Abstract
Background:
Although adjuvant chemotherapy in stage III colon cancer improves overall survival, prior studies have shown that it is underused. We analyzed different factors that may influence its use.
Methods:
This is a retrospective study of stage III colon cancer patients (n = 207,718) diagnosed between 2000 and 2011 in the National Cancer Data Base (NCDB). The NCDB contains ~70% of new cancer diagnosis from >1500 American College of Surgeons accredited cancer programs in the United States and Puerto Rico. The chi-squared test was used to determine any difference in characteristics of patients who did or did not receive chemotherapy.
Results:
A total of 35% of all stage III colon cancer patients, and 38% of stage III cases undergoing surgery, did not receive adjuvant chemotherapy. The use of chemotherapy had increased in recent years (64% in 2007–2011 versus 59% in 2000–2002; p < 0.0001). Its use was lower in whites (61%), females (60%), patients ⩾60 years (55%), patients with one or more comorbidities (55%), nonacademic centers (62%), those with medicare insurance (52%), lower education (61%) and income levels (59%, all p < 0.0001). The nonwhite and uninsured were more likely to be <60 years old.
Conclusion:
More than one-third did not receive adjuvant chemotherapy, although its use has increased in more recent years. Age was one of the most important determinants of chemotherapy use, which may explain higher rates in nonwhite and uninsured. In addition to patient characteristics, race, gender and socioeconomic factors influence chemotherapy use. These findings have important implications for healthcare reform.
Introduction
With an estimated 1.2 million new cases and 600,000 deaths per year, colorectal cancer is an important cause of morbidity and mortality worldwide [Jemal et al. 2011]. In the United States, 136,830 new cases of colorectal cancer and 50,310 deaths are estimated to have occurred in 2014 [Siegel et al. 2014b]. Although the incidence as well as the mortality of colon cancer has been declining in the United States over the past two decades, with a study reporting an average decrease of 3.4% and 3% per year, respectively, during 2001 to 2010 [Siegel et al. 2014a], it remains the third most common malignancy and third leading cause of cancer deaths [Siegel et al. 2014a].
Nearly a quarter of all colon cancer cases are stage III at diagnosis [Jullumstro et al. 2009; Edge et al. 2010]. Surgery is the primary curative modality in 70–80% of colon cancer patients who present with a nonmetastatic disease [Peeters and Haller, 1999; Siegel et al. 2014a]. However, recurrence is common and is seen in nearly 30% of stage III cases after 5 years [Tsikitis et al. 2014]. Adjuvant chemotherapy can eradicate micrometastases, prevent recurrence and improve overall survival. Several large-scale clinical trials have shown clear survival benefit with adjuvant chemotherapy in stage III colon cancer [Laurie et al. 1989; Moertel et al. 1990, 1995; Andre et al. 2004; Twelves et al. 2005; Kuebler et al. 2007].
Based on the encouraging results from the early trials, the National Institutes of Health (NIH) Consensus Conference in 1990 recommended that patients with stage III colon cancer receive adjuvant chemotherapy [NIH Consensus Conference, 1990]. While 5-fluorouracil (5-FU) based chemotherapy, including combination with leucovorin (LV) and oxaliplatin (FOLFOX) has been the cornerstone of treatment in adjuvant setting, newer drugs such as capecitabine and tegafur with uracil have shown promising results in different clinical trials and offer excellent alternatives for adjuvant chemotherapy [Buyse et al. 1988; Moertel et al. 1990; Wolmark et al. 1993, Marsoni, 1995; Andre et al. 2003, 2004; Chau et al. 2005; Haller et al. 2005; Poplin et al. 2005; Twelves et al. 2005; Lembersky et al. 2006].
Two large trials showed excellent benefits with combining oxaliplatin with 5-FU and LV, the FOLFOX and FLOX regimens, with significant improvement in disease-free and overall survival [Andre et al. 2004; Kuebler et al. 2007]. The maximum benefit is seen when chemotherapy is started within 8 weeks of surgery [Glimelius et al. 2005]. In the background of these results, FOLFOX for 6 months in adjuvant setting within 8 weeks of surgical resection and anastomosis has been adopted as the standard of care in stage III colon cancer patients [Engstrom et al. 2009]. Infusional 5-FU/LV or capecitabine alone may be considered in patients who are not fit to tolerate FOLFOX.
The National Comprehensive Cancer Network (NCCN) recommends colectomy with en bloc removal of regional lymph nodes followed by adjuvant chemotherapy for 6 months in stage III colon cancer [Engstrom et al. 2009]. While the panel recommends combination of oxaliplatin with infusional 5-FU and leucovorin (FOLFOX) as the standard of care, other options recommended in patients unfit for oxaliplatin therapy include either single agent capecitabine or a combination of 5-FU and LV.
Despite these compelling evidences of benefit and clear recommendations for the use of adjuvant chemotherapy in stage III colon cancer, it is still underused. Literature estimates its use in 62–74% of stage III colon cancer [Jessup et al. 2005; Phelip et al. 2010; Boland et al. 2013]. A study reported an increase in adjuvant chemotherapy use from 39% in 1991 to 64% in 2002 [Jessup et al. 2005]. Even more than two decades after being first endorsed by the NIH Consensus Conference, the practice has still not been completely embraced.
Our study is the largest and the most recent study to determine the use of chemotherapy in stage III colon cancer. We also analyzed different treatment related, demographic and socioeconomic factors that may influence its use.
Methods
This is a retrospective study of the NCDB of colon cancer patients diagnosed between 2000 and 2011. The NCDB contains nearly 70% of all new cancer diagnoses from more than 1500 American College of Surgeons accredited cancer programs in the United States and Puerto Rico. Certified tumor registrars at the Commission on Cancer accredited cancer program registries utilize nationally standardized data items and coding definitions to collect oncology data from patient charts and submit data using a specified transmission format. Data integrity and quality monitoring are closely verified [Bilimoria et al. 2008; Tsai et al. 2010].
We extracted data from the NCDB on 29 January 2014. As our study focused on the use of chemotherapy in stage III colon cancer; all calculations were done on stage III colon cancer cases only. Furthermore, the pattern of chemotherapy use according to different treatment-related, demographic and socioeconomic parameters was calculated as a percentage of the total stage III cases receiving surgery. Cases receiving no first course treatment as well as those receiving only nonsurgical treatment (n = 11,306) were excluded from these pattern calculations.
The treatment facilities in the NCDB are categorized into four categories, depending on the service provided and case volume: community cancer centers (100–649 cancer cases annually, may need referral for a portion of therapy); comprehensive community cancer centers (⩾650 cases annually, may need referral for a portion of therapy); academic comprehensive centers (associated with university medical schools or designated as National Cancer Institute Comprehensive Cancer Care Programs); and others [Conway et al. 2009]. The NCDB estimates the educational attainment for the patient’s area of residence by matching the zip code of the patient recorded at the time of diagnosis against files derived from US Census 2000 data.
The Institutional Review Board waiver was obtained from the University of Nebraska Medical Center Institutional Review Board.
Statistical analysis
Descriptive statistics were used to calculate the frequency of distribution of cases receiving chemotherapy according treatment-related, demographic and socioeconomic characteristics. The chi-squared test was used to determine any difference in characteristics of patients who did or did not receive chemotherapy.
Results
A total of 912,743 cases of colon cancer were reported to the NCDB by 1661 different hospitals, including 260 academic hospital programs, between 2000 and 2011. Nearly a quarter of these cases (n = 207,718) were in stage III; 95% of these stage III cases (n = 196,412) underwent surgery. However, almost 35% (n = 74,336) of all stage III colon cancer cases, and 38% of stage III colon cancer patients who underwent surgery, did not receive any chemotherapy. Among patients who underwent surgery, the use of chemotherapy had, however, increased in recent years with its use estimated at 59%, 62% and 64% for the time periods between 2000–2002, 2003–2006 and 2007–2011, respectively (p < 0.0001).
Patients’ demographic characteristics (age, sex, race and comorbidity) were important determinants for chemotherapy use. While its use was 82% in patients below 60 years of age, only 55% of those aged 60 years or above received it (p < 0.0001). A total of 65% of males and 60% of females with stage III colon cancer undergoing surgery received chemotherapy (p < 0.0001). Its use was 61% in whites, 65% in African Americans and 66% in Hispanics (p < 0.0001). Similarly, 67% of those without any significant comorbidity, compared to 55% of those with one or more comorbidity, received chemotherapy (p < 0.0001).
The use of adjuvant chemotherapy was also dependent on different socioeconomic factors. Its use stood at 64% for academic healthcare centers and 62% for nonacademic centers (p < 0.0001). More patients with private insurance (77%) received chemotherapy than those without insurance (73%) or those with Medicaid (70%) or medicare (52%) (p < 0.0001). Similarly, 63% of patients coming from neighborhoods where more than 88% of the population had a high school degree received chemotherapy; in comparison, the usage in neighborhoods that had either 70–88% or less than 70% of their population with a high school degree stood at 62% and 61%, respectively (p < 0.0001). Based on annual household income, 63% of those with $49,000 or more, 62% of those with between $28,000 and $48,999, and 59% of those with below $28,000 received chemotherapy (p < 0.0001). Of those patients who had to travel 100 miles or more to reach the treatment facility, 59% received chemotherapy compared with 62% of those who had to travel less than 100 miles (p < 0.0001) (Table 1).
Chemotherapy use in stage III colon cancer patients.

Discussion
Our analysis of the large NCDB demonstrated that more than one-third of stage III colon cancer cases, diagnosed between 2000 and 2011, did not receive adjuvant chemotherapy. The use of chemotherapy, however, had increased in recent years. These findings are consistent with prior studies, which estimated its use to range from 62% to 74% of the cases, with newer studies showing greater use [Jessup et al. 2005; Phelip et al. 2010; Boland et al. 2013]. The use of adjuvant chemotherapy improves cure rates in stage III colon cancer (Table 2) and is recommended as a standard practice by the NIH and NCCN [IMPACT investigators, 1990; Engstrom et al. 2009].
Adjuvant chemotherapy in stage III colon cancer.
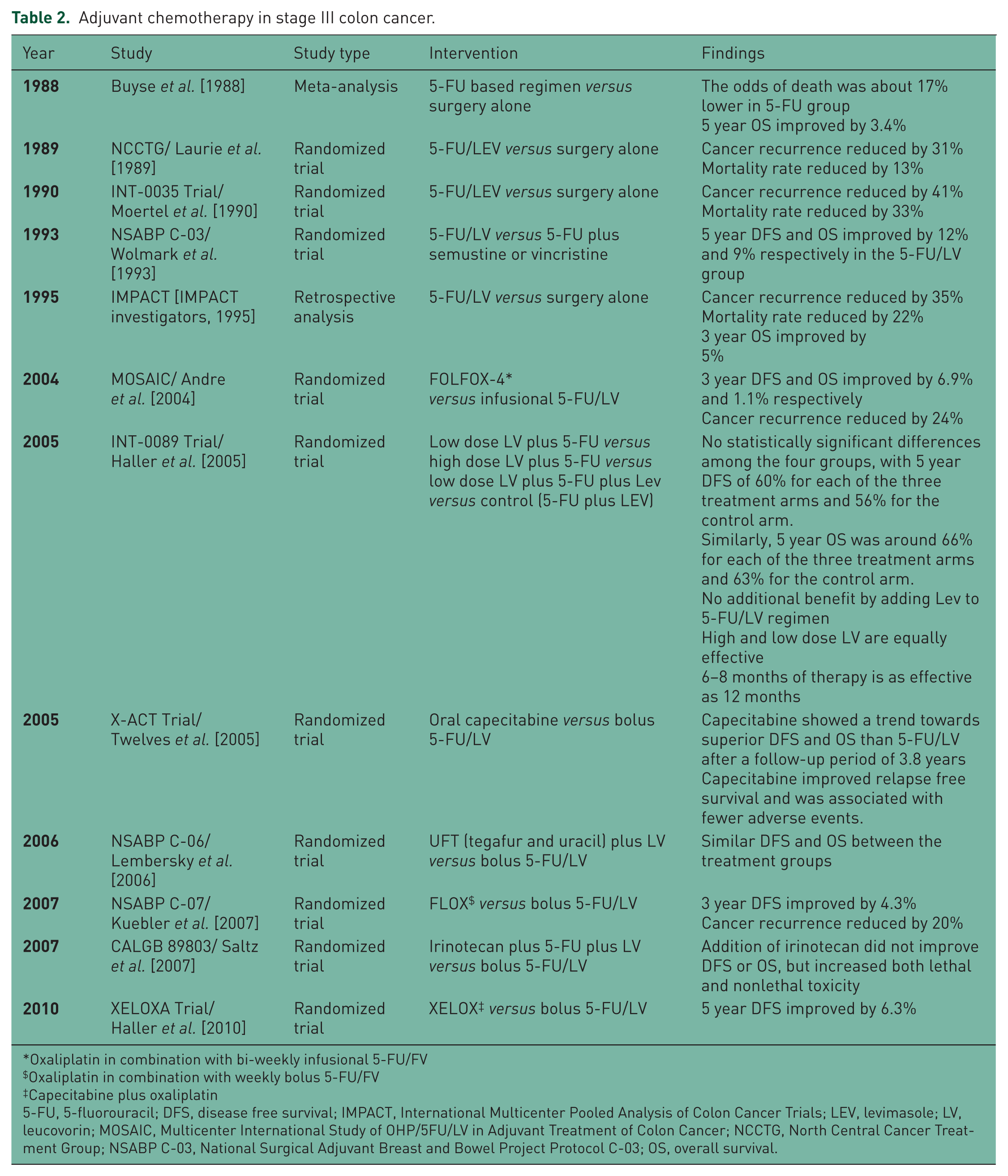
Oxaliplatin in combination with bi-weekly infusional 5-FU/FV
Oxaliplatin in combination with weekly bolus 5-FU/FV
Capecitabine plus oxaliplatin
5-FU, 5-fluorouracil; DFS, disease free survival; IMPACT, International Multicenter Pooled Analysis of Colon Cancer Trials; LEV, levimasole; LV, leucovorin; MOSAIC, Multicenter International Study of OHP/5FU/LV in Adjuvant Treatment of Colon Cancer; NCCTG, North Central Cancer Treatment Group; NSABP C-03, National Surgical Adjuvant Breast and Bowel Project Protocol C-03; OS, overall survival.
Age at diagnosis was one of the biggest determinants of chemotherapy use, with just 55% of patients ⩾ 60 years receiving chemotherapy compared with 82% of patients <60 years. This is consistent with prior reports [Schrag et al. 2001; Sundararajan et al. 2001; Potosky et al. 2002; Vaneenwyk et al. 2002; Jessup et al. 2005; Phelip et al. 2010]. A prior NCDB study (1990–2002) demonstrated similar findings with chemotherapy use in 62% of patients ⩾60 years compared with 82% of patients <60 years [Jessup et al. 2005]. Similarly, in another study, 55% of patients aged more than 65 years received adjuvant chemotherapy, with the rate declining steeply with further increase in age [Schrag et al. 2001].
Several factors may contribute to this low usage in the elderly patients. With increasing age, the burden of comorbidities becomes higher, which may complicate or even preclude the use of adjuvant chemotherapy. In our study, only half of the patients with significant comorbidities (versus 67% in patients without any comorbidity) received chemotherapy. In addition, nonmedical barriers such as poor financial status and family support may also contribute to limited access of older patients to chemotherapy. Although chemotherapy use was lower in patients with Medicare insurance, this likely reflects the older age group of these patients rather than their financial status. Many studies show survival benefit with adjuvant chemotherapy in the elderly population, without any significant increase in toxicity profile [Sargent et al. 2001; Hanna et al. 2012; Sanoff et al. 2012]. However, FOLFOX may not necessarily be the optimal adjuvant chemotherapy as a few studies suggest that adding oxaliplatin to 5FU/LV may not improve survival in patients above 70 years [Yothers et al. 2011; Sanoff et al. 2012; Tournigand et al. 2012; McCleary et al. 2013].
Academic hospitals were more likely to administer chemotherapy than nonacademic hospitals. Though women were as likely as men to benefit from adjuvant chemotherapy, males were slightly more likely to receive chemotherapy than females, consistent with prior studies [Mahoney et al. 2000; Jessup et al. 2005]. Chemotherapy use was slightly higher among Hispanics and African-Americans than Caucasians. This finding differs from the results of many other studies [Baldwin et al. 2005; Jessup et al. 2005]. This may be explained, at least partly, by a higher prevalence of older patients among Caucasians; 77% of Caucasians were ⩾60 years, compared with 62% of African-Americans and 61% of Hispanics. Patients from areas with higher educational or income status and those who required a shorter commute to the hospital were slightly more likely to receive chemotherapy. Several studies corroborated our findings of significant influence of different socioeconomic factors in the utilization of adjuvant chemotherapy in stage III colon cancer [Roetzheim et al. 2000; Vaneenwyk et al. 2002; Boland et al. 2013].
The retrospective study design and lack of patient level data for multivariate analysis are the major limitations of our study. In addition, the NCDB contains only the total number of cases in which systemic chemotherapy was used, without specifying whether such chemotherapy was used in adjuvant or neo-adjuvant setting; however, neo-adjuvant chemotherapy is not the standard of care in stage III colon cancer. Even though some of the differences between different subgroups in receipt of chemotherapy are statistically significant, the actual clinical difference may be small. This may be seen with inclusion of a large sample size, as in this study. Large databases, as in this study, may have a risk of coding error; however, the data in NCDB undergo extensive quality monitoring and evaluation for integrity. Despite these limitations, our study utilizes a very large national cancer database and is able to evaluate the impact of several treatment-related, demographic and socioeconomic on the receipt of chemotherapy in stage III colon cancer.
Conclusion
Mitigation of risk factors along with better screening and treatment modalities, including adjuvant chemotherapy, have resulted in a decline of the incidence and mortality of colon cancers. Despite compelling evidences of the benefit and clear recommendations for the use of adjuvant chemotherapy in stage III colon cancer, it is still underused. Different demographic, socioeconomic and treatment related factors continue to influence its use. Careful discussion about the risks and benefits of chemotherapy in older patients, use of geriatric assessment tools to anticipate the risk of associated toxicities in older patients [Extermann and Hurria, 2007; Ramjaun et al. 2013], greater involvement of patients >70 years old in future trials, improving physician awareness regarding healthcare disparities, provision of improved supportive care to avoid toxicities as well as utilizing multidisciplinary care to improve overall health can help in addressing some of these disparities. At institutional level, development of multidisciplinary assessment and care models, an institutional protocol, and measures to improve physician awareness can be beneficial. Further studies focusing on these disparities are necessary to improve our understanding of these issues.
Footnotes
Acknowledgements
This paper was presented as an abstract at the 50th Annual Meeting of the American Society of Clinical Oncology on 31 May 2014.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Peter T. Silberstein reports receiving payment for lectures from Bristol Myers and Celgene in the past. There are no conflicts of interest for any other authors.
