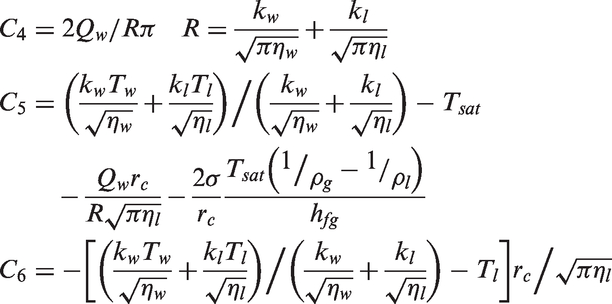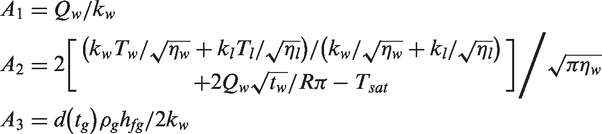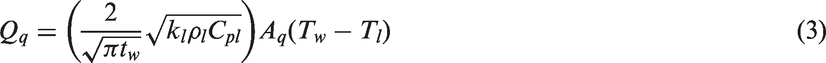Abstract
The main focus in the analysis of pool or flow boiling in saturated or subcooled conditions is the basic understanding of the phase change process through the heat transfer and wall heat flux partitioning at the heated wall and the two-phase bubble behaviours in the bulk liquid as they migrate away from the heated wall. This paper reviews the work in this rapid developing area with special reference to modelling nucleate boiling of cryogenic liquids in the context of computational fluid dynamics and associated theoretical developments. The partitioning of the wall heat flux at the heated wall into three components – single-phase convection, transient conduction and evaporation – remains the most popular mechanistic approach in predicting the heat transfer process during boiling. Nevertheless, the respective wall heat flux components generally require the determination of the active nucleation site density, bubble departure diameter and nucleation frequency, which are crucial to the proper prediction of the heat transfer process. Numerous empirical correlations presented in this paper have been developed to ascertain these three important parameters with some degree of success. Albeit the simplicity of empirical correlations, they remain applicable to only a narrow range of flow conditions. In order to extend the wall heat flux partitioning approach to a wider range of flow conditions, the fractal model proposed for the active nucleation site density, force balance model for bubble departing from the cavity and bubble lifting off from the heated wall and evaluation of nucleation frequency based on fundamental theory depict the many enhancements that can improve the mechanistic model predictions. The macroscopic consideration of the two-phase boiling in the bulk liquid via the two-fluid model represents the most effective continuum approach in predicting the volume fraction and velocity distributions of each phase. Nevertheless, the interfacial mass, momentum and energy exchange terms that appear in the transport equations generally require the determination of the Sauter mean diameter or interfacial area concentration, which strongly governs the fluid flow and heat transfer in the bulk liquid. In order to accommodate the dynamically changing bubble sizes that are prevalent in the bulk liquid, the mechanistic approach based on the population balance model allows the appropriate prediction of local distributions of Sauter mean diameter or interfacial area concentration, which in turn can improve the predictions of the interfacial mass, momentum and energy exchanges that occur across the interface between the phases. Need for further developments are discussed.
Keywords
Introduction
Cryogenic liquids are encountered in many industries. For the propulsion and thermal management for space systems, liquid hydrogen and liquid oxygen are usually used as fuel for RS-25 and J-2X rocket engines. 1 In the field of medical applications, liquid nitrogen and liquid helium are utilised as coolants for superconductors such as can be found in commercial MRI, MRT, NMRI and Large Hadron Collider.2,3 Natural gas, which is considered to be one of the clean and largest energy sources, requires its liquefied form to be stored and transported economically and safely in tanks and through pipelines in the oil and gas industries. Nowadays, all of the aforementioned technological developments have predicated an increased interest in the better understanding of the boiling of cryogenic liquids. It is apparent that boiling of ordinary liquids is rather different from boiling of cryogenic liquids. For an ordinary liquid such as water, it can remain as a liquid within a range of about 100 K. Nevertheless, cryogenic liquids only remain in their liquid form within a much narrower atmospheric liquid range (from melting point to boiling point at atmospheric pressure) of about 20 K (e.g. He – 6 K, H2 – 7 K, N2 – 14 K, liquefied natural gas – 20 K). Handling cryogenic liquids thus presents many challenges during the operations of two-phase cryogenic systems. For the case of the transportation of cryogenic liquids, small bubbles that may be generated from the boiling of cryogenic liquids due to the presence of inevitable heat leaks along the pipe can merge or coalesce to form larger bubbles and possibly reaching the same diameter of the pipe during flow boiling. These so-called Taylor bubbles can subsequently cause the blockage of pipelines which may result in the destruction of the pipes and valves as well as possible cavitation erosion to the cryogenic systems. Also, heat absorption from the surroundings into the storage tanks of liquefied natural gas may initiate the boiling off of liquefied natural gas during pool boiling. Such situation is undesirable since additional gas generation can dramatically increase the pressure within these storage tanks being already at their pressurised state.
Pool boiling or flow boiling is essentially a phase change process in which vapour bubbles are formed on a heated wall. The main difference between pool boiling and flow boiling is that the former takes place from a stagnant liquid on top of a submerged heated surface in a reservoir of liquid while the latter is produced through heating the liquid through conduits such as tubes or pipes. There are essentially different modes and regimes of boiling that can be observed in pool boiling and flow boiling.
In pool boiling, the different modes and regimes of boiling can be characterised by a typical saturated boiling curve depicted in Figure 1, which illustrates the relationship between the heat flux and the wall superheat temperature. As the heat flux and wall superheat temperature increases, nucleate boiling occurs at point a, which denotes the onset of nucleate boiling (ONB). The detachment of vapour bubbles from the heated surface is observed to be of spherical shape bubbles between points a and b but of slug and column shape bubbles between points b and c in the nucleate boiling regime. The heat flux finally reaches point c, which denotes the critical heat flux (CHF). After point c, the boiling process proceeds to transition boiling. The heat flux decreases as the wall superheat increases to point d, the Leidenfrost point, which signifies the minimum wall temperature required for film boiling.
Typical pool boiling curve and flow patterns.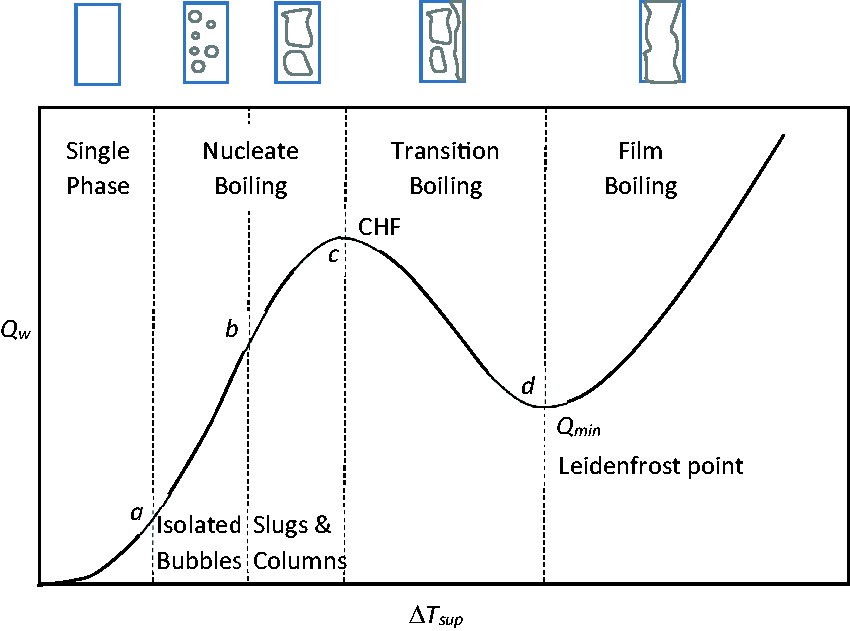
In flow boiling, there are, however, two distinct regimes, known as subcooled and saturated nucleate boiling, depending on the bulk temperature of the liquid moving past the heating surface. Figure 2 shows the evolution of the two-phase flow in a vertical heated pipe. During subcooled boiling, the temperature of the heating surface is above the saturation temperature, ebullition occurs in the immediate vicinity of the heating surface. Bubbles are produced and attached at the heating surface. As the bulk liquid is continually heated downstream, saturated nucleate boiling occurs with bubbles beginning to detach from the heating surface into the liquid stream. Further downstream, complex topologies of the two-phase flows persist throughout the entire vertical heated pipe such as flow transition regimes from bubbly to slug/churn to annular and disperse flows.
Evolution of the steam–water flow in a vertical heated pipe.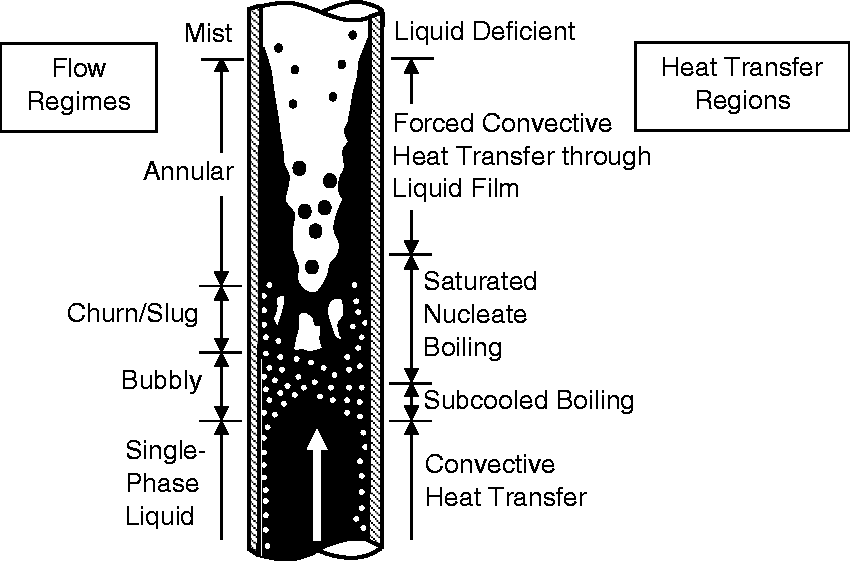
Based on the aforementioned observations prevalent in nucleate pool boiling or subcooled and saturated nucleate boiling flow of cryogenic liquids, it is clear that considerations for modelling requires the boiling phenomena to be predicted not only at the heating wall but also away from the heating wall. 4 In the framework of computational fluid dynamics (CFD), modelling subcooled or saturated nucleate boiling can be broadly classified into two categories: (i) heat transfer mechanistic modelling at the heating wall and (ii) two-phase flow and bubble behaviours in the bulk liquid away from the heating wall.
In general, the first category encompasses the development of suitable models to predict the heat transfer at the heated wall via partitioning the wall heat flux. On the basis of the review by Warrier and Dhir, 5 there are three approaches whereby the heat transfer rate can be determined. The first approach employs empirical correlations to predict the total wall heat flux given the wall superheat temperature in regions covering single-phase convection, partial nucleate boiling and fully developed boiling.6–9 The second approach considered utilising appropriate correlations to predict the different heat transfer mechanisms being considered in the wall heat flux partitioning and the fraction of the total heat flux that is transferred through each of these mechanisms.10,11 The third approach, which is the primary focus in this review, adopts the mechanistic modelling of the heat transfer mechanisms occurring during boiling, which can be readily employed for both the prediction of the wall heat flux as well as the partitioning of the wall heat flux between the liquid and gas phases. Almost all CFD investigations on flow boiling are mainly based on the use of these mechanistic models.12–14 The model partitions the wall heat flux into three heat flux components: single-phase convection, transient conduction and evaporation.
The second category involves the treatment of the fluid flow in the bulked liquid phase. Based on the interpenetrating media framework, the two-fluid model is considered to be the most detailed and accurate macroscopic formulation to resolve the thermal and hydrodynamic characteristics of the two-phase systems. In the so-called Eulerian–Eulerian approach, the field equations in the two-fluid model comprise two sets of conservation equations of mass, momentum and energy. One set is dedicated to resolve the gas phase while the other is solved for the liquid phase. Through an ensemble average of the transport equations, interphase exchange terms appear in the averaged equations, which represent the mass, momentum and energy transfers through the interface between the phases. These interphase exchange terms determine the rate of phase changes as well as mechanical and thermal non-equilibrium between phases. Most important of all, these terms provide the necessary closure to the two-fluid model formulation.
The prediction of local bubble sizes in cryogenic liquids is also expected to be strongly influenced by the complex bubble behaviours near the heated wall. In essence, complex mechanistic behaviours of bubble coalescence and bubble collapse due to condensation in subcooled nucleate boiling exist during the boiling of cryogenic liquids.15,16 Physical insights strongly indicate that the numerical solutions of two-phase cryogenic systems can be significantly improved through consideration of the bubble mechanistic behaviours in determining the local bubble size such as the Sauter mean bubble diameter, which appears prominently in the interphase exchange terms of mass, momentum and energy. Because of the importance of accurately determining the Sauter mean bubble diameter, which strongly governs the inter-phase heat and mass transfer through the interfacial area as well as the momentum drag, the potential to extend the modelling of boiling of cryogenic liquids in order to better predict the non-uniform bubble size distribution via the population balance approach17,18 is of enormous significance.
Description of the various models or correlations developed for the prediction of heat transfer and wall heat flux partitioning along with the formulation of governing equations for boiling of cryogenic liquids and population balance model is treated in the following sections.
Wall heat flux partition model
According to Kurul and Podowski
12
and Podowski,
13
the mechanistic modelling of local near-wall heat transfer applicable to saturated and subcooled boiling can be accounted based on partitioning the total wall heat flux into three components
Alternatively, Benjamin and Balakrishnan
19
have considered the heat to be removed from the heated surface by the boiling liquid according to
The difference in partitioning the total wall heat flux through this particular approach is the consideration of the volume of microlayer evaporated Vl which is given by
Closure relations
Empirical correlations for active nucleation site density.

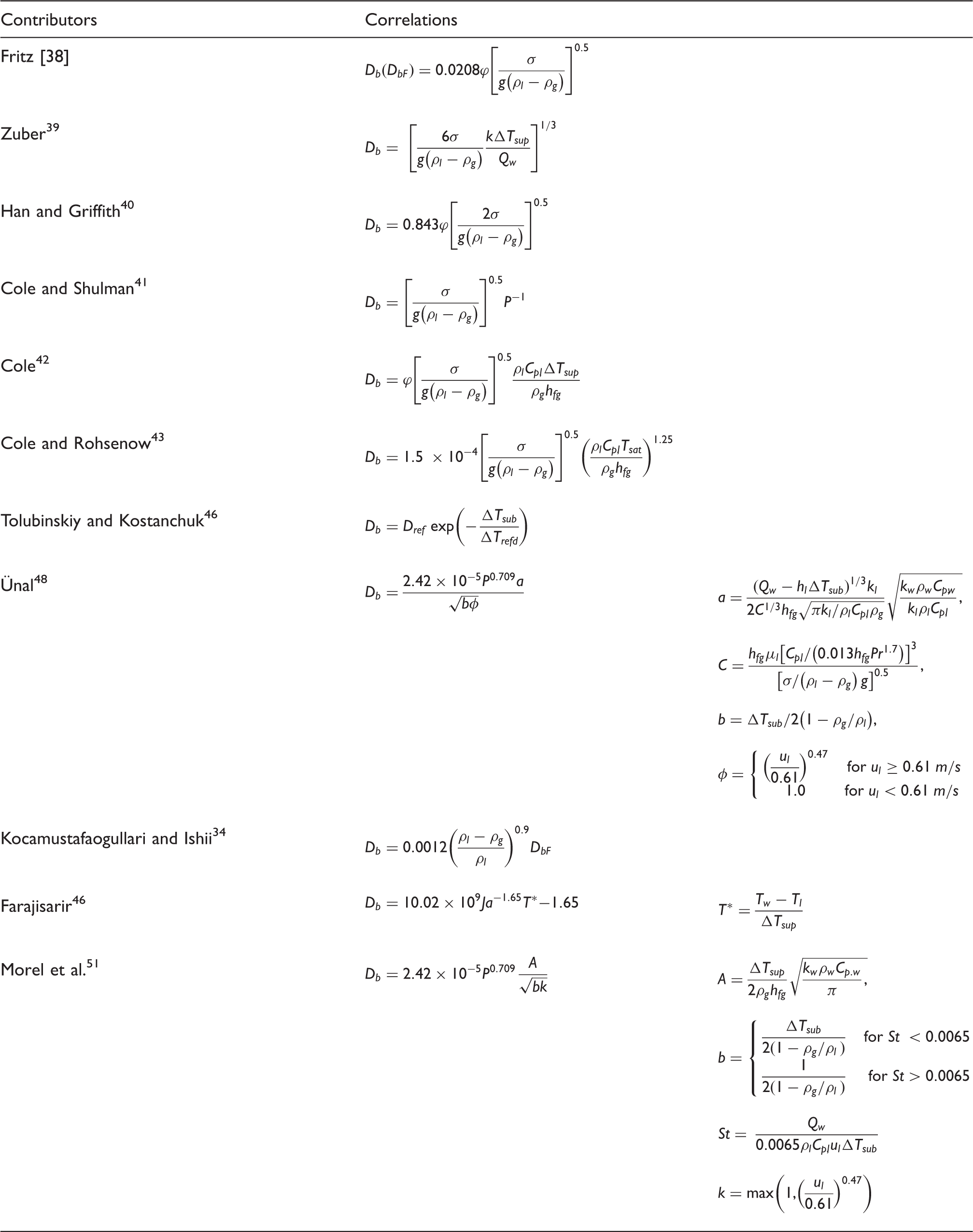
Empirical correlations for bubble departure diameter.
Empirical correlations for nucleation frequency.
Active nucleation site density
At the heated surface, not all the nucleation sites will be activated as the temperature of the surface exceeds the saturation liquid temperature at the local pressure. For nucleation sites to be activated, they are governed by the distributions of cavities on the wall surface, heater and liquid properties and contact angle between liquid and the wall. Active nucleation site density plays an important role in the evaluation of the heat flux components of the wall heat flux partition model. A large amount of research has been carried out to correlate suitable relationships of the active nucleation site density such as shown in Table 1.
Gaertner and Westwater 20 have formulated the active nucleation site density based on dissolution of nickel salts in water and observed the deposition on the copper-heater surface during boiling. An estimate of the active nucleation site density was obtained by counting the number of holes in the deposited nickel layer. The correlation for the active nucleation site density is expressed as a function of the wall heat flux. In their investigation, the exponent m matched the experimental data with a value of 0.48. Hsu and Graham 21 further reported that the range of the exponent m varies between 0.48 and 1. Westwater and Kirby 22 concluded that the range of the exponent m is 0.73 for glass and between 0.66 and 0.5 for metal. This demonstrated that the rate of activation was fairly steady regardless of the finish of the heated a wide range of cavity sizes exists.
Johov
23
related, however, the active nucleation site density to the critical cavity diameter (Dc) of the activated sites on the heating surface. For a pressure of 1 bar, the exponent m and C are taken to be 3 and
Cornwell and Brown 26 performed experiments for the pool boiling of saturated water on a copper surface at 1 bar. Using electron microscope, a systematic analysis was carried out to determine the active nucleation site density. They have correlated the active nucleation site density as a function of the wall superheat temperature (ΔTsup) with a value of 4.5 for the exponent m. The functional dependence on the local wall superheat temperature was justified by assuming that only conical cavities were present on the heated surface and before any nucleation could occur, vapour are required to be trapped in the cavities.
In a similar dependence on the wall superheat temperature, Lemmert and Chawla 27 investigated the active nucleation site density for experiments on pool boiling of saturated water with an additional consideration of a constant n. Končar et al. 28 have proposed to adopt values of n and m as 185 and 1.805 while Kurul and Podowski 12 suggested values for n and m as 210 and 1.805.
Recently, Krepper et al.29,30 proposed that the active nucleation site density has a strong influence on the wall superheat temperature, but has a small influence on the gas volume fraction and barely any influence on the liquid temperature. They suggested a similar correlation to the active nucleation site density correlation of Lemmert and Chawla
27
with additional consideration of the reference temperature (
Kirichenko et al.
33
proposed a correlation for the active nucleation site density for the nucleate boiling of cryogenic liquids. He has shown that the correlation has a dependency on not only the wall superheat temperature such has those proposed by the investigations above but also include the surface tension. The values of n and m depend on the pressure of the system:
Kocamustafaogullari and Ishii
34
performed a parametric study to correlate the active nucleation site density for the existing data of pool boiling in the literature as a function of bubble departure diameter as well as the surface tension. They have also extended the correlation to forced convection nucleate boiling by the use of an effective superheat (
Wang and Dhir
35
performed a systematic study of the effect of wettability on the density of active nucleation sites. Based on the pool boiling of water at 1 bar on vertical copper surfaces, the contact angle (ϕ) was varied from 18° to 90° by controlling the degree of oxidation of the surface. The shape of the cavities and the cumulative number density of them were obtained with an electron microscope, while the number of active sites was determined from still pictures. Hence, they correlated the active nucleation site density as a function of the contact angle as well as the critical cavity diameter. It should be noted that the correlation is valid for
Benjamin and Balakrishnan
19
investigated the nucleation site density during nucleate pool boiling through experiments on saturated pure liquids at low to moderate heat fluxes. The effect of surface–liquid interaction during the boiling phenomena was investigated on the nucleation sites. Different working mediums were used namely distilled water, carbon tetrachloride, n-hexane and acetone as liquid with the heated surface made of stainless steel and aluminium with different surface finishes. The active nucleation site density has been correlated as a function of the Prandtl number (⪻), surface–liquid interaction parameter (γ), dimensionless surface roughness parameter (ω) and the wall superheat temperature. For the dimensionless surface roughness parameter, Ra is the arithmetic average roughness which is defined as the average values of the peaks and valleys on the surface. The correlation is valid for
Hibiki and Ishii
36
mechanistically modelled the active nucleation site density by accounting the size and cone angle distributions of cavities existing on the heated surface. The active nucleation site density was correlated a function of the critical cavity diameter, contact angle and wall superheat temperature. This correlation gave fairly good predictions over the flow conditions:
Basu et al.
37
conducted experiments of flow boiling at subcooled conditions using a nine-rod (zircalloy-4) bundle and a flat plate with a copper surface to correlate the partitioning of the heat flux at the heated surface. Using a CCD camera, individual activated nucleation sites were manually accounted. The active nucleation site density was correlated with a dependence on the contact angle and wall superheat temperature. The range of parameters covered for the flat plate test surface were:
Bubble departure diameter
Most empirical correlations of bubbles departure diameter have been formulated as a function of the contact angle, Jacob number or thermo-hydraulic parameters in consideration of the particular boiling process. Table 2 depicts the various correlations for the bubble departure diameter.
Fritz 38 considered the static equilibrium between the adhesive and buoyancy forces acting on the bubble in order to predict the bubble departure diameter whereby the contact angle has been considered.
Zuber 39 assumed the existence of a thin superheated thermal layer near the surface for the occurrence of bubble growth. The correlation can be expressed as a function of the wall superheat temperature and wall heat flux.
Han and Griffith 40 derived a modified model based on existing bubble growth model. The bubble departure diameter could be obtained through the consideration of the bubble growth derivative set to zero, which has a similar form to the correlation of Fritz. 38
Cole and Shulman 41 proposed that the bubble departure diameter being inversely proportional to the absolute pressure of the boiling system. Subsequently, Cole 42 proposed another correlation as a function of the contact angle along with the consideration of the wall superheat temperature. Cole and Rohsenow 43 modified the correlation of Cole 42 by replacing the wall superheat temperature with a critical temperature at saturation.
Kocamustafaogullari and Ishii 34 correlated the bubble departure diameter with their experimental water data around atmospheric pressure. This correlation represents a modified version for higher pressures based on the evaluation of the correlation of Fritz 38 at low pressures.
Kirichenko et al.
44
and Bald
45
formulated the correlation for bubble departure diameter as a function to the critical cavity diameter due to the edge-break mechanism since small contact angles (
Tolubinsky and Kostanchuk
46
employed the local bulk temperature for at subcooled conditions in the evaluation of the bubble departure diameter. Krepper et al.
29
employed this correlation for their modelling and defined the reference temperature (
Ünal
48
investigated different sets of experiments to determine empirical correlations for bubble growth rate, maximum bubble diameter and maximum bubble growth time of subcooled boiling water. For calculating bubble departure diameter, the Rohsenow
49
superposition method was employed to determine the nucleate flow boiling heat transfer. At subcooled conditions. A similar method to Levenspiel
50
was then applied to calculate the heat transfer coefficient. The analysis showed that the proposed equation for bubble departure diameter has a good agreement with most of experiments within ±30% uncertainty. The correlation is valid for
Nucleation frequency
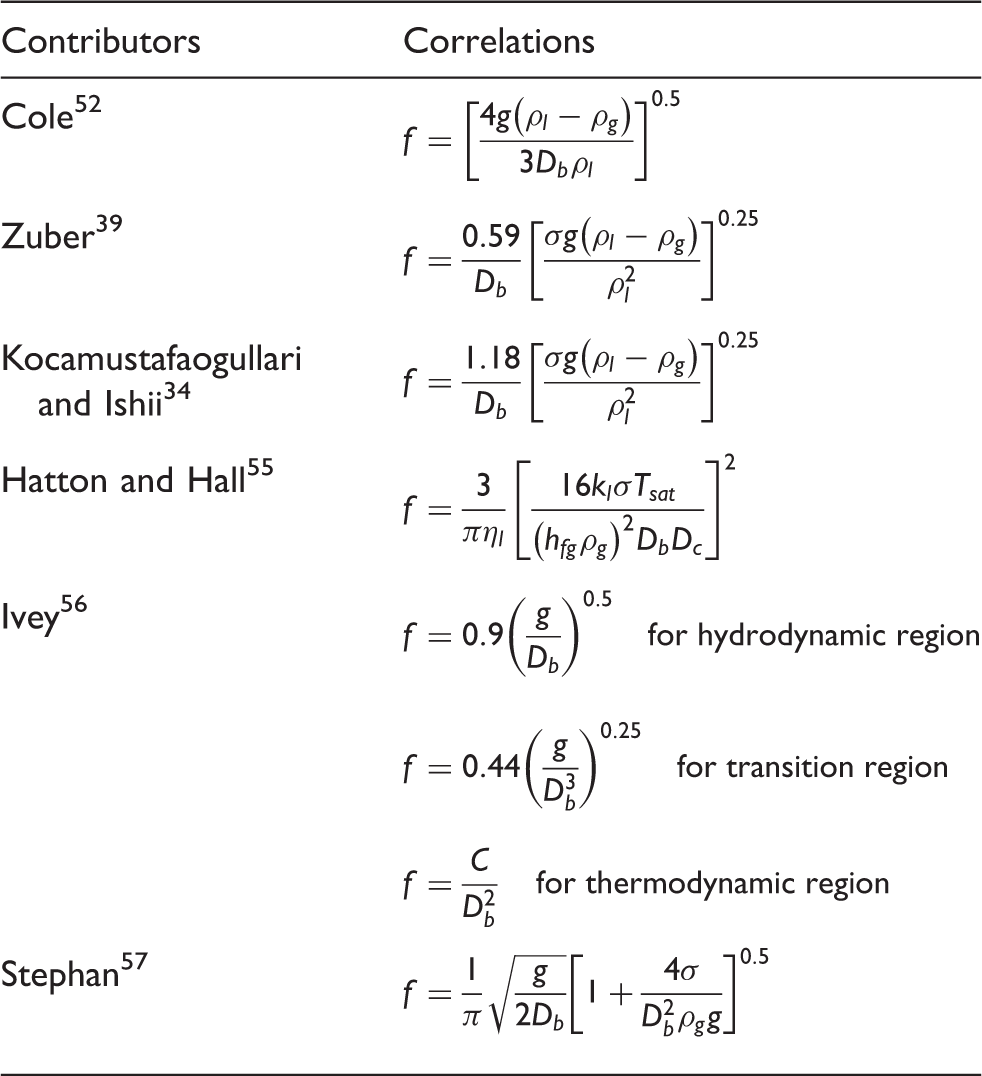
A number of investigations have been carried out to ascertain the nucleation frequency. Table 3 lists the various correlations for the nucleation frequency.
Cole 52 investigated the boiling phenomena in the vicinity of the critical heat flux. On the basis of the assumption that when successive bubbles leave the surface as they touch and coalesce, the nucleation frequency multiply by the bubble departure diameter at lift off can be taken to be equal to the rate that bubbles leave the surface.
Zuber 39 assumed the quotient of bubble departure diameter divided by the growth time can be taken to equal to the bubble rise velocity in a gravitational field. In the development of the nucleation frequency, he further assumed that bubble waiting time is almost equal to bubble growth time. Kocamustafaogullari and Ishii 34 applied the same correlation for their investigative study in a boiling channel albeit with a higher nucleation frequency due to a larger constant being taken.
For cryogenic liquids, the correlations usually take the form similar to those of Cole 52 or Zuber. 39 Correlation by McFadden and Grassman 53 has shown to agree well with the experimental data of liquid nitrogen. Kirichenko et al. 44 have developed a semi-empirical correlation, based on the experimental data, through correcting the contact angle of the detached bubble. In the developed pool nucleate boiling, Jin et al. 54 have nonetheless observed that the detachment frequency of coalesced bubbles remained almost unchanged with high wall heat fluxes and different heating surface materials in cryogenic liquids. For simplicity, the nucleation frequency can be regarded as a constant.
Hatton and Hall 55 correlated the nucleation frequency in terms of the square power of bubble departure diameter and critical cavity diameter with the experimental data from pool boiling of water at pressure of 0.0162, 0.0662 and 0.101 MPa. The correlation has been formulated based on the assumption that the waiting time was negligible in comparison to the growth time.
Ivey 56 proposed three correlations, relating the nucleation frequency for three different regions: (1) hydrodynamic region in which the buoyancy and drag forces predominate, correlated with water and methanol data; (2) transition region where the buoyancy, drag and surface tension forces are of the same order, correlated with water, methanol, isopropanol and carbon tetrachloride data; and (3) thermodynamic region where bubble growth dominates, correlated with water data.
Stephan 57 investigated the heat transfer coefficient in boiling systems and reported that nucleation frequency could be estimated from a correlation by Malenkov. 58
Mechanistic closure
It is recognised that not one single dedicated correlation is expected to be universally valid for all types of flow conditions to determine the three important parameters of active nucleation site density, bubble departure diameter and nucleation frequency in the wall heat flux partitioning model. In order to remove the application uncertainty of correlations empirically determined, a more mechanistic approach in determining the closure for the wall heat flux partitioning model may be required to improve the CFD predictions for cryogenic liquids.
Fractal model
The fractal distribution of sizes of active cavities on heated surfaces being considered is strongly dependent on not only the wall superheat as has been typified in most correlations for the active nucleation site density but also on other parameters including the liquid subcooled temperature, fractal dimension, minimum and maximum active cavity sizes, contact angle and physical properties of the adjacent fluid including the bulk velocity. From a mechanistic consideration, this model demonstrates enormous potential in appropriately determining the active nucleation site density distribution. Yeoh et al.59,60 have recently applied the fractal model to predict the active nucleation site density on the heated surface for a specific vertical subcooled flow boiling configuration.
According to Xiao and Yu,
61
the generated active cavities on heated surfaces for pool or flow nucleate boiling can be considered to be similar to the existence of pores in porous media. The cumulative number of active cavities with diameters equal to and greater than a particular active cavity diameter (Dc) can be described by

The total number of active nucleation sites per unit area (sites/cm2) from the smallest to the largest active cavity diameter can thus be obtained as
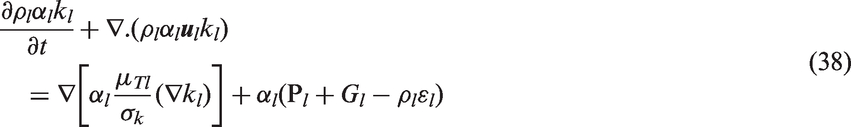
The smallest and largest active cavity diameters for nucleation site distribution can be evaluated based on Hsu
62
according to
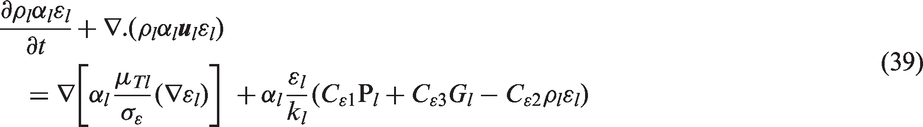
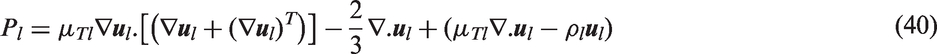
The fractal dimension 

Force balance model for bubble detachment from cavity site and bubble lift off
A force balance model developed in the context of CFD framework by Yeoh and Tu
64
has demonstrated that the bubble growth and its maximum detached bubble size contributed significantly towards the growth of void fraction and heat transfer within the bulk liquid. The development of the force balance model concentrates primarily on the various forces that influence the growth of a bubble in the directions parallel and normal to a heating surface. These forces are formulated according to the studies performed by Klausner et al.
65
and Zeng et al.66,67 As illustrated in Figure 3, the forces acting on the bubble in the x- and y-directions for a vertical heating surface are
Schematic views illustrating the forces acting on a growing vapour bubble along the vertical and horizontal heating surfaces.
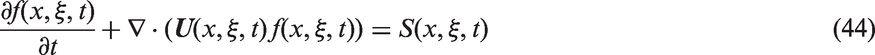

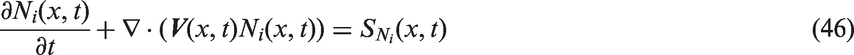
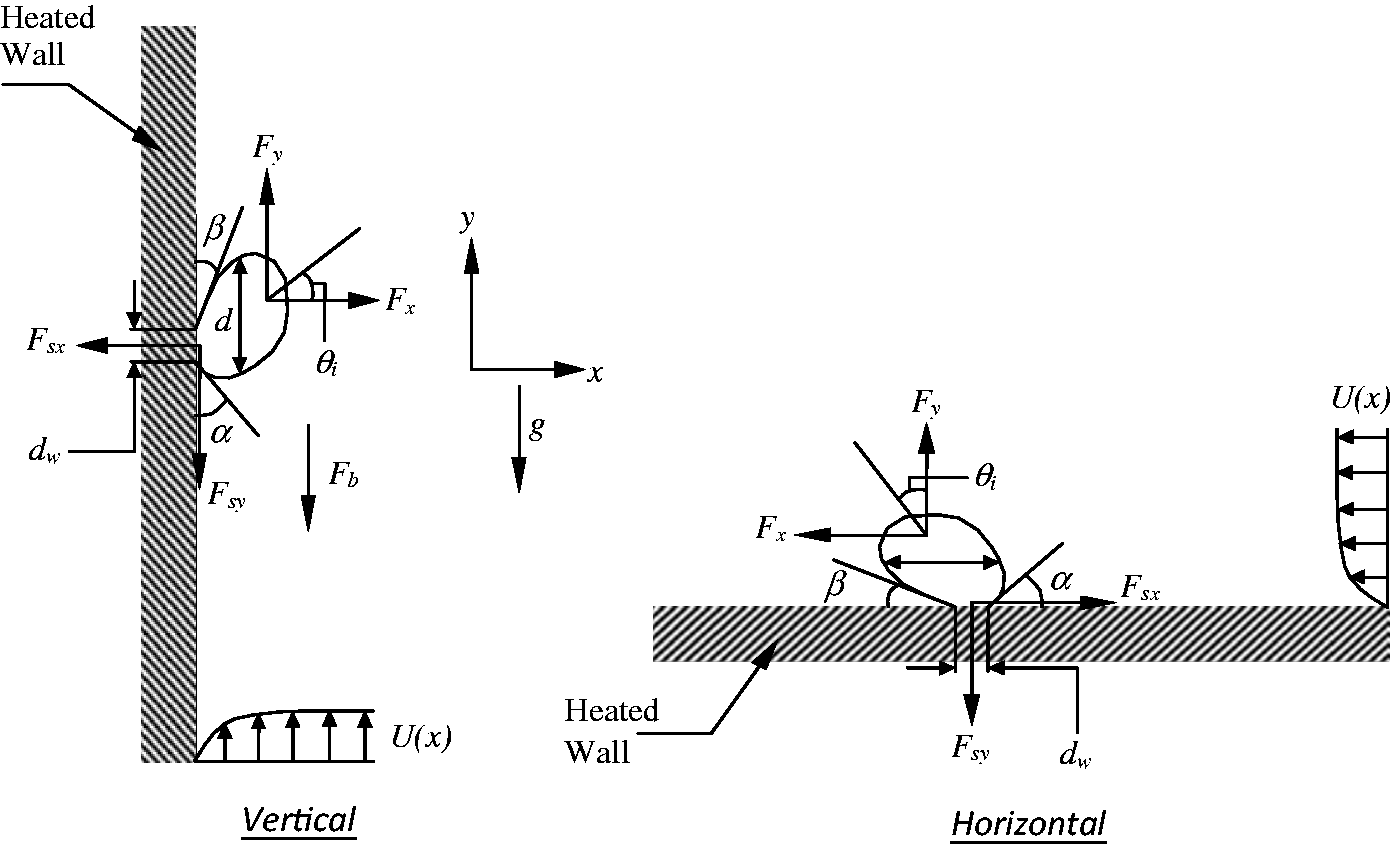
Forces acting on a growing bubble on the heated wall.
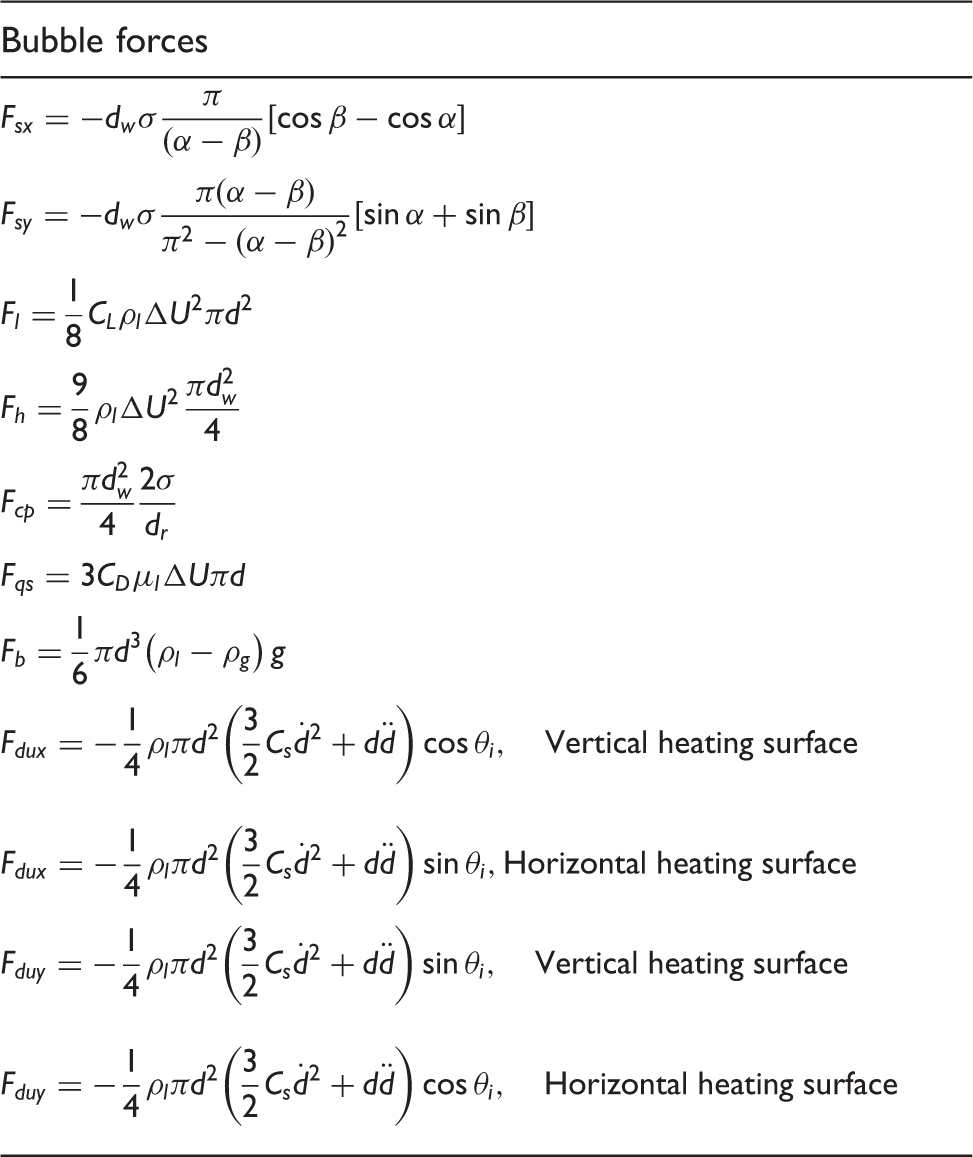
The drag and shear lift coefficients in Table 4 are determined according to the relationships proposed by Klausner et al.
65
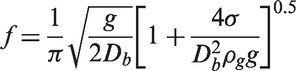
While a bubble remains attached to the heated surface, the sum of the parallel and normal forces must satisfy the following conditions:
Bubble growth time and bubble waiting time
Based on the description of an ebullition cycle during nucleate boiling, the nucleation frequency can be mechanistically determined from
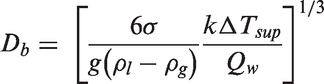
The bubble growth time can be determined through a diffusion-controlled bubble growth solution proposed by Zuber
68
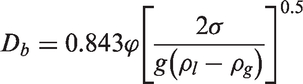
The bubble waiting time can be evaluated through the occurrence of transient conduction when a bubble slides or lifts off of which the boundary layer gets disrupted and cold liquid comes in contact with the heated wall. It is estimated as
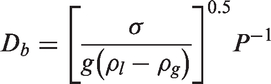
In equation (23), C1 and C2 are 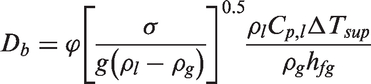
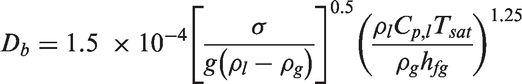
According to Basu et al., 69 the factor F indicates the degree of flooding of the available cavity size and the wettability of the surface. If the contact angle θ → 0, all the cavities will be flooded. Alternatively, as θ → 90°, F → 1, all the cavities will not be flooded (i.e. they contain traces of gas).
Alternatively, Podowski et al.
70
formulated mechanistic expressions for both the waiting time and the growth time. The bubble waiting time can be derived based on the balance of the transient heat transfer inside the wall and fluid near the heated surface
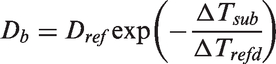
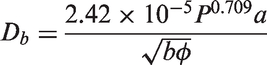
The bubble growth time is however obtained through the consideration of the critical bubble size and an energy balance during the bubble growth
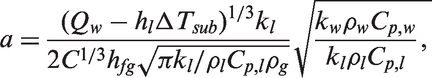
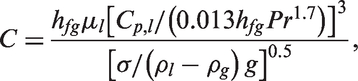
Two-fluid model for nucleate boiling
Away from the heated surface, ensemble-averaged of mass, momentum and energy transport equations are considered for each phase in the Eulerian–Eulerian framework to resolve the two-phase boiling and bubble behaviours in the bulk liquid. The liquid is generally taken as to be the continuum phase while the vapour (bubbles) is treated as the disperse phase. These equations can be derived as
Mass conservation of liquid phase
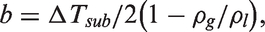
Mass conservation of vapour phase
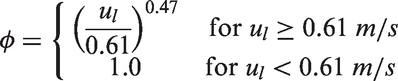
Momentum conservation of liquid phase
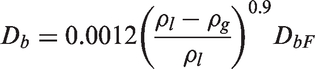
Momentum conservation of vapour phase
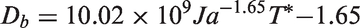
Energy conservation of liquid phase
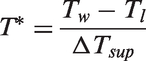
Energy conservation of vapour phase
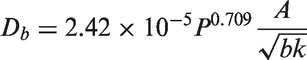
Interfacial momentum forces.

In Table 5, the drag force is a result of the shear and form drag of the fluid flow. Coefficient CD appearing in the drag force, normally known as the drag coefficient, can be determined based on the correlations proposed by Ishii and Zuber
74
for different flow regimes (bubbly, slug or churn-turbulent). As bubbles rise in a liquid, they are subjected to a lateral lift force which acts perpendicular to the direction of relative motion between the two phases. Lopez de Bertodano
75
and Takagi and Matsumoto
76
have proposed a value of
On the basis of the eddy viscosity hypothesis, the effective viscosity in the momentum liquid phase transport equation can be taken as the sum of the molecular viscosity and turbulent viscosity of each phase. With the inclusion model for bubble-induced turbulence by Sato et al.,
83
the effective viscosity is thus considered as the total of the shear-induced turbulent viscosity and the bubble-induced turbulent viscosity. The effective viscosity of the liquid phase in equation (30) can thus be expressed as
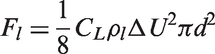
In equation (34), the liquid turbulent viscosity is given by
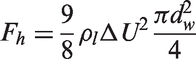
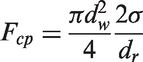
For the vapour phase, the turbulent viscosity can be simply evaluated as
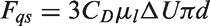
In order to evaluate the liquid turbulent viscosity in equation (35), the two-phase two-equation k–ɛ turbulence model, which is mere extension of its single-phase counterpart, may be employed to resolve the turbulent flow associated with the continuous liquid phase.
84
The transport equations for the turbulent kinetic energy of the liquid phase (kl) and dissipation of the turbulent kinetic energy of the liquid phase (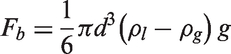
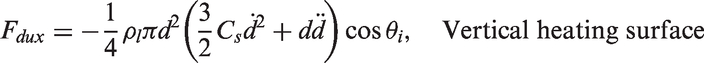
The shear production of turbulence Pl is given by
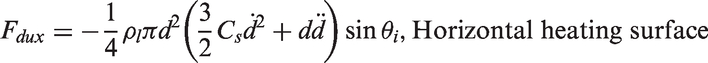
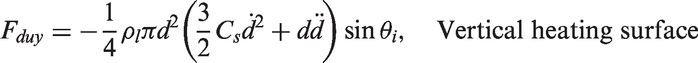
Appropriate values for
In the case of subcooled boiling, the interfacial mass transfer rate due to condensation in the bulk liquid in equation (28) can be formulated as
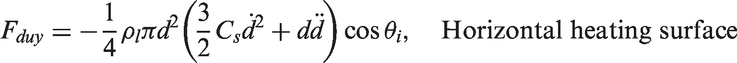
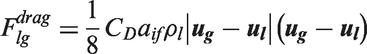
For nucleate boiling, the wall nucleation rate is generally treated as a specified boundary condition, which can be treated as the boundary mass flux imposed on the heating surface.
Population balance model
CFD coupled with an appropriate population balance model are increasingly being employed to predict the evolution of the particle (bubble) size distribution in the gas–liquid systems. This is particularly important especially in resolving the interfacial momentum transfer in the two-fluid model where the interfacial forces appearing in the momentum transport equations require the knowledge of the bubble size (Sauter mean diameter) or interfacial area concentration, which is crucial to the overall prediction of the local phase distribution. Some earlier studies have simplified the two-phase problem with a prescribed single bubble size (or so-called mono-dispersed bubble size).88,89 Such assumption is obviously invalid beyond bubbly flow conditions such as can be found in practical gas–liquid systems. Mounting interests have spurred numerous studies in attempting to better synthesise the behaviour of bubbles and track their dynamical evolution within the two-phase flow domain.
In accordance with Cheung et al.
90
and Yeoh et al.,
91
the population balance equation can be expressed in an integro-differential form as
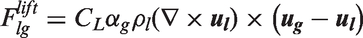
In practice, the transport of particle (bubble) number density needs to be solved within the framework of CFD. Following the formulation proposed by Prince and Blanch,
92
Luo
93
and Luo and Svendsen,
94
the continuous size range of particles represented by the particle (bubble) number density distribution can be discretised into a series number of discrete size classes. By taking the volume Vp as the independent internal coordinate, the population balance equation for the discrete particle (bubble) number density defined by
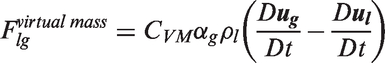
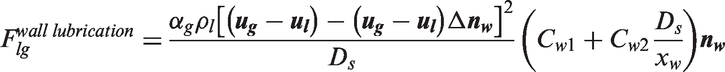
The population balance equation for the average particle (bubble) number density defined by
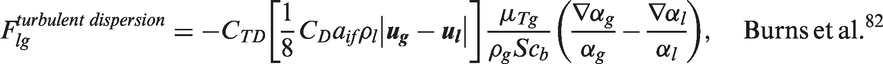
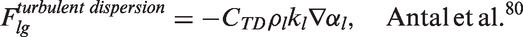
Population changes in equations (46) and (48) are accommodated through mechanistic models being formulated for the source terms within these transport equations for particular gas–liquid systems.
Considerable attention has been focused on the description of the temporal and spatial evolution of the geometrical structure caused by the effects of coalescence and break-up through the interactions among bubbles as well as between bubbles and turbulent eddies in turbulent flows. For bubbly flow, appropriate mechanistic models for the source terms in equations (46) and (48) have been established: (i) Coalescence through random collision driven by turbulent eddies; (ii) Coalescence due to the acceleration of the following bubble in the wake of the preceding bubble; and (iii) Break-up due to the impact of turbulent eddies. Schematic illustrations of these mechanisms are illustrated in Figure 4. Table 6 tabulates three mechanistic models that have been considered for the consideration of the average particle (bubble) number density equation while Table 7 lists some of the more popular mechanistic models that have been applied to characterise the coalescence and break-up of bubbles for the discrete particle (bubble) number density equation. More details can be found in Yeoh et al.
91
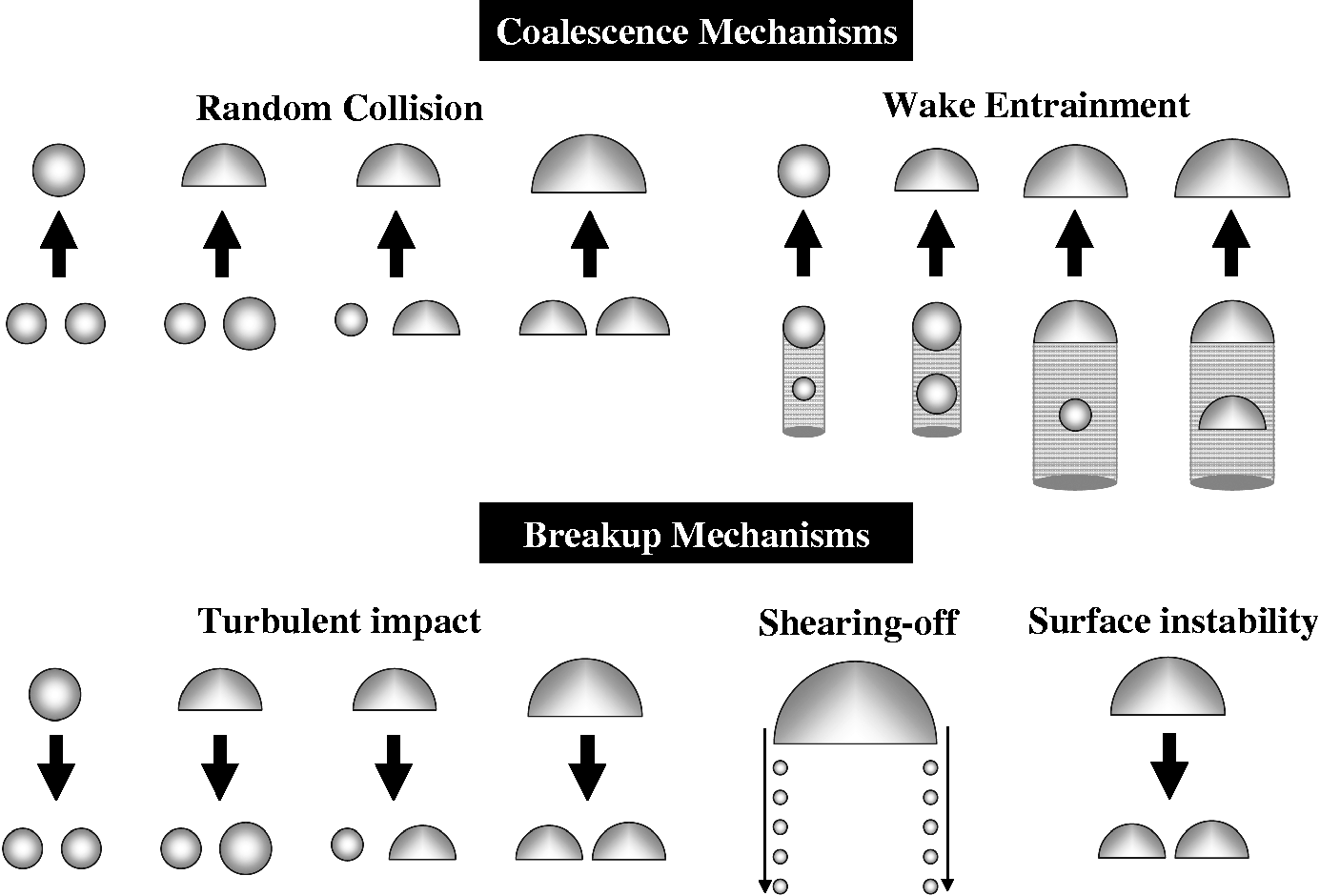
In Table 6, Bubbles undergoing coalescence and break-up in gas–liquid systems. Coalescence and break-up mechanisms for averaged particle (bubble) number density. Coalescence and break-up mechanisms for discrete particle (bubble) number density. Beyond bubbly flow, the requirement of accommodating other bubble sizes and shapes involves the intra-group and inter-group interactions for spherical and cap/slug bubbles. The probable mechanisms of these interactions that need to be considered are: (i) Coalescence due to random collisions driven by liquid turbulence; (ii) Coalescence due to wake entrainment; (iii) Break-up due to the impact of turbulent eddies; (iv) Shearing-off of small bubbles from cap/slug bubbles; and (v) Break-up of large cap bubbles due to flow instability on the bubble surface. For intra-group mechanisms for spherical bubbles, the usual coalescence and break-up processes due to random collisions and turbulent impact can be adopted. The intra-group mechanisms for cap/slug bubbles results in a more complex consideration where the bubble coalescence is due to random collisions and wake entrainment while the bubble break-up is attributed by turbulent impact, shearing-off and surface instability. Referring to Figure 5, the wake entrainment for cap/slug bubbles predominantly governs the cap/slug bubble number, which significantly affects the flow structure and intensiveness of inter-group interactions. Here, when bubbles enter the critical wake region of a preceding bubble, they can accelerate and coalesce with this preceding bubble according to the mechanisms based upon the critical distance of a probability approach such as proposed by Sun et al.
102
Nevertheless, the cap/slug bubble disintegration due to surface instability can be significantly enhanced by the high turbulent intensity and active eddy-bubble interaction in the wake region of the slug bubbles. This occurs when the volume of the resulting bubble from bubble coalescence exceeds the maximum bubble stable limit; it becomes unstable and disintegrates. The mechanism for shearing-off effect is reflected by the fact that the bubbles shearing-off from the cap/slug bubbles contribute as a primary source for the spherical bubbles and ultimately the interfacial area concentration. More details on the mechanisms for cap/slug bubbles can be found in Hibiki and Ishii,
103
Fu and Ishii,
104
Hibiki and Ishii
105
and Cheng et al.
106
In addition to the intra-group interactions, it has been demonstrated through experiments performed by Hibiki and Ishii
103
that the inter-group interactions also contribute significantly to the transport of the interfacial states. Similar mechanisms from the above considerations for intra-group interactions may be derived accordingly for the inter-group interactions.
Schematic drawings of the mechanisms associated with the two-group bubble interactions.
105
The boiling behaviour of cryogenic liquids is considered to be different from the boiling behaviour of ordinary liquids. In comparison to an ordinary liquid such as water, cryogenic liquids can only remain in their liquid form within a much narrower atmospheric liquid range. A review on the use of empirical correlations and theoretical development of mechanistic models to handle nucleate boiling of cryogenic liquids is presented. In spite of the prospect of adopting a mechanistic approach in modelling the heat transfer mechanisms occurring during the complex boiling process, which can be readily employed for both the prediction of the wall heat flux as well as the partitioning of the wall heat flux between the liquid and gas phases, limitations still persist due to the inadequacy of empirical correlations in fully achieving the necessary closure. The proposal of the fractal and force balance models to mechanistically determine the active nucleation site density and bubble diameters lifting off from the heated wall can in some way elevate the deficiencies that are prevalent in the use of empirical correlations. In addition, the necessary closure for the inter-phase mass, momentum and energy exchanges that are required to be accounted in the two-fluid model requires constitutive relations which can be realised through the use of mechanistic population balance model in predicting the local changes of the Sauter mean diameter or interfacial area concentration. Nevertheless, most mechanistic models for bubble coalescence and break-up that have been developed thus far in the population balance model are established mainly for the bubbly flow regime. Extensive work is still required to identify proper bubble mechanisms and develop models for flow regimes that may be filled with complex shape bubbles such as slugs and columns as the wall heat flux increases to CHF. The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The author(s) received no financial support for the research, authorship, and/or publication of this article.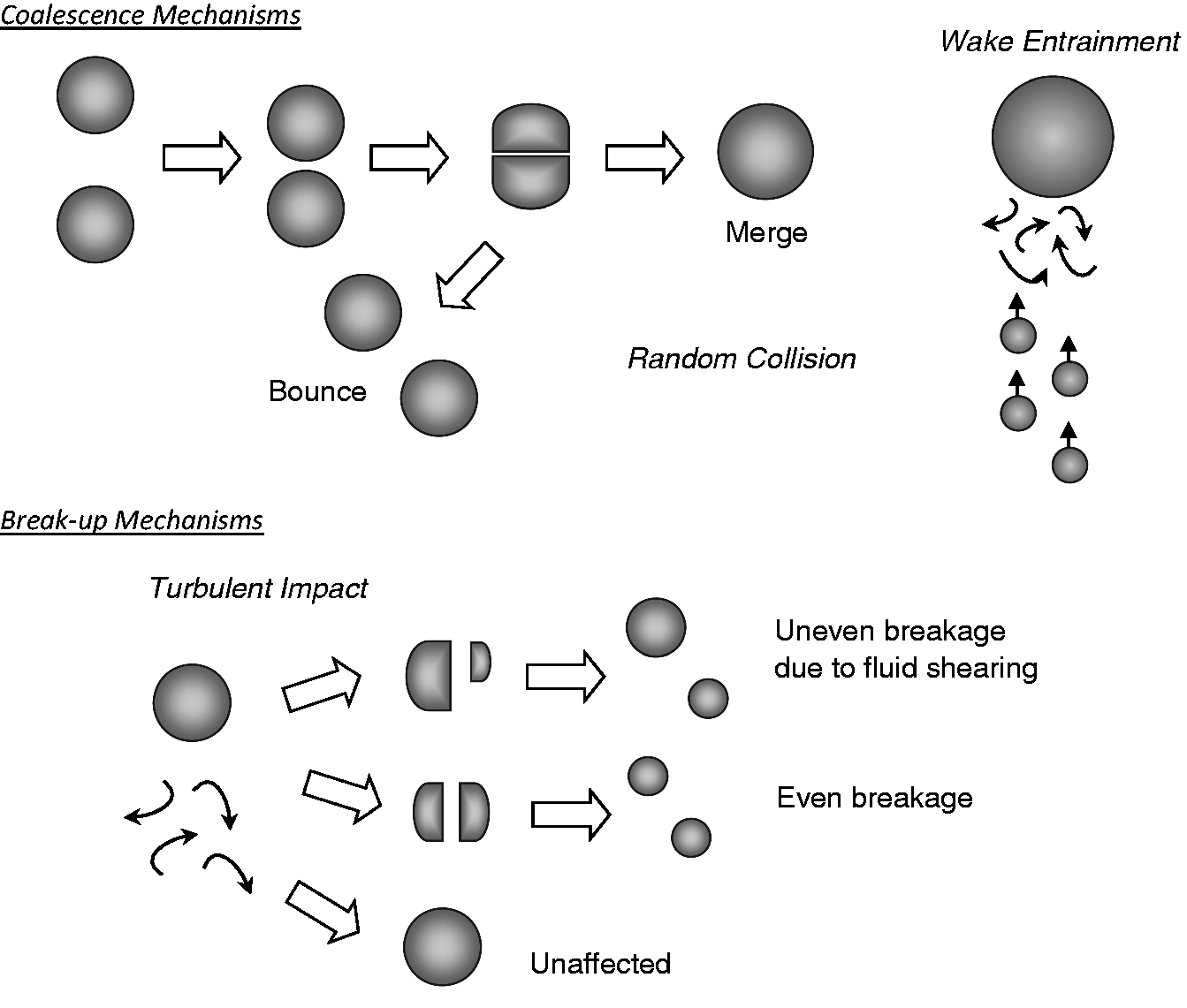


Concluding remarks
Footnotes
Declaration of conflicting interests
Funding
Appendix
References