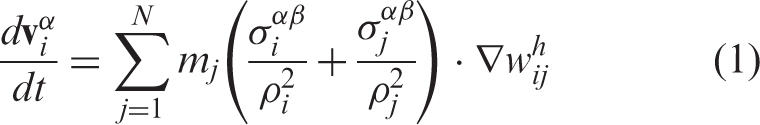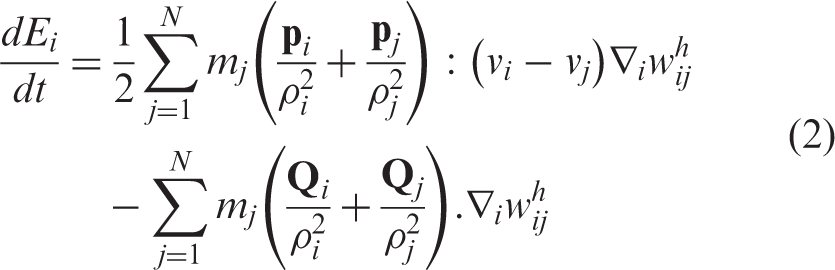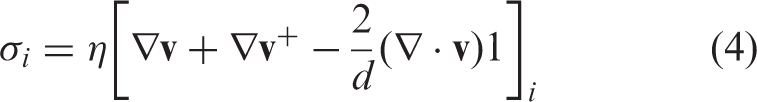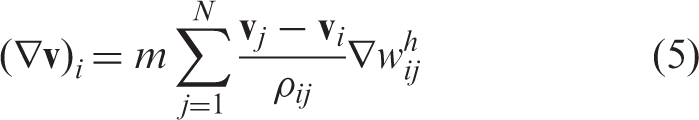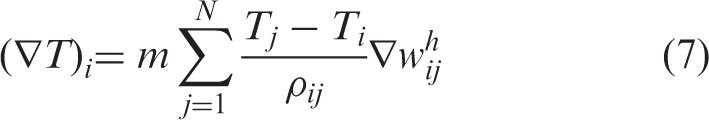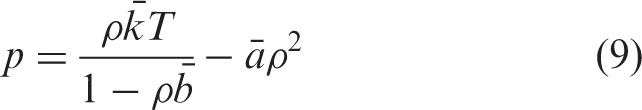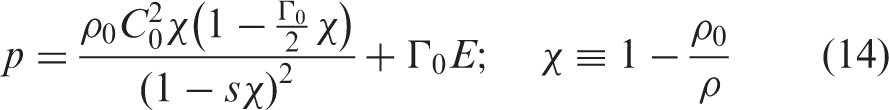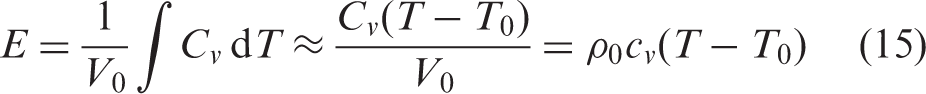Abstract
It is proposed here a hydrodynamic formalism in order to obtain an approximate numerical solution to the Navier–Stokes equations. This formalism consists in the interpolation of the properties of the system of particles. The Smoothed Particle Hydrodynamics formalism (SPH) is employed when the system exhibits big deformation and large velocities because it is mesh-free in nature. In this study, it is simulated for the first time the condensation and formation of a spherical van der Waals drop by means of particles in three-dimensional space with the SPH formalism. When the drop is formed, starting from a gas box, the distribution of density implies that there are zones where the mass of gas is converted to a liquid state, but out of the droplet radius, some particles remain in the gas form.
Introduction
Smoothed Particle Hydrodynamics is a method to solve approximately the equations of fluid dynamics, replacing the fluid by a set of particles. The SPH method was simultaneously invented by Lucy 1 and Gingold and Monaghan 2 to solve astrophysical problems. This method has been used to study a range of astrophysical topics including: the formation of galaxies, stars, supernovas, stellar collisions, and so on.
The advantage of the SPH method is that interface problems can be easily modeled while they are hard to model using methods based on finite differences. An additional advantage is that SPH can be considered as a bridge between continuous and fragmented material, a feature that adapts well to study problems of fragmentation in solids. 3 Another feature which makes the SPH method attractive is that the solutions yielded by this method depend on space and time, making it very versatile for the treatment of a wide variety of problems in physics. Currently, there are several applications of SPH in different areas related to fluid dynamics, such as: incompressible flows, elastic flows, multiphase flows, supersonic flows, shock wave simulation, heat transfer, explosive phenomena, and so forth. 3
Ucar and Erbil 4 determined that the equations derived for the diffusion controlled drop evaporation processes can be successfully applied to the condensation rate of water droplets on polymer surfaces having a surface temperature just below the dew point. The differences between the growth rates of condensed isolated droplets on five different polyolefin surfaces, whose surface free energies were in a close range of 30–37 mJ/m2, were attributed to the difference in surface roughness which alters the droplet nucleation rate, drop pinning effect and initial contact angles. The drop radius of the individual isolated droplets was found to grow according to a power law with exponent 1/3 except polypropylene similar to previous reports. These authors also compared the volume increase of an isolated single droplet with the volume increase of another similar sized single droplet which was surrounded by neighboring droplets and found that when neighboring droplets are present in close proximity, the condensation rate was 14–40% lower than that of a single isolated droplet due to blocking of lateral water vapor diffusion. This effect was more pronounced on substrates having high surface roughness.
Kalyuzhny and Kulacki 5 studied the heat transfer coefficients for condensing FC-72 vapor on vertical copper and Teflon plates as a function of sub-cooling at one atmosphere. Results include the evolution of the average heat transfer coefficient with time and a visual record of droplet formation and coalescence owing to non-condensable gas. Experiments are run for a Reynolds number of 15.1, wall heat flux of 0.92–2.88 W/cm2, and 6–44 K sub-cooling. Film condensation heat transfer coefficients compare reasonably well with those of prior studies run under different convective conditions. High-resolution video captures evolution of droplet size (average diameter) and number density. A correlation is shown to exist between overall heat transfer coefficients and droplet size and number density. When the droplet number density exceeds 10 cm2 and droplet area exceeds 1.5 mm2, average heat transfer coefficients approach a limiting value.
Van der Hage6,7 reported the drop formation on insoluble particles. In these studies it is shown that one absorbed monolayer contains enough liquid to account for the number of droplets observed in condensation experiments. Furthermore, it is shown that there are no thermodynamic objections against spontaneous drop formation on an adsorbed layer as a result of continued adsorption at a critical saturation ratio. Hienola et al. 8 compare four different H2O mass flux descriptions in simulations of the evolution of mixed water/ice aerosol populations undergoing condensational growth. It is shown that the Maxwellian description, ignoring condensation heat release, overestimates the growth rates severely when compared with an exact numerical description. The well-known Mason equation predicts the growth rates somewhat better, but still in an inadequate manner as far as cloud drop formation is considered: after 100 s of growth, the drop radii are overestimated by up to a factor of 3 at 30℃. Many important features of the condensation in such diverse systems can be captured by the nonlinear Schroedinger model.
Zakharov and Nazarenko 9 within this model develop a statistical description in which the condensate is nonlinearly coupled to wave turbulence described by a kinetic equation. This study was on the strong-condensate regime in which the three-wave interaction replaces the four-wave process operating on the preceding stages of an explosive condensate formation and its initial growth. In the strong-condensate regime, the condensate growth accelerates and becomes quadratic in time. This regime will proceed until the wave dispersion drops below a critical value and the state of dispersionless acoustic turbulence forms. Rao and Murthy 10 proposed a model for heat transfer during dropwise condensation based on the assumption that heat transfer takes place through the bare surface in between drops to form nuclei at nucleation sites during waiting period required for nucleation. The authors consider the dynamics of drop formation and surface renewal, and the presence of non-condensable gases in the vapor phase.
Yin et al. 11 conducted numerical experiments to investigate the effects of giant cloud condensation nuclei (CCN) on the development of precipitation in mixed-phase convective clouds. The results show that the strongest effects of introducing giant CCN occur when the background concentration of small nuclei is high, as that in continental clouds. Under these conditions, the coalescence between water drops is enhanced due to the inclusion of giant CCN, resulting in an early development of large drops at the lower parts of the clouds. It also leads to the formation of larger graupel particles and to more intensive radar reflectivities. When the background concentration of small nuclei is low, as in maritime clouds, the effect of the giant CCN is smaller and the development of precipitation is dominated by the droplets formed on large nuclei. Scheludko 12 studied the effect of line tension on condensation of liquid from supersaturated vapors onto solid spherical nuclei. A negative line tension leads to a decrease in critical supersaturation and a new mechanism of barrierless phase formation. Krakovskaia and Pirnach 13 made simulations of mixed supercooled clouds to see as to what extent the different microphysical processes such as collection, aggregation, freezing, accretion, riming, etc. and thermodynamical conditions such as surface temperature and updrafts can impact on the development of cloud and precipitation. They have shown that the liquid, as well as solid, precipitation from supercooled mixed clouds may be significant, especially at surface temperatures greater than 0℃. The influence of updraft values on liquid precipitation is significant, while the surface temperature affects liquid phase precipitation slightly. The opposite holds for solid precipitation; the temperature is a principal factor, while the updrafts affect solid precipitation only at temperatures above 0℃. In this study, the different mechanisms of cloud and precipitation formation show that all such mechanisms are important. If one is absent, others compensate and can form the precipitation successfully. The obtained spectra of cloud droplets and ice crystals conform to a γ-distribution and spectra of the raindrops correspond to the power distribution.
The numerical diffusion of clouds can be totally suppressed by the volume-of-fluid (VOF) method, which is applied by Hinneburg and Knoth 14 in connection with an atmospheric model. The cloud phase is distinguished by prognosing the partial cloud volume in all grid cells near the cloud boundary. Adopting elementary geometrical forms for the intracellular cloud volume and simple diagnostic rules of their alignment, the standard transport fluxes can be used in the new equation. Separate variables for the cloud and environmental phase complete the transport scheme. The VOF method and its realisation are described in detail in this study. Advection, condensation, evaporation and turbulent diffusion are considered within the VOF framework. The variation of the grid resolution and turbulence conditions for a rising thermal leads to striking arguments in favour of the VOF method, resulting in higher intensity, lifting, and lifetime as well as clear boundaries of the simulated clouds (even for low grid resolution).
Hoover 15 and Colagrossi and Landrini 16 used the SPH method to model immiscible flows and found that the standard formulation of SPH proposed by Gingold and Monaghan 2 creates an artificial surface tension on the border between the two fluids. Colagrossi and Landrini 16 put forward an SPH formulation for the simulation of interfacial flows, that is to say, flow fields of different fluids separated by interfaces. Tartakovsky and Meakin 17 have shown that the artificial surface tension that emerges from the standard formulation of the SPH method 2 could be eliminated by using SPH equations based on the number density of particles rather than the density of particles in the fluid, which are used in standard formulations of SPH.
In this paper, the smoothing function used was the cubic B-spline kernel, 18 and the equation of state considered was a general Mie-Gruneisen form of EOS for the van der Waals drops and the aluminium solid surface. The aim of this work is to simulate the condensation and formation of van der Waals drops in 3D with the smoothed particle hydrodynamics method. Hence, a 3D hydrodynamics formalism is proposed here that allows the modeling of the condensation of the droplet starting from a 3D box of a van der Waals gas. As a hydrodynamical test of the SPH code implemented in this paper it is show the collision between the van der Waal drop and a solid aluminium surface.
Numerical scheme
In the SPH formalism, the velocity and acceleration fields are given by
3
The internal energy evolution is given by the expression
3
In the above equation,
In these equations, pi is the pressure, η and ηv are the shear and bulk viscosities, respectively, and d = 3 is the spatial dimension. The notation “+” means transposition and
In this equation, Ti is the temperature associated with particle i, and κ is the heat conductivity and ρ ij = (ρ i + ρ j )/2.
In the present work, the model is performed in three dimensions and the cubic b-spline kernel used was
18
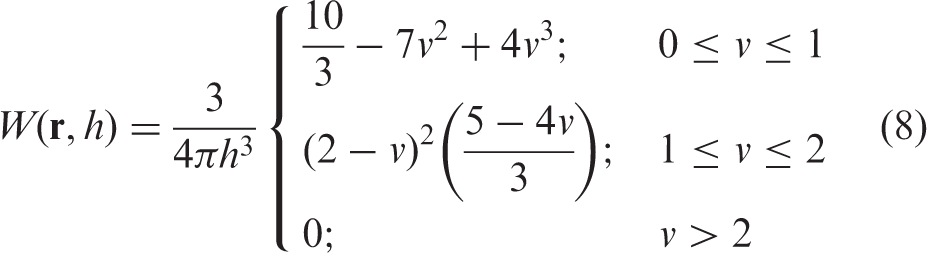
In this paper, a van der Waals fluid is considered
20
and the equation of state in the hydrodynamics code was
In the above equation
Furthermore
Formation and condensation of spherical drops
In all calculations, the following values for the constants are used: N = 4096 particles, length of the 3D box L = 12.0 with smoothing lengths h = 5.0. Reduced units were used here
21
; mass m = 1, η = 1, ηv = 0.1, κ = 5.0 and the time step inside the hydrodynamics SPH code was Δt = 0.05. In Figure 1 can be seen the condensation of a van der Waals drop in t = 22.0 s and reduced temperature of 0.2. Initially, at t = 0.0 s, the mass of gas is a T = 1.05 and when the dynamics run one can see that this temperature is decreased with time until the formation of a van der Waals drop at t = 22.0 s and T = 0.2. At intermediate stages of the calculation, it can be seen a polyhedral structure with a spherical mass of liquid and gas inside. Figure 2 shows the radial distribution of density at t = 22.0 s. One can see that there are zones where the fluid reaches the liquid state and the drop is surrounded by a vapor mass of the van der Waals fluid.
Stable drop configurations for the reduced temperatures 0.2 (t = 22.0 s), 0.3 (t = 19.0 s), 0.4 (18.0 s), 0.6 (t = 15.0 s), 0.87 (t = 10.0 s) and 1.05 (t = 0.0 s) of a van der Waals fluid. N = 4096 SPH particles were used in a 3D box of width L = 12. 0 with a smoothing length of h = 5.0. SPH radial mass–density dependence ρ (r), where r is the distance from the centre of mass. All quantities are given in reduced units.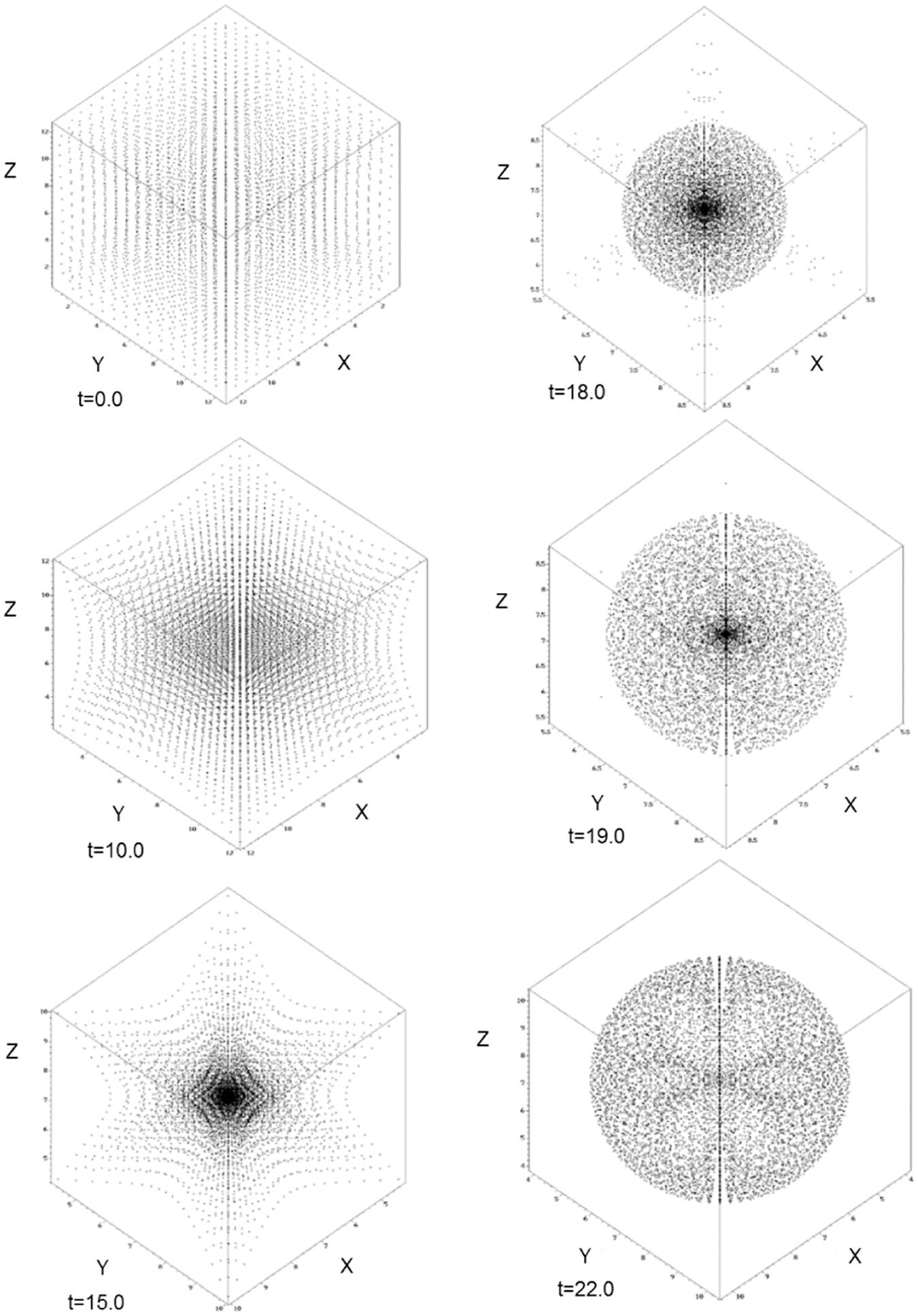
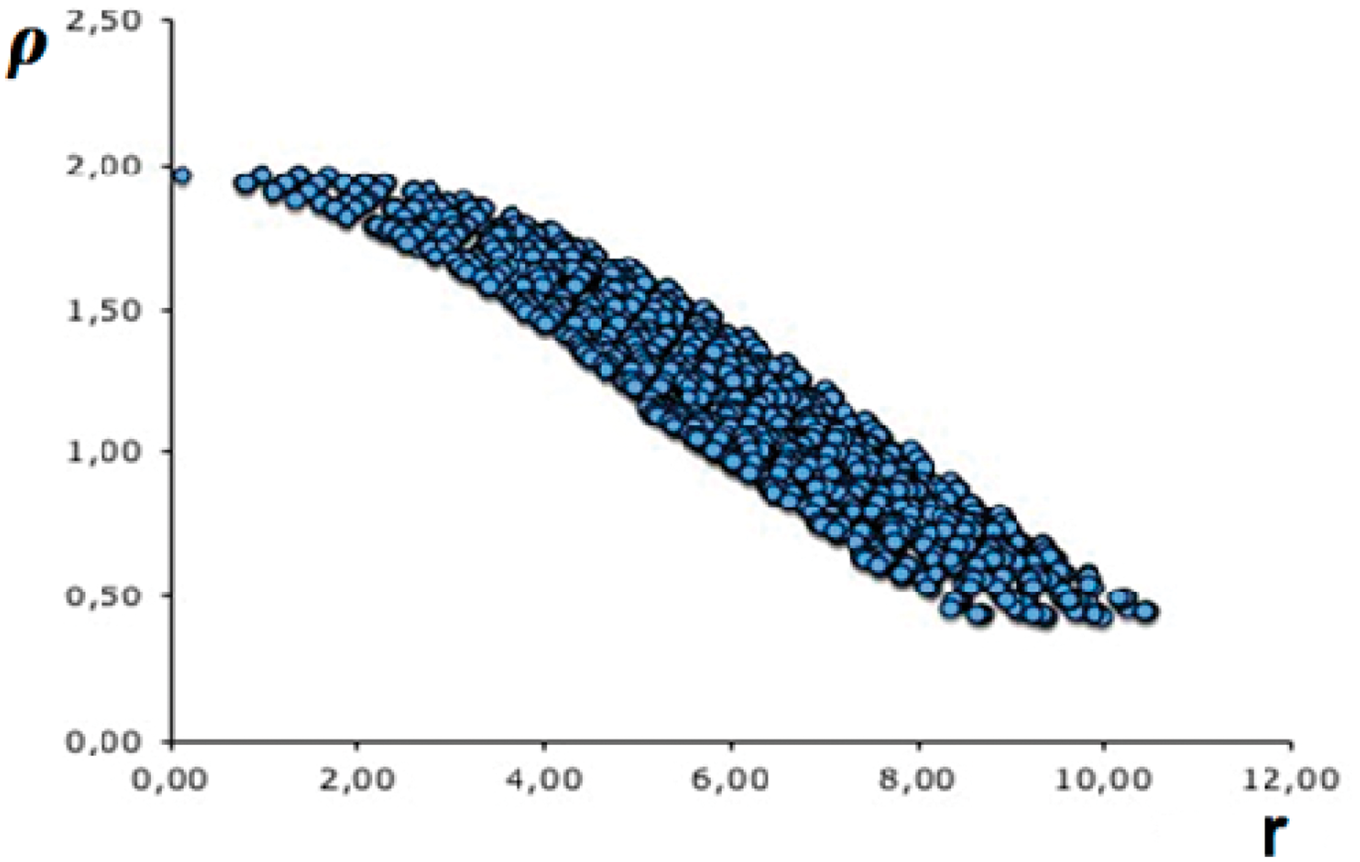
In the following calculations, an SPH system with the droplet obtained previously is constructed that collides with a planar solid surface of aluminium. The number of SPH particles for the n-Hexane droplet (radius of 30 µm) was 4456 and 37303 for the aluminium surface.
In order to model the aluminium surface, was used inside the hydrodynamical code the Mie–Gruneisen equation of state, which is frequently used for solids
It can be observed in Figure 3 the collision between one n-hexane droplet and the aluminium surface with a velocity of 30.0 mm/ms. When the calculation begins, there is a stretching of the droplet surface and a planar section appears in the zone of contact between the drop and the aluminium surface (see Figure 3 at t = 8.5 × 10−5 ms). The surface tension forces are not enough to preserve the droplet in this stage of the calculation; in fact, the inertial forces produce more deformation of the drop until this droplet is transformed in a thin liquid film over the aluminium surface (see Figure 3 at t = 1.4 × 10−3 ms).
The collision between one van der Waals droplet (n-Hexane) and aluminium solid surface. Time is given in milliseconds. The collision velocity is 30.0 mm/ms.
In Figure 4 can be observed the velocity vector field inside the n-hexane droplet at t = 2.6 × 10−4 ms. It can be seen that in the zone of contact between the drop and the solid surface, the velocity has an increment with a value around 40 m/s.
Velocity vector field of the van der Waals (n-Hexane) droplet. Time is given in milliseconds.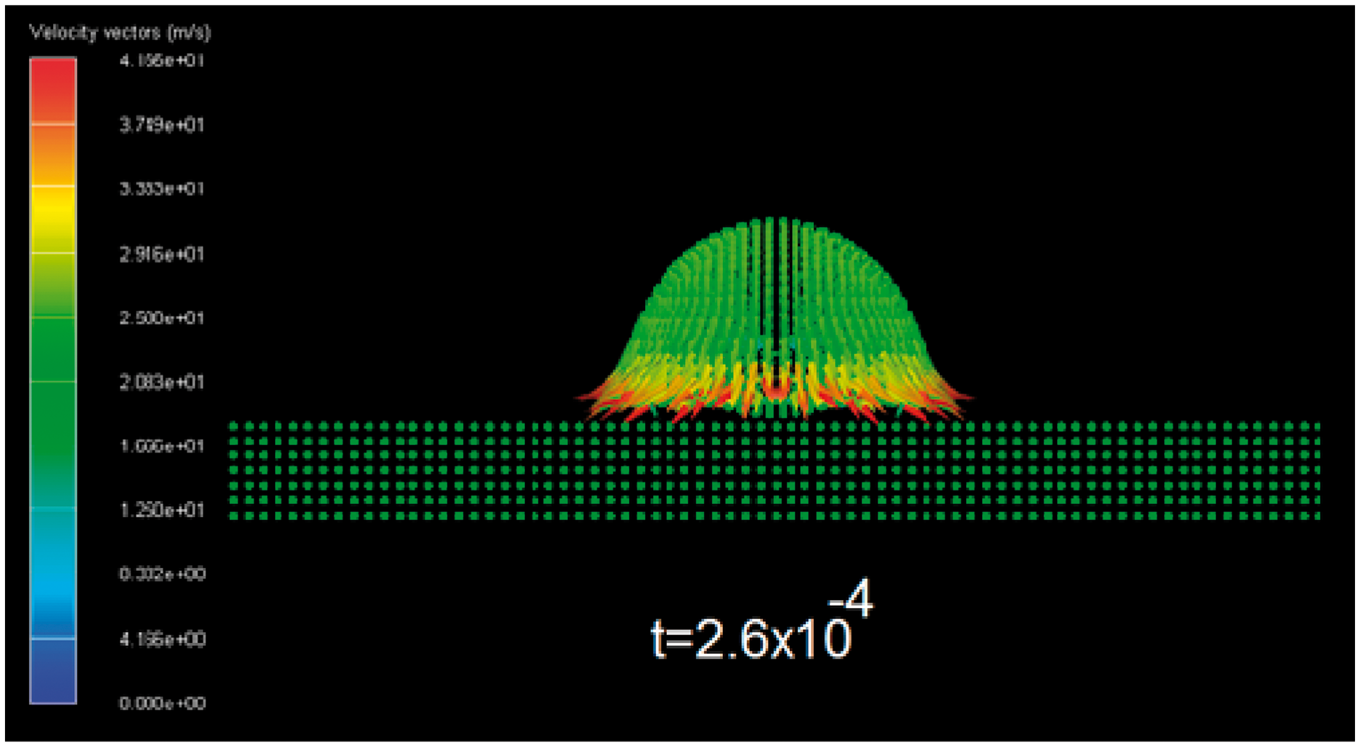
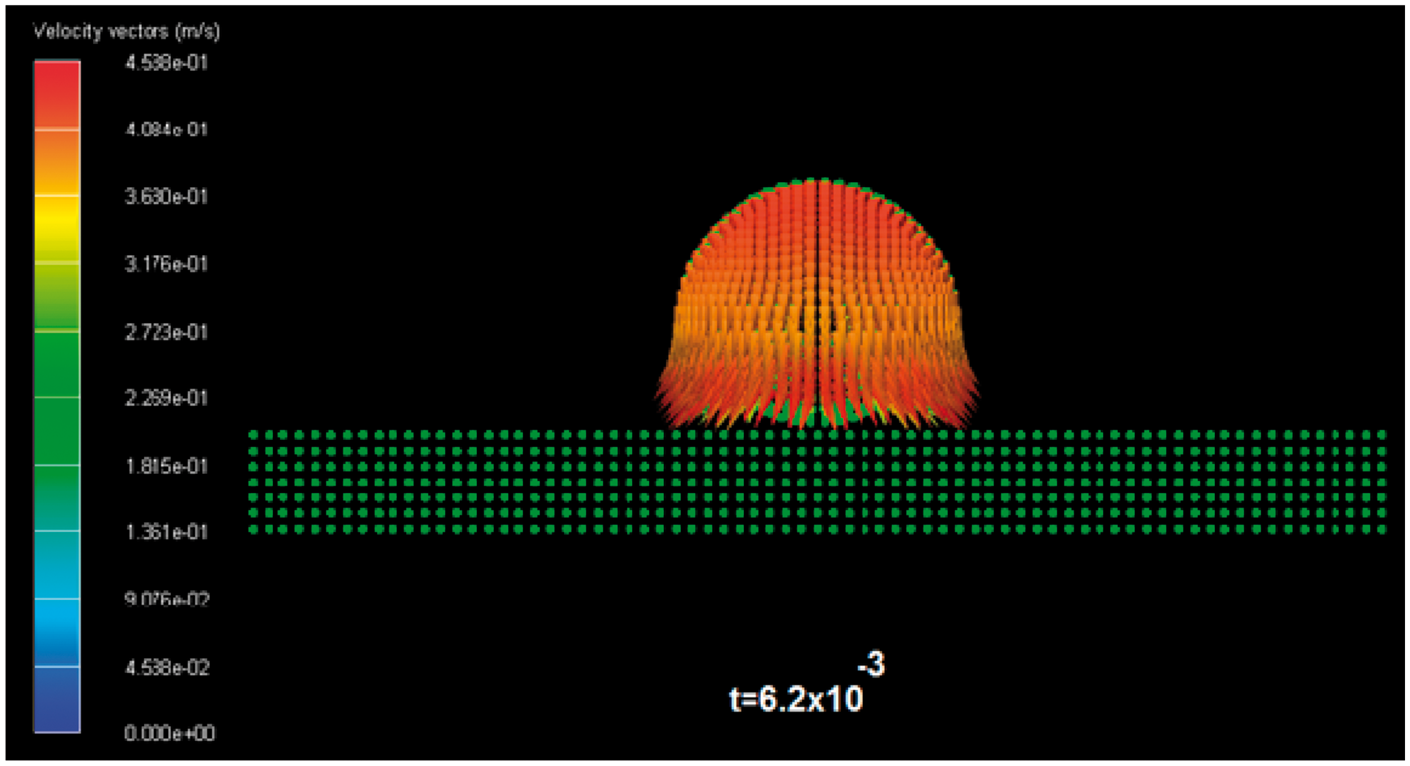
In Figure 5, the collision process between the n-hexane droplet and the aluminium surface is shown. In this case, the velocity of the droplet v = 0.5 m/s is smaller than the previous calculation. Here the surface tension forces prevailing over the inertial forces and the droplet deform its surface and maintain this form until the end of the calculation. In Figure 6 can be seen the velocity vector field inside the n-hexane drop, in this case the velocity is lower than the initial velocity value of 0.5 m/s; in fact, the velocity reaches a value around 0.4 m/s.
The collision between one van der Waals droplet (n-Hexane) and aluminium solid surface. Time is given in milliseconds. The collision velocity is 0.5 mm/ms. Velocity vector field of the van der Waals (n-Hexane) droplet. Time is given in milliseconds.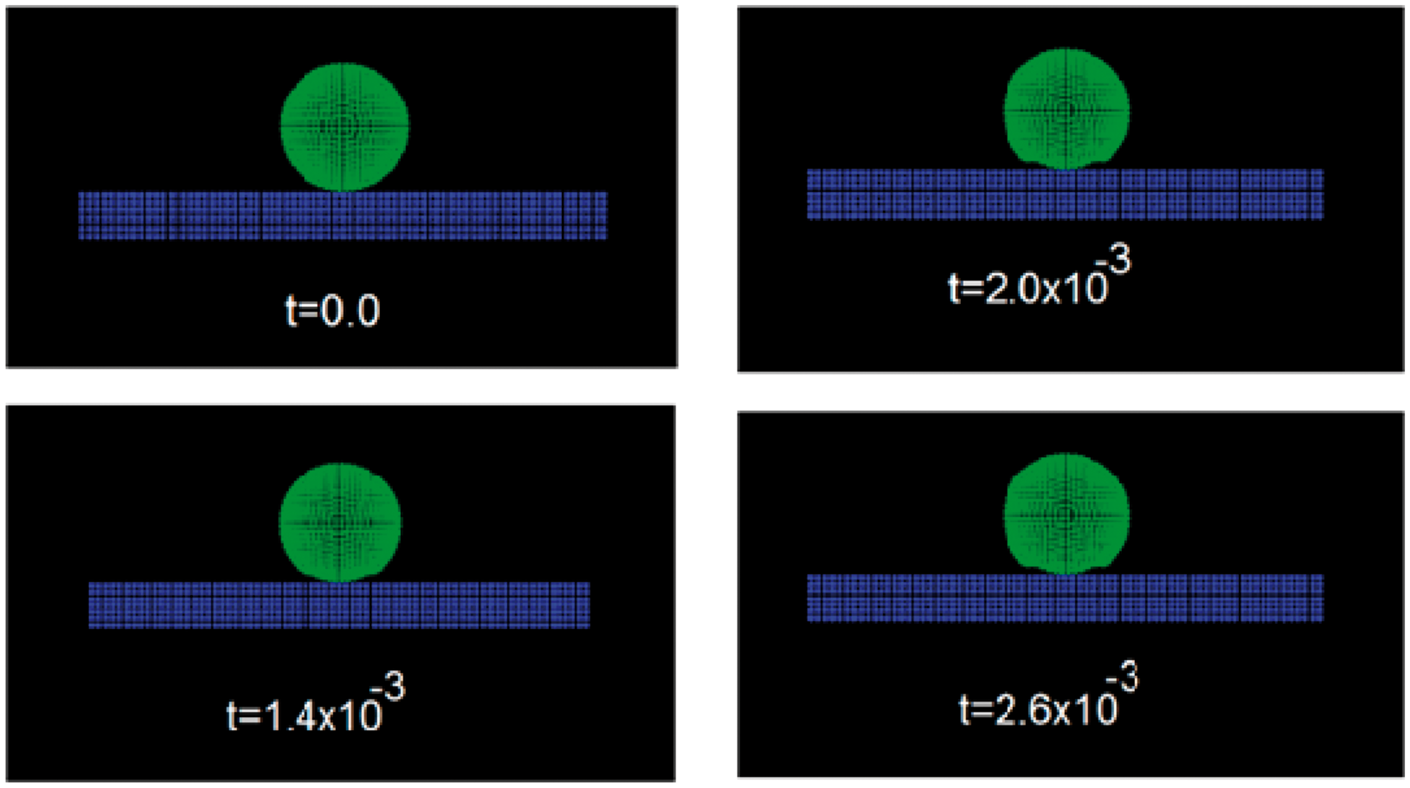
Conclusions
In this work was proposed a hydrodynamic formalism in order to obtain an approximate numerical solution to the Navier–Stokes equations. This formalism consists in the interpolation of the properties of the system of particles. The Smoothed Particle Hydrodynamics formalism was employed and this method is very useful when the system exhibits big deformation and large velocities because it is mesh-free in nature. In this study it was simulated for the first time the condensation and formation of a spherical van der Waals drop by means of particles in three-dimensional space with the SPH formalism. When the drop is formed, starting from a gas box, the distribution of density implies that there are zones where the mass of gas is converted to a liquid state, but out of the droplet radius, some particles remain in the gas form.
On the other hand, two tests are presented with the drop obtained in the first calculation. In these calculations can be seen that there are two regimes for the collision process of the droplet with the solid surface. If the inertial forces prevail, the drops are converted in a thin liquid film, but if the surface tension forces prevail the drop only deforms its surface with a certain contact angle with respect to the aluminium surface.
When the inertial forces prevail, the velocities around the contact zone between the drop and the solid surface are bigger than the initial velocity and the opposite behavior is obtained when the surface tension forces prevail in the model.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest withrespect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.