Abstract
Incubation is a fundamental component of avian reproductive biology. One element of avian incubation, egg turning, is nearly universal among avian taxa and is vital for optimal embryonic development, hatch success, and chick quality. Despite its importance, egg turning behaviors are poorly understood. Egg turn parameters, such as rate and angle, are subject to wide variation among and within taxa and may vary over the incubation period. In this study, we used data loggers in artificial eggs to measure the rate and angle of egg turns from wild Scarlet Macaw (Ara macao macao) hens nesting within artificial cavities in southeast Peru. Our results from four of these hens indicate that egg turn rates averaged 11.5 turns h−1, turn angles averaged 42.2° turn−1, and these parameters varied significantly among hens. Egg turn rates were dynamic over the incubation period, peaking during the middle third of incubation, whereas egg turn angles increased linearly over the incubation period. Comparison with the literature suggests that the highly altricial Scarlet Macaws turned their eggs at a higher rate than the more precocial taxa studied with similar methods. Our study is the first to characterize the egg turn parameters of any wild parrot using data loggers and may prove useful for discerning larger taxonomic patterns in incubation. We found wild Scarlet Macaw egg turn rates to be much higher than published recommendations for the artificial incubation of parrot eggs, suggesting this work could be used to inform captive breeding and other conservation techniques.
Keywords
Introduction
Incubation is a fundamental component of avian reproductive biology that provides conditions critical for embryonic development. 1 Among birds, incubation is almost universally marked by direct contact between the egg and incubating bird. 1 This form of incubation, known as contact incubation, augments the ability of incubating birds to behaviorally modulate the four essential elements of incubation: temperature, gas exchange, water exchange, and egg turning.1,2 One of these elements, egg turning, involves changes in the location or orientation of eggs, usually mediated through various incubation behaviors. 3 Although egg turning prevents embryonic adhesion to the inner shell membrane surface, its predominant purpose appears to be facilitating the utilization of albumen. 4 Suboptimal egg turning may cause embryonic death or otherwise impair embryonic development, hatch success, and chick quality.3–5 Despite its importance, egg turning is the least studied of the four main components of incubation, and the rates and angles of egg turns have historically been difficult to measure in the wild. 2 Early egg turning studies relied on sub-standardized observations,3,6 but technological developments have given rise to telemetric eggs 7 and egg loggers 8 that can measure egg turn parameters of wild birds with minimal disturbance. The latest egg logger models contain triaxial accelerometers and magnetometers that can provide a complete and accurate log of egg turning events in three dimensional space over the incubation period. 9
Egg turning behaviors are subject to wide variation. 3 Taxa that entirely lack egg turning behaviors include megapodes (Megapodiidae), which have foregone contact incubation altogether, 10 and palm swifts (Cypsiurus spp.), which glue their eggs to their nests with saliva.11,12 Altricial taxa generally turn their eggs at a higher rate than precocial taxa. 13 Within a taxon, egg turning behaviors may vary by reproductive experience, sex, and hormonal condition of the incubating bird; embryonic vocalizations; clutch size; time of day; and weather conditions.3,9,14–21 Parental behaviors during incubation, such as incubation exchange between mates, deliberate cooling of eggs, and defensive movements in response to predators and conspecifics have also been suggested to influence egg turning.14,22,23 Furthermore, egg turning behaviors have also been reported to vary over the incubation period for some taxa but not for others.2,3,6,14,15,24 When it does occur, temporal variation in egg turning behavior may be related to a critical period during the first third of incubation for which egg turning is disproportionately important. 4 Alternatively, it may be related to dynamic trade-offs between incubation and self-maintenance behaviors.6,25,26
The Psittaciformes (parrots) are among the most threatened avian orders.27,28 Parrots face population declines from habitat loss, fragmentation, and degradation, as well as poaching for the illegal wildlife trade. 29 The future persistence of many parrot species rely on conservation efforts, both in in-situ and ex-situ contexts. In-situ conservation efforts for parrots often involve nest management techniques, which can be improved with in-depth knowledge of parrot reproductive ecology and behavior. 23 Ex-situ conservation techniques include captive breeding, which is recommended for 49 different parrot species (11.6% percent of parrot species). 30 Captive breeding frequently involves artificial incubation, as it can facilitate multiple egg laying cycles in a single breeding season and rescue embryos from incompetent parents.8,31 Unfortunately, artificial incubation may also decrease hatch success for many avian taxa. 32 Artificial incubation techniques can likely be improved by characterizing and implementing the incubation parameters of wild pairs, as hatchability in an artificial incubator is largely determined by temperature and egg turning parameters.3,4,8,33 This has been demonstrated for the Asian Houbara (Chlamydotis macqueenii): implementing egg turn parameters measured from wild houbara hens led to a 2.7% increase in the hatch weight of chicks in a captive breeding program for this species. 2 In this study, we used data loggers in artificial eggs to study the egg turning behaviors of wild Scarlet Macaw (Ara macao macao) hens nesting in artificial nest cavities located within the Tambopata National Reserve of southeastern Peru. We characterized the rate and angle of egg turns and analyzed the variation of these parameters among individual hens and over the incubation period.
Methods
Study site
This study was conducted in the Collpa Colorado sector of the Tambopata National Reserve (13° 07’ S, 69° 36’ W; 250 m in elevation), located near the border between the Tambopata National Reserve (275,000 ha) and Bahuaja-Sonene National Park 1,091,416 ha) in the Department of Madre de Dios, in southeastern Peru. The site contains tropical moist forest34,35 including mature floodplain forest, upland forest, successional floodplain forest, successional riparian forests, Mauritia flexuosa (Arecaceae) palm swamps, and bamboo stands.34,36–39 The area receives 3,200 mm of rainfall annually with a dry season between April and October36,39 and has an average temperature of 24.3 ± 1.48°C. 36 The site is near a 500-m-long, 30-m-high clay lick (Collpa Colorado) used by up to 1,000 macaws and other species of parrots daily, resulting in high psittacine densities in the area. 36
Study species
The Scarlet Macaw (Ara macao macao) nests within large pre-existing tree cavities or artificial nest boxes.40,41 At the Tambopata National Reserve, females typically lay from two to four eggs from October to December. 23 The Scarlet Macaw exhibits assisted uniparental incubation, with the females solely responsible for incubation while being provisioned by the male. Highly altricial chicks hatch asynchronously after an incubation period of 24–28 days. 23 One or two chicks normally fledge per nest about 86 ± 4 days after hatching.22,23,42 If nests fail early in the breeding season, pairs sometimes produce a second clutch.
Egg loggers
We collected data on the rates and angles of egg turns using data loggers within artificial eggs (hereafter, “egg loggers”). Egg loggers were custom constructed following Shaffer 9 and were provided for this study by the Scott Shaffer laboratory. Software was developed by Geoff Bower and Alex Naimen, and 3D-printed shells were fabricated by CJW. Egg loggers consisted of a temperature thermistor, triaxial accelerometer, magnetometer, micro-SD memory card, lithium-ion battery, and white plastic 3D-printed shell. Temperatures recorded by three egg loggers placed in an incubator were valid within less than 0.5°C but units required individual calibration. The triaxial accelerometer had typical zero-g offset accuracy of ±60 mg, with functional tests indicating accuracy within 1.5° of rotation. The sensors, memory card, and battery were enclosed within the 3D-printed eggshells, which closed via a threaded coupling mechanism. These shells were designed to match the dimensions and coloration of natural Scarlet Macaw eggs and were strong enough to withstand the crushing power of their beaks. 43 Egg loggers were not intentionally weighted because the weight of an egg logger (19.8 g) was already close to the weight of a sampled Scarlet Macaw egg (23.7 g). Furthermore, the natural egg weight of this species can vary within and among clutches. 44 Temperature and positional data were logged continuously every second and minute, respectively; and data were saved to the internal memory card every hour.
Field work
Field work was conducted during two consecutive breeding seasons: October 2016 to April 2017 (the 2016 breeding season) and October 2017 to April 2018 (the 2017 breeding season). We accessed active Scarlet Macaw nests using single rope ascension techniques.45,46 To determine when the first eggs were laid, nests were climbed or checked via installed cameras every one to four days starting in October. Once the first eggs were spotted, batteries were charged, and the egg loggers were added to the clutches the following day. We deployed egg loggers in 16 nests during the 2016 breeding season and in eight nests during the 2017 breeding season. For both breeding seasons, we deployed egg loggers in a variety of nest sites, including artificial cavities made of PVC or wood as well as natural cavities. To provide a baseline for egg logger noise in the absence of incubating macaws, we deployed control egg loggers in two unoccupied nests. Although the battery could power the sensors for up to 28 days in laboratory conditions, they typically discharged much more quickly in the field. Consequently, we usually replaced them with a second egg logger approximately two weeks into the incubation period. Egg loggers were removed once the natural eggs began to hatch, and logger data was extracted from the memory card with custom MATLAB scripts provided by the Shaffer lab.
Data processing
Egg loggers recorded rotations as low as 0.03°, which are unlikely to be biologically relevant. This was demonstrated by the control egg loggers placed in the two unoccupied nests, which regularly recorded rotations less than 5°. Because egg rotations less than 5° could not be conclusively attributed to incubating hens, we defined egg turning events as egg logger rotations greater than 5°. Similarly, rotation angle thresholds of 5°, 10°, and 15° have been used in other studies to define egg turning events.2,6,9,14–17,20,24
We excluded egg turning data from three egg loggers due to logger malfunction. Two egg loggers recorded corrupted data that could not be extracted with the custom MATLAB scripts. The other egg logger recorded clearly inaccurate temperature data, with immediate and regular jumps of >10°C. We also excluded data collected by egg loggers from nests that suffered predation events. The persistence of egg loggers in nests following predation events created a biologically unrealistic scenario. Comparing egg logger data with external data sources indicated that all the egg logger sensor clocks ran slow. To maintain the temporal integrity of our data, we adjusted our datasets using adjustment factors calculated for individual egg logger sensors. For manageability, temperature data were reduced to one data point per minute prior to analysis.
We excluded data collected from nests where hens failed to adequately attend the egg loggers, which could occur if the hen failed to incubate the clutch generally or the egg logger specifically. Camera systems installed within some of these nests frequently documented the latter, showing systematic exclusion of the egg logger from the rest of the clutch during incubation. This was reflected in the data collected by these excluded egg loggers, which showed strong day-night cyclicity of temperature and very few egg turns, similarly to the data recorded by the control egg loggers. To address this, we classified hours of egg logger data as “attended” (>1 change in the temperature trend direction and >0 egg rotations) or “unattended”. We excluded datasets with <90% attended hours (N = 22 datasets). This exclusion criterion was validated by the camera system footage, which showed systematic egg logger exclusion in nests with attendance <90%, and high egg attendance during incubation has previously been reported for parrots. 47 Hatching success from the nests with excluded egg loggers was comparable to that of the other nests, indicating that inadequate attendance was specific to the egg logger, not the real clutch.
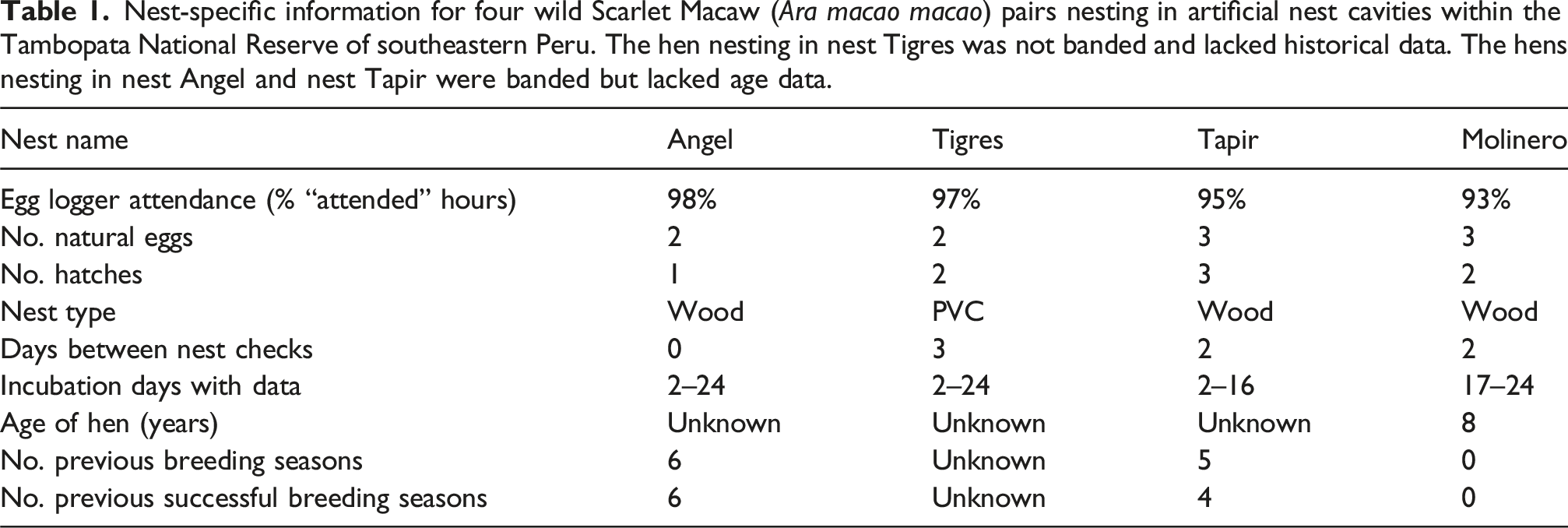
Nest-specific information for four wild Scarlet Macaw (Ara macao macao) pairs nesting in artificial nest cavities within the Tambopata National Reserve of southeastern Peru. The hen nesting in nest Tigres was not banded and lacked historical data. The hens nesting in nest Angel and nest Tapir were banded but lacked age data.
Breeding pair histories
Of the four wild Scarlet Macaw hens included in this study, three were banded and had recorded reproductive histories (Table 1). The hen that nested in nest Angel was the most experienced with six previous breeding seasons, all with successful hatches. The hen that nested in nest Tapir had five seasons of experience, four of which included successful hatches. The hen that nested in nest Molinero was a new parent, with no previous recorded breeding. No historical data was available for the hen that nested in nest Tigres.
Data analysis
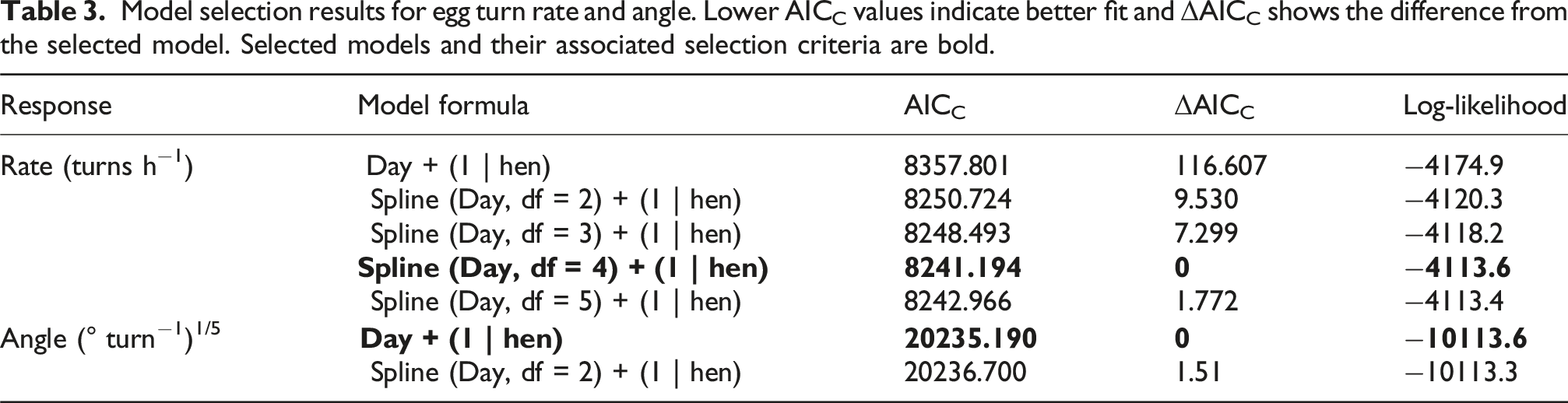
All statistical analyses were conducted in R version 4.4.1., 48 and scripts are available upon request to the corresponding author. Egg turn rate was quantified as the count of egg turning events recorded per hour of data, and egg turn angle as the magnitude of the egg turn in degrees, calculated as the minimum distance the egg logger was rotated about all three axes (pitch, roll, and yaw) using Euler angles. 9 Egg turn rate was approximately normally distributed, but egg turn angle was extremely right skewed and was made more symmetrical through a fifth root transformation before modeling. For both egg turn rate and egg turn angle, we used the glmmTMB package 49 to fit linear mixed models (LMMs) with incubation day as a fixed effect and hen as a random factor to account for repeated measures from each hen. To account for potentially nonlinear relationships between our predictor and response variables, we created LMMs with spline terms of increasing degrees of freedom to see if they reduced AICC and increased model performance. 50 To avoid overparameterization, degrees of freedom were only added until the AICC reached a local minimum. For both egg turn rate and egg turn angle, we selected the model represented by local AICC minimum as our preferred model. 51 For all model selection, we used a rotation angle threshold of 5°. Selected LMMs were checked for normality and homoscedasticity. To maximize comparability with other studies, we used the selected models to then model egg turn rate and egg turn angle with rotation angle thresholds of 10° and 15°.
To estimate central tendency for both egg turn rate and angle, we averaged the estimated marginal means (EMMs) calculated for each incubation day (2–24) using the emmeans package. 52 For egg turn angle, the average EMM was transformed back to the degree scale by raising to the fifth power. To depict variation in egg turn rate and angle over the incubation period, we plotted their EMMs and 95% confidence intervals for each day (2–24) of incubation. To test for statistically significant differences in egg turn rate and angle among the four hens, we conducted likelihood ratio tests (LRTs), where we fitted a second model without the hen random effect and tested for a significant increase in model deviation with a χ2 test. 53 To investigate the biological significance of the hen fixed effect, we estimated the standard deviation of the random effect intercept and calculated its 95% confidence interval.
Results
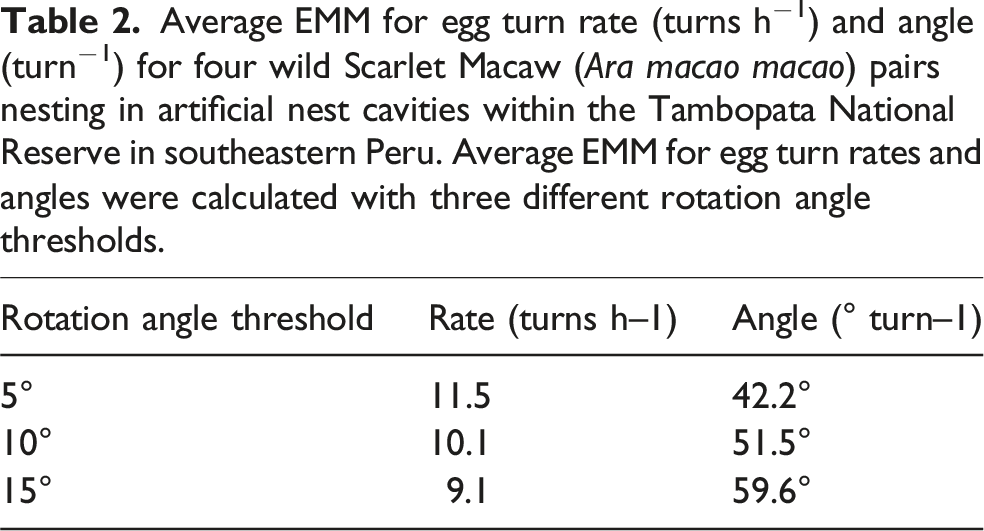
Average EMM for egg turn rate (turns h−1) and angle (turn−1) for four wild Scarlet Macaw (Ara macao macao) pairs nesting in artificial nest cavities within the Tambopata National Reserve in southeastern Peru. Average EMM for egg turn rates and angles were calculated with three different rotation angle thresholds.
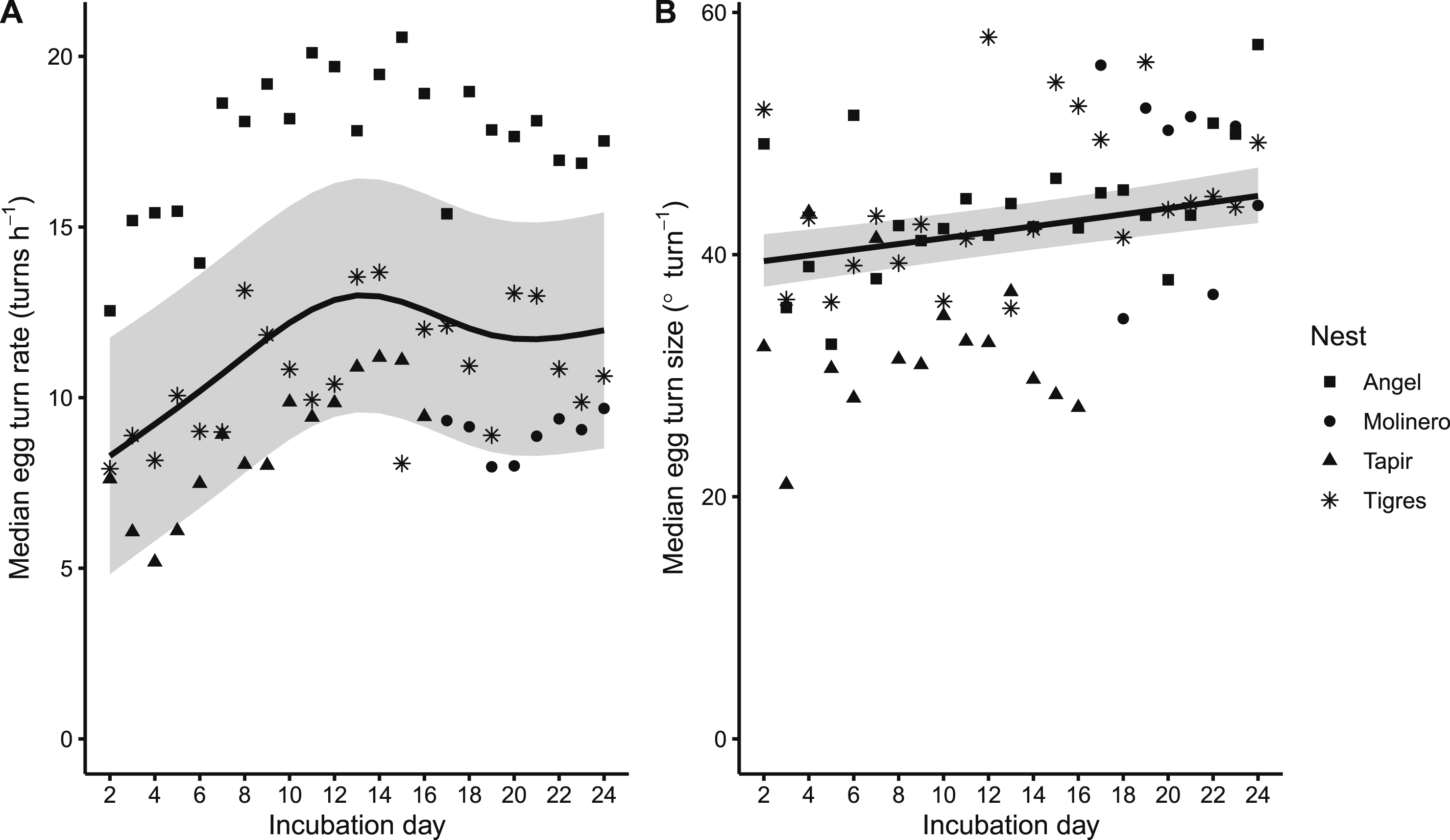
Egg turn rates (turns h−1, A) and angles (° turn−1, B) over the incubation period (days 2–24) for four wild Scarlet Macaw (Ara macao macao) pairs nesting in artificial nest boxes within the Tambopata National Reserve of southeastern Peru. Egg turns are defined with a movement angle threshold of 5°. Black lines and gray ribbons are plotted EMMs with their 95% CIs. Points are median egg turn rates and sizes for each nest during each day of incubation.
Model selection results for egg turn rate and angle. Lower AICC values indicate better fit and ΔAICC shows the difference from the selected model. Selected models and their associated selection criteria are bold.
Discussion
Average egg turn rates (turns h−1) reported from egg logger studies of other species compared to the average egg turn rates of four wild Scarlet Macaw (Ara macao macao) pairs nesting in artificial nest cavities within the Tambopata National Reserve in southeastern Peru. Average egg turn rates for the macaws were calculated with three different rotation angle threshold values (5°, 10°, and 15°) to match the threshold values used in the studies of the other species.
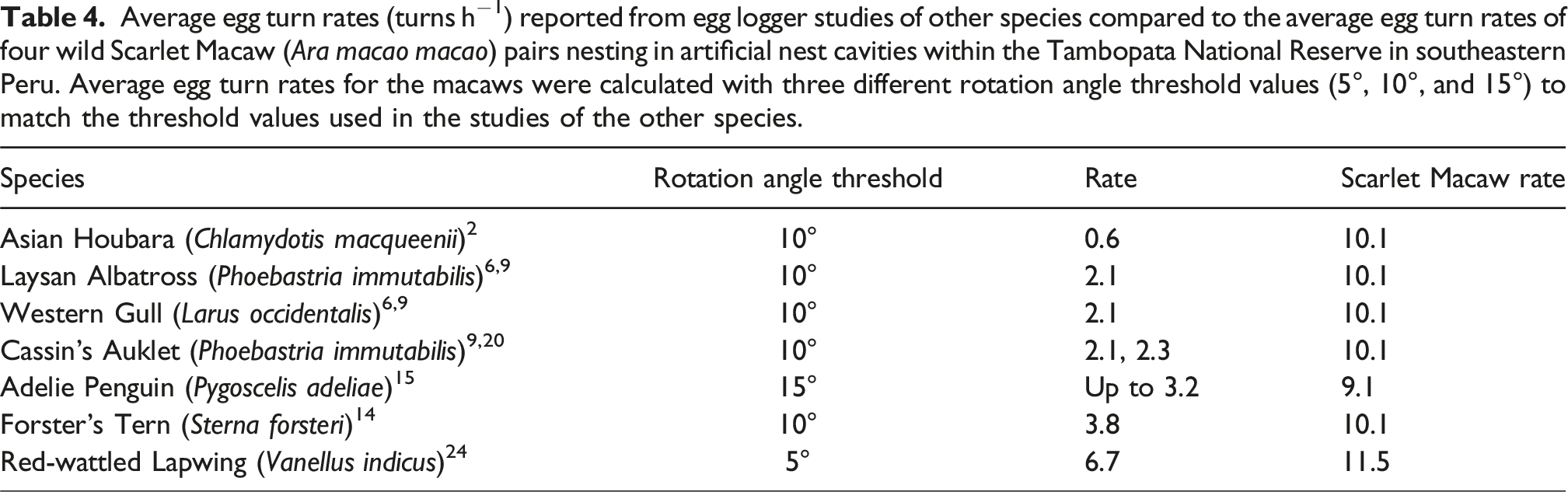
Average egg turn angles (° turn−1) reported from egg logger studies of other species compared to the average egg turn rates of four wild Scarlet Macaw (Ara macao macao) pairs nesting in artificial nest cavities within the Tambopata National Reserve in southeastern Peru. Average egg turn angles for the macaws were calculated with two different rotation angle threshold values (5° and 10°) to match the threshold values used in the studies of the other species.

Comparison of egg turn rate and angle trends from the first to second halves of the incubation period reported from egg logger studies.
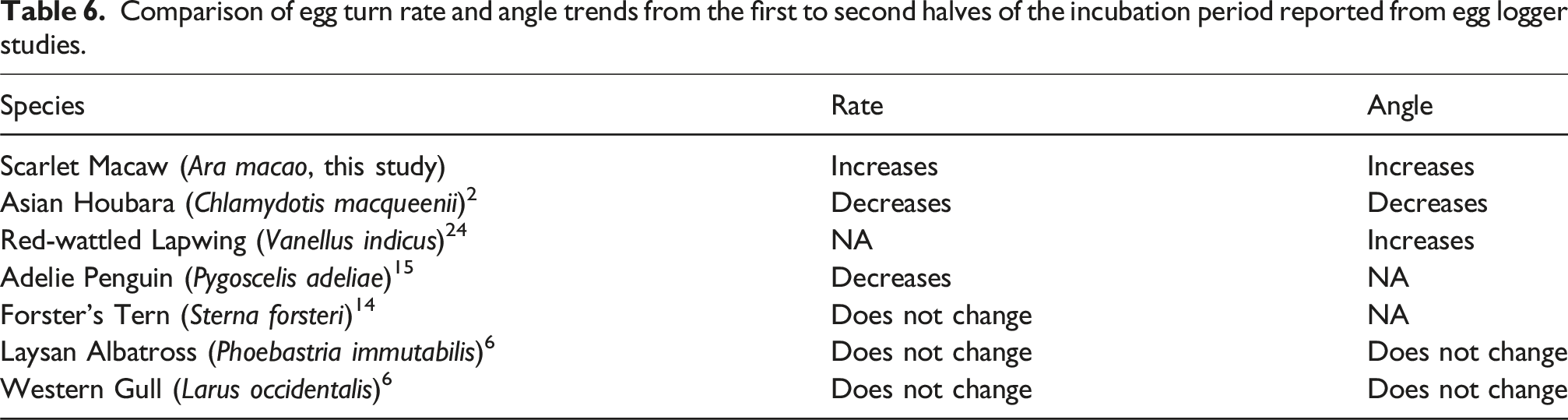
In our study, there was significant variation in egg turning among the different Scarlet Macaw hens. The hen that nested in nest Angel turned her eggs at a remarkably high rate. Interestingly, this hen also had the most recorded previous reproductive experience, but the relationship between reproductive experience and egg turning remains unclear. A study on Forster’s Terns predicted that average egg turn rate would increase with increasing reproductive experience, but found the opposite pattern. 14 Reproductive inexperience has been implicated as a cause for hatch failure in the Scarlet Macaw, 23 but it remains to be investigated if this is associated with differential egg turning specifically. A camera system installed within nest Angel showed the occupying hen to be highly active, leaving then rapidly returning to her clutch more frequently than other recorded hens. Previous studies have suggested that increasing the rate of incubation disturbances may increase the number of egg rotations caused when incubating adults move on and off the eggs. 14 Breeding Scarlet Macaws within the Tambopata National Reserve vigorously compete for nest cavities and often must defend their nest cavities from other pairs. 23 These intense nest fights likely cause incubation disturbances22,23 that may manifest in the data as increased egg turn rates.
The high rate of egg logger exclusion in this study highlights the need for researchers to reduce the noticeability of egg loggers within clutches. In this study, the quantitative addition of egg loggers to clutches may have enabled hens to distinguish and systematically exclude the egg loggers from their clutches. In other, unpublished work with parrots, researchers have replaced real eggs with egg loggers instead of simply adding egg loggers to clutches (Cornejo pers. com.). This may have resulted in better acceptance of the egg loggers, as there was no mention of systematic egg logger exclusion. However, this study was conducted with captive birds in a captive breeding center, which can affect incubation behavior. In our study, the 3D-printed egg logger shells lacked the shininess typical of macaw eggs and became increasingly conspicuous as they accumulated dirt. By the 2017 breeding season, egg logger exclusion reached levels that prevented the inclusion of any data collected that season. Future efforts to match egg logger shells and natural eggs (especially in terms of bird vision) and manage overall clutch sizes may help decrease noticeability of egg loggers and increase their acceptance.
Our study has several applications to ex-situ and in-situ conservation for threatened parrots. Artificial incubation guidelines for captive breeding programs typically suggest an egg turn rate of one turn per hour, 4 and psittacine aviculture guides similarly recommend egg turn rates ranging from five turns per day to one turn per hour.65,70 Interestingly, some aviculturists note that one turn per hour is inadequate and recommend additional turns. 71 Future studies could test if higher egg turn rates improve parrot embryonic development, hatch success, and chick quality in captive breeding programs.2,4 Effective in-situ conservation regimes for parrots typically involve nest management, which can be augmented with egg loggers. One of the egg loggers from this study was the only “egg” remaining following a predation event. The egg logger was used to maintain and validate the hen’s incubation behavior until it was ready to receive a foster chick, which would go on to fledge successfully. 72 Egg loggers may be useful to help maximize reproductive output by maintaining and validating the incubation behavior of hens when manipulating egg laying or incubation.
Footnotes
Acknowledgements
We thank the Scott Shaffer and the Shaffer lab from San Jose State University for assisting initial construction, programming, and trial deployment of loggers. We thank the field leaders Dr. Dylan Whitaker, Anna Hackinson, and Liz Villanueva Paipay; the nest camera operators Jorge Leon and Lana Austin; and Dr. Maria Belen Aguirre and Dr. Sharman Hoppes for their veterinary work. We also thank the many macaw assistants for their hard work in the field and to the staff of the Tambopata Research Center for their support. We especially thank Dr. Janice Boyd.
Ethical considerations
This research was conducted under permits from SERNANP - Peru (Resolución Jefatural de la Reserva Nacional Tambopata N022-2016-SERNANP-JEF) and the Texas A&M IACUC (AUP# 2015-0200).
Author contributions
CJW conceived the study and GV-T, DJB, CJW designed the methodology. GV-T led fieldwork, data collection, and data curation. ACB and CJW analyzed the data. ACB wrote the manuscript with input from all authors. All authors approved the final version for publication.
Funding
This work was supported by the Schubot Center for Avian Health, Amigos de las Aves USA, Phoenix Landing, Raleigh-Durham Caged Bird Society, Rainforest Expeditions, and private donations.
Declaration of conflicting interests
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Data availability statement
The data and scripts from this study are available on request to the corresponding author. The data are not publicly available due to permit restrictions from the National Service of Protected Areas of Perú (SERNANP).
