Abstract
Objectives:
Survival for oesophagogastric adenocarcinoma (OGA) patients varies globally and clinical trial results are seldom replicated in clinical practice. We sought to examine the efficacy and toxicity of salvage paclitaxel chemotherapy for patients with advanced OGA at our institution.
Methods:
Advanced OGA patients treated with paclitaxel between June 2011 and February 2014 were identified from the electronic record at the Royal Marsden Hospital (RMH), London. Chart review was performed to obtain demographics, performance status (PS), laboratory parameters, radiological response and dates of progression, death and last follow up. Overall survival (OS) and progression-free survival (PFS) were estimated using the Kaplan–Meier method. Multivariate Cox regression analysis examined the interaction between clinical and laboratory parameters and survival.
Results:
Fifty-seven patients were identified; OS and PFS were 5.8 and 2.6 months respectively. From first-line chemotherapy, median OS was 14.3 months. Two-year and three-year survival rates from diagnosis were 26% and 13%. More than or equivalent to Grade 3 neutropenia occurred in 13% of patients. In multivariate analysis, PS more than or equal to 2, alkaline phosphatase (ALP) more than or equal to100 U/L, and previous rechallenge with platinum were independent prognostic factors for OS.
Conclusions:
OGA cancer patients treated at RMH with salvage paclitaxel had an OS equivalent to patients in clinical trials with more (33%) PS = 2 patients treated and less haematological toxicity than Asian patients. Eastern Cooperative Oncology Group (ECOG) PS more than or equal to 2, ALP more than or equal to 100 U/L, and prior platinum rechallenge were associated with poor survival. However, the proportion of patients surviving more than or equal to two years from diagnosis demonstrates a clinically relevant improvement from historical controls.
Keywords
Introduction
Survival for patients diagnosed with unresectable, advanced or metastatic oesophagogastric adenocarcinoma has historically been poor: optimal first-line triplet-combination chemotherapy in fit (ECOG 0–1) patients yields a survival of 9 to11 months [Ajani et al. 2007; Cunningham et al. 2008; Siegel et al. 2012]. Randomized evidence also supports the use of second-line chemotherapy with either paclitaxel, docetaxel or irinotecan for patients who have progressed following first-line therapy [Thuss-Patience et al. 2011; Kang et al. 2012; Hironaka et al. 2013; Ford et al. 2014]. More recently, the results of the REGARD and RAINBOW trials suggest that ramucirumab, a fully humanized VEGFR2 monoclonal antibody, may also be utilized, either as a single agent (with equivalent benefit to cytotoxic chemotherapy), or in combination with paclitaxel in the second-line setting [Fuchs et al. 2014; Wilke et al. 2014].
Gastro-oesophageal cancer is notable for geographic variation in survival, in particular a marked East–West divide [Shah and Kelsen, 2010; Bickenbach and Strong, 2012]. Patterns of anatomical location, stage at presentation and therapeutic approaches vary significantly along these axes, and regional pharmacogenetic diversity may also play a role [Shah and Kelsen, 2010; Bickenbach and Strong, 2012; Saijo, 2013]. As paclitaxel had previously only been investigated in randomized trials in Asia and the COUGAR–02 study had evaluated only docetaxel in European patients, we sought to examine whether the survival of second-line therapy in oesophagogastric cancer patients treated with paclitaxel at our institution was commensurate in terms of efficacy and toxicity, with patients treated using the same regimen in Asia and with docetaxel in the COUGAR–02 study.
Patients and methods
Patients and study design
We retrospectively identified all patients with oesophagogastric adenocarcinoma (OGA) treated with salvage weekly paclitaxel between June 2011 and February 2014 from the electronic patient record at the Royal Marsden Hospital, London. Patients eligible for inclusion in the analysis were those with (a) histologically proven unresectable advanced or metastatic adenocarcinoma of the oesophagus or stomach and (b) progressive disease following prior treatment not including a taxane. The following data were collected: patient demographics, metastatic sites, resection status, response or duration of response to prior chemotherapy, ECOG PS, haemoglobin, albumin, alkaline phosphatase (ALP), neutrophil to lymphocyte ratio, levels of carcinoembryonic antigen (CEA) and carbohydrate antigen 19.9 (CA19.9), Royal Marsden Hospital prognostic score [Chau et al. 2004], CT scan response and date of disease progression, death or last follow up. CT response was annotated in the study database as complete response (CR) or partial response (PR), or stable or progressive disease; all patients with a radiological response had imaging reviewed retrospectively to confirm response according to RECIST 1.1 criteria. The study was reviewed and approved by the institutional review board of the hospital prior to initiation.
Treatment schedule
Paclitaxel at a dose of 80 mg/m2 was administered intravenously on days 1, 8, and 15, every 4 weeks. Treatment continued for either 6 cycles or until event of unacceptable toxicity or disease progression. In case of grade 3 or higher haematological or nonhaematological toxicity, dose of paclitaxel was reduced by 25% or cycle delays were permitted until recovery. Use of haematopoietic growth factors was not recommended.
Treatment evaluation
Performance status, toxicity and laboratory assessments were evaluated before each treatment. Tumour response was assessed using contrast-enhanced CT scan of the chest and abdomen every 12 weeks. Adverse events were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE, version 4.0).
Statistical analysis
The primary endpoint of the study was to evaluate the median overall survival (OS) of all patients, defined as time from the first cycle of paclitaxel to death as a result of any cause. Secondary endpoints were progression-free survival (PFS), defined as time from the first paclitaxel cycle to disease progression or death resulting from any cause; response rate defined as CR or PR from RECIST version 1.1; median number of cycles administered per patient; toxicity assessment; and assessment of clinical and pathological factors associated with prognosis. Survival estimates were conducted using the Kaplan–Meier method, and median rates are presented with 95% confidence intervals (95% CIs). Univariate Cox regression analysis (presenting hazard ratios with 95% CIs) was performed on OS and PFS, testing the following clinical and laboratory parameters: age, ECOG PS, radiological response to previous treatment, neutrophil to lymphocyte ratio, ALP, CEA, CA19.9, sites and numbers of metastases, PFS on first-line therapy, rechallenge with first-line therapy and the Royal Marsden Hospital prognostic score. Any factors significant with a p value of less than 0.05 were entered into a multivariate Cox regression analysis to investigate independent association of the factors with survival. Statistical analyses were performed using SPSS, statistics version 22.
Results
Patient characteristics
A total of 57 patients with OGA treated with salvage weekly paclitaxel at the RMH during the time period between June 2011 and February 2014 were identified for the study. Baseline characteristics are summarized in Table 1. Most patients (74%) were male, and the median age was 64 years. All patients had progressed following a prior platinum-fluoropyrimidine (PF) regimen; 2 patients (3.2%) were treated in the third-line setting (Table 2). The most common first-line chemotherapy was EOX (epirubicin, oxaliplatin, and capecitabine). A minority of patients had previously undergone resection of their primary tumour (23%). Most patients (more than 80%) had derived a clinical benefit [CR or PR, or stable disease (SD)] from first-line chemotherapy and less than half (44%) progressed within less than or equal to 6 months of commencement of first-line treatment. Four (7%) patients had been rechallenged with a platinum-based regimen (based on a long disease-free interval following prior platinum therapy) prior to commencing paclitaxel. Two thirds of patients had an ECOG performance status of 0–1 at initiation of treatment and 88% of patients had either a good- or moderate-risk Royal Marsden Hospital prognostic score.
Patient characteristics.

Previous treatment and response.

14 patients received monoclonal antibodies in conjunction with their first-line chemotherapy. These were trastuzumab (7 patients), rilotumumab (1 patient), cetuximab (1 patient), panitumumab (4 patients), and onartuzumab (1 patient).
CapeOx, capecitabine, oxaliplatin; Carbo-5FU, carboplatin, 5-fluorouracil; CR, complete response; CX, cisplatin, capecitabine; ECX, epirubicin, cisplatin, 5-fluorouracil; ECARBOX, epirubicin, carboplatin, capecitabine; EOX, epirubicin, oxaliplatin, capecitabine; FOLFOX, folinic acid (leucovorin) fluorouracil (5-FU) OX – Oxaliplatin; PD, progressive disease; PR, partial response; SD, stable disease.
Exposure to paclitaxel
The median number of cycles of paclitaxel delivered was three (range, one to eight). One patient discontinued treatment due to a grade 4 allergic reaction. The proportions of patients who experienced dose reductions or dose delays due to toxicity were 14% and 33%, respectively. Eleven (19%) patients were HER2 positive, and six of those patients received trastuzumab plus paclitaxel as second-line therapy. Thirteen patients (23%) received further chemotherapy on disease progression following paclitaxel therapy. Nine patients were treated with irinotecan or FOLFIRI chemotherapy, one patient received a carboplatin-capecitabine doublet and three were enrolled in clinical trials including an FGFR inhibitor, an AKT inhibitor, and a dual TORC inhibitor.
Toxicity
Aggregated toxicity data are shown in Table 3. Regarding haematological toxicities, 6 patients (11%) and 7 patients (13%) experienced leukopenia and neutropenia of grade 3 or 4, respectively. The most common grade 3 or higher nonhaematological toxicities were fatigue (11%), hyponatremia (14%), and liver-function disturbance (9%). Grade 3 sensory neuropathy was observed in 1 patient (2%) and 1 patient (2%) had a severe allergic reaction. Two deaths occurred within 30 days of the last administration of paclitaxel; both of these were due to disease progression.
Toxicity.

GGT, gamma-glutamyltransferase; ALT, alanine aminotransferase.
Response and survival
Forty-nine patients were evaluable for response. No CRs were observed. Nine (18%) patients experienced a PR, and SD was observed in 15 (31%) patients, giving a disease-control rate of 49%. Median follow-up time for the study was 13.6 months. Median OS was 5.8 (95% CI, 4.8 to 6.8) months (Figure 1), and the median PFS was 2.6 (95% CI: 1.9–3.2) months (Figure 2). Survival was not significantly different for patients who were HER2 positive or those who were treated with trastuzumab. From commencement of first-line chemotherapy, median OS was 14.3 (95% CI, 11.1 to 17.6) months. Two-year and three-year survival rates were 26% (95% CI, 13.2 to 38.6) and 13% (95% CI, 1.1 to 25.4), respectively.

Overall survival from initiation of paclitaxel.

Progression-free survival from initiation of paclitaxel.
Univariate & multivariate analysis for independent prognostic factors
Univariate analysis of PFS and OS are presented in Tables 4 and 5.
Univariate and multivariate analysis of clinicopathological factors associated with progression-free survival.
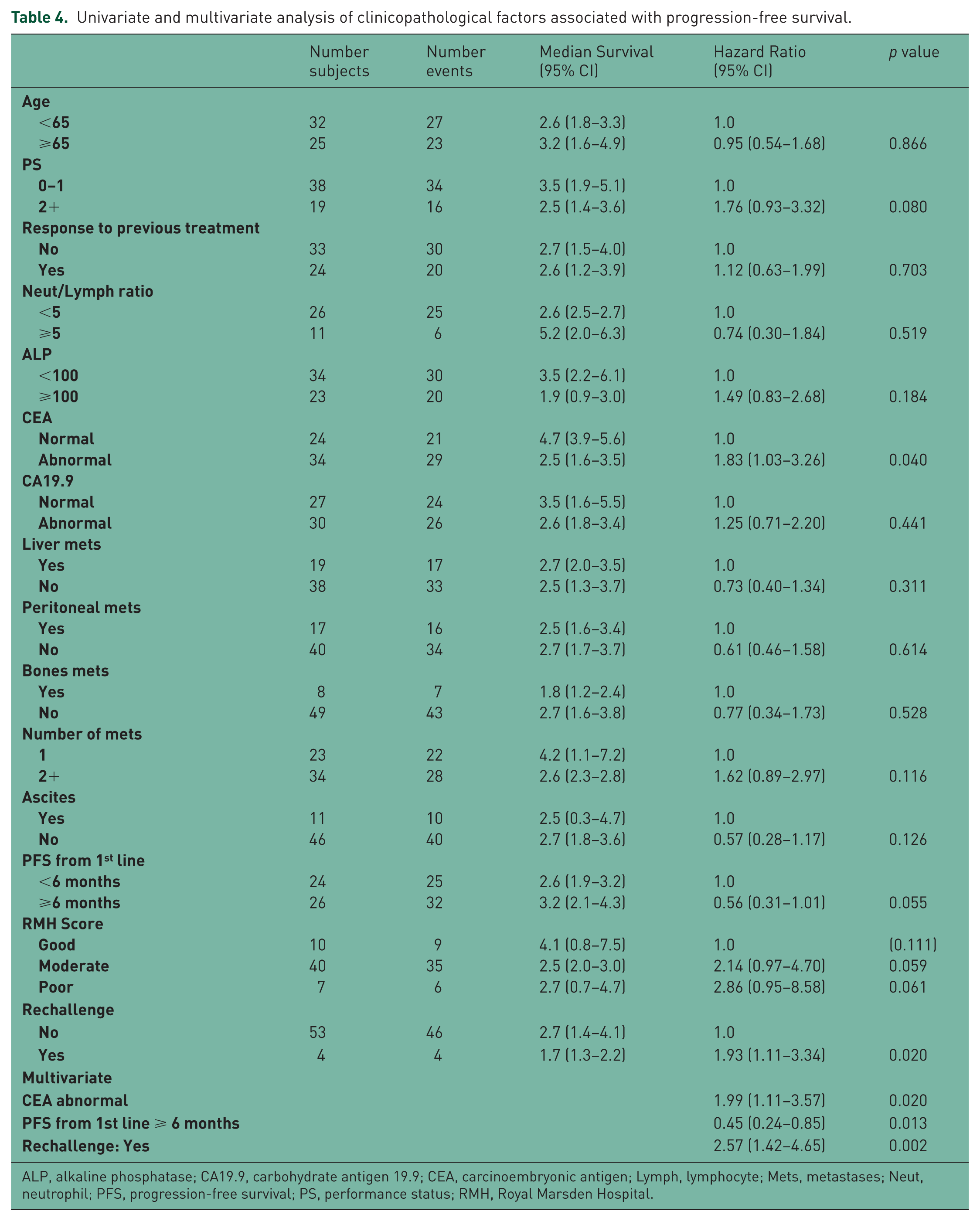
ALP, alkaline phosphatase; CA19.9, carbohydrate antigen 19.9; CEA, carcinoembryonic antigen; Lymph, lymphocyte; Mets, metastases; Neut, neutrophil; PFS, progression-free survival; PS, performance status; RMH, Royal Marsden Hospital.
Univariate and multivariate analysis of clinicopathological factors associated with overall survival.
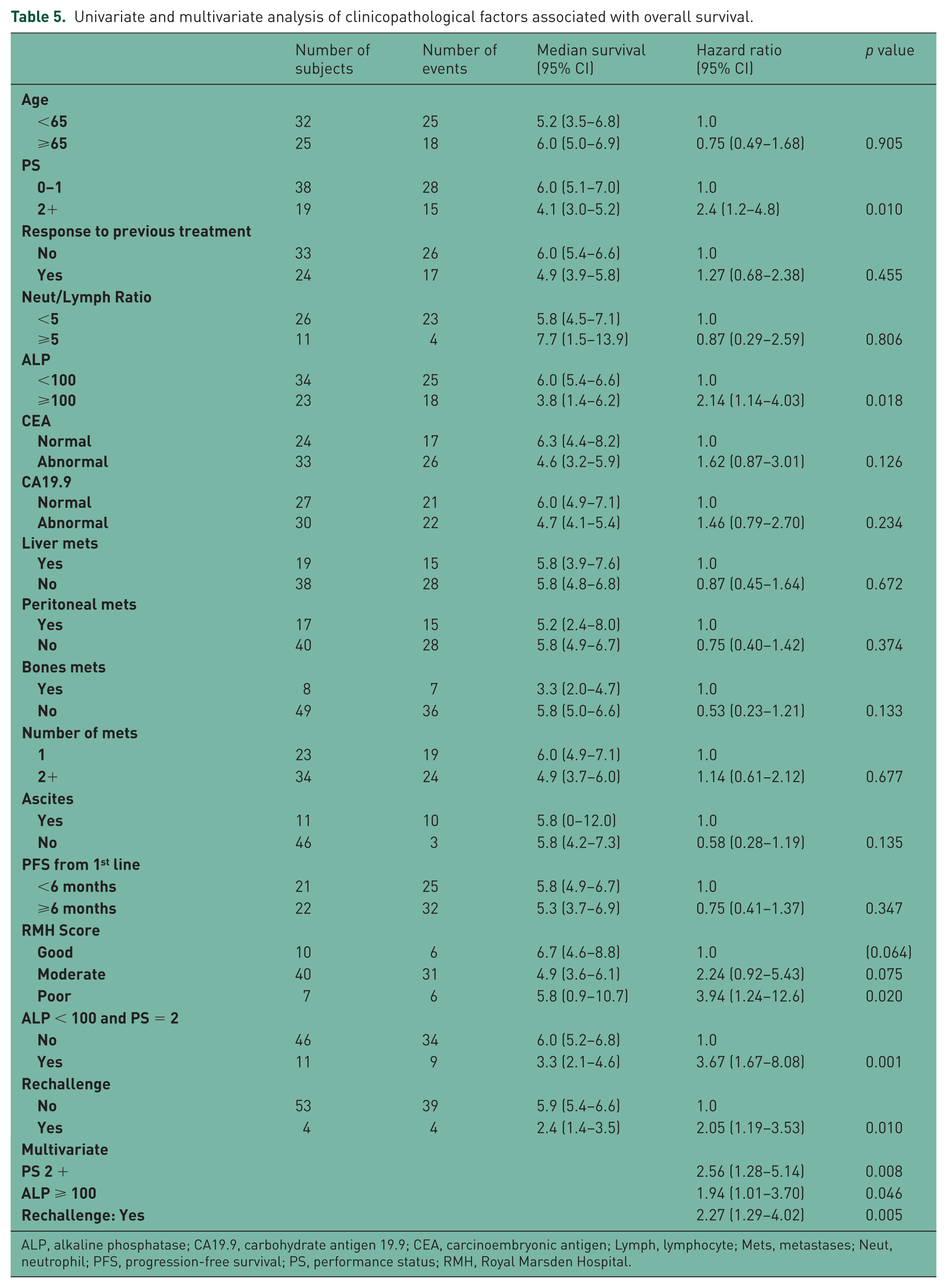
ALP, alkaline phosphatase; CA19.9, carbohydrate antigen 19.9; CEA, carcinoembryonic antigen; Lymph, lymphocyte; Mets, metastases; Neut, neutrophil; PFS, progression-free survival; PS, performance status; RMH, Royal Marsden Hospital.
Factors significantly associated with PFS on multivariate analysis were: abnormal CEA and prior rechallenge with platinum regimen (negative association, see Table 4), and progression of more than or equal to 6 months following completion of first-line chemotherapy (positive association).
For OS, three factors were negatively associated with survival outcomes: these were PS 2, ALP more than or equal to100 U/L, and a history of a previous rechallenge with a platinum-based regimen (Table 5). Median OS for patients with ECOG PS 2, ALP more than or equal to100 U/L, and rechallenged patients with a platinum regimen was 4.1, 3.8 and 2.4 months, respectively.
Discussion
Our study evaluated the survival of patients treated with salvage weekly paclitaxel for advanced oesophagogastric cancer at our institution over a three-year period from 2011 to 2014. Agreeably, we found that both median PFS and OS were consistent with those previously demonstrated in randomized trials in this setting, with the exception of Hironaka and colleagues, who demonstrated a median OS of 9.5 months in paclitaxel-treated Japanese patients (however 90% of those patients were subsequently treated with third-line chemotherapy) [Kang et al. 2012; Hironaka et al. 2013; Ford et al. 2014]. This is notable, as our patient cohort is reflective of all those eligible for treatment with paclitaxel who may not have otherwise met stringent trial eligibility criteria and we therefore believe that these results are generalizable to a broad patient population. Furthermore, median survival from commencement of first-line chemotherapy was 14.3 months, while 2-year and 3-year survival from commencement of first-line chemotherapy for advanced disease were 26% and 13%, respectively. These long-term survival figures are a significant advance on those previously demonstrated in large randomized trials of first-line chemotherapy and demonstrate considerable incremental benefit attributable to salvage chemotherapy in appropriate patients.
Despite these clinically relevant improvements in OS it must be acknowledged that not all patients will benefit from salvage chemotherapy. We performed a regression analysis in order to improve our ability to select patients for treatment. We identified a prolonged (more than or equal to 6 months) PFS whilst receiving first-line chemotherapy as a positive predictor for PFS to paclitaxel, implying that chemosensitivity to first-line chemotherapy is likely to be carried into the second-line setting. We also found that patients with an ECOG PS 2 or greater had significantly shorter OS when treated with paclitaxel than those with ECOG PS 0–1 (median OS 4.1 compared with 6.0 months, respectively). Notably, although ECOG PS 2 patients were eligible for both COUGAR–02 and WJOG 4007 trials, only eighteen such patients were treated on both studies combined, making our series the largest one of PS 2 patients treated with paclitaxel in this setting to date [Hironaka et al. 2013; Ford et al. 2014]. In COUGAR–02, although the confidence intervals surrounding the hazard ratio for PS 2 patients were wide due to the small number of patients treated (n = 14), no statistically significant heterogeneity between the PS subgroups was demonstrated, and therefore it was concluded that salvage chemotherapy provided a benefit for all patients [Ford et al. 2014]. Interestingly, in the WJOG 4007 study, which randomized patients between salvage paclitaxel and irinotecan, subgroup-survival analysis demonstrated a strong trend towards favouring paclitaxel for patients of lower performance status [Hironaka et al. 2013]. Taken together, we believe that weekly paclitaxel chemotherapy is an acceptable regimen for patients with an ECOG PS 2, however, given the restricted median survival in this group a frank discussion with patients regarding the benefits of this approach is warranted.
Interestingly, in our study, patients previously treated with a rechallenge of platinum-based chemotherapy had a shorter PFS and OS when treated with paclitaxel chemotherapy. The reasons for this are not clear. We have previously demonstrated that platinum-sensitive patients with a disease-free interval of more than or equal to three months appear to derive benefit from rechallenge with platinum-based chemotherapy and this concept is broadly established across tumour types [Okines et al. 2010]. Hypothetical explanations for these findings include cumulative toxicity from prior therapies or more advanced disease at initiation of paclitaxel. However, as this group of patients was small (four in total), we are reluctant to draw any firm conclusions from this data.
In our United Kingdom study population, we found weekly paclitaxel therapy to have less haematological toxicities than reported in previous Asian trials. In our analysis, 13% of patients experienced more than or equivalent to grade 3 neutropenia. This contrasts with a rate of approximately 30% more than or equal to grade 3 neutropenia in two randomized Asian studies using the same regimen [Hironaka et al. 2013; Shitara et al. 2014]. These findings are comparable to those in non-small cell lung cancer in which pharmacogenetic differences in drug metabolism have led to differential toxicities and survival across regions in clinical trials. In a study which examined genotype profiles in Japanese and American patients across four randomized trials in NSCLC, Gandara and colleagues demonstrated variability in allelic frequencies of variants in enzymes relating to paclitaxel and cisplatin metabolism that were associated with both toxicity and patient outcomes [Gandara et al. 2009]. Notably, the rate of more than or equal to grade 3 neutropenia in the paclitaxel-only arm of the globally-recruited RAINBOW study was 19%, which is intermediate between that seen in Asian and Western studies and is consistent with this hypothesis [Wilke et al. 2014].
The strengths of our analysis include a homogenously treated patient population, which is representative of oesophagogastric cancer patients in general and therefore externally valid. As a tertiary referral cancer centre, patients treated at the RMH may participate in clinical trials that could potentially extend their survival. However, this would not impact on the survival during paclitaxel chemotherapy as evidenced by the almost identical PFS and OS in our study when compared to recent randomized trials in the same setting. Finally, although our study is retrospective in nature, all data were collected from the electronic record, which was recorded contemporaneously with patient assessment, and although not comparable to a clinical-trial dataset, is fairly representative of the treatment pathway of each patient.
In conclusion, we have demonstrated that weekly paclitaxel is a well tolerated salvage regimen in European patients with advanced oesophagogastric cancer and at our institution led to survival directly comparable to that reported in the literature. We are encouraged by the improvements in two-year and three-year survival for these patients compared with historical controls and believe that greater incremental gains are possible with the addition of targeted agents such as ramucirumab or extended lines of chemotherapy [Kang et al. 2012; Wilke et al. 2014]. However, for patients with poor prognostic factors, the potential disadvantages of treatment require careful discussion due to the reduced benefits observed in that patient group.
Footnotes
Funding
The authors acknowledge funding support from the NIHR RM/ICR BRC. Noelia Tarazona holds an ESMO Translational Fellowship Grant from the European Society for Medical Oncology.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Cunningham declares research funding from Amgen, Celgene, Sanofi, Merck Serono, Astra Zeneca, Medimmune and Merrimack.
