Abstract
Background:
Treatment of patients with severe liver dysfunction including hyperbilirubinemia secondary to liver metastases of gastrointestinal (GI) cancer is challenging. Regimen of oxaliplatin and fluoropyrimidine (FP)/folinic acid (FA) ± a monoclonal antibody (moAb), represents a feasible option considering the pharmacokinetics. Clinical data on the respective dosage and tolerability are limited and no recommendations are available.
Methods:
Consecutive patients with severe hyperbilirubinemia [>2 × upper limit of the normal range (ULN) and >2.4 mg/dl] due to liver metastases of GI cancer without options for drainage receiving oxaliplatin, FP/FA ± moAb were analyzed. To collect further data a review of the literature was performed.
Results:
A total of 12 patients were identified between 2011 and 2015. At treatment start, median bilirubin level was 6.1 mg/dl (>5 × ULN, range 2.7–13.6). The majority of patients (n = 11) received dose-reduced regimen with oxaliplatin (60–76%) and FP/FA (0–77%), rapidly escalating to full dose regimen. During treatment, bilirubin levels dropped more than 50% within 8 weeks or normalized within 12 weeks in 6 patients (responders). Median overall survival was 5.75 months (range 1.0–16.0 months) but was significantly prolonged in responders compared to nonresponders [9.7 and 3.0 months, p = 0.026 (two-sided test); 95% confidence interval (CI): 1.10–10.22]. In addition, case reports or series comprising a further 26 patients could be identified. Based on the obtained data a treatment algorithm was developed.
Conclusion:
Treatment with oxaliplatin, FP/FA ± moAb is feasible and may derive relevant benefits in patients with severe liver dysfunction caused by GI cancer liver metastases without further options of drainage.
Introduction
Gastrointestinal (GI) cancer, particularly colorectal, gastric, esophageal and pancreatic malignancies, are among the most common cancer types worldwide [Ferlay et al. 2014]. Moreover, the high rate of metastatic disease accounts for the overall high mortality rates. Due to anatomical location and venous drainage the liver is the predominant organ for metastatic manifestation of tumors arising from the GI tract. Thus, impaired liver function and finally liver failure are common among GI cancer patients. On the other hand, the approved treatment combinations are based on clinical trials of selected patients with good liver function and normal values of bilirubin [<1.5 times upper limit of normal (ULN)] and transaminases.
Metastatic liver involvement may result in liver dysfunction, indicated by increased bilirubin (hyperbilirubinemia), increased level of cholestatic parameters (gamma-glutamyl transferase and alkaline phosphatase) and transaminases as well as impaired liver synthesis (e.g. low albumin). Pretreatment evaluation of liver dysfunction is important to ensure treatment tolerability and thus efficacy. Within the above-mentioned laboratory values, bilirubin is often used for a rough estimate of liver function and the consecutive treatment selection. Hyperbilirubinemia may have different reasons, in the case of severe liver involvement without options for biliary drainage, active and tolerable systemic treatment may reduce tumor burden and alleviate symptoms. By contrast, hyperbilirubinemia due to extrahepatic cholestasis may be managed by drainage and bilirubin will recover over time. Thus, tolerability will be the central concern of treatment administration.
Several systemic agents are available for GI cancer (e.g. irinotecan, fluoropyrimidine, taxanes and platin compounds), which differ highly in terms of pharmacokinetics, particularly regarding hepatic metabolism. For example, bioactivation of irinotecan and detoxification of its metabolite SN-38 occurs predominantly in the liver and thus drug clearance is diminished in patients with hepatic dysfunction [Rivory and Robert, 1995; Gagne et al. 2002]. The tolerability of irinotecan in patients with hepatic dysfunction (bilirubin greater than 2 mg/dl) has not been assessed sufficiently and no recommendations for dosing can be made. Fluoropyrimidines (FP) are eliminated primarily by hepatic metabolism, with less than 5% of the drug excreted in the urine in normal individuals [Schalhorn and Kuhl, 1992; Schilsky, 1998]. Capecitabine, a prodrug of 5-Fluororuracil (5-FU) is activated through three enzymatic reactions. Although high activities of drug metabolizing enzymes are expressed in human liver, the involvement of the liver in capecitabine metabolism is not fully understood and the use in patients with hepatic failure is controversially discussed [Twelves et al. 1999; Saif and Tejani, 2007]. Available reports showed no clinically significant influence on the pharmacokinetics of capecitabine and its metabolites by modestly impaired hepatic function [Twelves et al. 1999; Saif and Tejani, 2007]. Taxanes (docetaxel or paclitaxel) are predominantly eliminated by hepatobiliary extraction and are thus already contraindicated in case of modest liver dysfunction [Bruno et al. 2001; Hooker et al. 2008; Minami et al. 2009; Eckmann et al. 2014]. In contrast to irinotecan, fluoropyrimidines and taxanes, oxaliplatin is rapidly cleared from plasma by binding to tissue and renal elimination [Graham et al. 2000; Jerremalm et al. 2009]. Urinary excretion (53.8 ± 9.1%) is the major route of platinum elimination. In addition, oxaliplatin undergoes rapid and extensive nonenzymatic biotransformation [Graham et al. 2000].The favorable pharmacokinetic profile makes oxaliplatin a systemic agent of particular interest in patients with liver dysfunction.
Clinical data on the use of systemic chemotherapy in hepatic dysfunction are limited, particularly for current standard combination regimen including monoclonal antibodies (moAbs). Available data mainly confer to small case series on the use of oxaliplatin in combination with infusional 5-FU or capecitabine for either metastatic colorectal (mCRC) or gastric cancer or single agent cetuximab [Fakih, 2004; Hwang et al. 2006; Walia et al. 2008; Mizota et al. 2011; Elsoueidi et al. 2014; Tural et al. 2014].
In order to establish a dose recommendation for administration of current combination regimen including moAbs, bi-institutional case series and a literature review was performed.
Materials and methods
Consecutive patients with severe liver dysfunction and hyperbilirubinemia (>2 × ULN, >2.4 mg/dl) due to liver metastases of GI malignancies from two German institutions treated between August 2011 and April 2015 were analyzed for outcome, adverse events, liver function tests and blood count. Patients with hyperbilirubinemia were screened for options of internal or external biliary drainage by different imaging modalities (computed tomography, magnet resonance tomography or ultrasound) and discussed within the multidisciplinary tumor board. Only patients with hyperbilirubinemia caused by diffuse metastatic liver involvement after exclusion of further options for biliary drainage and no extrahepatic cholestasis were included in the analysis. Patients with known nonmalignant liver diseases like hepatitis, fibrosis or cirrhosis were excluded. The treatment had to contain any FP, folinic acid (FA) and oxaliplatin with or without moAbs.
Patients were stratified into two groups according to the respective decrease in bilirubin levels following treatment.
Responders were defined as those with a reduction of bilirubin levels of at least 50% within the first 4–8 weeks or normalization up to week 12. The remaining patients were defined as nonresponders.
For the literature review relevant data from published trials, reports and abstracts presented at selected oncology association meetings [American Society of Clinical Oncology (ASCO), European Society for Medical Oncology (ESMO), European CanCer Organisation (ECCO)] and Medline/Pubmed until July 2015 were reviewed.
Results
Detailed results of the literature review are shown in Table 1. Overall 11 case reports or case series about hyperbilirubinemia in patients (a total of 26 patients) with GI cancers (25 with mCRC) and one with gastric cancer were found.
Results of literature review.

5-FU, 5-Fluororuracil; nk, not known; red, reduced; LV, leucovorin; Ox, oxaliplatin; OS, overall survival; pts, patients.
Elevated bilirubin levels due to liver metastases ranged from 2.6 mg/dl to 29.8 mg/dl before treatment start. The majority of patients (n = 18) had not undergone any prior systemic treatment. After treatment start, bilirubin decreased in 16 of 25 patients (missing n = 1). Only one patient with mCRC received an irinotecan based chemotherapy regimen (FOLFIRI), in combination with bevacizumab as first line treatment [Yeh et al. 2014]. In this patient initial bilirubin was 5.94 mg/dl and dropped to 0.78 mg/dl after 4 cycles; overall survival (OS) was 9 months.
Shitara and colleagues reported on seven heavily pretreated patients with mCRC treated with cetuximab alone [Shitara et al. 2010]. In these patients median bilirubin at treatment start was 7.4 mg/dl. In five of seven patients bilirubin dropped less than 50% of the initial value. Treatment with cetuximab alone was feasible, however outcome was poor with a median OS of 2.4 months.
Eighteen patients were treated with oxaliplatin-based chemotherapy regimens (with any FP; with or without moAb; no moAb: n = 11, bevacizumab: n = 3, cetuximab: n = 1, panitumumab: n = 1) [Fakih, 2004; Hwang et al. 2006; Walia et al. 2008; Grenader et al. 2009; Mizota et al. 2011; Terasawa et al. 2013; Elsoueidi et al. 2014; Tural et al. 2014; Kasi et al. 2015] with bilirubin ranged from 3.2–29.8 mg/dl at treatment start, that dropped in 14 patients by more than 50%. OS ranged from 1.5 months to 18 months. All treatment regimens were feasible without any severe toxicity (grade ⩾3).
In our case series, 12 patients were identified (Table 2): 9 male and 3 female, with a median age of 59 (range 46–74) years. Tumor types were colorectal (n = 6), biliary tract (n = 5) and gastroesophageal cancer (n = 1). In addition to diffuse liver metastases, they also presented lung (n = 3), bone (n = 3) and lymph node (n = 6) metastases. Before the malignant liver involvement none of the patients had any preexisting liver diseases. In all patients, drainage by stent placement or percutaneous transhepatic biliary drainage was evaluated and if feasible done before systemic treatment with 5-FU/FA, oxaliplatin ± moAb. Thus, hyperbilirubinemia was solely caused by diffuse malignant liver infiltration with all other potential causes ruled out. Prior systemic chemotherapy, either in the adjuvant or metastatic setting, was administered in eight patients. In four patients 5-FU/FA, oxaliplatin ± moAb was the firstline treatment.
Bilirubin levels and individual dosing strategies of systemic treatment with oxaliplatin/ fluoropyrimidine, with or without moAbs.

not application because of death.
Bev, Bevacizumab; Bili, bilirubin in mg/dl (normal value <1.2 mg/dl); BL, baseline; Cape, capecitabine; Cet, Cetuximab; f, female; FP, 5-Fluororuracil; m, male; mets, metastases; OS, overall survival; Ox, oxaliplatin; Tras, Trastuzumab ; Tx, treatment.
Oxaliplatin was given as weekly FUFOX (oxaliplatin 50 mg/qm), every 2 weeks mFOLFOX6, FOLFOX7 or FLO (oxaliplatin 85 mg/qm) or FOLFOX6 (oxaliplatin 100 mg/qm); and every 3 weeks XELOX (oxaliplatin 130 mg/qm).
5-FU (5-Fluororuracil) was given as FUFOX (24 h infusion; 2.0 g/qm) weekly; mFOLFOX6 (48 h infusion; 2.4 g/qm) every 2 weeks; FOLFOX7 (48 h infusion; 3.0 g/qm) every 2 weeks or FLO (24 h infusion, 2.6 g/qm).
Capecitabine was given as CAPOX with 1000 mg/qm twice a day for 14 days.
Bevacizumab, every 2 weeks with 5 mg/kg.
Cetuximab, loading dose 400 mg/qm, weekly 250 mg/qm, every 2 weeks 500 mg/qm.
Trastuzumab, 4 mg/kg.
Liver dysfunction was documented prior to systemic treatment (but after maximal drainage) by a median bilirubin level of 6.1 mg/dl (range 2.7–13.6 mg/dl), median alkaline phosphatase (AP) level of 571.0 U/l (range 192–938 U/l), median gamma-glutamyl transpeptidase (gGT) 422.75 U/l (range 165–1446 U/l), median glutamic-pyruvic transaminase (GPT) level of 76.0 U/l (range 29–283 U/l) and median level of glutamic-oxaloacetic transaminase (GOT) 140.0 U/l (range 82–441 U/l). Liver synthesis function was relevantly impaired, shown by low levels of albumin (median level of 31 g/l, range 20–38) and elevated international normalized ratio (INR) of prothrombin time (median 1.3, range 0.92–1.84).
Oxaliplatin was administered with infusional 5-FU/FA once a week (n = 5) or every two weeks (n = 7) or with capecitabine every three weeks (n = 1). Chemotherapy was combined with bevacizumab (n = 4), trastuzumab (n = 1) or cetuximab (n = 1).
Upfront dose reductions were applied in five patients for oxaliplatin (60–76% of full dose) and in 10 patients for 5-FU (0–77% of full dose). In one patient, treatment was started with fully dosed chemotherapy.
Only in one patient, a dose escalation of oxaliplatin to full standard doses was done. However, the weekly FUFOX regimen was changed to every two weeks FOLFOX in two patients. Escalation of 5-FU/FA to full standard doses was done within median 3.8 weeks, respectively. Individual dosing strategies are displayed in Table 2.
Bilirubin levels dropped within 4–8 weeks or normalized within 12 weeks in six patients, (responders). The remaining six patients showed no bilirubin response to systemic treatment (nonresponders). Responders compared with nonresponders were younger (median age of 55.5 years versus 64 years) and had higher bilirubin levels at baseline (9.6 mg/dl versus 4.7 mg/dl).
Median overall survival after start of chemotherapy was overall 5.75 months (range 1.0–16.0 months) but was significantly longer in responders compared with nonresponders [median 9.7 and 3.0 months, p = 0.026 (two-sided test); 95% CI: 1.10–10.22]. Bilirubin level at treatment start was not correlated to response rate, side effects or survival. Comparing patients with and without the combination with moAb, median OS was 6.8 months and 6.5 months and thus not statistically different (p = 0.92).
Apart from severe nausea and emesis (grade 3) in one patient the treatment with 5-FU/FA, oxaliplatin with or without moAb was well tolerated, even after escalation to full dose. No correlation between the severity of liver dysfunction, hyperbilirubinemia and treatment tolerability was noted.
Discussion
The presented case series clearly demonstrate the feasibility of systemic treatment with oxaliplatin/fluoropyrimidine combinations, with or without moAbs. In regard to the low percentage of escalation to a full dose regimen, and the overall relatively poor outcome of these patients, a more aggressive dosing strategy and treatment intensity should be considered in eligible patients, as outcome may be correlated to adequate dosage. Similarly, recent guidelines (e.g. in mCRC) recommend an upfront rather intensive treatment approach in the case of symptomatic or rapid progressive disease (ESMO group 2), as commonly applies for patients with hyperbilirubinemia [Schmoll et al. 2012]. However, choosing an intensive approach with a quick dose escalation might not be feasible for all patients and harmful. Therefore, a cautious pretreatment evaluation is important. In case of an underlying not cancer-associated reduced ECOG performance status (PS) of two or more or a far advanced and already heavily pretreated disease, the suggested approach may not be reasonable.
Of note, patients without bilirubin response after 4–8 weeks do not seem to derive any benefit from systemic treatment and may thus receive early treatment discontinuation and referral to best supportive care. In contrast, patients responding to treatment as shown by an early decline in bilirubin levels may have a longer survival.
The addition of moAbs was feasible, which seems to be in line with the known pharmacokinetics in these drugs [Lu et al. 2008; Azzopardi et al. 2011; Zhi et al. 2011]. As recently shown, epidermal growth factor receptor (EGFR) antibodies seem to be of particular interest in terms of application as single agent or in combination in RAS wildtype mCRC with severe liver dysfunction [Shitara et al. 2009; Mizota et al. 2011; Kasi et al. 2015].
The current case series and the respective analyses are limited by the small number of patients, the retrospective character and the evaluation of patients from only two centers. Therefore, a review of literature was performed to collect further case series.
Based on the published data and our case series, we suggest the following treatment algorithm for patients with GI cancer and severe liver dysfunction (Figure 1).
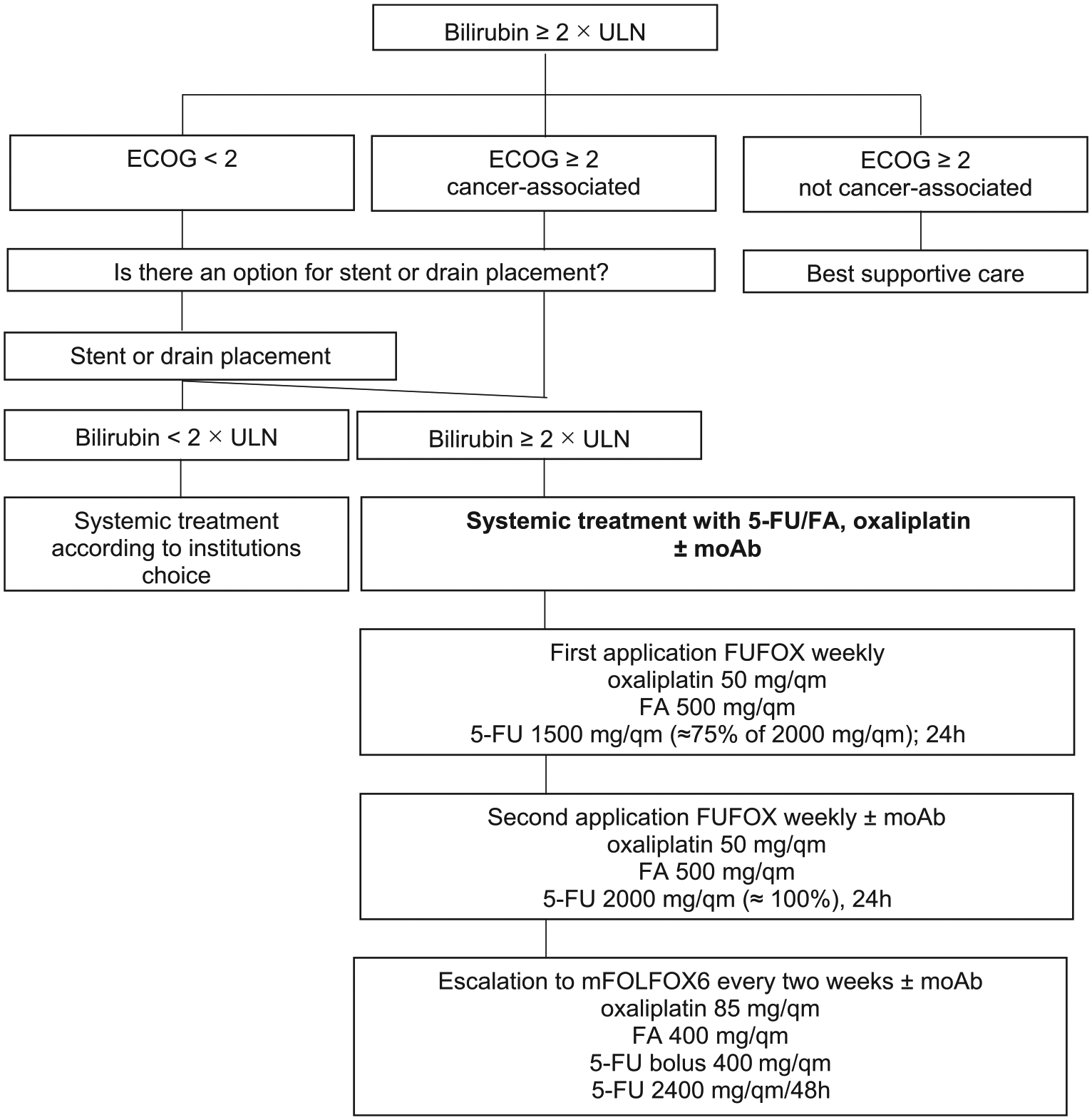
Treatment algorithm for patients with GI cancer and severe liver dysfunction.
In case of elevated levels of bilirubin (>2 × ULN) due to liver metastases, the option for interventional drainage should be evaluated first. If bilirubin remains high (or drainage is technically not possible) and ECOG PS is adequate, systemic treatment should be started with a dose-reduced regimen, preferably FUFOX weekly, with the doses of oxaliplatin 50 mg/qm (100%), FA 500 mg/qm (100%) and 5-FU 1500 mg/qm (75% of the original dosage) for the first application [Grothey et al. 2002]. If treatment is well tolerated, dosages should also be elevated to 100 (2000 mg/qm) given for the second application. Thereafter, a switch to a biweekly regimen with higher single doses (like FOLFOX) is possible. The combination with moAbs is feasible and may thus be considered in eligible patients to increase efficacy in this poor prognosis patient population.
Conclusion
Liver dysfunction resulting from liver metastases in patients with GI malignancies should not lead to therapeutic nihilism. In contrary, these patients may derive relevant benefit from chemotherapy with FA and oxaliplatin with or without the addition of a moAbs. Careful upfront patient selection based on ECOG PS, comorbidity and disease characteristics is important to identify patients able to still tolerate such an aggressive treatment approach. In regard of the limited data further studies are necessary. The recommended approach will further be evaluated in a prospective registry.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
