Abstract
Background
Conflicting guidance exists regarding the characterization of nebulization as an aerosol-generating procedure and subsequent risk of transmission of SARS-CoV-2 to healthcare workers.
Aim/Objective
This study sought to address whether SARS-CoV-2 viral load distribution was impacted by bronchodilator delivery via nebulizer versus metered-dose inhaler (MDI).
Methods
Adults infected with COVID-19 were enrolled and received a single dose of albuterol sulfate nebulized solution (2.5 mg/3 mL via breath-actuated nebulizer with filtered mouthpiece) or albuterol sulfate hydrofluoroalkane inhalation aerosol (90 µg/actuation; two puffs via MDI with spacer) in a randomized crossover sequence. Air and surfaces were sampled at predefined locations within patients’ hospital rooms to assess SARS-CoV-2 dispersion over three periods (baseline, MDI, and nebulizer).
Findings/Results
Eleven patients received crossover therapy. Six patients had detectable SARS-CoV-2 RNA during one treatment period (n = 3 each for MDI and nebulizer) and one during both treatment periods. No viral RNA was found in the rooms of four (36.4%) patients. Overall, few environmental samples (17/397; 4.3%) contained detectable viral RNA, with no meaningful differences in positivity rate across periods; RNA genome copy numbers were low in positive samples. No correlation between dispersion and patient clinical status or environmental parameters was observed.
Discussion
In this first prospective trial evaluating viral load distribution following use of nebulizer versus MDI in hospitalized patients with COVID-19, low environmental contamination was found regardless of administration method. Findings support the use of either device when needed to treat patients with COVID-19.
Background
The Centers for Disease Control and Prevention (CDC) and World Health Organization classify the administration of nebulized treatments as an “uncertain” aerosol-generating procedure that may be infectious based on limited evidence (Centers for Disease Control and Prevention, 2022a; World Health Organization, 2021). The emergence of COVID-19 has led to concerns that nebulizer therapy may increase the risk of SARS-CoV-2 transmission. These concerns are due in part to a case series from Hong Kong suggesting that nebulizer treatments facilitated nosocomial spread during the 2003 SARS outbreak (Lee et al., 2003; US Department of Veterans Affairs, 2020; updated in April 2021). In response to the COVID-19 pandemic, the American Thoracic Society, American Association for Respiratory Care, and American College of Chest Physicians advised against using nebulizer treatments when feasible and suggested other protective measures including personal protective equipment (PPE) use, or instead implementing metered-dose inhalers (MDIs) (American Association for Respiratory Care, 2021; Katz et al., 2021; Pasnick et al., 2020).
Scientific evidence regarding the potential risk of viral infection from nebulization is limited (Tashkin and Barjaktarevic, 2020; Tran et al., 2012; US Department of Veterans Affairs, 2020; Wan et al., 2004; updated in April 2021; Woods, 2021). A recent systematic review encompassing 22 studies found no conclusive evidence that nebulizer treatment was associated with an increased risk of SARS-CoV-2 transmission (Goldstein et al., 2021). To date, no prospective studies have specifically evaluated the risk of SARS-CoV-2 transmission to healthcare workers delivering medication to virally infected patients via nebulizers versus MDIs. While nebulization may generate high quantities of fugitive aerosols in ambient air, this does not necessarily correlate with virus transmission (Li et al., 2022). Medical aerosols generated during nebulization do not contain pathogens unless the nebulizers are contaminated by the patient or healthcare worker (Dhand and Li, 2020; Fink et al., 2020). The use of a filtered mouthpiece and breath-actuated nebulizer may further mitigate these risks (Dhand and Li, 2020; Fink et al., 2020). On the other hand, MDI use may stimulate a patient’s cough (Chang et al., 2020), and bioaerosols that are exhaled through cough or normal exhalation are neither avoided nor contained with the use of inhalers (Fink et al., 2020). Medication delivery via nebulization may be preferable to MDI when considering a patient’s clinical condition, poor lung function, cognitive impairment, supply shortages, or manual dexterity challenges (Talwar et al., 2021).
The aim of this study was to measure the SARS-CoV-2 RNA load in aerosols and droplets dispersed from patients with COVID-19 during administration of albuterol using a breath-actuated nebulizer with a filtered mouthpiece compared to an MDI with a spacer.
Methods
Study design and patients
This was a randomized, open-label, two-period crossover study (NCT04681079) to evaluate COVID-19 viral dispersion from a breath-actuated nebulizer versus an MDI by collecting environmental samples from hospitalized patients with mild-to-moderate COVID-19. The trial was conducted according to the principles of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use Guideline for Good Clinical Practice and the code of ethics of the World Medical Association’s Declaration of Helsinki. The protocol was reviewed and approved by the University of Kansas Medical Center Institutional Review Board (IRB #STUDY00146336, Human Research Protection Program, Kansas City, KS, USA). Written informed consent was obtained from all patients. Healthcare staff and clinical trial team members entering patients’ rooms were required to wear PPE including an N95 or higher-level respiratory protection, eye protection, gloves, and a gown.
Eligible patients were ≥18 years old and had COVID-19 infection as confirmed by a polymerase chain reaction (PCR) test on samples from nasopharyngeal or oropharyngeal swabs or a saliva specimen taken within the previous 10 days. To confirm COVID-19 positivity throughout the study day, nasal swab and saliva samples were collected at baseline, and saliva samples were collected prior to each subsequent period. Patient exclusion criteria are listed in the Supplement.
Procedures
All patients underwent a baseline air and surface sampling period during which no study therapies were administered. Patients then underwent two 2-h treatment periods in a randomized crossover sequence, with a 2-h washout period between treatments (Figure S1). At the start of each treatment period, patients received a single dose of albuterol sulfate nebulized solution (2.5 mg/3 mL) administered via inhalation using a breath-actuated nebulizer with a filtered mouthpiece (AEROECLIPSE® II BAN™, Monaghan Medical) or albuterol sulfate hydrofluoroalkane inhalation aerosol administered via MDI with a spacer (90 µg per actuation; two puffs). Each patient completed all study periods in a single day.
Aerosol and surface sampling were used to assess viral loads in the rooms of patients with COVID-19. Aerosol sampling for SARS-CoV-2 was performed by cascade impactors (Sioutas SKC, Inc, Eighty Four, PA, USA) at baseline and at initiation of inhaled therapy with either the nebulizer or MDI for 2 h. Impactors were mounted 1 ft above the patient’s head and 3-ft and 6-ft distances from the patient (Figure 1). Surface sampling was performed after the 2-h air sampling period. (a) Hospital room sampling layout, (b) number of samples with SARS-CoV-2 RNA detected by period, and (c) percent of SARS-CoV-2 viral positive samples per period and location. ft, feet; neb, nebulizer; MDI, metered-dose inhaler.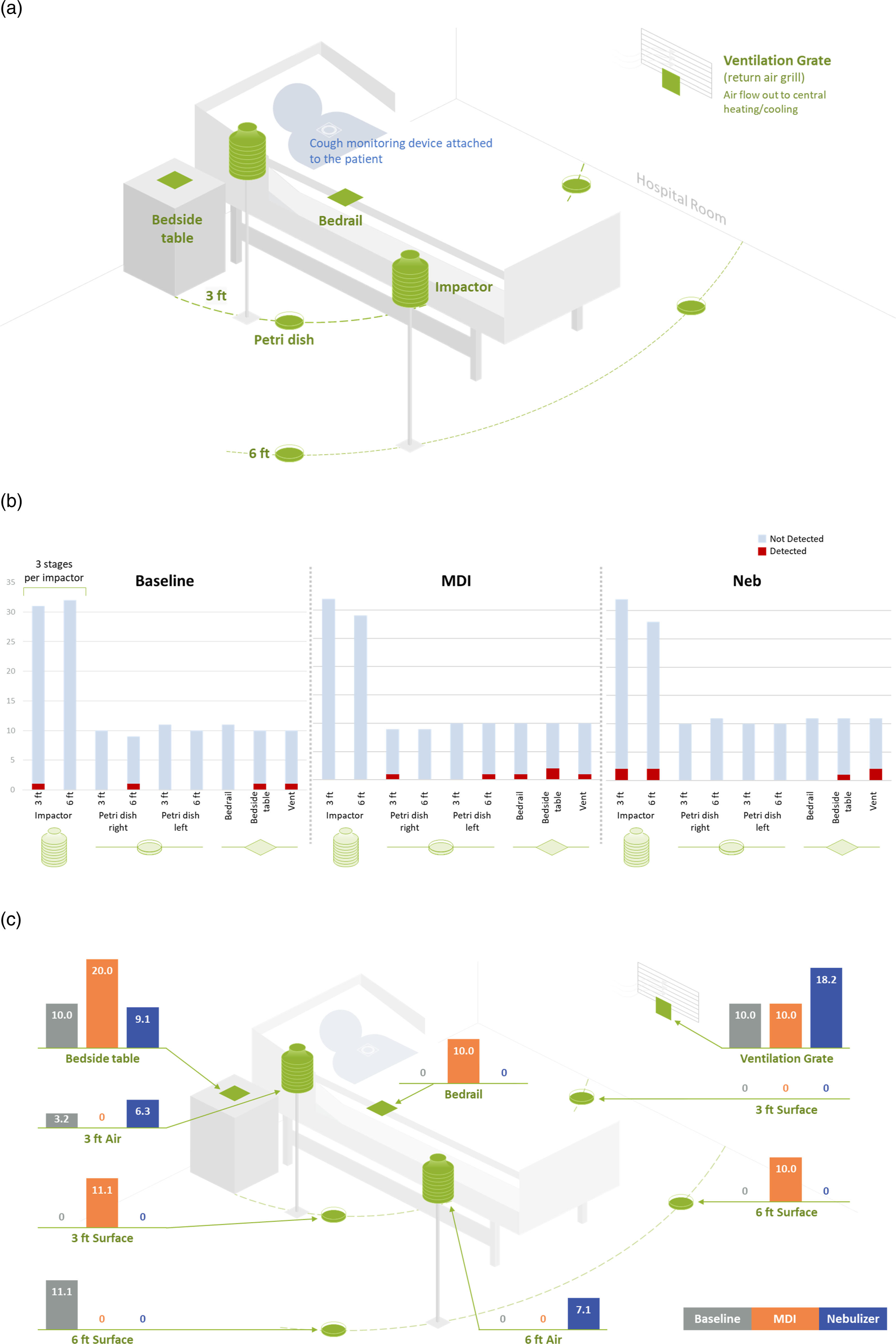
Patients were evaluated for cough at baseline and throughout each collection period using a VitoloJAK II recording device (Vitalograph, Lenexa, KS, USA) with a sensor and microphone. Data were uploaded to the Vitalograph web portal and analyzed by a team of expert cough analysts.
Sample collection
Impactors were calibrated to a flow rate of 9 L/min. Aerosols were collected onto polytetrafluoroethylene (PTFE) filters fractioned into three size groupings: <1.0, 1.0 to 2.5, and >2.5 µm. Open Petri dishes with PTFE filters were used to collect aerosols and droplets that settled from the air at four specified locations within the patients’ rooms (Figure 1a). The distances between the patient and Petri dishes and impactors were consistent across all hospital rooms. However, the distances of the swab surface areas from the patient varied (but were consistent throughout the study periods). Collected patient and environmental samples were immediately stored on dry ice prior to storage at −70°C. Additional collection details are in the Supplement.
Methodological assays
An extraction-free sample preparation method (for nasopharyngeal swabs and environmental samples) was developed by Celerion Laboratories (Lincoln, NE, USA) and validated with a standard curve fit for 100 to 200 copies/µL using the Applied Biosystems SimpliAmp Thermal Cycler (ThermoFisher Scientific, Waltham, MA, USA). The 2019-nCoV CDC EUA Kit (Integrated DNA Technologies, Coralville, IA, USA; limit of detection: 10 copies/µL [Tombuloglu et al., 2022]) was used with Reliance One-Step Multiplex RT-qPCR Supermix (Bio-Rad Laboratories, Hercules, CA, USA). For each run, 20 µL of environmental samples or nasopharyngeal swab media was used along with 2019-nCoV_N and Hs_RPP30-positive control samples, human specimen controls (buccal swab, collected at Celerion Laboratories), and no-template controls. A standard curve was included in each run with heat-inactivated 2019-nCoV standards (0.5–10,000 genome copies/µL). The 2019-nCoV_N1 reverse transcription-PCR (RT-PCR) and RNAse P RT-PCR reaction mix consisted of 7.5 µL 4x Reliance One-Step Multiplex RT-qPCR Supermix and either 2.24 µL 2019-nCoV_N1 or equivalent volume of RNAse P. A final volume of 10 µL of these reaction mixes was added to appropriate wells. PCR details are included in the Supplement.
Variable controls
All study rooms were single occupancy and had non-negative pressure. Variables that could impact the dispersion of SARS-CoV-2 aerosol particles were documented (room dimensions, number of air exchanges, date/type of last housekeeping visit, and entry of personnel). Other variables (room temperature and humidity) were recorded at baseline and prior to each period start. Rooms had air exchange rates of four or twelve times/hour. If a patient was on supplemental oxygen, the flow rate was recorded and any changes documented throughout the study day. Any non-study activity was minimized (including entry of personnel) within the patient’s room during the 2-h viral collection period. Additional procedures are described in the Supplement.
Endpoints and statistical analyses
The planned primary endpoint of change in viral load from baseline at each sample period was not assessed due to undetectable virus levels in most baseline samples. Additional planned study endpoints included percentage of positive patient samples at each sampling period, copies of SARS-CoV-2 viral genomes detected, and the relationship between patient clinical characteristics or environmental factors and virus in room air and on surfaces. All endpoints of this pilot study were descriptive and not statistically powered. Additional statistical analysis details are described in the Supplement.
Results
Patients
A total of 11 patients were enrolled in the study from June to November 2021. Patients had a mean age of 55.5 years, 81.8% were female, 54.5% were White, and the mean body mass index was 31.9 kg/m2 (Table S1). Most patients were not vaccinated (6/9 [66.7%]; data not available for two patients), and median hospital stay was 3 days (range, 1–6). Supplemental oxygen was required for seven (63.6%) patients, with similar mean supplemental oxygen flow rate across all periods for each individual patient. The oxygen flow rate varied greatly between users (range, 2–50 L/min). Most patients (63.6%) were treated with remdesivir. Nine and two patients occupied rooms with four and twelve air exchanges/hour, respectively.
SARS-CoV-2–positive samples
No viral RNA was found in the rooms of 4/11 patients. Samples from three patients contained detectable SARS-CoV-2 RNA in a single period (baseline, MDI, or nebulizer), and samples from four patients contained detectable SARS-CoV-2 RNA in multiple periods (Table S2). A total of four patients had detectable SARS-CoV-2 RNA during the baseline period, four patients had detectable viral RNA during the MDI period, and four patients had detectable viral RNA during the nebulizer period. Six patients had detectable SARS-CoV-2 RNA during one treatment period (MDI or nebulizer, but not both), and one patient had detectable SARS-CoV-2 RNA during both treatment periods (MDI and nebulizer).
There were no meaningful differences in the number of patient positive samples for each period, and most environmental samples did not have detectable SARS-CoV-2 RNA (Figure 1(b)); only 4.3% (17/397) of total environmental samplings contained detectable SARS-CoV-2 RNA. During the baseline period, 3.0% (4/134) of environmental samples were positive; during the MDI and nebulizer periods, 4.7% (6/129) and 5.2% (7/134) of samples, respectively, were positive. The amount of viral RNA detected in any positive environmental sample was low, with a mean of 0.14 viral RNA copies/µL (range, 0.03–1.00 copies/µL). Upon contingency analysis of pooled samples, no difference in detection of SARS-CoV-2 RNA was found between nebulizer and MDI periods (odds ratio, 1.00; 95% CI, 0.134–7.466; p = 1.00).
Only a slight variation in the distribution of positive samples was observed around the room by period. SARS-CoV-2 RNA was found most often on the bedside table (9.1% [1/11]–20.0% [2/10] across periods) and on the ventilation grate (10.0% [1/10]–18.2% [2/11]) for all three periods (Figure 1(c)). Viral RNA was detected on the bedrail during the MDI period and was detected in the Petri dishes at 6 ft during the baseline period and 3 and 6 ft during the MDI period. SARS-CoV-2 RNA was detected in the impactor samples from the baseline and nebulizer periods, and viral RNA was found in all three particle size groupings (<1 µm, 1–2.5 µm, and >2.5 µm).
Viral dispersion by status/factor
Viral dispersion and patient clinical status.
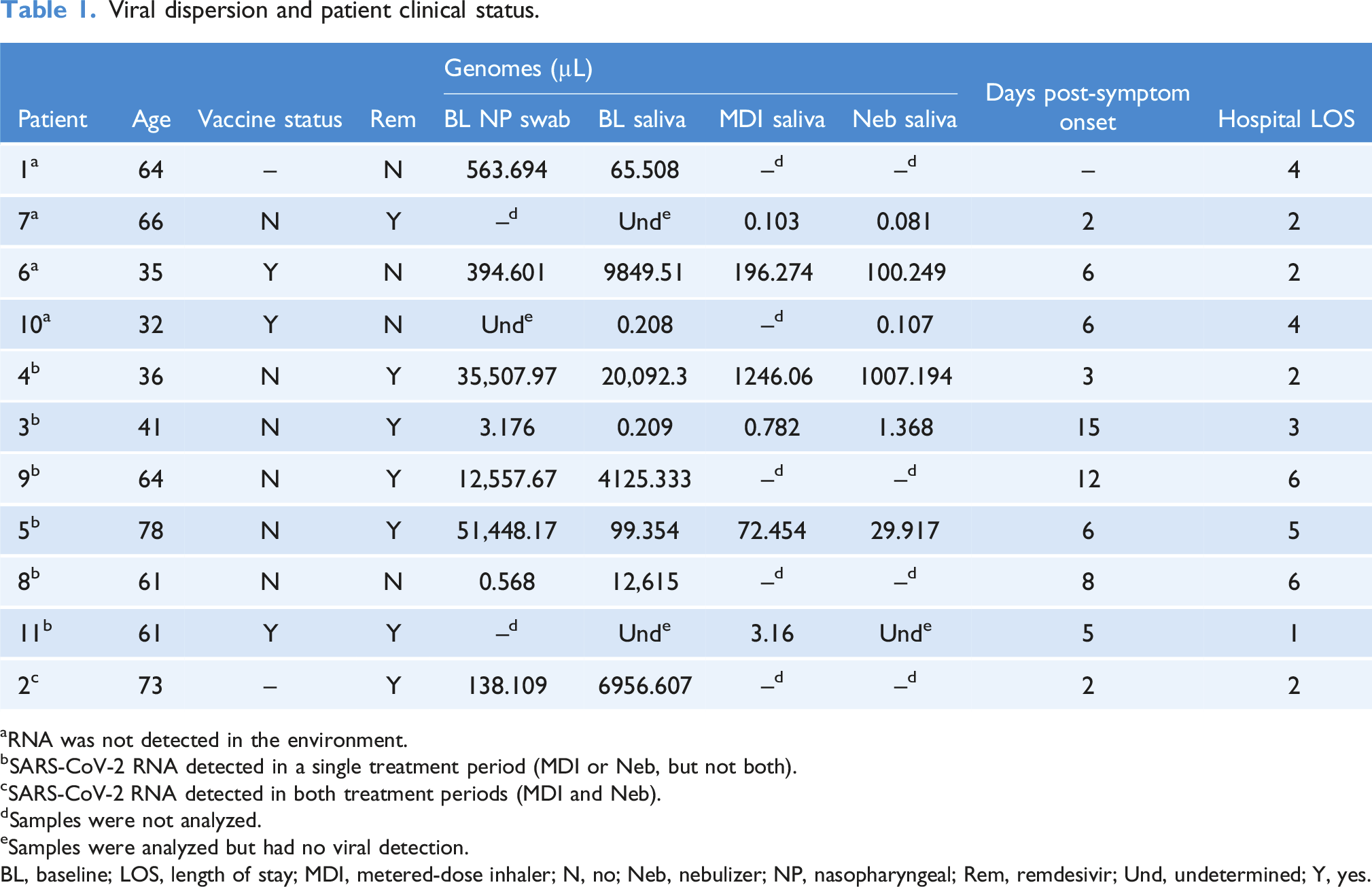
aRNA was not detected in the environment.
bSARS-CoV-2 RNA detected in a single treatment period (MDI or Neb, but not both).
cSARS-CoV-2 RNA detected in both treatment periods (MDI and Neb).
dSamples were not analyzed.
eSamples were analyzed but had no viral detection.
BL, baseline; LOS, length of stay; MDI, metered-dose inhaler; N, no; Neb, nebulizer; NP, nasopharyngeal; Rem, remdesivir; Und, undetermined; Y, yes.
Discussion
In this novel, randomized, open-label, multiple-period crossover study, SARS-CoV-2 viral load distribution was not affected by the method of bronchodilator administration—nebulizer versus MDI. Eleven patients were enrolled, from whom nearly 400 evaluable samples were collected; the crossover study design allowed for within-participant testing, thereby minimizing the risk of confounders owing to inter-patient variability. The planned primary endpoint of change in viral load from baseline at each sample period was abandoned due to a low number of baseline samples with detectable viral RNA across all patients (3.0%). Viral RNA was detected during all periods, but there were no differences between the number of positive samples via nebulizer versus MDI administration. SARS-CoV-2 viral shedding appears to be patient specific and not related to a particular delivery method.
Detectable levels of SARS-CoV-2 RNA were found in at least one period in all sampled locations in the room, except for the Petri dish positioned at 3 ft away from the patient (left bedside). Viral RNA was also found on the ventilation grate during all three periods, even though the ventilation grate was the sampling location furthest from the patient (up to 12 ft horizontal distance). Viral detection during the MDI period was unexpected and demonstrates that this delivery device is not without risk, similar to previous SARS-CoV-2 environmental sampling studies within rooms of hospitalized patients with COVID-19 (in the absence of aerosol-generating procedures) (Ong et al., 2020; Santarpia et al., 2020). This suggests that bioaerosols are pulled towards the ventilation grate based on the airflow patterns in the hospital room (Jayaweera et al., 2020).
These data demonstrate that SARS-CoV-2 viral presence in the environment was not impacted by medication delivery method. This is the first prospective, randomized study to suggest that there is no increase in bioaerosol transmission with a breath-actuated nebulizer with a filtered mouthpiece compared to MDI (Tashkin and Barjaktarevic, 2020; Tran et al., 2012; US Department of Veterans Affairs, 2020; Wan et al., 2004; updated in April 2021). Healthcare workers should exercise clinical judgment and use appropriate PPE when administering nebulization or MDI therapy to virally infected patients (Centers for Disease Control and Prevention, 2022a; Woods, 2021).
There are several key study limitations to consider. SARS-CoV-2 virus was detected by RT-PCR, which is highly sensitive but does not differentiate infectious virions from non-infectious RNA viral fragments. Enrolled patients had low-to-medium viral load levels, which is consistent with reports of viral peak prior to symptom onset (He et al., 2020); however, the viral dispersion data may not reflect that of “super spreaders” or those with higher viral load levels. Additional studies are needed to help inform clinicians on the management and treatment of SARS-CoV-2, especially in those with persistent viral shedding and varying clinical characteristics (e.g., viral load, different variants, cough, and vaccination status) (Batra et al., 2022). Due to the prevalence of the delta SARS-CoV-2 variant during the time of patient enrollment period (Centers for Disease Control and Prevention, 2022b), it is believed to be the main variant; however, this was not directly assessed and findings may not be generalizable across variants, which vary in transmissibility (Jones et al., 2021). Finally, it is unclear if these findings are generalizable to other nebulization devices.
Use of a breath-actuated nebulizer with a filtered mouthpiece did not lead to increased dispersion of SARS-CoV-2 RNA compared with MDI in patients with COVID-19, with low environmental exposure observed regardless of administration method. Detection of viral RNA was also independent of patient clinical status and environmental factors, and instead seemed to be patient dependent. Nebulized therapy should not be withheld in patients with COVID-19 who may benefit from this treatment method.
Supplemental Material
Supplemental Material - Impact of nebulization versus metered-dose inhaler utilization on viral particle dispersion in patients with COVID-19
Supplemental Material for Impact of nebulization versus metered-dose inhaler utilization on viral particle dispersion in patients with COVID-19 by Brian M Clemency, Candice Clay, Renoj Varughese, Carli Kennedy, Wayne Yates, Asma Lat, Ai Ling Ching, Doug Clark, David Lewin, Xianyi Chen, and Mario Castro in Journal of Infection Prevention.
Footnotes
Acknowledgments
Writing and editorial support were provided by Hilary Durbano, PhD, of AlphaBioCom, a Red Nucleus Company, and funded by Theravance Biopharma US, Inc.
Author contributions
All authors substantially participated in the creation of this work, approved the final version of the manuscript, and take responsibility for the integrity of the work. Investigation: B.M.C., R.V., C.K., and M.C. Project administration: B.M.C., C.K., W.Y., A.L.C., and D.C. Supervision: B.M.C. and M.C. Conceptualization: C.C., C.K., W.Y., A.L.,A.L.C., D.C., and M.C. Formal analysis: C.C. and D.L. Data curation: C.K., D.L., and X.C. Methodology: C.K., D.L., and M.C. Validation and visualization: C.K. and D.L.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.M.C. and R.V. have received grant funding from Covis and NIH. C.C. is a former employee of Theravance Biopharma US, Inc. C.K. and A.L. declare no conflicts of interest. W.Y. and X.C. are employees of Theravance Biopharma US, Inc., and shareholders of Theravance Biopharma US, Inc. A.L.C. is an employee of Erasca, Inc., and a former employee of Theravance Biopharma US, Inc. D.C. is an employee of Aligos Therapeutics and a former employee of Theravance Biopharma US, Inc. D.L. is a contractor for Theravance Biopharma US, Inc. M.C. has received personal fees from 4D Pharma, AstraZeneca, Aviragen, Boston Scientific, Boehringer Ingelheim, Elsevier Genentech, Mallinckrodt, Neutronic, Nuvaira, Sanofi-Aventis, Teva, Therabron, Regeneron, Vectura, VIDA, and Theravance Biopharma US, Inc.; and received grant money from ALA, AstraZeneca, Chiesi, Boehringer Ingelheim, GSK, NIH, Novartis, PCORI, and Sanofi-Aventis.
Funding
The author(s) disclosed receipt of the following financial support for the research, medical writing support, and/or publication of this article: Theravance Biopharma US, Inc. This study was sponsored by Theravance Biopharma US, Inc., who was involved in the experimental design, collection, analysis, interpretation of the data, and development of this manuscript.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
