Abstract
Background
Proximal humerus fractures (PHFs) are common in the elderly, often requiring acute arthroplasty, such as reverse shoulder arthroplasty (RSA) or hemiarthroplasty (HA). Understanding long-term mortality trends in these patients is crucial for improving care and informing health economic analyses.
Methods
Data from the Australian Orthopaedic Association National Joint Replacement Registry (2004–2022) were analysed. Patients who underwent primary RSA or HA for PHF were included. Mortality rates were calculated using standardised mortality ratios (SMRs) and compared to the general Australian population, with subgroup analyses based on age, gender, body mass index (BMI), and American Society of Anaesthesiologists (ASA) score.
Results
A total of 7868 RSA and 3006 HA procedures were reviewed. PHF patients undergoing RSA or HA had significantly higher mortality rates than the general population, with an increasing trend over 15 years. Higher mortality was associated with younger age, male gender, extreme BMI, and higher ASA scores.
Conclusion
PHF patients receiving acute arthroplasty face higher long-term mortality, influenced by demographic and clinical factors. These findings underscore the need for optimised care and healthcare policy adjustments for this vulnerable group. Further research is required to establish causality and improve outcomes.
Keywords
Introduction
Proximal humerus fractures (PHFs) represent a common pattern of injury, particularly amongst the elderly (>65 years) population.1,2 Previous studies report that PHF are the third most common non-vertebral fracture in this age group, after neck of femur and distal radius fractures. 3 Acute management options for PHF include non-operative treatment, surgical fixation with open reduction internal fixation, and acute arthroplasty. 4 Amongst the arthroplasty-based management options, reverse shoulder arthroplasty (RSA) and hemiarthroplasty (HA) are the predominant choices amongst most shoulder surgeons. 5
Considering that PHF is associated with frailty and comorbidities such as osteoporosis, it stands to reason that mortality rates in this cohort will be higher compared to the general population.6,7 Understanding the relationship between PHF patients and mortality is significant for several reasons. From a clinical perspective, if PHF is found to be a pre-morbid condition, then this should drive an impetus to treat PHF patients like other fragility fracture patients – with an emphasis on bone density optimisation, falls prevention, and medical review in collaboration with orthogeriatric specialists. 8 However, from an economic perspective, changes in mortality rates within this cohort necessitate an adjustment of how economic analyses are performed to evaluate the cost-effectiveness of acute arthroplasty as a treatment for PHF.9,10
Conventional health economic analyses calculate quality-adjusted life years (QALYs) after a treatment intervention. 11 The unit of a QALY is calculated by multiplying the change in utility (quality of life) values induced by the treatment intervention with the duration of treatment effect. 12 In patients who received acute arthroplasty as treatment for PHF, the duration of treatment effect is influenced by the years of life remaining for the patient. For example, a patient who lives 5 years after surgery will ceteris paribus benefit more than a patient who only lives 1 year after surgery. Consequently, an accurate understanding of long-term mortality in PHF patients who undergo arthroplasty will refine the precision of economic analyses amongst this cohort and improve the healthcare and policy decisions that are made based on this information. 13
Therefore, the aim of this study was to calculate the standardised mortality ratios (SMRs) of PHF patients who undergo acute arthroplasty in comparison to the general population. In addition, temporal trends in mortality rates up to 15-years after index procedure are presented to demonstrate change in mortality rate over time in this cohort.
Methods
Registry background
The AOANJRR began data collection on 1 September 1999 for all hip and knee arthroplasty procedures performed in Australia. Data collection was expanded to include shoulder arthroplasty procedures in April 2004 and has documented almost all shoulder arthroplasty procedures Australia-wide since November 2007. The AOANJRR now has data on 99.0% of hip, knee and shoulder joint replacement procedures performed in Australia. 14 These data are externally validated against patient-level data provided by all Australian state and territory health departments. A sequential, multilevel matching process is used to identify any missing data which are subsequently obtained by follow-up with the relevant hospital. Each month, in addition to internal validation and data quality checks, all primary procedures are linked to any subsequent revision involving the same patient, joint and side. Data are also matched bi-annually to the Australian National Death Index (NDI) data to identify patients who have died.
Study design
We used prospectively collected data from the AOANJRR between 16 April 2004 and 31 December 2022. All patients who had a primary RSA or HA procedure using a stemmed prosthesis performed for fracture, were included in the study. Procedures revised for glenoid fracture or post-traumatic arthritis were excluded. Additional patient characteristics such as age, gender, body mass index (BMI) category, and American Society of Anaesthesiologists (ASA) score were also included in the study. Patients aged <55 years were excluded due to inadequate sample size.
Outcome measures
The primary outcome measure of this study was the long-term (up to 15-years after index procedure) mortality rates of patients with PHF who have undergone acute RSA or HA. Only the first procedure (in patients with bilateral or multiple arthroplasty procedures) were included for analysis. Mortality rates are presented as SMRs, which is a statistical measure of mortality comparing a specific group (PHF patients with RSA or HA) to the general Australian population. SMR values greater than 1.0 represent a mortality rate that is higher in the PHF cohort compared with the general population, whereas SMR values less than 1.0 represent a mortality rate that is lower. We have separated the primary outcome measure into three distinct groups; RSA only, HA only, and RSA/HA combined. Secondary outcome measures include a sub-analysis of SMR by procedure (RSA vs. HA), age, gender, BMI, and ASA score. An additional comparison of SMR by indication (PHF vs. osteoarthritis [OA]) for RSA is presented to provide a baseline comparator of excess mortality associated with PHF as an acute injury event.
Data analyses
Observed deaths in the RSA and HA population were counted for the entirety of the follow-up period. The mortality rate was calculated using the indirect method. 15 This method calculated expected deaths by using estimated annual mortality rates from national age-sex-specific mortality rates standardised to the 2004 Australian population cohort, as published in the ABS. 16 The observed deaths were then compared against the expected deaths in the total cohort and for each subgroup. The 95% confidence interval for the SMR was calculated using the standard Poisson approximation. 17 Bonferroni corrections were used to calculate overall significance of the effect for each patient or surgical factor evaluated. 18 A p value of 0.05 or less was considered statistically significant. Statistical analyses were performed using software package R (RStudio Team 2022).
Results
Patient characteristics
There were 7868 primary RSA procedures and 3006 HA primary procedures (10,874 RSA or HA procedures) included in the analysis. This comprised of 10,069 unique patients, of whom 2859 patients had died at the time of data analysis. The mean age of the combined cohort was 74.2 years (standard deviation: 8.8 years), with patients aged over 85 years representing 13.1% of the total cohort. Females were significantly over-represented in this cohort, representing 82.6% of all patients. Further details of patient characteristics are presented in Table 1.
Patient characteristics by procedure type.
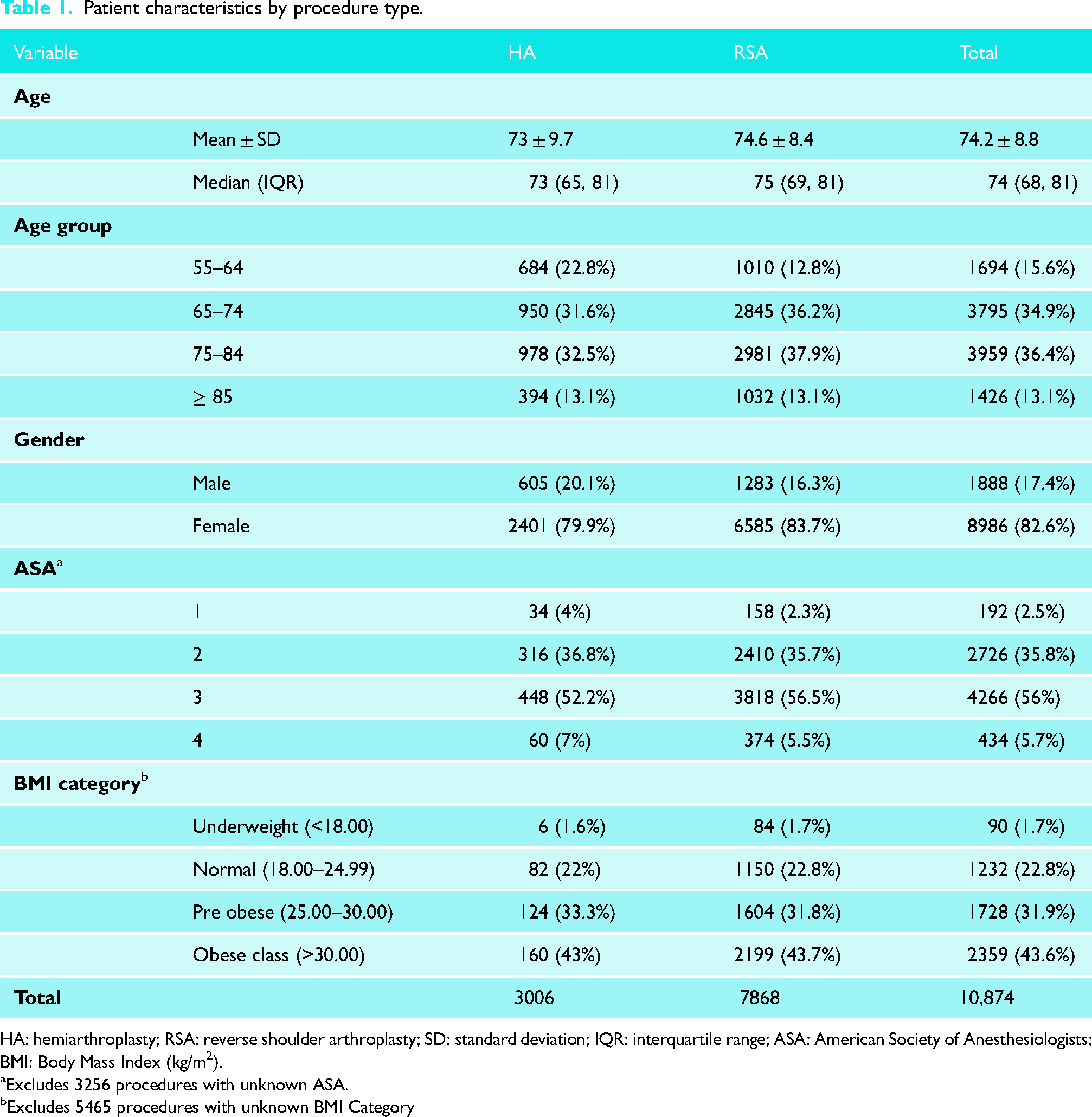
HA: hemiarthroplasty; RSA: reverse shoulder arthroplasty; SD: standard deviation; IQR: interquartile range; ASA: American Society of Anesthesiologists; BMI: Body Mass Index (kg/m 2 ).
Excludes 3256 procedures with unknown ASA.
Excludes 5465 procedures with unknown BMI Category
Mortality rates for RSA and Ha
In general, PHF patients who underwent RSA or HA were found to have a significantly higher mortality rate compared to the general population. The overall SMR of the full cohort was 1.26 (95% confidence interval [95% CI] 1.21–1.31). A temporal increase in SMR was also found in this cohort, with the SMR at one year after index procedure being 1.15 (95% CI 1.08 to 1.21) in contrast to the SMR at 10-years after index procedure being 1.35 (95% CI 1.25 to 1.46) (Figure 1). Patients who received an RSA reported lower mortality rates compared those who received an HA up to 10-years post-index procedure (Figure 2). The 10-year SMR for RSA was 1.31 (95% CI 1.13–1.52) compared with the 10-year SMR for HA of 1.37 (95% CI 1.25–1.50). Within the first year after index procedure, the RSA cohort demonstrated no significant differences in mortality compared with the general population (SMR immediately after procedure: 1.01, 95% CI 0.92–1.12).
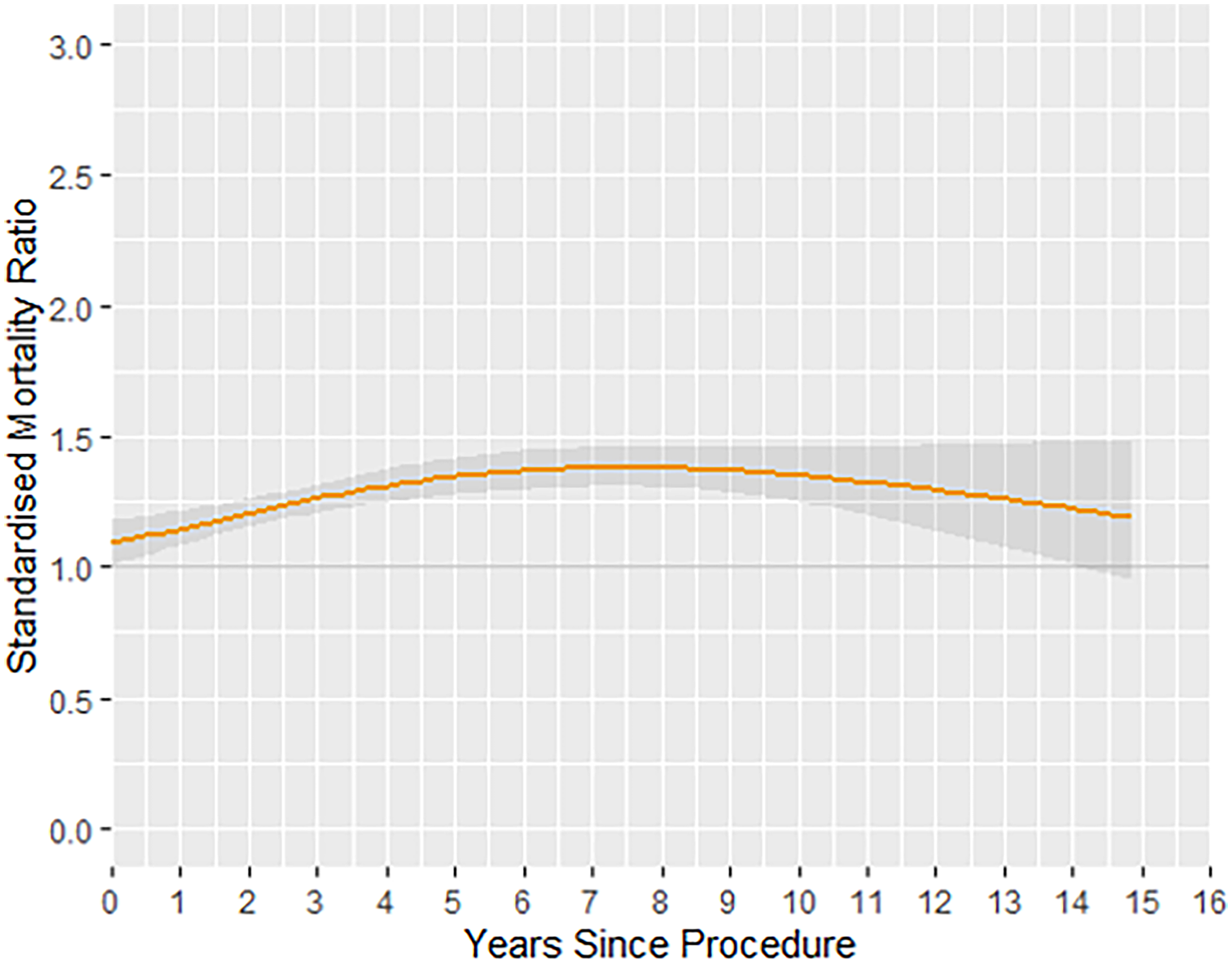
Temporal changes in mortality rate (SMR) for full cohort of RSA and HA. SMR: standardised mortality ratio; RSA: reverse shoulder arthroplasty; HA: hemiarthroplasty.
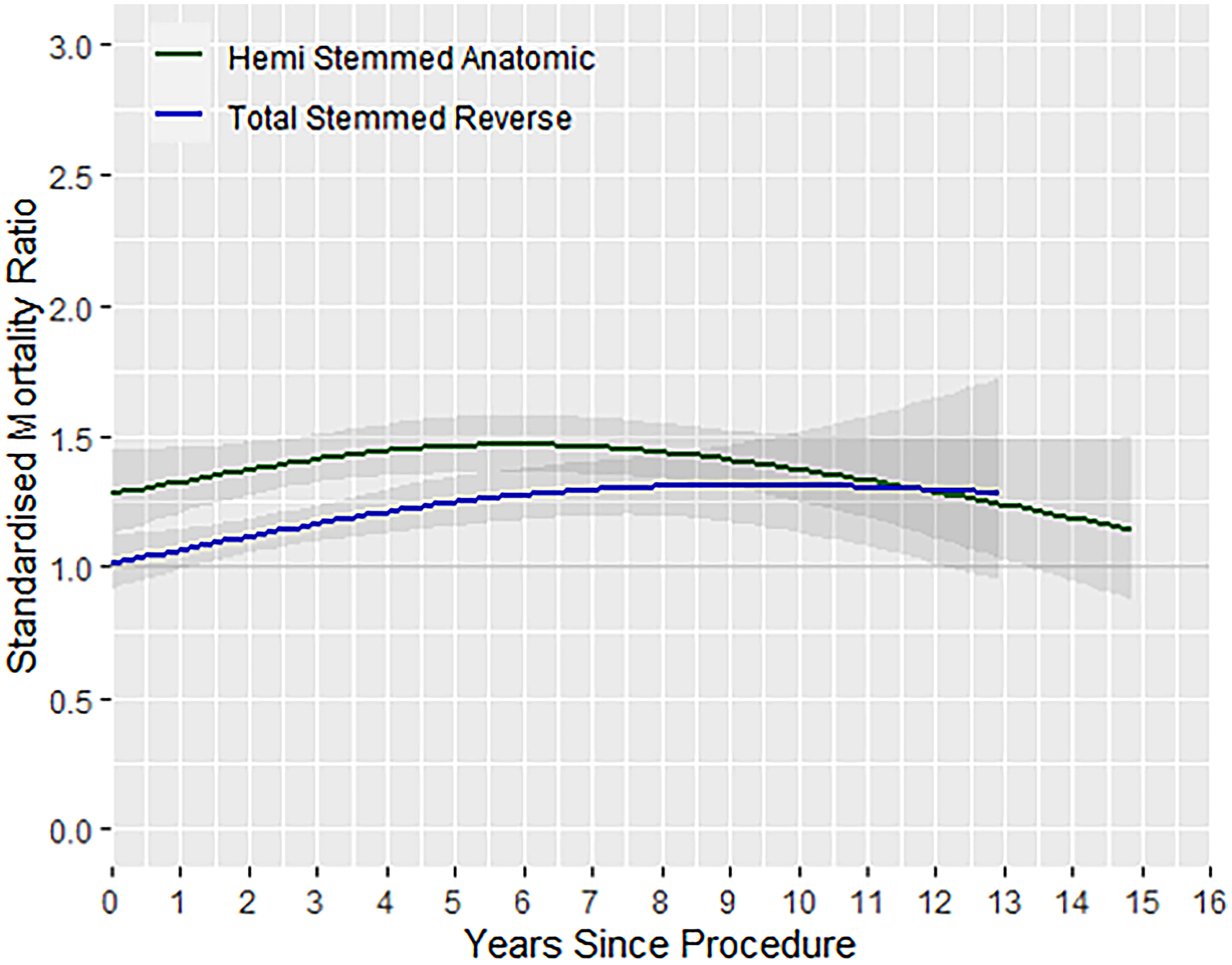
Temporal changes in mortality rate (SMR) for RSA and HA cohorts (separate). SMR: standardised mortality ratio; RSA: reverse shoulder arthroplasty; HA: hemiarthroplasty.
Mortality rates by age, gender, BMI, and ASA
Younger patients (aged 55–64 years) had the highest mortality amongst all age groups (Figure 3A and 3B). Within the HA cohort, patients aged 55–64 years reported an SMR of 3.96 (95% CI 2.71–5.78) within the first year after index procedure. This decreased to an SMR of 2.72 (95% CI 2.12–3.50) at 10-years post-index procedure. In comparison, patients of the same age in the RSA cohort reported an SMR of 5.46 (95% CI 3.90–7.65) within the first year after index procedure, with a reduction in SMR to 3.12 (95% CI 2.17–4.50) after 5-years from the index procedure. With respect to gender, both the RSA and HA cohorts independently found higher mortality rates up to 5-years post-index procedure for males compared to females. Furthermore, for both RSA and HA cohorts, patients with higher ASA scores reported higher mortality rates across the entire follow-up period. In the RSA cohort, underweight patients (BMI < 18) had increased mortality rates across the entire follow-up period, whereas obese patients (BMI >30) had increased morality rates beyond 2 years.
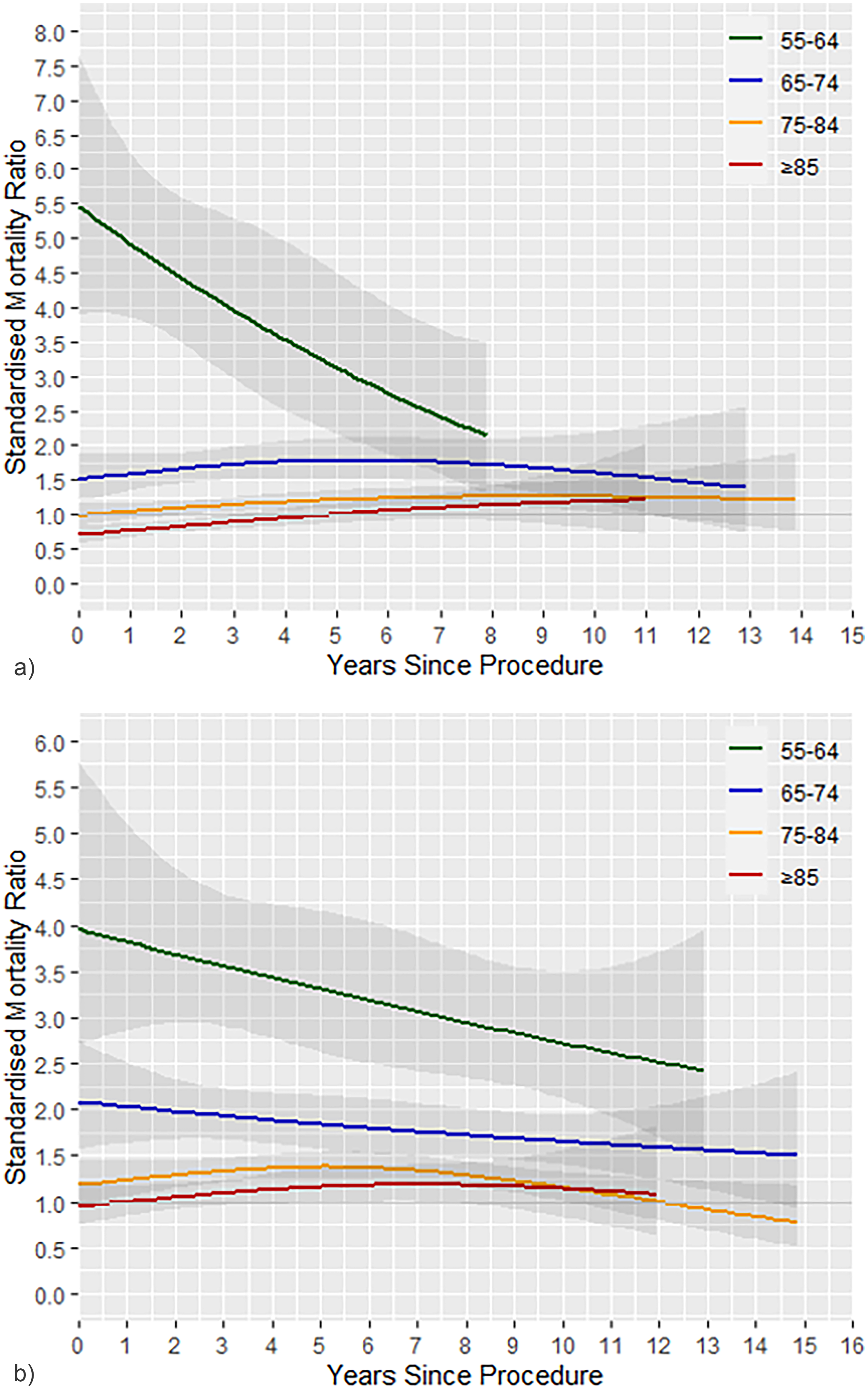
Temporal changes in mortality rate (SMR) for (A) RSA and (B) HA cohorts. SMR: standardised mortality ratio; RSA: reverse shoulder arthroplasty; HA: hemiarthroplasty.
Mortality rates of RSA for PHF vs. OA
When comparing mortality rates of primary RSA performed for PHF and OA, those who had RSA for PHF demonstrated higher mortality compared to those who had RSA for OA up to 14 years after surgery (Figure 4). Within a year since the primary procedure, PHF patients demonstrated an SMR of 1.04 (95% CI 0.94–1.15) directly following the procedure. This is in comparison to patients with OA who had an SMR of 0.43 (95% CI 0.39–0.47) for the same timepoint.
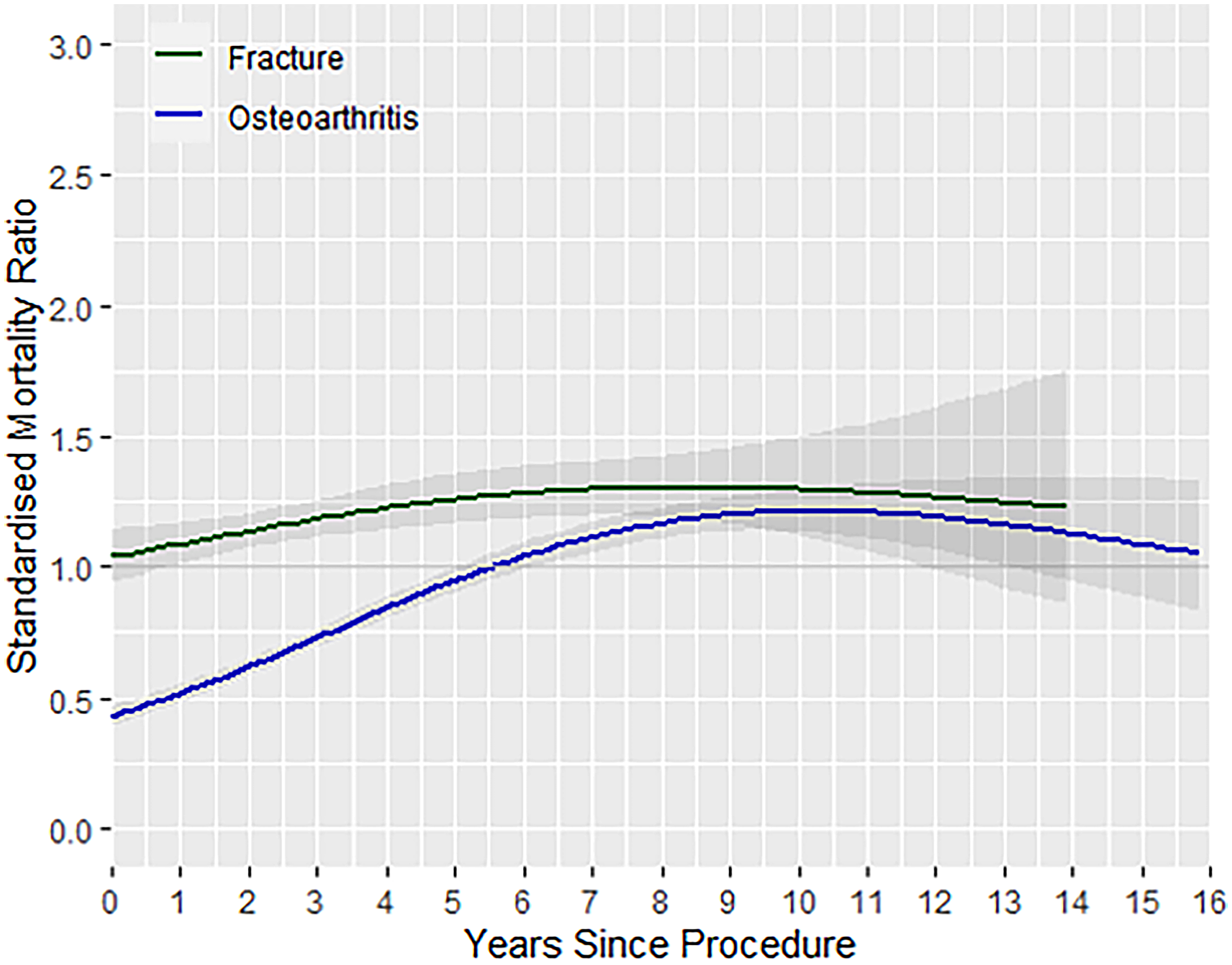
Temporal changes in mortality rate (SMR) by indication for RSA. SMR: standardised mortality ratio; RSA: reverse shoulder arthroplasty.
Discussion
This study analysed the long-term mortality trends of patients with PHF who underwent RSA and HA in comparison to the general population. Across all timepoints, PHF patients demonstrated higher mortality rates compared to the general population, suggesting that this injury pattern combined with acute arthroplasty as management is associated with increased mortality. Furthermore, specific risk factors are associated with higher mortality; notably the HA procedure, younger age group (55–64 years), male gender, being underweight or obese, and higher ASA score. Given the observational nature of the study, it is important to emphasise that these results should not be interpreted to infer causality between PHF (and RSA/HA for PHF) with mortality. However, the results remain significant as they describe the underlying mortality risk observed in PHF patients using national joint registry data. The results reported in this study should be used to drive an impetus to optimise clinical care of these patients in the peri-operative and long-term recovery following acute arthroplasty procedures. Furthermore, the results can be used to improve the precision of health economic analyses for this unique population.
When interpreting the results of this study, it is important to understand the potential sources of bias that may influence mortality rates seen in this population. 19 Most obvious is the understanding that patients who have been selected for RSA or HA to treat their PHF injury are likely to be fitter than those who have been determined to be unfit for operative management. From this perspective, the SMRs reported in this study may be an underestimate of the true mortality rate seen in the full PHF cohort. An additional factor to consider are mortality effects from the PHF patient cohort who are younger and healthier, where surgical fixation may have been selected as their acute management option. This may explain the high mortality rates seen in those aged 55–64 years in this study; where the reported SMR was inflated due to the exclusion of fitter patients where surgical fixation rather than arthroplasty was deemed more appropriate. An alternative inflator of mortality rates seen in the male and younger (55–64 years) populations may be the differential mechanisms of injury; with males who are younger more likely to sustain PHF from high energy trauma. 20
The issue of selection bias may also explain the differences in mortality rate seen in patients who received RSA for PHF compared with OA. In this sub-analysis, patients who had surgery for OA reported a mortality rate less than half of a comparable age-gender adjusted general population within a year of their primary procedure. In this example, the probable effect of selecting patients who meet a minimum set of health criteria to undergo elective surgery is the likely explanation for these findings. However, it is also worth noting that healthier patients have higher demands and therefore may present for correction of their disability as is the case for OA arthroplasty.9,19,21 PHF in this context is a “no choice” presentation, where a wider mix of patient health is presented. This can be measured as a proxy by the distribution of ASA and BMI in these two patient cohorts.
Previous studies that have investigated mortality rates in the PHF population have focused on short-term or peri-operative mortality rates; with no studies to our knowledge reporting long-term mortality rates in PHF patients treated with arthroplasty. 22 With respect to short-term mortality rates, Bergdahl et al. 23 reported the 1-year mortality rate for all operatively and non-operatively managed PHF patients based on data from the Swedish Fracture Registry. In the Bergdahl study, the 1-year mortality rate was reported at 7.83%, with non-operative management considered an independent risk factor for increased mortality rate. A similar study from the United Kingdom reported that 1-year mortality rate of PHF patients (operatively and non-operatively managed) aged over 65 years was 10%. 24
Although short-term mortality studies are important to understand the immediate impact such injuries can have on patients, longer-term mortality rates are necessary to inform health economic analyses. In this study, temporal changes in mortality have been observed amongst all sub-groups and sub-analyses, indicating that the mortality rates of this unique group may differ significantly with time after surgery. The consequence of these temporal changes in mortality are that health economic analyses should not simply use general population lifetables to calculate life expectancy of these patients (during the calculation of QALYs), but instead use adjusted lifetables based on mortality associations observed in patients with PHF. 25
The strengths of this study predominantly are found within the comprehensive capture of data using a national joint registry. Given the legislative support of the AOANJRR to conduct its activities, and the opt-out consent process, the capture rate of RSA and HA procedures (including for fracture) is greater than 99%. 14 Therefore, the mortality rates observed in this study cohort are likely to be representative of the full Australian context. In addition, the AOANJRR is linked at a patient-level to the NDI to provide accurate longitudinal mortality rates. 26
However, there are limitations to the study that must also be acknowledged. First, as an observational study, the mortality rates reported can only be reported as association between the study population and the general Australian population. 27 Causal inferences require a higher level of scrutiny with respect to confounders and this can only be ascertained with confidence through randomised clinical trials.28,29 Second, as with all registry-based studies, the accuracy of the results are dependent on the accuracy of documentation. 30 Although in the AOANJRR, the indication ‘fracture’ is commonly considered to represent acute fracture, we cannot guarantee that surgeons have not included other entities such as non-united PHFs or failed surgical fixation as the true indication for arthroplasty. Accuracy of the study has been improved through the exclusion of glenoid fractures (n = 64) as the indication for surgery. Third, previous studies have suggested that timing of acute arthroplasty for PHF may have an influence on outcomes, including mortality.31,32 Unfortunately, due to the nature of the data, we were unable to distinguish between patients who received acute arthroplasty (<6 weeks from injury) versus patients who received delayed arthroplasty (>6 weeks from injury). Finally, the distinction between mortality associated with the injury (PHF) compared with mortality associated with the surgery (RSA or HA) cannot be made with this study. Additional studies that compare long-term mortality rates in PHF patients managed operatively and non-operatively should be performed in future to make this important distinction. 32
Conclusions
This study provides valuable insights into the long-term mortality trends of patients with PHF who undergo acute arthroplasty procedures. The findings indicate that PHF patients undergoing these procedures exhibit higher mortality rates compared to the general population, with significant variations based on age, gender, BMI, and ASA score. These results underscore the importance of optimising clinical care for PHF patients and highlight the need for tailored interventions addressing specific risk factors associated with increased mortality. This study also provides essential data to refine health economic analyses and inform healthcare policies aimed at improving outcomes in this vulnerable population. Further research, including randomised clinical trials and comparative studies between operative and non-operative management, is warranted to enhance our understanding of mortality outcomes in PHF patients.
Supplemental Material
sj-docx-1-sel-10.1177_17585732251359475 - Supplemental material for Long-term mortality rates in patients with proximal humerus fracture managed acutely with reverse shoulder arthroplasty and hemiarthroplasty
Supplemental material, sj-docx-1-sel-10.1177_17585732251359475 for Long-term mortality rates in patients with proximal humerus fracture managed acutely with reverse shoulder arthroplasty and hemiarthroplasty by Yushy Zhou, Samuel J Lynskey, Peiyao Du, David RJ Gill, Sophie Corfield, Marc Hirner and Brett Moreira in Shoulder & Elbow
Footnotes
Acknowledgements
We thank the AOANJRR staff, orthopaedic surgeons, hospitals, and patients whose data made this work possible.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
The AOANJRR is approved by the Commonwealth of Australia as a Federal Quality Assurance Activity (F2022L00986) Part VC of the Health Insurance Act 1973 (HIA) and Part 10 of the Health Insurance Regulations 2018. All AOANJRR studies are conducted in accordance with ethical principles of research (the Helsinki Declaration II).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: YZ is a recipient of PhD stipend funding from the HCF Research Foundation and The University of Melbourne Research Training Programme. Furthermore, YZ is the recipient of a New Zealand Orthopaedic Association Wishbone Foundation grant. The AOANJRR is funded by the Commonwealth of Australia's Department of Health and Aged Care.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
