Abstract
Background
An antimicrobial stewardship programme (AMS) aims to combat antimicrobial resistance and healthcare-associated infections (HCAIs). While studies in developed countries show AMS’s effectiveness in reducing AMR and HCAIs, its impact in African countries, given differing socioeconomics, remains unclear.
Objectives
To review the impact of AMS on HCAIs, antibiotic prescriptions, cost of antimicrobial procurement, and compliance with diagnostic measures for detecting resistant HCAIs in African countries.
Methods
Two reviewers (S.S. and U.C.) searched databases like CINAHL, Medline, and PubMed for studies on AMS interventions in African countries, focussing on their impact on healthcare-associated infections (HCAIs) and antibiotic prescriptions. We excluded studies on outpatients, children, or those not in English and conducted a meta-analysis using data collected from changes in HCAIs before and after intervention using a random-effects model.
Results
The search identified 1153 studies, of which 14 were included in the review, while four eligible studies were included in the meta-analysis. Thirteen of the 14 studies were designed using pre- and post-study methods, and one study employed a case-control method. AMS interventions effectively reduce antibiotic consumption, the cost of antibiotic procurement, and improve diagnostic measures for the detection of resistant microorganisms. The forest plot suggested a 34% reduction in HCAIs.
Conclusions
In Africa, AMS interventions, whether combined or single, reduce HCAIs and antibiotic prescriptions in healthcare settings. Surgical antibiotic prophylaxis lowers HCAIs in hospitals by one-third. However, findings are cautiously generalised due to the varied quality of studies and the limited number of African countries involved.
Keywords
Introduction
Healthcare-associated infections (HCAIs) are infections caused by pathogenic organisms which are acquired by patients in the process of receiving care for other illnesses within hospitals, community healthcare centres, clinics, or care homes that appear within 48 hours to 30 days after receiving care (Revelas, 2012; Haque et al., 2018; World Health Organisation [WHO], 2018).
HCAIs include ventilator-induced pneumonia, surgical site infections, bloodstream infections, and urinary tract infections (Al-Tawfiq and Tambyah, 2014). Ventilator-associated pneumonia is a lung infection that affects a ventilator user (Centres for Disease Control and Prevention [CDC], 2025). A central line-associated bloodstream infection develops when a bacteria or virus enters the bloodstream through the central line (CDC, 2025). A urinary tract infection is an infection of the urinary tract system, including the urethra, ureter, kidney, and bladder (CDC, 2025). Also, a surgical site infection develops when bacteria enter the human body through an opening in the surgical site (CDC, 2025). Antimicrobial-resistant microorganisms are often the causes of HCAIs, leading to longer hospital stays, disability, and increased cost of healthcare services (Ahmed et al., 2021; Benammar et al., 2018; WHO, 2019). These microorganisms acquired these resistance traits because of repeated exposure to suboptimal doses of antimicrobials (Al-Tawfiq and Tambyah, 2014).
Several studies have demonstrated disparities in the distribution of healthcare-associated infections (HCAIs) across high-income countries (HICs) and low- and middle-income countries (LMICs). Due to insufficient record-keeping in many African countries, an accurate estimate of the global burden of HCAIs is challenging. However, studies have shown that for every 100 hospitalised patients in a healthcare setting, an estimated 7% of patients in HICs and 10% in LMICs will acquire at least one type of HCAIs (Cassini et al., 2016; Danasekaran et al., 2014; Haque et al., 2018, 2019)
Additionally, a study by Haque et al. (2018) states that 2,609,911 new cases of HCAIs occur in the European Union (EU) annually, leading to approximately 2,506,091 DALYs (Disability-Adjusted Life Years) yearly. DALY can be defined as the sum of life lost due to premature mortality and the years lived with a disability due to a health condition (Cassini et al., 2019).
Importantly, studies from two systematic reviews concluded that there is a high risk of patients acquiring HCAIs in healthcare centres in LMICs (Arabi et al., 2008; Zaidi et al., 2005). Also, a systematic review by Tadesse et al. (2017) on antimicrobial resistance in Africa shows that 40% of African countries do not have data on antimicrobial resistance (AMR), and the resistance level to commonly prescribed antibiotics is significant. Subsequently, during the 2014 Sixty-Seventh World Health Assembly Meeting in Geneva, Switzerland, antimicrobial stewardship (AMS) was adopted as one of the five global responses to contain AMR and HCAIs (WHO, 2014; WHO, 2015).
The Infectious Disease Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA) defined AMS as ‘coordinated interventions designed to improve and measure the appropriate use of antimicrobial agents by promoting the selection of the optimal antimicrobial drug regimen including dosing, duration of therapy, and route of administration’ (IDSA and SHEA, 2012). The AMS programme can be categorised into two core interventions: 1. Prospective audit with intervention and feedback and 2. Formulation restriction with pre-authorisation. However, there are other supplementary AMS interventions such as de-escalation of therapy based on culture results, dose optimisation, intravenous to oral medication switch, education, antimicrobial order forms, antimicrobial cycling, combination antimicrobial therapy, procalcitonin, and antibiograms (BSAC, 2023; Dellit et al., 2007). Additionally, based on the IDSA/SHEA AMS establishment guidelines, effective AMS programmes involve monitoring of antibiotic use and collaboration with various hospital committees such as the infection prevention and control (IPC) team, drug and therapy team, and safety committee. This is important because AMS programmes are multidisciplinary, requiring effective expert commitment and collaboration. Hence, the guidelines emphasise the importance of a multidisciplinary AMS team comprising an infectious diseases physician, a clinical pharmacist trained in infectious diseases, a clinical microbiologist, an information system expert, an IPC expert, and an epidemiologist (Dellit et al., 2007).
Furthermore, several studies have also demonstrated the positive impact of AMS interventions on HCAIs and AMR. For instance, studies by Moffa et al. (2018), Mijović et al. (2018); Aldeyab et al. (2012) concluded that the rate of Clostridium difficile infections significantly reduced during the implementation of AMS interventions. However, all of these studies were from developed countries. Similar studies focussing on the African context did not concentrate on HCAIs and antibiotic prescription in adult inpatients. Nevertheless, given the variability and difficulties in implementing AMS interventions, it is currently unknown what impact various AMS interventions have on the rate of HCAIs and antibiotic prescriptions among adult inpatients in African countries. To provide more information on AMS effectiveness, this systematic review was conducted to identify and evaluate the impact of different AMS interventions on HCAIs and antibiotic prescription among adult inpatients in African countries.
Methods
This systematic review and meta-analysis were conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Page et al., 2021). The completed PRISMA checklist is included as Supplemental Material in Appendix 1. The review protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) Database.
Databases and search strategy
Search terms and strings.

AMS, antimicrobial stewardship; HCAIs, healthcare-associated infections.
Eligibility criteria
Studies describing the impact of AMS interventions in healthcare settings in African countries were included. The inclusion criteria based on the Population, Intervention, Comparison, Outcome and Study design (PICOS) for the systematic review and meta-analysis were as follows: - Population: Adult patients in African countries and 18+. - Intervention: Antimicrobial stewardship interventions in hospitals, healthcare or care home settings such as pre-authorisation of named anti-infectives, prospective audits and feedback, de-escalation of therapy based on culture results, dose optimisation, intravenous to oral medication switch (switching medications from injectables to oral form), education, antimicrobial order forms, antimicrobial cycling, combined antimicrobial therapy, enhanced surveillance, procalcitonin, and drug prescription guidelines and antibiograms. - Comparison: AMS pre- and post-implementation, before/after intervention, control and intervention group, and standard of care before AMS implementation. - Outcome: Care-related outcomes: Change in rate of HCAIs or reduction in antimicrobial consumption or prescribed (i.e. defined daily doses and number of prescriptions or costs) Clinical and ecological outcomes: Change in the rate of resistant HCAIs detected.
We excluded studies that did not target antimicrobial optimisation (i.e. infection control strategies), targeted outpatients, and focused on the paediatric population. Others were studies conducted outside Africa, and those that did not report relevant outcome measures. We also excluded studies not written in English, conference papers, reviews, abstracts, and studies published before 1 January 2013.
Study selection
Two reviewers (S.S. and U.C.) independently screened the deduplicated titles and abstracts against the eligibility criteria. Abstracts were considered for full-text review if they fulfilled all the requirements, and those not conforming to the eligibility criteria were excluded. Where there were discrepancies, the two reviewers discussed and compared results to resolve the disagreement. However, where no resolution was reached, studies were sent to the third (J.S.) reviewer. Conversely, when uncertainty remained, studies were carried forward to the full-text review. The full text of the primary studies that remained after the title and abstract screening was sourced and downloaded into a Reference Manager, EndNote (version 20). When the full text of any study could not be sourced from the library databases, the authors of the papers were emailed to assist in retrieving the full text. Two reviewers (S.S. and U.C.) read the full texts of the retrieved studies independently and assessed whether they met the inclusion and exclusion criteria. Any disputes between reviewers were sent to the third (J.S.) reviewer for resolution in discussions where consensus was sought.
Data extraction
A modified data extraction form for this study was created to collect data from the selected studies. The following data were extracted by two reviewers (S.S. and U.C.): author’s name, year of publication, study location, study design, participants’ characteristics (age, gender, sex, etc.), intervention details, target illness (from the primary study result), study duration, data summary (gotten from the results), outcomes (risk ratios, odd ratios, risk difference, mean difference, etc.), effect size (with confidence interval), p-value, and study conclusion Appendix 3. The outcomes measurement relevant to the AMS included identifying (a) changes in the rate of HCAIs, (b) a reduction in the number of antimicrobials prescribed, (c) changes in the rate compliance measures in the diagnosis detection of resistant HCAIs, and (d) a decrease in the cost of procurement of antimicrobials pre- and post-intervention. Also, the two reviewers (S.S. and U.C.) extracted the data for the meta-analysis, while any discrepancies were reconciled by discussion and consensus.
Quality assessment
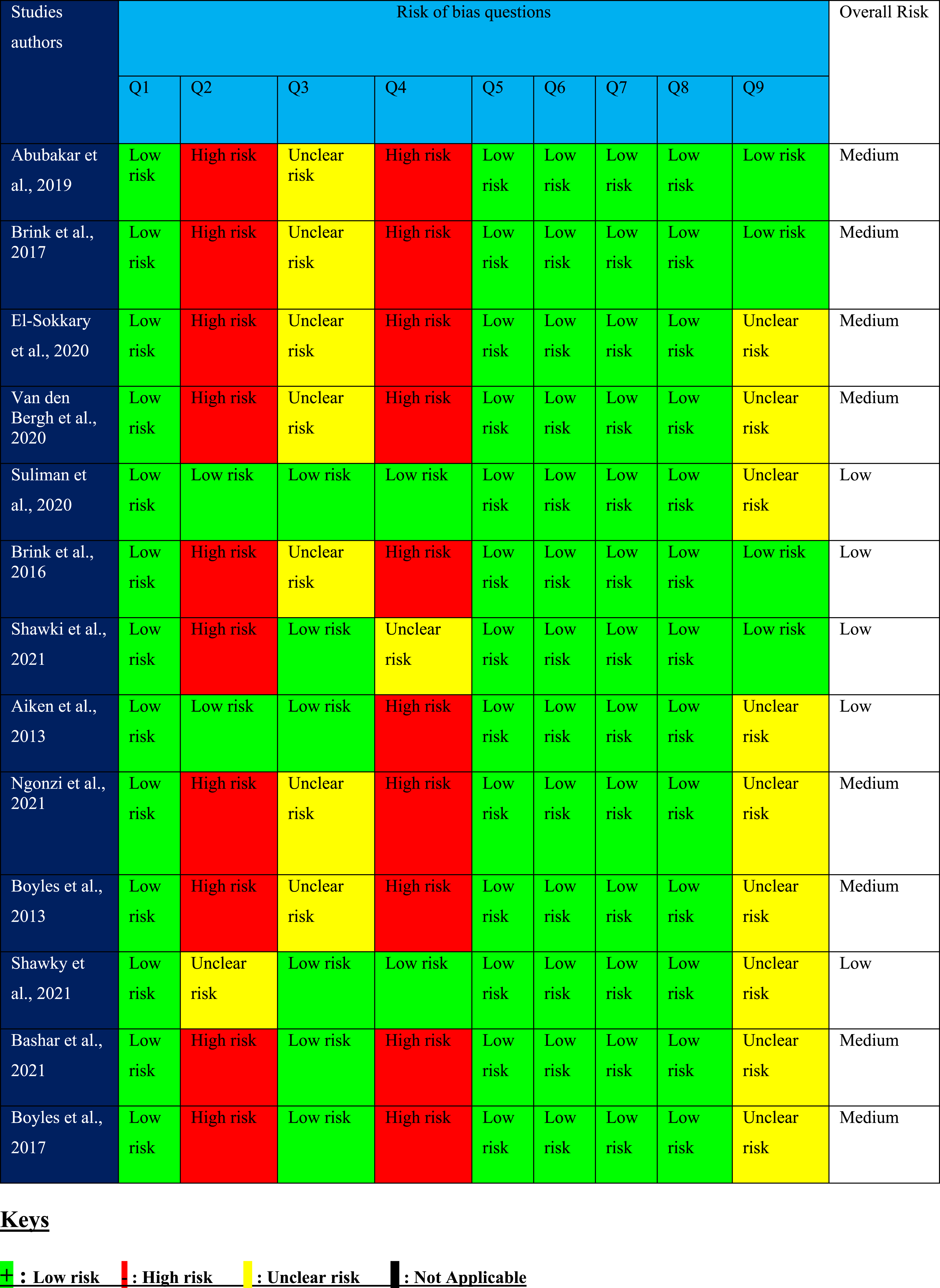
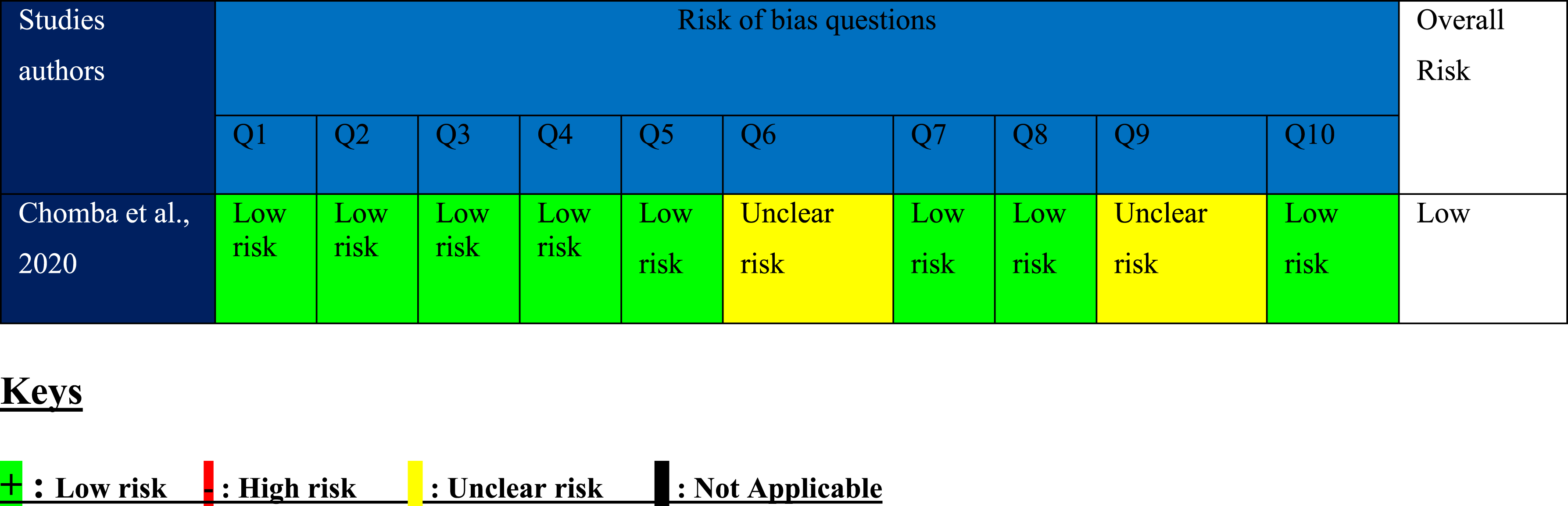
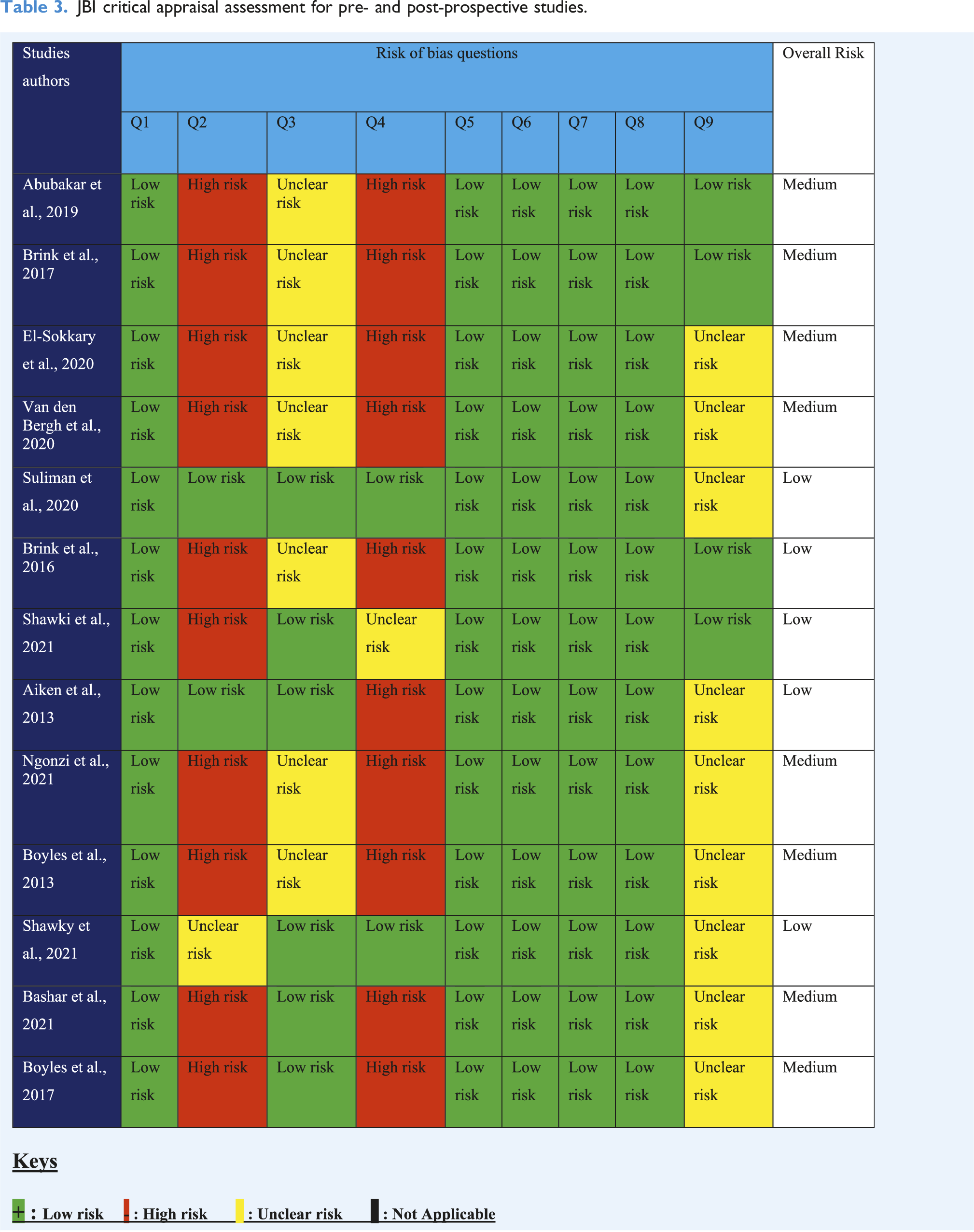
The risk of bias (RoB) for quasi-experimental and case-control studies was assessed according to the Joanne Briggs Critical Appraisal Tools (JBI) or guidelines (Barker et al., 2023). The studies were assessed by two reviewers (S.S. and U.C.), and any disagreement in the quality assessment was resolved by discussion with the third reviewer (J.S.). The JBI tool for assessing the quasi-experimental study is in the form of a chart Appendix 4, as well as that of a case-control study Appendix 5. The tools consist of questionnaires with nine and 10 questions for quasi-experimental and case-control studies. Each question for quasi-experimental and case-control studies can be mapped to the domains of bias as specified in the Cochrane RoB2 tool. These domains of bias include selection, attrition, performance, and detection bias (Barker et al., 2023). The questionnaires can be answered ‘yes’, ‘no’, ‘unclear’, or ‘not applicable’. If the answer to a question is ‘yes’, then that study is at ‘low risk’ of bias for that question. A ‘no’ answer indicates the study is at ‘high risk’ of bias. Additionally, if the answer is unknown, the study has an ‘unclear risk’ related to that question.
The overall risk assessment follows the rules: (a) Studies with less than two high and one unclear risk will have a ‘low’ overall risk. (b) Studies with two high risks and one unclear risk and above will have an overall risk ‘medium’. (c) Studies with the above three high risks will have an overall risk ‘high’.
Statistical analysis
The outcomes measurement relevant to the AMS included identifying (a) changes in the rate of HCAIs, (b) a reduction in the number of antimicrobials prescribed, (c) changes in the rate of diagnosis of resistant HCAIs, and (d) a decrease in the cost of procurement of antimicrobials pre- and post-intervention. The study characteristics (publication date, study locations, intervention details and target illness) were analysed using descriptive statistics and summarised in a table. Studies that reported the same outcomes were pooled and a meta-analysis was performed using the random-effect model and Mantel-Haenszel analysis. For each study, the Odds Ratios (O.R.) were calculated using the change in rate of HCAIs reported for both intervention and control groups. The pooled estimates were presented as a Forest plot with a 95% confidence interval (CI). P-values were presented as reported in the studies, and values <0.05 were considered significant. The I2 test was used to assess the statistical heterogeneity, with values less than 40% representing low heterogeneity, those between 40% and 60% moderate heterogeneity, and values greater than 60% high heterogeneity (Higgins et al., 2003). The meta-analysis was performed using the Review Manager software version 5.4 (Cochrane Collaboration, 2020). However, subgroup analysis was not performed because of insufficient data.
Results
Search results and study selection
The initial search retrieved 1,133 studies relevant to AMS and HCAIs from the databases, which were exported into the EndNote software (version 20). An additional 20 studies that met the eligibility criteria were found from references of similar systematic reviews, totalling 1153 studies. Twelve identified articles were removed as duplicates from EndNote, narrowing down the total to 1141. A further 1,084 studies were excluded following title and abstract screening because they did not meet the eligibility criteria. However, 12 of the studies were not retrievable due to journal subscription and restrictions. The remaining 45 studies were evaluated for eligibility by reading the full articles. Thirty-one studies were excluded because of the different study designs, interventions, and outcomes that did not meet the inclusion criteria. Fourteen studies that met the inclusion criteria formed the dataset for this systematic review Figure 1. PRISMA flow diagram.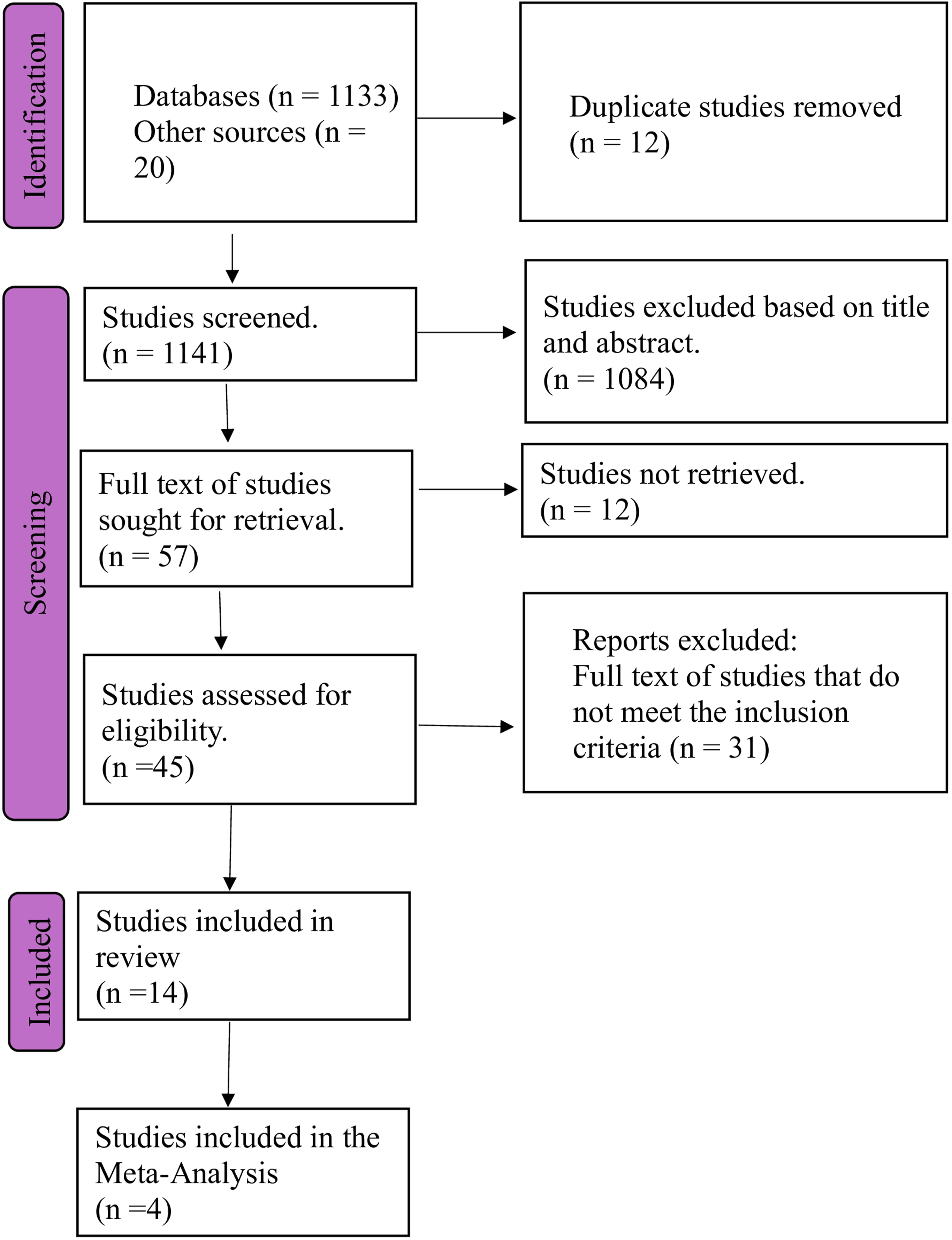
Characteristics of the included studies
Types of study
See Supplemental Material Appendix 6 shows the types of studies; one of the studies was a prospective case-control, and the remaining 13 were quasi-experimental studies (pre- and post-prospective). The case-control study was from Chomba et al. (2020), while the remaining 13 studies were from Shawki et al. (2021); Brink et al. (2016); Suliman et al. (2020); Boyles et al. (2013); Boyles et al. (2017); Bashar et al. (2021); Brinks et al. (2017); Abubakar et al.., 2019; El-Sokkary et al. (2020); Ngonzi et al. (2021); Shawky Abdelgawaad et al. (2021); Van Den Bergh et al. (2020); Aiken et al. (2013).
Study location, setting, and participants
Studies location, settings, and participants.
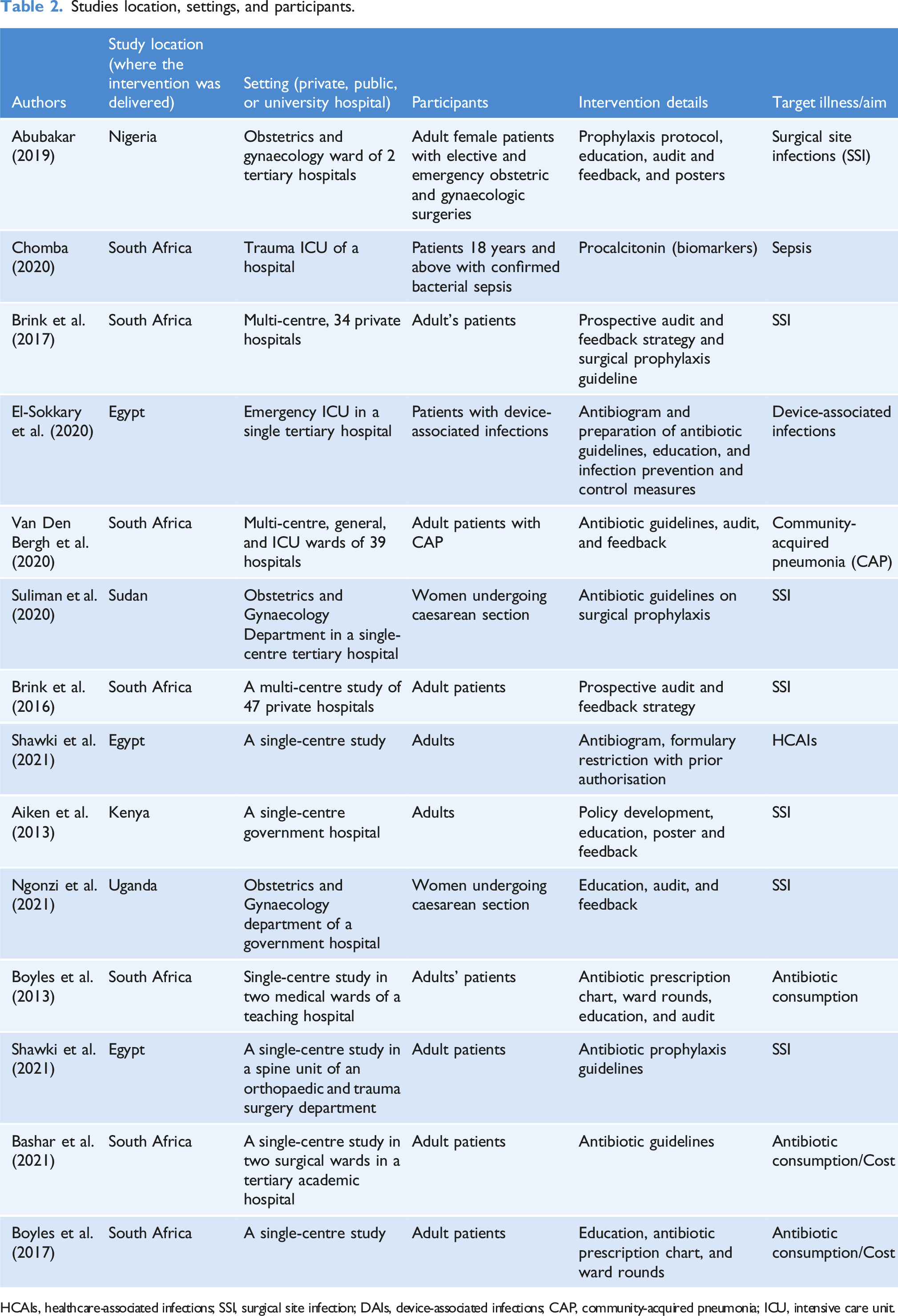
HCAIs, healthcare-associated infections; SSI, surgical site infection; DAIs, device-associated infections; CAP, community-acquired pneumonia; ICU, intensive care unit.
Quality assessment
JBI critical appraisal assessment for pre- and post-prospective studies.
JBI critical appraisal assessment for case-control study.

AMS intervention
AMS interventions used by the selected studies.

Meta-analysis results
Data from studies included in the meta-analysis.
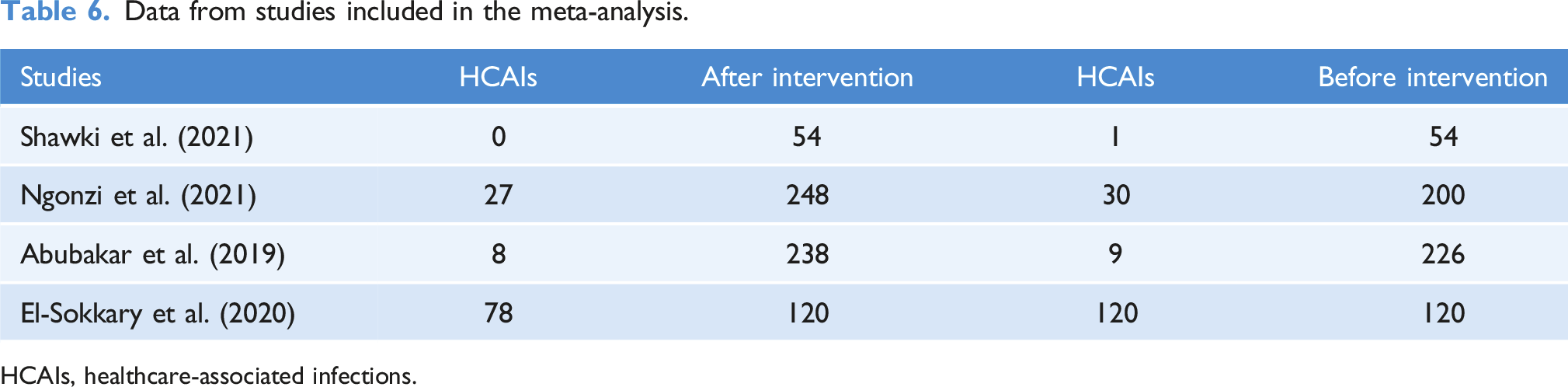
HCAIs, healthcare-associated infections.
Impact of AMS on healthcare-associated infections
The review indicates that AMS can have a positive impact on reducing the rate of HCAIs (Abubakar et al., 2019; Brink et al., 2017; El-Sokkary et al., 2020; Ngonzi et al., 2021; Shawki et al., 2021). For stances, Abubakar et al. (2019) show that the used AMS bundle decreases the rate of HCAIs (4.0%–3.4%, p-value = .722) in 8 months. Brink et al. (2017) also show that the AMS bundle decreases the rate of mean HCAIs (2.46 to 1.97%, CI = 1.27–2.15, p-value = .0029) in 30 months. Similarly, El-Sokkary et al. (2020) also show that the AMS bundle conducted in an ICU ward decreases HCAIs from 121 to 78 in 17 months. Furthermore, Ngonzi et al. (2021) also reported a decrease in HCAIs from 14.5% to 10.5%, p-value = .198 in 3 months. Likewise, a study by Shawki et al. (2021) that used a single AMS intervention (guideline) showed a decrease of 1.85% in HCAIs within 3 months. All these studies indicated that single or AMS bundle interventions positively impact HCAIs. Subsequently, a meta-analysis was conducted by pooling four of the studies’ risk ratios. The meta-analysis overall pooled effect was 0.66 (CI = 0.58–0.75), with heterogeneity (I2) = 0% and tau (ԏ2) = 0% and p-value = .89, as shown in Figure 2. Forest plot of comparison: AMS versus no-AMS, outcome: HCAIs.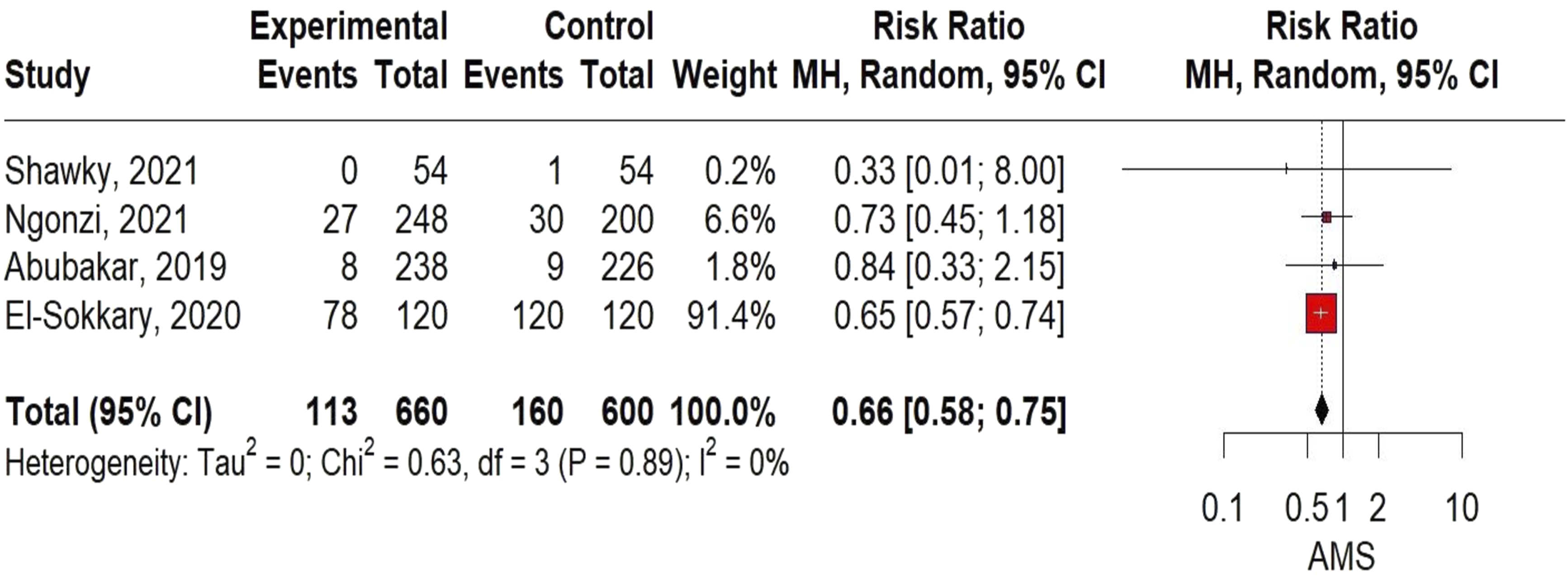
Impact of AMS on antibiotic consumption
Similarly, the review also indicates that single or combined AMS interventions positively impact antibiotic consumption in Africa (Abubakar et al., 2019; Bashar et al., 2021; Boyles et al., 2013, 2017; Brink et al., 2016; Chomba et al., 2020; Shawki et al., 2021; Suliman et al.., 2020). For instance, Abubakar et al. (2019) study shows that the AMS intervention decreases antibiotic consumption from 16.6 to 12.8 DDD/procedure, p-value = .001 in 7 months. Likewise, Shawki et al. (2021) also show that the use of an AMS bundle decreases antibiotics consumption from 75.1 DDD/100 bed days to 64.65 DDD/100, p-value = .001 in 19 months. Furthermore, Boyles et al.'s (2013) study also shows that using an AMS bundle decreases antibiotic consumption from 592.0 DDD/1000 to 475.8 DDD/1000 inpatient days in 12 months. Although all these studies have different durations, all indicated a positive impact on antibiotic consumption. Boyles et al. (2017) also showed that the use of an AMS bundle decreases antibiotic consumption from 1046 DDD to 868 DDD/1000 patient days in 60 months. On the other hand, studies using single AMS intervention, such as Chomba et al. (2020), reported an increase in antibiotic-free days from 3.8 to 7.7 days after the intervention, p-value = .10. A similar study was conducted by Suliman et al. (2020) using a single AMS intervention, which also showed decreases in antibiotic consumption. Likewise, a study by Brink et al. (2016) also shows a decrease in antibiotic consumption from 101.38 DDD/100 to 83.04 DDD/100 patient days, p-value 0.0001. Finally, a study by Bashar et al. (2021), which used a single AMS intervention, also shows a decrease in antibiotic consumption from 739.30 DDD/1000 to 564.93/1000 patient days in 12 months. Although most of these studies used different metrics of measurement like patient days and bed days, as well as DDD, all indicated decreases in antibiotic consumption as shown in Appendix 6.
Impact of AMS on the cost of procurement of antibiotics
Four of the studies show that single and combined AMS intervention reduces the cost of antibiotics (Abubakar et al., 2019; Aiken et al., 2013; Boyles et al., 2013; Suliman et al., 2020). For example, Abubakar et al.'s (2019) study used an AMS bundle, which shows a differential decrease in the cost of antibiotic procurement by $4.2/procedure, p-value = .001 in 8 months. Additionally, Aiken et al.'s (2013) study used a single AMS intervention and showed a differential decrease in the cost of prophylactic antibiotics by $2.50/operation in a period of 17 months. Boyles et al.'s (2013) study used an AMS bundle (Antibiotic prescription chart, education, and audit) and showed a decrease in the cost of antibiotics from R 1,068,325 to R 694,705 in 12 months. Likewise, Suleiman, Yousef, and Hamadelni’s study (2020) employed a single AMS intervention (antibiotic guidelines of surgical prophylaxis) and showed a reduction in the cost of prophylactic antibiotics from SP735 to SP505/prescription in 3 months. Even though these studies were from different countries and used different AMS interventions, they all indicated a reduction in the cost of antibiotics after implementing AMS, as shown in Appendix 6.
Impact of AMS on rate of diagnosis of resistant microorganisms
The review also indicates an increase in compliance with diagnostic measures for detecting resistant microorganisms. For instance, a study by Van Den Bergh et al. (2020), which used an AMS bundle (Antibiotic guidelines, audit, and feedback), showed an increase in compliance with diagnosis measures for the detection of resistant microorganisms from 49.1% to 54.6% (p-value, 0.0001) in 12 months. Although this study is statistically significant, there is a need to be cautious about its significance as it is a single study.
Discussion
Summary of findings
This systematic review and meta-analysis assessed the effect of AMS interventions in Africa on HCAIs and antibiotic prescriptions. Many of these AMS interventions have significant effects on these specific outcomes. AMS intervention focussing on preoperative prophylaxis demonstrated the impact of AMS in reducing HCAIs. Through the meta-analysis, we were able to reveal that implementation of AMS intervention could decrease HCAIs (Abubakar et al., 2019; El-Sokkary et al., 2020; Ngonzi et al., 2021; Shawki et al., 2021). As seen in the result of the meta-analysis, the pooled estimate was 0.66, with a CI ranging from 0.58 to 0.75. Additionally, the I2 statistic was used to test for heterogeneity with (I2) = 0%, tau (ԏ2) = 0%, and p-value = .89, indicating that the included studies are not different from each other, and meta-analysis can be performed. This finding suggests that AMS interventions (preoperative prophylaxis) can lead to a modest change (decrease) in the rate of HCAIs in Africa.
Additionally, the review’s findings also suggest that implementing a single AMS intervention or bundle substantially decreased antimicrobial consumption. Some of the studies that used AMS bundle and those that used a single AMS intervention also show a decrease in the consumption of antimicrobials and prescriptions, which indirectly reduces the cost of antibiotics in healthcare centres. Finally, a study by Van Den Bergh et al. (2020), which used AMS bundle, also showed an increase in compliance with diagnosis measures for the detection of resistant organisms after implementing guidelines within a healthcare setting.
Similarities and differences of findings with previous research
Four studies that combined AMS interventions (Abubakar et al., 2019; Brink et al., 2017; El-Sokkary et al., 2020; Ngonzi et al., 2021) and a study by Shawki et al. (2021) (single AMS intervention) all reported on HCAIs. However, only one was significant (p-value = .0029), while the others were not significant (p-value = .722 and .198). The significance that Brink et al. (2017) reported could be due to the long duration of the study (29 months), while Abubakar et al. (2019) and Shawki et al. (2021) have a short duration (6 months each) as the length of study could also affect the significant of a result. However, the Meta-analysis conducted involved four eligible studies (Abubakar et al., 2019; El-Sokkary et al., 2020; Ngonzi et al., 2021; Shawki et al., 2021) with an overall pooled effect size of 0.66, showing that AMS reduced the risk of HCAIs by 34% with CI (0.58–0.75). Nevertheless, with this CI, the reduction in HCAIs ranges from 25% to 42%. Also, with I2 = 0 and tau2 = 0, this indicated that the studies were not heterogeneous or different from each other. Additionally, these findings align with previous reviews from Siachalinga et al. (2022) and Honda et al. (2017) on HCAIs since the reviews from Siachalinga et al. (2022) and Honda et al. (2017) reported positive effects of AMS in reducing HCAIs and antibiotic consumption. In terms of comparison of the review findings with studies conducted in HICs, this review’s findings were comparable with a similar study in North America (Monmaturapoj et al., 2021). The Monmaturapoj et al., 2021 study findings show a reduction in the duration of antimicrobial therapy, mortality, hospital stays and HCAIs, which is similar to this review. Hence, this indicates that AMS decreases the rate of HCAIs.
Additionally, four studies that combined AMS interventions (Abubakar et al., 2019; Boyles et al., 2013, 2017; Shawki et al., 2021), and four other studies that used a single AMS intervention (Bashar et al., 2021; Brink et al., 2016; Chomba et al., 2020; Suliman et al., 2020), reported a decrease in the rate of antibiotic consumption. The duration of all the selected studies ranges from 6 months to 40 months, as the duration could affect the significance of a study. However, five of the studies reported a p-value, while three did not. However, three of the p-values (p-values = 0.001, 0.0001, and 0.0001) were significant, while one (p-value = .10) was not significant. This indicated that AMS decreases antibiotic consumption, which aligned with a similar study by Van Dijck, Vlieghe, and Cox (2018) and Monmaturapoj et al. (2021). This also suggests that AMS bundles are more effective than a single AMS intervention. Therefore, implementing two or more AMS interventions in a health institution would be effective.
Likewise, three studies that combined AMS intervention (Abubakar et al., 2019; Aiken et al., 2013; Boyles et al., 2013) and a study that used a single AMS intervention (Suliman et al., 2020) reported on the effect of AMS on the cost of antibiotics procurement. However, only one of the studies reported a p-value = .001, which is significant. All three other studies only indicated a reduced cost of antibiotics consumed. This indicated that the implementation of AMS interventions reduced the cost of prescribed antibiotics. The studies also suggested a reduction in the number of antibiotics prescribed, indicating a reduction in antibiotic consumption as reported in a similar study by Van Dijck, Vlieghe, and Cox. (2018). However, there is a need for future research to focus on studies using a single AMS intervention to determine the AMS intervention responsible for such an effect.
Lastly, a single study by Van Den Bergh et al. (2020) that used a combined AMS intervention also reported a significant increase in the diagnosis rate of resistant microorganisms with a p-value = .0001. This finding from this study also aligned with a similar study by Van Dijck, Vlieghe, and Cox (2018) in LMICs. However, more research is needed in this field, as the evidence was based on a single study conducted in South Africa.
Practical implications for policy
The high level of poverty, limited resources, and the high burden of infectious diseases in African countries underscore the need for the AMS programme (Tadesse et al., 2017; Abo et al., 2022; Mzumara, Mambiya, and Iroh Tam, 2023). Consequently, the implementation of an effective AMS programme is essential for achieving WHO’s SDGs 1 and 3. SDG 1 relates to the eradication of poverty (end poverty in all its forms everywhere), and SDG 3 relates to good health and well-being (ensure healthy lives and promote well-being for all at all ages). As this review has shown that single AMS interventions can be effective, countries in Africa can adopt and adapt single AMS interventions to reduce HCAIs, such as surgical perioperative prophylaxis (Abubakar et al., 2019; Brink et al., 2017; El-Sokkary et al., 2020; Ngonzi et al., 2021; Shawki et al., 2021). This may be a cost-effective starting point for introducing AMS, which can subsequently be upgraded in combination with other supplemental AMS interventions, such as procalcitonin, antibiograms, and the two main core interventions. Implementation of the AMS intervention (surgical perioperative prophylaxis) is recommended as a starting point, as it is evident that the intervention was effective in reducing HCAIs. However, implementation should be based on the hospital’s capability and resources because AMS is multidisciplinary and needs commitment from the AMS team. Additionally, sensitisation and educating healthcare workers on the consumption of antibiotics could go a long way in reducing the use of antibiotics. Government and hospitals need to enforce antibiotic surgical prophylaxis guidelines in all hospitals, combined with hospital infection prevention and control programs.
Future studies
Future studies should focus on randomised control trials, quasi-experimental research with control, and time series study design because of their high quality and the need to establish different hospital contexts where the studies were done. This will help to strengthen the evidence base in this field. Additionally, most of the included studies were from six (11%) out of 54 African countries, with seven studies (50% of the review dataset) derived from South Africa. This indicated that the current review is limited in its ability to represent the impact of AMS on the Continent of Africa. More research needs to be conducted on the impact of AMS in different African regions to help strengthen the quality of future research on a systematic review and meta-analysis of AMS in Africa.
Strength of the review
A key strength of this review is that it followed a systematic process to identify and select relevant literature so that the evidence is based on a comprehensive understanding of the research area. Additionally, the included studies were evaluated for quality. The assessment showed that the studies were rated medium to high quality. On this basis, none of the 14 selected studies were rejected. These studies included several different AMS interventions, such as prospective audit and feedback, antibiogram, formulation restriction and pre-authorisation, antimicrobial guideline, education, antibiotic prescription form, pre-authorisation, and procalcitonin, in the hopes that it might be generalised across all of Africa. The inclusion criteria limited the selected studies to 10 years, and the study designs were quasi-experimental designs and case-control studies, which allowed for an analysis of recent and high-quality studies. Lastly, the review was undertaken by three reviewers to minimise researcher bias.
Limitations of the review
Firstly, this review was limited by the search strategy, which identified only English language articles from several databases. This might have resulted in missing articles, which may exist but are written in a language other than English. This could produce selection and publication bias in the resulting dataset. Also, this was a decade review of studies published from 2013 to 2024. Any relevant articles published before 2013 have not been identified, reducing the evidence database.
Secondly, the review only focused on the impact of AMS interventions on HCAIs, antibiotic prescription and cost. Hence, only outcomes related to the review’s objectives were extracted and synthesised. This may have meant that other impacts of AMS have not been fully considered.
Thirdly, the empirical studies were all undertaken in different hospital contexts and wards, for example, Abubakar et al. (2019) and Suliman et al. (2020) were in O & G. Brink et al. (2016), a multi-centre study and many other studies were conducted in different contexts. Subsequently, different treatment and visitation regimens would operate in each context, which may have affected the results. Therefore, more context standardisation is needed to better understand AMS interventions impact better.
Fourthly, as some of the AMS interventions were combined, it was not always possible to disentangle the effects of specific types of intervention or the combinations of specific interventions to understand their role in reducing HCAIs. More research is needed to focus on AMS interventions in Africa using single interventions or specific combinations. This would enable the positive (and negative) impact of AMS interventions on HCAIs or antibiotic prescriptions to be fully understood.
Conclusion
This review’s findings indicate that both single and combined Antimicrobial Stewardship (AMS) interventions have a positive effect on reducing antibiotic consumption and healthcare-associated infections (HCAIs). However, while some studies showed significant results, others exhibited considerable heterogeneity. Notably, the studies included in this review were limited to only six African countries, representing just 11% of countries on the African continent, and nearly 50% of the studies were conducted in South Africa. One effective AMS intervention, the use of prophylactic surgical antibiotics, was found to reduce HCAIs by approximately one-third (34%) in healthcare centres across African countries. Additionally, combined AMS interventions (as noted in studies by Abubakar et al., 2019; Brink et al., 2017; El-Sokkary et al., 2020; Shawki et al., 2021; Boyles et al., 2013; Boyles et al., 2017) proved to be more effective than single AMS interventions (as highlighted in studies by Shawki et al., 2021; Chomba et al., 2020; Suliman et al., 2020; Brink et al., 2016; Bashar et al., 2021) in reducing both the rate of HCAIs and antibiotic prescriptions among adult inpatients in African nations. However, these findings should be generalised with caution due to the quality of the included studies, the predominance of studies written in English, the varying intervention settings, the different combinations of interventions employed, and the limited representation of African countries.
Supplemental Material
Supplemental Material - Impact of antimicrobial stewardship on healthcare-associated infections and antibiotics prescription in African countries: Systematic review and meta-analysis
Supplemental Material for Impact of antimicrobial stewardship on healthcare-associated infections and antibiotics prescription in African countries: Systematic review and meta-analysis by Sikiru Shuayb, Judith Sixsmith, and Chika Robinson Okoyeuche in Journal of Infection Prevention.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.