent androgens within the tumor microenvironment, and would in theory not be affected by CYP17 inhibition. To that end, drugs that impair these transporters’ activity, such as statins, have begun to be explored [Harshman et al. 2015]. In this review, we will summarize the role of intratumoral androgens in driving CRPC cell growth, with a focus on the role of androgen biosynthesis and steroid transporters.
Intracrine androgen production
Data from CRPC studies from both localized prostate and rapid autopsy tissues have supported the hypothesis that intracrine androgen production may drive castration-resistant growth [Mohler et al. 2004; Montgomery et al. 2008]. Mohler and colleagues compared tissue androgen levels in biopsies from men with locally recurrent prostate cancer on ADT with androgen levels in benign prostate specimens from untreated men [Mohler et al. 2004]. They found that testosterone levels were comparable between these samples, while dihydrotestosterone (DHT) levels were lower in CRPC specimens. Given that persistent prostate-specific antigen (PSA) expression was observed in the CRPC specimens, it was concluded that intratumoral androgen levels were sufficient to activate the AR transcriptional program. Montgomery and colleagues have shown that in men who died with CRPC, androgen levels within metastatic deposits were significantly higher compared with normal (i.e. benign) control tissue obtained from the same patients [Montgomery et al. 2008]. In addition, when mRNA transcript levels of steroidogenic enzymes were compared between primary prostate tumors obtained from eugonadal men and metastatic tumors from men with CRPC, upregulation of several important enzymes (i.e. 3βHSD, CYP17A1, AKR1C3 and SRD5A2) were observed.
In vitro and in vivo models of CRPC have lent further support to the putative role intracrine hormone production plays in maintaining intratumoral androgens, and thus sustaining AR activation. AKR1C3, an enzyme able to convert the androgen precursor DHEAS into testosterone and DHT, has been shown to mediated steroidogenesis, leading to resistance to AR signaling inhibitors [Desmond et al. 2003; Cai et al. 2011; Liedtke et al. 2013; Liu et al. 2015]. Conversely, indomethacin, a nonsteroidal drug that has the off-target effect of inhibiting AKR1C3, has been shown to synergize with abiraterone and enzalutamide in resistant prostate cancer cell lines [Cai et al. 2011; Liu et al. 2015]. As such, clinical trials designed to more completely impair AR signaling by including an AKR1C3 inhibitor are currently under development.
Another key steroidogenic enzyme, 3βHSD, also plays an important role in catalyzing the conversion of androgen precursors (i.e. androstenediol) to more potent androgens (i.e. testosterone), with its enzymatic activity mediating the rate-limiting step that leads to the conversion of DHEA to DHT [Sharifi, 2013]. Interestingly, a 3βHSD gain-of-function mutation, which may be selected for in a castration-resistant state, has been shown to lead to increased DHT production [Chang et al. 2013]. Furthermore, increased 3βHSD activity can result in persistent intratumoral androgen production in the face of CYP17 inhibition, potentially driving abiraterone resistance. Whether targeting 3βHSD is a viable treatment strategy remains to be seen; however, efforts to effectively target its activity are likely forthcoming [Evaul et al. 2010]. It should be noted that preclinical data demonstrate that abiraterone is a weak 3βHSD inhibitor, and one strategy being pursued to target persistent intratumoral androgen production entails the administration of dose-escalated abiraterone [ClinicalTrials.gov identifier: NCT01503229] [Scher et al. 2012].
Speaking to the complexity of the metabolic processes at play within the tumor microenvironment, 3βHSD has also recently been shown to catalyze the conversion of abiraterone to Δ4 abiraterone (D4A), a compound able to potently inhibit the steroidogenic enzymes CYP17, 3βHSD and SRD5A, and to effectively antagonize the AR (at a level comparable to enzalutamide) [Li et al. 2015]. Given that 3βHSD has been implicated in resistance to abiraterone implies that the scales may be tipped towards 3βHSD catalyzing more DHT production relative to D4A; however, more research to understand the relative contribution of DHT and D4A in promoting resistance and response, respectively, are needed.
Androgen transporters
Organic anion-transporting polypeptides (OATPs), which are encoded by SLCO genes, are a family of transport proteins that are responsible for the influx of a number of substrates, including androgens and other steroids, bile acids, peptides and a number of pharmaceutical agents [Sissung et al. 2010; Pressler et al. 2011]. In total, there are 11 OATPs, which are classified into six families and subfamilies [Obaidat et al. 2012]. OATPs are ubiquitously expressed normal tissues, and have been found to be upregulated in several malignancies, including prostate cancer, and to associate with clinical outcomes in patients with prostate cancer [Obaidat et al. 2012].
The OATP1B3 (encoded by SLCO1B3) and OATP2B1 (encoded by SLCO2B1) transporters have both been shown to actively transport androgens and androgenic substrates. Not surprisingly, SLCO genes are more highly expressed by tumors from men with CRPC compared with primary specimens, and SLCO polymorphisms that associate with enhanced androgen uptake have been shown to associate with prostate cancer clinical outcomes [Hamada et al. 2008; Mostaghel et al. 2010; Wright et al. 2011; Yang et al. 2011]. These observations are both consistent with the hypothesis that OATP transporters may be able to maintain intratumoral androgen concentrations, thus sustaining AR signaling. For instance, in one study cos-7 cells were transfected with either wild type (WT) SLCO1B3 (334T/699G) or various combinations of the allelic variants 334T>G and 699G>A, which were identified through genotyping NCI-60 cell lines. Testosterone influx was then determined for the T/G, G/G, T/A and G/A variants, with diminished androgen transport activity observed only in cells expressing the SLCO1B3 G/A variant [Hamada et al. 2008]. To assess for the clinical importance of this finding, genotyping was performed on germline DNA from 180 patients with CRPC enrolled onto several prospective clinical trials. Those possessing the SLCO1B3 GG/AA haplotype were shown in this post hoc analysis to have improved survival compared with those with the TT/AA or TG/GA haplotypes (10-year survival: 42% versus 23%, p < 0.023), consistent with preclinical experiments.
A larger study assessed for an association between 18 SLCO single nucleotide polymorphisms (SNPs) and clinical outcomes [Yang et al. 2011]. From a cohort of 538 patients treated with ADT, three SNPs in the SLCO2B1 gene (rs12422149G>A, rs1789693A>T and rs1077858A>G) were found to associate with time to PSA progression in univariate and multivariate analyses. Of these SNPs, only SLCO2B1 rs12422149G>A (Arg312Gln) is exonic. To explore the biologic function of this SNP, LNCaP cells were transfected with either SLCO2B1-312Gln or SLCO2B1-312Arg, and their respective DHEAS uptake efficiency was characterized. While both SLCO2B1 variants were able to transport DHEAS, the SLCO2B1-312Arg variant was able to do so more efficiently. Because DHEAS is a precursor of the potent androgens DHT and testosterone, its influx into prostate cancer cells may be an important mechanism by which intracrine androgen production is mediated, thus providing a plausible biologic explanation for the association observed between this SNP and diminished time to PSA progression [Tamae et al. 2015].
Statins and androgen transporter activity
As mentioned above, the OATP transporters have a number of substrates in addition to androgens. Notably, statins are also substrates of the SLCO2B1 transporter, an observation that provided the motivation for evaluating if there was an association between statin use and time to PSA progression with frontline ADT [Harshman et al. 2015]. Harshman and colleagues evaluated the effect of a number of statins (i.e. atorvastatin, fluvastatin, pravastatin, and simvastatin) on DHEAS uptake in LNCaP and 22RV1 cells, both androgen responsive and expressing the SLCO2B1 transporter. They found that pravastatin more effectively inhibited DHEAS transport in both cell lines in vitro; although all of the statins tested were able to inhibit DHEAS influx to some extent. Atorvastatin, the most commonly prescribed statin, was also shown to inhibit DHEAS-induced prostate cancer cell growth in vitro. Importantly, a separate group was able to show that oral simvastatin administration was associated with decreased castration-resistant progression and lower intratumoral androgen levels in LNCaP xenografts compared with controls [Gordon et al. 2016].
To evaluate for a potential clinical effect of statin use on prostate cancer growth, Harshman and colleagues next performed a retrospective analysis to assess for an association between statin use and time to PSA progression. Using an institutionally maintained database of patients with prostate cancer, a cohort of 926 men receiving ADT was assembled. They found that the median time to PSA progression for those taking a statin was 27.5 months versus 17.4 for those not receiving one (p < 0.001). Importantly, this association persisted after multivariate analysis adjusted for predefined prognostic factors.
While there is a strong suggestion that the clinical correlation between statin use and delayed time to PSA progression is a result of interfering with SLCO2B1-mediated DHEAS uptake, the possibility remains that this effect could be due to lowering cholesterol levels, a substrate for all steroid hormones, through inhibiting 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase activity [Ramos and Yu, 2015]. A retrospective analysis by Allott and colleagues specifically evaluated the risk of biochemical recurrence (i.e. PSA relapse) following prostatectomy in 843 patients who had never received statins prior to their surgery. They found that elevated triglycerides were associated with increased risk of recurrence for the entire cohort, and that an elevated total cholesterol level was associated with an increased risk for those with hyperlipidemia [Allott et al. 2014]. While this finding is consistent with statins exerting a beneficial antitumor effect through lowering lipids, it does not specifically address whether or not statins modulate SLCO transporter activity. Ultimately, a prospective randomized study will be needed to evaluate the ability of statins to impair intratumoral androgen transporters and to determine if they are a useful adjunctive prostate cancer treatment.
Statin use and cancer risk
Because statins are generally well tolerated and have documented health benefits, there is considerable motivation to explore their use as repurposed chemopreventive agents. In addition to lowering the risk of major vascular events, epidemiologic data indicate that they may be effective at preventing a number of malignancies [Poynter et al. 2005; Mihaylova et al. 2012; Nielsen et al. 2012]. For instance, a population-based observational study out of Denmark evaluated the effect of statin use on a subsequent cancer diagnosis. Upon multivariate analysis, they found that statin use was associated with a 15% risk reduction in all-cause and cancer-specific mortality, including prostate cancer [Nielsen et al. 2012]. Similarly, a prospective population-based cohort study (n = 1001) evaluating the effect of statin use on prostate cancer outcomes found that statin use at the time of diagnosis was associated with decreased prostate cancer specific mortality [Geybels et al. 2013]. In contrast, a meta-analysis from 27 large randomized statin trials did not find that statin use was associated with a decreased cancer risk, calling into question the result of several observation studies, and raising the possibility that the chemoprotective benefits of statins may be merely a reflection of the inherent bias associated with retrospective studies [Emberson et al. 2012].
Several epidemiologic and retrospective studies have been completed to specifically evaluate for an association between statin use and the development of prostate cancer [Flick et al. 2007; Boudreau et al. 2008; Chang et al. 2011; Allott et al. 2014; Jespersen et al. 2014; Chan et al. 2015; Cuaron et al. 2015; Kantor et al. 2015; Luo et al. 2015; Sun et al. 2015; Zapata et al. 2015]. In general, these studies have supported the notion that statins may function as a prostate cancer chemoprotective agent; however, some data conflict with this assertion.
To further explore for a potential association between statin use and the development of prostate cancer, two large meta-analyses were conducted [Bonovas et al. 2008; Bansal et al. 2012]. Bansal and colleagues reported the results of a meta-analysis combining 27 observational studies that assessed the association between statin use and the risk of developing prostate cancer, and included 1,893,571 men in their final analysis [Bansal et al. 2012]. They found that statin use was associated with a reduction in total [relative risk (RR) = 0.93, 95% confidence interval (CI): 0.87–0.99, p = 0.03) and advanced prostate cancer (RR = 0.80, 95% CI: 0.70–0.90, p < 0.001) risk. In contrast, Bonovas and colleagues analyzed the data from six randomized controlled trials and 13 observation studies, which included over 880,000 male subjects [Bonovas et al. 2008]. This study did not find an association between statin use and total prostate cancer incidence (RR = 0.95, 95% CI: 0.73–1.23). The authors noted that there was high heterogeneity between the observational studies, and those studies that were specifically designed to examine statin use in relation to the risk of developing advanced prostate cancer found that statin use had a protective association (RR = 0.77, 95% CI: 0.64–0.93). These conflicting results highlight the need for randomized studies to evaluate the utility of statin as a chemoprotective agent.
Future directions
A number of strategies are being explored to target intracrine androgen synthesis or transporter-mediated androgen influx into the tumor microenvironment. Compelling preclinical data supported both of these strategies and a number of the drugs in our current ‘therapeutic toolbox’ may allow us to accomplish these goals, thus decreasing the long lead-in time required to launch most translational clinical trials. As mentioned above, agents warranting further clinical investigation include high-dose abiraterone, indomethacin and, as a class, the statins.
Indeed, a query of ClinicalTrials.gov demonstrates that a number of trials evaluating the therapeutic effect of statins on prostate cancer have either recently been activated or are planned to open in the near future (Table 1). It should be noted that the majority of studies are being developed in the presurgical or biochemically recurrent space. While there is certainly a rationale for developing statins as a therapy for hormone-sensitive prostate cancer, available evidence indicates that steroidogenesis may be key mechanism underlying the emergence of a castration-resistant phenotype [Mohler et al. 2004; Montgomery et al. 2008]. Additional studies evaluating the role of statins, ideally in combination with other inhibitors of intratumoral steroidogenesis (e.g. abiraterone or indomethacin), in treating CRPC are therefore justified.
Ongoing or planned trials evaluating statins in men with prostate cancer.
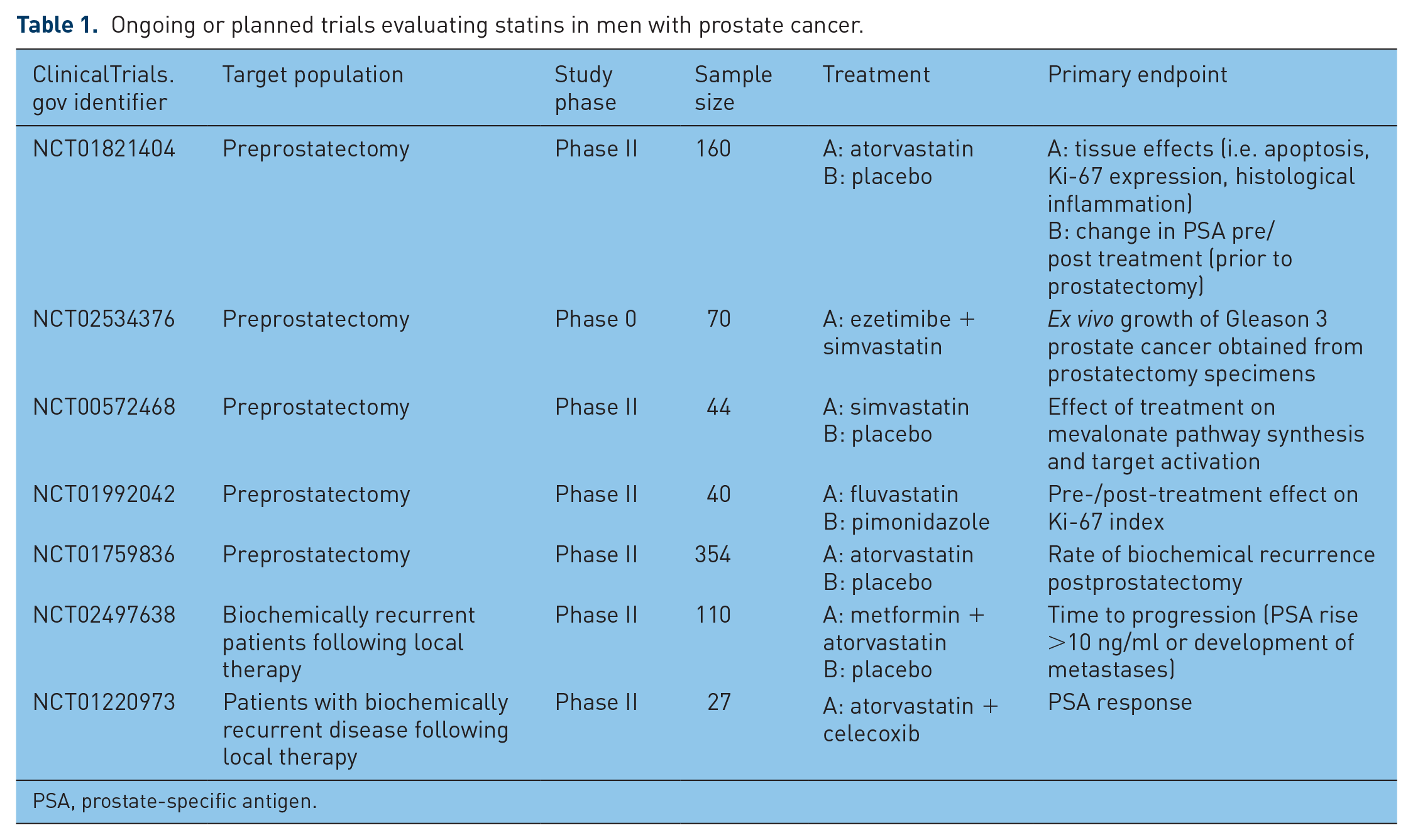
PSA, prostate-specific antigen.
Conclusion
While the availability of statins may tempt one to proceed with prescribing them off label in an effort to more effectively treat prostate cancer, caution should be taken. Even in the face of compelling preliminary data, randomized trials are still needed to establish new treatment standards. This point was highlighted by the SELECT trial: a large (n = 35,533), randomized chemoprevention trial testing vitamin E with or without selenium versus placebo [Lippman et al. 2009; Klein et al. 2011]. This study was launched on the basis of prior randomized trials that reported improved prostate cancer outcomes for those on vitamin E and selenium as a secondary outcome, presumably due to their antioxidant effects. Ultimately, this trial reported that vitamin E was associated with the unintended consequence of increasing the risk of developing prostate cancer. Given their long safety track record, it seems unlikely that a randomized prostate cancer trial testing statins would result in such an unexpected outcome; however, exposure to unnecessary medications should be limited whenever possible due to the potential for side effects (e.g. rhabdomyolysis, liver function test abnormalities).
Prostate cancer is a worthy foe, constantly adapting to maintain persistent AR signaling. Our understanding of the intracrine and other microenvironmental processes that can lead to persistent intratumoral androgens is still in its infancy; however, mounting evidence supports the assertion that these are viable targets by which prostate cancer growth can be further attacked. With drugs like statins readily available, clinical trials to formally assess the utility of impairing the mechanisms that sustain intratumoral androgens should proceed without delay.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
