Abstract
This study monitored the temporal pattern of the diurnal foraging behaviour of yellow-wattled lapwing Vanellus malabaricus Boddaert, 1783 (Charadriiformes: Charadriidae) in different seasons (breeding and non-breeding) and habitats (uncultivated, cultivated, ponds and rivers). The focal observation method was employed to investigate the foraging behaviours of yellow-wattled lapwings. Foraging techniques showed non-significant (p = 0.85) variation in different habitat types; however, walk-halt pecking (WHP) was observed to be the most commonly utilized foraging technique across all habitats. Furthermore, foraging success varied significantly in seasons (p <0.05) as well as in habitat types (p <0.05). Due to the availability of ample food items, foraging success reached its peak in uncultivated habitats. We documented considerably high feeding rates in the uncultivated habitat in the breeding season due to the high prey abundance. Additionally, seasons and habitat both greatly affected the rate of walking. Uncultivated habitat represented the highest value of % successful attempt
Introduction
The Vanellus malabaricus, yellow-wattled lapwing (Boddaert, 1783 ), 1 is a medium-sized wader and belongs to the family Charadriidae of the avian order Charadriiformes. The term “lapwing” refers to their wavering flight and “walk-halt-walk” movement, which transforms into a “run-halt-tilt” forward-ground pricking” action during feeding. 2 They have long legs, rounded wings, a fairly large head and a short straight bill.3,4
Even though the yellow-wattled lapwing is regularly seen in a range of habitat types in the Indian subcontinent, including bare ground, dry stubbles, fallow fields and the edges of wetlands, 5 it is less reliant on aquatic water bodies. 6 This species extends from southern Sind in Pakistan and stretches throughout the Indian subcontinent up to West Bengal in North India, comprising Bangladesh. It occurs over the whole South Indian Peninsula, in arid zones in Ceylon, and sometimes in the valley of Nepal. 6
According to Jerdon, 7 they usually eat termites, beetles, and other arthropods that are entrapped on the ground. Yellow-wattled lapwings get their prey at the substrate boundary layers by collecting minute arthropods and other food items from low plant cover or from the surface. 8 They typically run swiftly, stop abruptly, glance around, and peck when foraging.
Foraging reflects behavioural activities that consist of search, pursuit, assessment and handling, which result in the final consumption of food. 9 In animals, food searching is a natural necessity, and it plays a vital function with respect to their survival, fitness and reproduction. 10 Depending on biotic (body size, age, sex, and fitness) and abiotic (temperature, humidity, light, and physicochemical qualities) conditions, many wading birds catch their food in various ways. 11
A review of the literature showed that temporal pattern has been described as behavioural changes through time in birds. 12 Temporal pattern in relation to diurnal foraging activity have been described in a few species of waders.13,14 Many insectivores birds exhibit a common pattern of foraging activity, which includes a high frequency of foraging in the early morning, a decrease in activity during midday, and a gradual increase in the late afternoon. 15
Animals decide to alter their foraging behaviour whenever there is a problem with the availability of food sources. 16 It's possible that resources aren't always accessible at all scales of time and space. 17 In shore birds, the indispensable energy, home range and available time for foraging can fluctuate.18,19 The optimal foraging theory proposes that foraging should be the outcome of spatial and temporal adjustments within an organism’s behaviour in response to non-uniform availabilities of prey in space and time. 20
Multiple variables influence habitat quality, including the availability of food, water depth, foliage cover, the presence of predators, the ease of defence, the distance from populated areas, and many other abiotic factors like temperature, humidity, light, and physicochemical qualities. 21 The habitat structure may also affect birds' foraging pattern through various sources of food items, their availability and vital controls on acquiring them. 22 Consequently, the timing of feeding and foraging in a specific habitat is critical to the lifetime strategy. 23
In this study, we looked into the temporal pattern of diurnal foraging behaviours (technique and success) of yellow-wattled lapwings in four different habitats during breeding and non-breeding seasons. In this regard, we assumed the hypothesis that there are differences in foraging behaviours (technique and success) in relation to habitats and different seasons.
Materials and methods
Study area
The temporal pattern of diurnal foraging behaviour in yellow-wattled lapwings was observed during the breeding (March to June)24,25 and non-breeding (August to November) seasons in Lucknow district (26.7863° N, 80.8987° E), Uttar Pradesh (India), in the years 2021 and 2022. Based on frequent sightings of lapwing, we randomly selected five study sites and their Global Positioning System (GPS) coordinates (Bakshi Ka Talab, BKT: 26°56'47.58"N 80°56'53.69", Post Graduate Institute, PGI: 26°44'37"N 80°57'15"E), Gosaiganj: 26°46'34.56"N 81°4'36.02"E, Malihabad: 26°50'34.11"N 80°45'38.73"E, Nigohan: 26°33'24.06"N 81°1'1.43"E) were noted down (Figure 1). On the basis of topography of the study area, the field observation was conducted in four different habitats: uncultivated, cultivated, pond and river. GIS map of the study area.
Bird observation
To observe foraging behaviours, we employed the focal observation method. 26 We used binoculars (12×42 magnifications), stopwatches and video recorders (a Nikon digital camera with a 60×zoom capability). All focal observations were conducted from a seated position, 50–100 meters away from the focal bird. While moving to a new vantage point, we waited 5 minutes for the lapwing to settle. Furthermore, we considered a bird observation session of 5-minutes with an interval of 15-minutes. Hence, the total observation session of yellow-wattled lapwings constituted 4320-minutes (15min/hour, 90 min/day, 270 min/month, 2160 min/year). In each habitat, focal observations were carried out from 6:00 am to 18:00 pm in triplicates. Temporal pattern of diurnal foraging behaviour was observed during a random walk in the study sites, which typically began one or two hours after sunrise and lasted until mid-day. Lapwings were detected by sounds or by systematic scanning of the study sites using binoculars. Overall, we recorded the videos of 40-focal individuals of lapwings at different study sites. All foraging behaviour was estimated either by observing them directly at the site or indirectly by the observation of recorded video. The gregarious behaviour of lapwings, however, allowed us to track several individuals at once, which further reduced the possibility of sampling the same individual more than once. Since the study sites were homogeneous, which means all four habitat types were found at each study site, all the data were pooled together.
Data collection
In this study, foraging behaviour consisted of two components, foraging technique and success. Pecking and probing were the main foraging techniques in yellow-wattled lapwings. Pecking was defined as the bill penetrating the substrate for less than one-quarter of its total length, whereas probing was defined as the bill penetrating the substrate for more than one-quarter of its total length. 27 Foraging techniques were further re-categorized as walk-halt (WH), walk-halt-pecking (WHP), walk-halt-multiple pecks (WHMP), walk-halt-probe (WHPro) and walk-halt-multiple probes (WHMPro). 27
Moreover, foraging success was further classified into four components: (1) walking rate: steps per minute taken by the lapwing during the observation; (2) foraging or feeding rate: total number of feeding attempts (pecks or probes) made by individual birds per minute; (3) foraging success rate: number of prey items taken per bird per minute; and (4) percent attempt success: calculated as the number of feeding attempts that resulted in prey consumption per lapwing per minute divided by the total number of feeding attempts made per bird per minute and then multiplied by 100 (with the help of original data).
The video sequences were analysed in detail (in slow motion) on a computer using the VLC 2.2.6 media player. We recorded foraging parameters such as pecks or probe/minute and the number of prey items captured/minutes. A digital stopwatch and hand tally counter were used to estimate the number of paces, probes, captures and prey successes. 28 To quantify temporal variation in relation to diurnal foraging behaviours (foraging techniques and success), we calculated the average hours per day spent in each activity for each yellow-wattled lapwings. 29 The frequency (Mean±SE) of foraging techniques and success was calculated in different habitats across all study sites.
Statistical analysis
The data were checked for homogeneity and normality using Kolmogorov–Smirnov tests and Levene’s test, respectively. Once the normality was tested, the data were statistically analysed by the parametric test, while non-parametric test was used for the data that did not meet the assumptions. Once normality was checked, we employed parametric tests. In this field investigation, we utilized one-way ANOVA (parametric) followed by tukey’s post hoc test to statistically analysed foraging behaviour (foraging techniques and success) in different habitats. In addition, we used t-test (parametric) to examine foraging behaviour (foraging techniques and success) in different seasons (breeding and non-breeding).
Multifactorial ANOVA (MANOVA) was also applied to find out the effects of “time of the day” on foraging behaviours with respect to frequency of foraging techniques and success in different habitats and seasons. The statistical analysis was performed by SPSS (version 16.0).
Results
Foraging behaviour (technique and success)
During the field survey, 92-yellow-wattled lapwing individuals were monitored in five different study sites that actively engaged in foraging. We documented that there was no significant (F=24,94, df=3, p=0.85) difference in foraging techniques across all habitats (Figure 2). Similarly, we found that there was no significant (t=1.08, p=0.73), variation observed in foraging techniques within different seasons (Figure 2). Estimation of foraging techniques (mean ± S.E.) of yellow-wattled lapwing in different habitats and seasons: (a) uncultivated; (b) cultivated; (c) pond; and (d) river. WH= walk-halt; WHP = walk-halt-peck; WHMP = walk-halt-multiple pecks; WHPro = walk-halt-probe; WHMPro = walk-halt-multiple probes.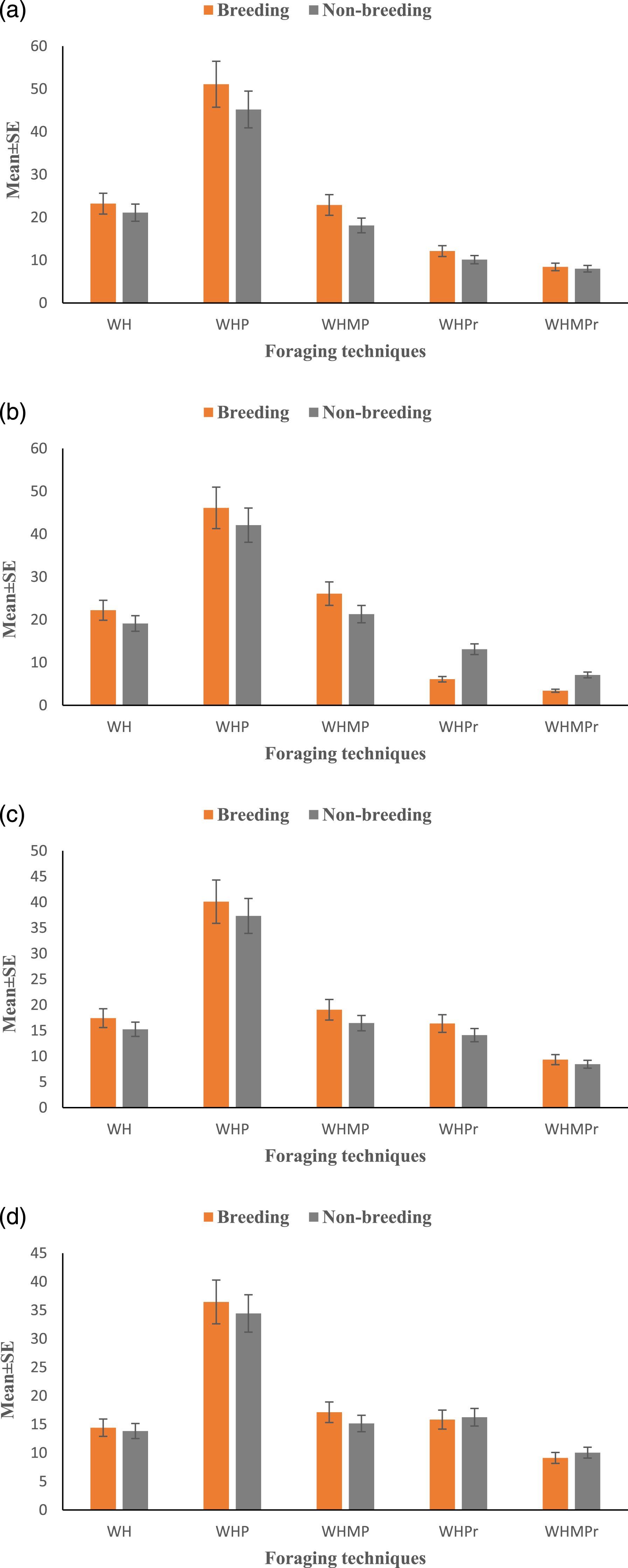
In nutshell, walk halt pecking (WHP) was observed to be the most utilized foraging technique in yellow-wattled lapwings. Moreover, WHPro and WHMPro techniques were prominently displayed in aquatic habitats (pond and river) (Figure 2).
Foraging success (mean ± S.E.) of yellow-wattled lapwing in different habitats and seasons during the study period (2019 and 2021).

Furthermore, the highest feeding rates were recorded during the breeding season (t = 10.78, p <0.05) and in uncultivated habitat (F = 26.92, df = 3, p <0.05). Moreover, we estimated the highest feeding rate (8.15 ± 3.78 pecks or probes/minute) in uncultivated habitat during the breeding season, while the lowest (1.45 ± 2.33 pecks or probes/minute) in river habitat during the non-breeding season (Table 1).
Additionally, we found that the foraging success rate was statistically significant across all habitat (F = 32.07, df = 3, p <0.05) and all seasons (t = 19.61.08, p <0.05) (Table 1). We recorded the highest foraging success rate (5.92 ± 2.44 prey captured/ minute) in uncultivated habitat during breeding season and the lowest (0.84 ± 1.85 prey captured/minute) in river habitat during non-breeding season (Table 1).
Uncultivated habitat represented significantly (F = 48.25, df = 3, p <0.05) highest value of % successful attempt, while the lowest value of % successful attempt was recorded in river habitat (Table 1). Moreover, the percentage of successful attempts was significant (t = 22.13, p <0.05) in the breading season as compared to the non-breeding season (Table 1).
Temporal pattern
Results of MANOVA showed that the factor “time of the day” produced a significant effect on the daytime foraging activity of the yellow-wattled lapwing with respect to frequency of technique (F = 38.32, df = 4, p < 0.05) and success (F = 46.12, df = 3, p < 0.001) (Figure 3). MANOVA test exhibited that time of day also has a significant effect on foraging activity in reference to seasons (F = 42.22, df = 1, p <0.05) and different habitat types (F = 49.31, df = 3, p <0.05). Temporal pattern of daytime foraging behaviour (technique and success) of yellow- wattled lapwings in different seasons (breeding and non-breeding) and habitat types ((a) uncultivated; (b) cultivated; (c) pond; and (d) river).
Across all habitat categories, a bimodal pattern in daytime foraging activity with reference to frequency of foraging techniques (WH, WHP, WHMP, WHPro and WHMPro) as well as success (movement rate, feeding rate and success rate) was observed during the breeding and non-breeding seasons (Figure 3). One peak of modality was recorded in the morning (09:00-11:00 am) and another peak was evident in the evening (15:00-18:00 pm) for all foraging techniques as well as success during breeding and non-breeding seasons in different habitat categories (Figure 3). In a nutshell, out of four habitats, uncultivated habitats showed the highest peak of modality in the morning (09:00-11:00 am) and evening (15:00-18:00 pm).
Discussion
In this study, a wading bird species, Vanellus malabaricus was selected to monitor their diurnal temporal pattern of foraging behaviour (technique and success) in relation to seasons and habitat types. Based on immediate or future necessity, foraging is the simple activity of collecting food material from natural habitats. Additionally, different techniques of avifauna depend on their morphology, which allows a species to take advantage of unique food items. 22
Foraging techniques
Generally, foraging techniques in wading bird species can be broadly categorized into two types: probing and pecking applicable for tactile and visual foraging, respectively. 30 Pecking conceivably involves a superficial peck (a single bit of the bill tip into the sediment), a rapid peck (an incessant peck over the surface) and an unsuccessful peck (a peck arrested in mid-air before contact with the ground). 31 Furthermore, probing comprises insertion of the bill into the sediment, permitting the capture of invertebrates that live under the sediment surface (infauna prey types)31,27
In this study, we found that yellow-wattled lapwings adopted different foraging techniques and consumed invertebrates’ prey at dissimilar rates in different habitats. In this species, the specific habitat types were associated with different foraging techniques and success due to temporally varying food resources. A similar observation was documented in river lapwing (Vanellus duvaucelii). 27
In both seasons, WHP was the most frequently used technique for yellow-wattled lapwings across all habitat categories. This observation coincides with previous field research in river lapwings. 27 However, WHP, WHMP and WH techniques were also prominent in uncultivated habitats as compared to other habitat categories in the breeding seasons. Being insectivore, 32 they preferably capture surface-active prey (termites, beetles, and other arthropods). 33 When deciding where to go foraging, waders assess benefits and expenses. 34
The difference in peck rates between wet features and marsh habitats could reflect differences in prey encounter rates, such as whether prey may be present but difficult to locate in vegetation or whether the vegetation structure on the marsh may constrain foraging efficiency.35,36 Waders make their foraging decisions based on rewards and costs. 34 Foraging birds engage in rapid pecking behaviours that seem to depend more on tactile cues than visual cues, 30 whereas during slow pecking, birds move at a consistent pace over the narrowed area. 37 The previous study suggested that during rapid pecking, birds would focus their efforts on a large area. 37 To determine where birds feed within fields, elements like sward shape, soil moisture and food richness will be crucial. 36 Furthermore, probing can cost more than pecking in terms of time and energy 38 or there is a probable risk of bill attrition due to friction with sediment particles. 39
Our results have indicated that probing techniques (WHPro and WHMPro) were most frequently used in aquatic habitats (river and pond) throughout the years. During the non-breeding period, WHPro, WHMPro were the most frequently used techniques in yellow-wattled lapwing. This could have been observed due to the presence of numerous aquatic insects, particularly in rainy season. Similar findings were displayed in curlew sandpipers 40 and river lapwing. 27 This field study revealed that pecking was the most frequent foraging style in yellow-wattled lapwings, but probing technique were also employed in other habitats occasionally since they fed on invertebrates below the sediment surface. A group of researchers has reported a similar pattern of foraging techniques in common redshank. 41
Foraging success
Our results revealed that, during breeding season, uncultivated habitats had the highest foraging success rate, while river habitats had the lowest in non-breeding. Similar trends were reported in Golden Plovers (Pluvialis apricaria) and Lapwings (Vanellus vanellus). 42 The optimal foraging theory suggests that animals decide and select optimal (efficient and beneficial) patches of habitat for foraging to maximize the cost benefit ratio. 43 Additionally, the relationship between prey abundance and foraging rates of wading birds has displayed positive indications in many studies. 44 However, variations in food availability change foraging behaviour in birds. 27
The findings of this study indicated that seasons and habitats have both greatly affected the rate of walking. The earlier study suggested that the increase in the rate of walking resulted in an increase in prey search. 45 Waders have been identified for scooping tiny invertebrates out of mud or exposed soil while feeding on the ground 46 despite the fact that their biology depends greatly on terrestrial movement. 46 Since yellow-wattled lapwings have comparatively short legs, their rate of walking is incumbered by long, dense vegetation. 47 Additionally, high foliage could make it difficult for the parents to keep an eye on their chicks.
In this field investigation, we documented the highest feeding rates in uncultivated habitat during the breeding season. In bare or uncultivated areas, soil may provide abundant surface-active prey. 36 This field observation coincides with a previous study. 27 The earlier study has shown that short swards may improve foraging rates by increasing food accessibility and reducing predation risk, as well as mobility costs.36,48 Our results showed that feeding rate was significant in breeding season as compared to non-breeding season. Non-breeding season consisted of the rainy and winter season; hence, low temperatures might have reduced the invertebrate’s abundance, 49 consequently reducing foraging time. 50
Results revealed that the foraging success rate was highest in uncultivated habitat during breeding seasons. A similar observation was noticed in river lapwing (Vanellus duvaucelii). 27 Studying the foraging success of birds is preferred to direct measurements of food abundance as a method of evaluating habitat suitability as it reflects the resources available to foragers. 51 There are a number of factors, such as foraging surface area, different prey assemblages between habitats, prey availability, mobility and vigilance rates, that may explain this pattern 36 Our field research has indicated a low foraging success rate in river habitat, mainly in the non-breeding season, due to the fact this season-habitat combination was not used in yellow-wattled lapwing. 52
Foraging success may be influenced by many known factors such as the availability of prey items and geographical features of the habitat. 53 Moreover, many other factors, like social interactions, 54 predation 55 and disturbance by humans 56 may also affect foraging success. Foraging costs and the provision of food may have good impact on the selection of foraging sites when there is scarcity of food items. 57 It has been widely documented that shorebirds select those habitats to feed, where they experience the greatest foraging success. 58 It has been observed that the availability of prey items is essential for determining the selection of habitats59,53 and foraging success 60
Temporal pattern
Across all habitats, the temporal pattern shows that the time-of-the-day (clock hours) had a significant impact on daytime foraging activity concerning frequency of foraging technique and success during the breeding and non-breeding seasons. Similar trends were recorded in the Indian pond heron (Ardeola grayii) 22 and in foliage-gleaning passerines. 61
One peak of modality was registered in the morning and another peak was evident in the evening, showing a bimodal pattern during daytime foraging activity in different habitats and seasons. The continued existence of a decline in foraging activity around noon is ostensibly unrelated to food supply and may be caused by thermoregulatory issues or the fact that the birds are excessively foraging and becoming satiated at this time. 62 The afternoon's rise in temperature may be associated with maintenance behaviours and inactivity, which may have an impact on foraging in open spaces. 63 This finding coincides with earlier field researchers22,14,15 in different species of waterbirds. Furthermore, Datta et al. 64 have suggested that foraging was predominant in the morning, afternoon and midnight, while resting during mid-day.
Based on the findings of several studies,65,66 spatial and temporal differences in the availability and quality of prey may be influential sources of variation in foraging behaviour. Thus, alterations in natural factors like availability of food items, day-night cycle and temperature in the study sites can play a vital role in determining the temporal pattern of feeding activity of wading birds.67,68
Footnotes
Acknowledgments
The authors would like to thank the Head, Department of Zoology, University of Lucknow, for providing facilities and administrative support. Prateek is thankful to the CSIR-UGC, New Delhi, for the UGC-Junior Research Fellowship [Ref. No. 211610126959]. Prateek is thankful to Anshu Mishra, Ph.D. research scholar, who actively helped during the field visits and to the local people of the study area who have given secondary information about behavioural aspects and the occurrence of yellow-wattled lapwing. Finally, the authors are also grateful to both reviewers for their valuable suggestions, which helped in the improvisation of the manuscript.
Author’s contribution
Prateek (conceptualization, performed field survey, data collection, statistical analysis, and writing), HM (data collection and interpretation), VK (data collection and interpretation), AK (conceptualization, planning research and interpretation).
Declarations of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
