Abstract
Background
Healthcare workers (HCWs), especially in high tuberculosis (TB) incidence countries, are at risk of Mycobacterium tuberculosis infection and TB disease, likely due to greater exposure to TB cases and variable implementation of infection control measures.
Aim
We aimed to estimate the prevalence of tuberculin skin test (TST) positivity, history of TB and to identify associated risk factors in HCWs employed at a tertiary referral hospital in Bandung, Indonesia.
Methods
A cross-sectional study was conducted from April to August 2018. A stratified sample of the HCWs were recruited, screened by TST, assessed for TB symptoms, history of TB disease and possible risk factors. Prevalence of positive TST included diagnosis with TB after starting work. HCWs with TB disease diagnosed earlier were excluded. Survey weights were used for all analyses. Possible risk factors were examined using logistic regression; adjusted odds ratios and 95% confidence intervals (CI) are presented.
Results
Of 455 HCWs recruited, 42 reported a history of TB disease (25 after starting work) and 395 had a TST result. The prevalence of positive TST was 76.9% (95% CI 72.6–80.8%). The odds increased by 7% per year at work (95% CI 3–11%) on average, with a rapid rise in TST positivity up to 10 years of work and then a plateau with around 80% positive.
Discussion
A high proportion of HCWs had a history of TB or were TST positive, increasing with longer duration of work. A package of TB infection control measures is needed to protect HCWs from Mycobacterium tuberculosis infection.
Background
Healthcare workers (HCWs), especially in high tuberculosis (TB) incidence countries, are at increased risk of Mycobacterium tuberculosis (M. tuberculosis) infection and TB disease due to greater exposure to TB cases and variable implementation of infection control measures (Apriani et al., 2019; Joshi et al., 2006; Uden et al., 2017; Zwerling et al., 2012). Their risk of M. tuberculosis infection is approximately two to three times higher than the general population (Uden et al., 2017). Several systematic reviews on M. tuberculosis infection and TB disease among HCWs have been conducted previously (Apriani et al., 2019; Grobler et al., 2016; Joshi et al., 2006; Menzies et al., 2007; Nasreen et al., 2016; Uden et al., 2017). In our systematic review (Apriani et al., 2019), which included 53 tuberculin skin test (TST) studies, the prevalence of M. tuberculosis infection in HCWs in low- and middle-income countries (LMICs) was shown to be high, especially in high TB incidence countries, with nearly 50% of HCWs having a positive TST. Occupational factors associated with M. tuberculosis infection involved more direct contact with a TB patient or longer duration of health care work, indicating that more effort needs to be done to protect staff (Apriani et al., 2019).
Indonesia has the third highest number of incident TB cases in the world with approximately 320 per 100 000 population per year in a country of around 264 million people (World Health Organization, 2018). In 2014, using mathematical modelling, it was also estimated that around 120 million people (46%) in Indonesia were infected with M. tuberculosis – making Indonesia the third highest M. tuberculosis infection burden country in the world after China and India (Houben and Dodd, 2016). Despite these documented high rates, the information on the burden of M. tuberculosis infection and TB disease in HCWs is absent. We therefore aimed to estimate the prevalence of M. tuberculosis infection as measured by tuberculin skin test (TST) positivity and to identify associated risk factors in HCWs in Bandung, Indonesia.
Methods
Study design and population
We conducted a cross-sectional study from April to August 2018 of a stratified sample of HCWs employed in a tertiary referral hospital. The hospital has 969 beds and has had, since 2007, the facilities to carry out TB case detection, screening, diagnosis and treatment using directly observed treatment short-course (DOTS). In 2011, it was designated as one of 13 referral hospitals for multidrug-resistant TB (MDR-TB) patients in Indonesia and the only one in West Java province. In 2017, 258 pulmonary TB cases were diagnosed at the hospital – approximately 5% of the total pulmonary TB cases notified to the Bandung City Municipal Health Office (unpublished data).
Of approximately 3000 permanent workers at the hospital, two-thirds have direct patient care (e.g. doctor, dentist, nurse, midwife, laboratory personnel, pharmacist, nutritionist and radiographer); the other third are management, administration and support workers. In Indonesia, as recommended by the World Health Organization, 2018 (WHO), Bacillus Calmette–Guérin (BCG) vaccination is given soon after birth as part of the childhood immunisation programme. The majority of HCWs would have received a BCG vaccination in infancy; BCG vaccination is not given on hiring. Our study included hospital workers who have direct patient care, stratified according to job category to allow sufficient numbers for description and comparison by category. The total number providing health services (n = 2045) was divided by seven job categories to give 71 people required from each category to reach the minimum sample size of 500 (Supplementary Table 1). As there were around, or less than, 71 in the midwives, nutritionist and radiographer categories, all workers in these categories were invited to participate. From each of the remaining categories, 85 workers were randomly selected.
Study procedures
A formal letter was sent to the hospital director and the head of departments or units to inform them and seek permission to carry out the study. HCWs were identified, and selected, from the hospital database. Scheduled visits to each department or unit and meeting with selected HCWs were arranged at which time information about the study was provided and HCWs invited to participate. The enrolment process required more than one visit due to shift work of the HCWs. Written informed consent was sought from all participants prior to undertaking the questionnaire and administering TST. Any staff absences, reasons provided for non-consent or refusal for TST were noted.
A questionnaire was developed using examples from previously published studies to collect data and to identify factors associated with M. tuberculosis infection (Menzies et al., 2007; Nasreen et al., 2016; Rutanga et al., 2015; Zhang et al., 2013). This was piloted in the local context before finalisation. Questions included information about the following: 1) demographic characteristics: age, gender, ethnicity and educational status; 2) medical history: BCG vaccination, HIV status, other immunocompromised conditions, smoking, alcohol consumption and body mass index (BMI); 3) occupational TB exposure: profession, working area or procedure, service years as a HCW, experience working in a TB clinic/pulmonary ward/isolation ward, any direct contact with a TB patient and direct contact with a colleague with TB; 4) non-occupational TB exposure: any direct contact with family or friend diagnosed with TB; and 5) any formal TB training with the National TB program or informal TB training. Questions required either a yes/no answer or selection of one answer from several options.
A trained research nurse administered a single step TST using 0.1 mL Tubersol® bioequivalent to 5 tuberculin units (TU) on the volar side of the forearm. The skin reaction was read 48–72 hours later. An induration size ≥10 mm was considered positive (Jensen et al., 2005). Participants were provided with their result in a sealed envelope, in person. Those with a positive result were assessed for symptoms of TB disease and referred for free diagnostic evaluation for TB disease and appropriate treatment if necessary. At the time of implementing this study, according to local and international recommendations, no TB preventive treatment was offered to HCWs with a positive TST result (World Health Organization, 2015). For HCWs, the WHO recommends a package of prevention and care interventions, including HIV prevention, antiretroviral therapy and isoniazid preventive therapy (IPT) only for those who are HIV-positive (World Health Organization, 2009). Based on the very low rate of HIV positivity in Indonesia, HIV testing was not offered to all workers, but was offered to anyone who was diagnosed with TB.
Ethics approval was obtained from the University of Otago Ethics Committees, New Zealand (HE17/023) and Hasan Sadikin Hospital Ethics Committees Bandung, Indonesia (No LB.04.01/AO5/EC/O14/I/2018).
Sample size considerations, data management and statistical analyses
Allowing for 10% non-response, a sample of 500 was estimated to provide the prevalence of M. tuberculosis infection with a margin of error of 5% (half the width of a 95% exact binomial confidence interval [CI]), taking into account the study sampling frame. Data from a randomised controlled trial in M. tuberculosis infection in HCWs in Bandung suggested that the TST positivity might be around 60–65% (unpublished data). For evaluation of the factors associated with M. tuberculosis infection, we calculated that if the prevalence of exposure were between 20% and 60%, the study would have 80% power to detect a prevalence ratio of 1.26 (odds ratio [OR] of 2.2) at the two-sided 5% level. All data were cross-checked and entered into a database created in RedCap® (Harris et al., 2009). Data were analysed using Stata® version 14.2 (Stata Corp LP, Texas, US). All statistical analyses accounted for the stratified sampling and the total population size. Sampling weights were used and were calculated from the selection probabilities of the HCWs in each job category.
Noting the focus on M. tuberculosis exposure since commencing work and that prevalent TST positivity tends to reflect cumulative incident infection, we assumed that those who had a history of TB were TST positive without testing. We excluded HCWs with TB disease diagnosed before starting work at the health care facility from the analysis. Prevalence of positive TST includes all HCWs who had a positive TST in the study plus those who had a history of TB treatment after starting work in the hospital in the numerator (the latter group did not have a TST, but we assumed they would have been positive if they had). The prevalence is presented with a 95% CI. The factors associated with TST positivity were examined using univariate logistic regression. Estimates are presented as odds ratios and adjusted for characteristics and exposures obtained using modified backwards-stepwise regression. Age at hiring, gender and job categories were retained in the multivariate model, and other variables were retained if p < 0.2. The TB case rate, presented as the total cases per 1000 people per year, was calculated as the following:
Results
A total of 455 HCWs were recruited. The response rate was high for nurses (100%), nutritionists (94.6%) and pharmacists (89.4%), but less for laboratory personnel (66.7%), midwives (65.8%), doctors and dentists (50.0%). For radiographers (13.0%), the response rate was low due to a reluctance of the unit coordinator to arrange a meeting with staff.
Forty-two HCWs had a history of TB disease (Figure 1), of whom 17 were diagnosed before, and 25 after, starting work in this hospital (past TB n = 23; current TB n = 1; newly identified pleural TB n = 1, diagnosed by a physician external to the study). The TB case rate was 4.7 cases per 1000 person per year. Of the 413 HCWs with no TB disease, 18 refused TST (mostly because of fear of needles), leaving 395 who received the TST, of whom 298 (75.4%) were TST positive and 97 (24.5%) negative. Two HCWs who had cough for more than 2 weeks were tested by sputum examination and chest X-ray, both were negative for TB disease. Study profile: recruitment and tuberculin skin test results (N = 395). TB = tuberculosis, TST = tuberculin skin test. HCWs with history of TB disease were asked when they had TB disease, whether it happened before or after starting work in this hospital.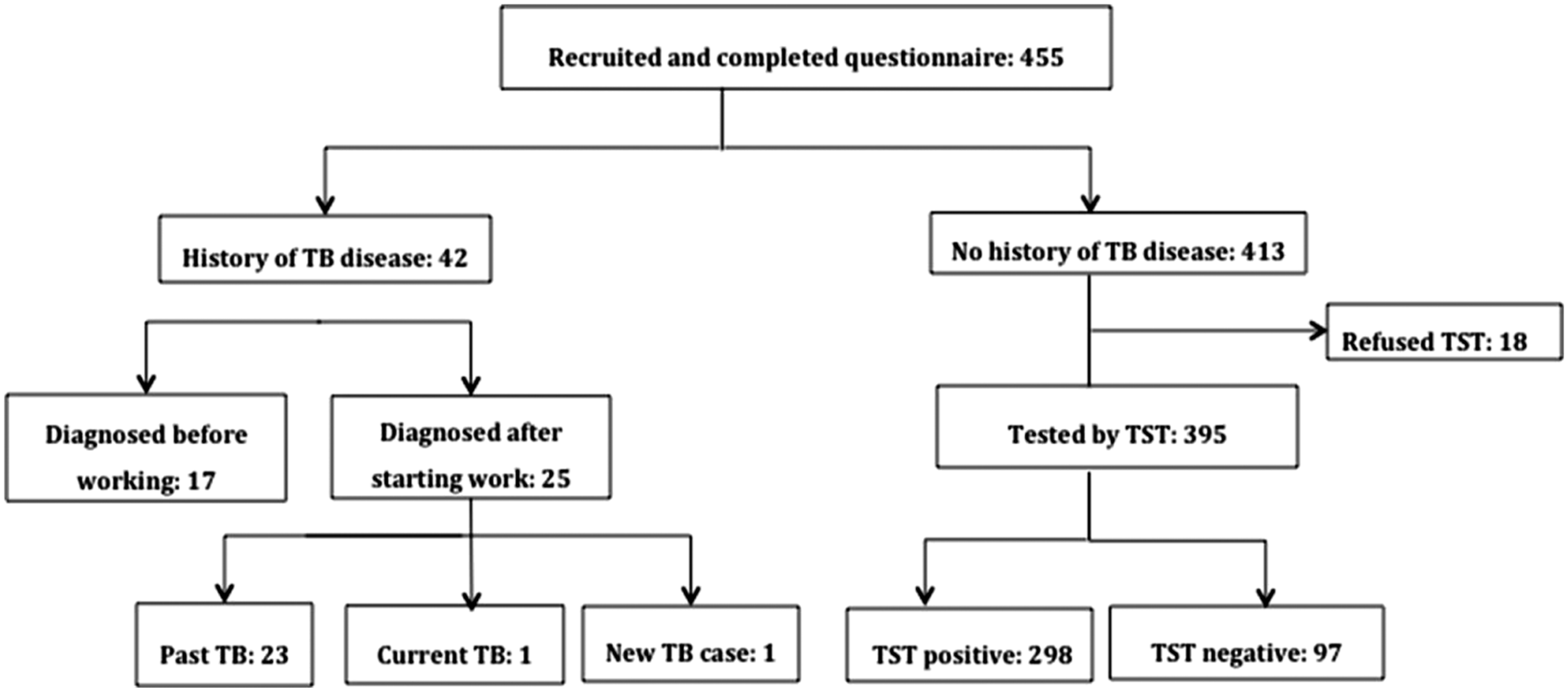
Demographic, medical history and exposure to TB of health care workers included in the study (N = 455).
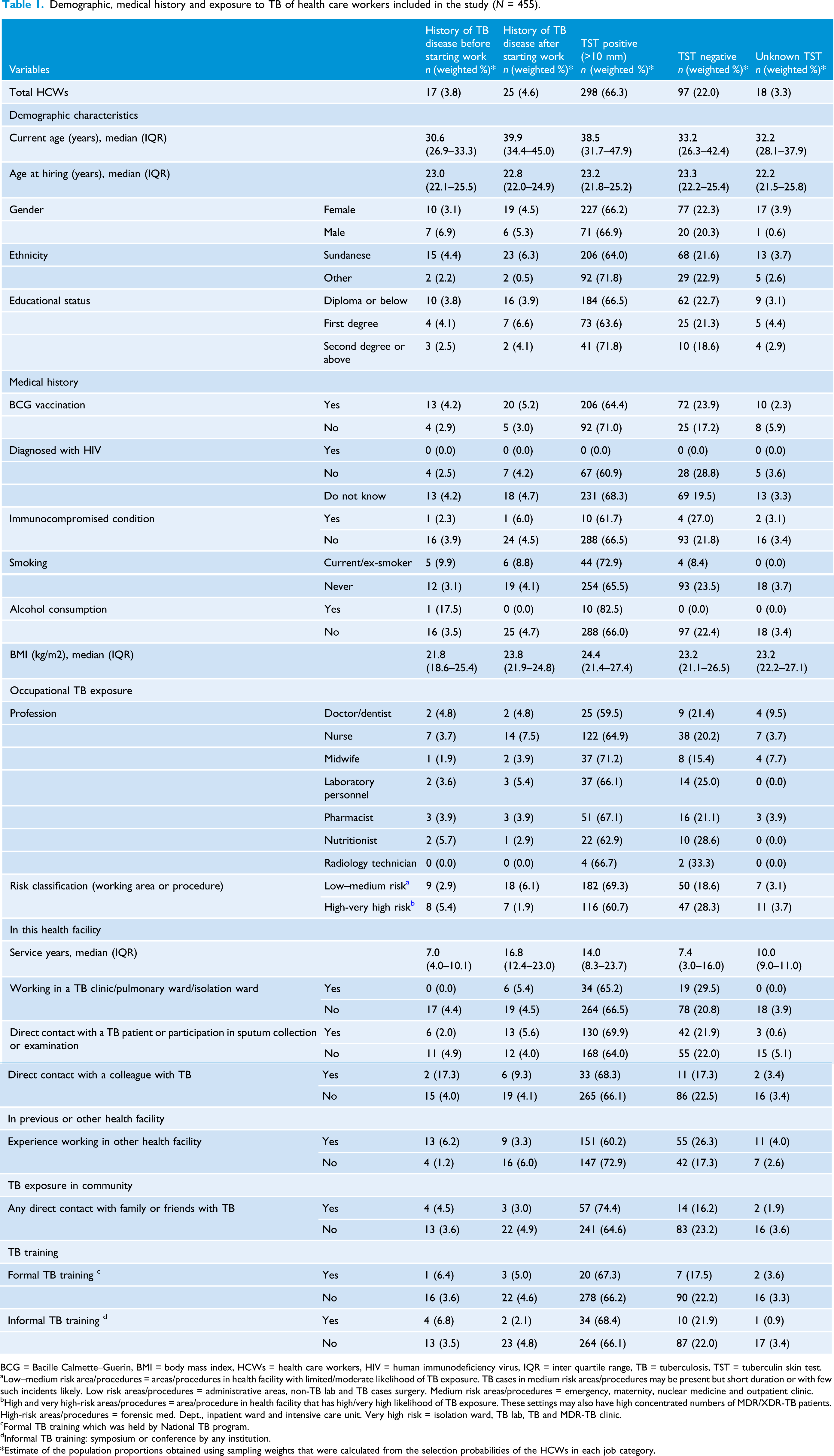
BCG = Bacille Calmette–Guerin, BMI = body mass index, HCWs = health care workers, HIV = human immunodeficiency virus, IQR = inter quartile range, TB = tuberculosis, TST = tuberculin skin test.
aLow–medium risk area/procedures = areas/procedures in health facility with limited/moderate likelihood of TB exposure. TB cases in medium risk areas/procedures may be present but short duration or with few such incidents likely. Low risk areas/procedures = administrative areas, non-TB lab and TB cases surgery. Medium risk areas/procedures = emergency, maternity, nuclear medicine and outpatient clinic.
bHigh and very high-risk areas/procedures = area/procedure in health facility that has high/very high likelihood of TB exposure. These settings may also have high concentrated numbers of MDR/XDR-TB patients. High-risk areas/procedures = forensic med. Dept., inpatient ward and intensive care unit. Very high risk = isolation ward, TB lab, TB and MDR-TB clinic.
cFormal TB training which was held by National TB program.
dInformal TB training: symposium or conference by any institution.
*Estimate of the population proportions obtained using sampling weights that were calculated from the selection probabilities of the HCWs in each job category.
Eight different types of professionals were recruited. The median service years in the current facility ranged from 7 to 17 years. Fifty-five (13.4%) had experience working in a TB clinic/pulmonary ward/isolation ward, and 189 (35.0%) were exposed to a high or very high TB risk work area or procedure. A total of 194 (61.3%) had some direct contact with a TB patient or participation in sputum collection or examination, 54 (9.6%) had some direct contact with a colleague with TB and 80 (17.5%) previously had direct contact with a family or friend who was diagnosed with TB. Half of the HCWs had experience working in another health facility, and the majority had not had any formal (95.3%) or informal TB training (92.0%).
Prevalence of positive tuberculin skin test and tuberculosis rate in healthcare workers
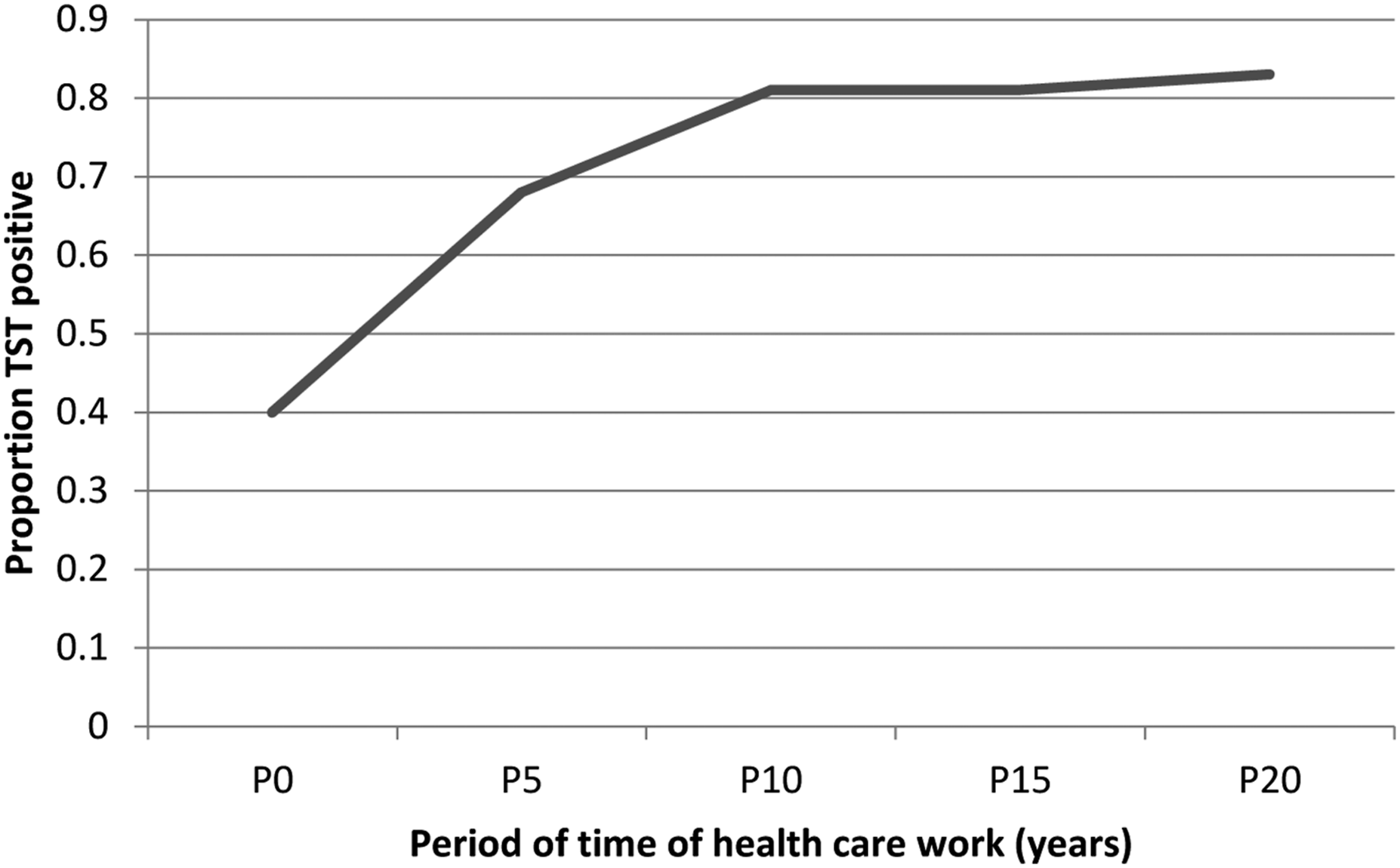
Three-quarters of HCWs had a TST induration ≥10 mm (Figure 2). With this cutoff, the prevalence of positive TST in the hospital was 76.9% (95% CI 72.5–80.8%, n = 420). There was a rapid rise in prevalence up to 10 years of work and then a plateau at around 80% TST positivity (Figure 3). The estimated TB rate in HCWs was 47 cases per 1000 per year. Tuberculin skin test (TST) reaction size (mm) using a moving 3 point average. Proportion of health care workers who were TST positive by length of time in health care work (N = 395).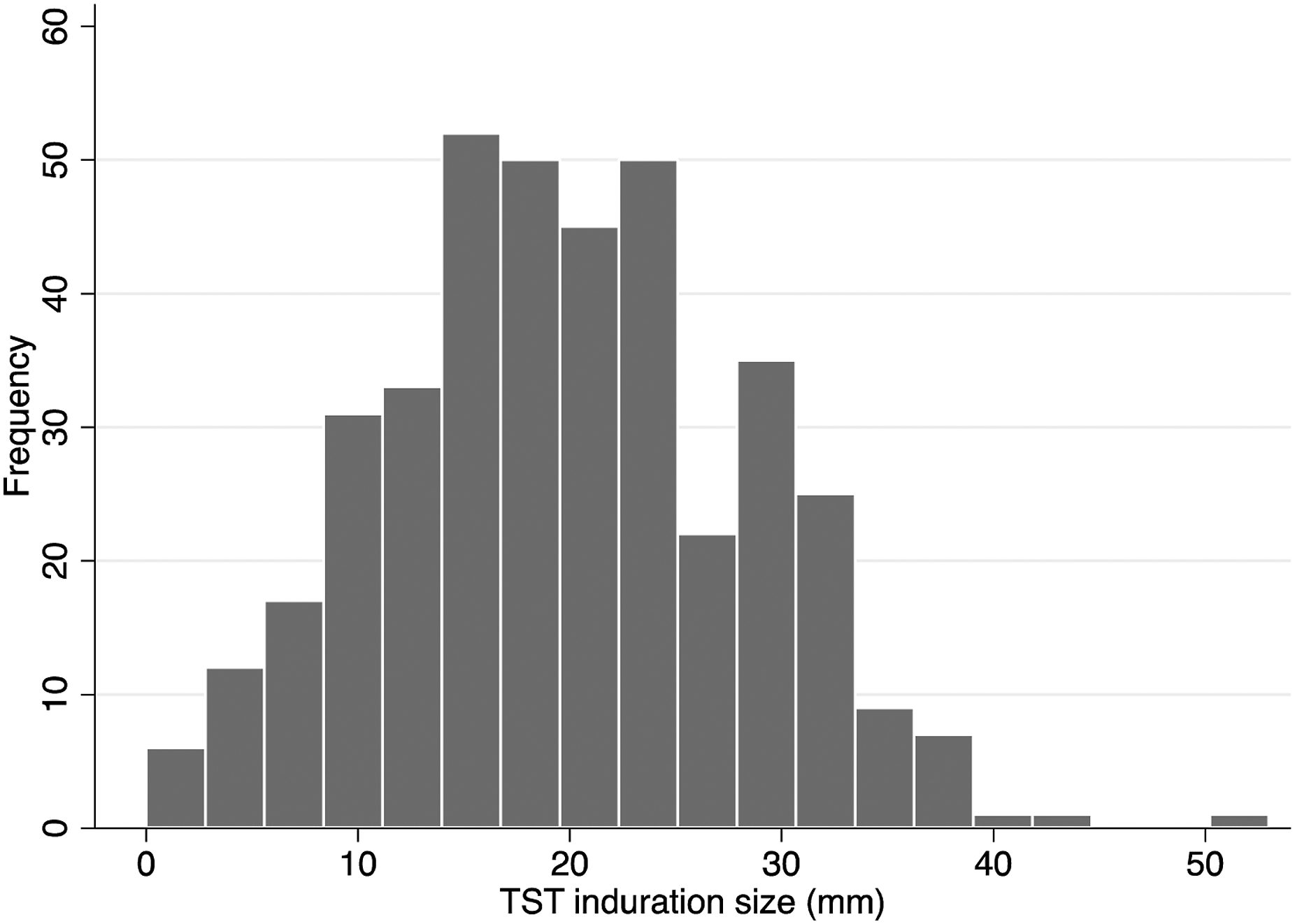
Risk factors associated with tuberculin skin test positivity
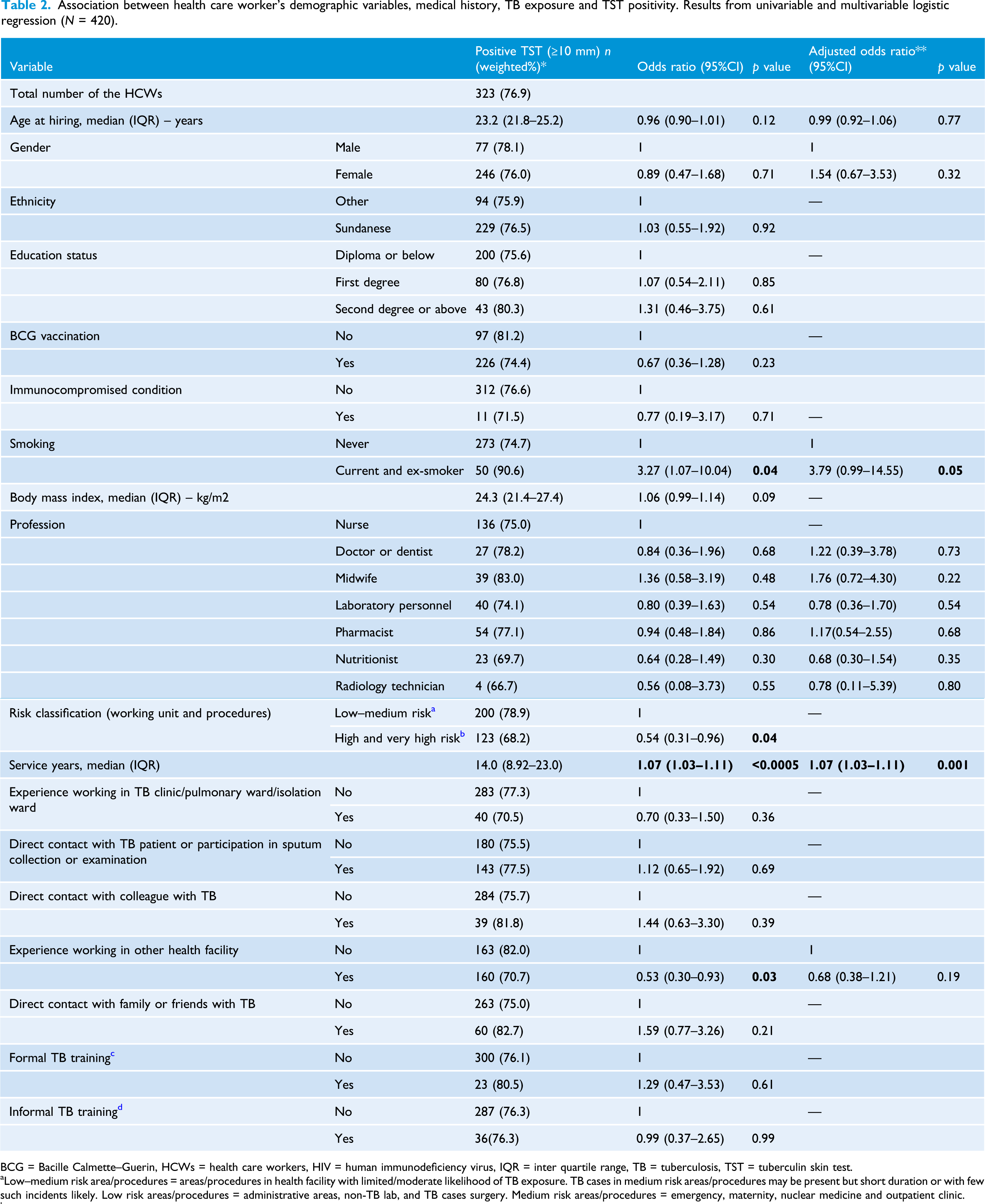
Association between health care worker’s demographic variables, medical history, TB exposure and TST positivity. Results from univariable and multivariable logistic regression (N = 420).
BCG = Bacille Calmette–Guerin, HCWs = health care workers, HIV = human immunodeficiency virus, IQR = inter quartile range, TB = tuberculosis, TST = tuberculin skin test.
aLow–medium risk area/procedures = areas/procedures in health facility with limited/moderate likelihood of TB exposure. TB cases in medium risk areas/procedures may be present but short duration or with few such incidents likely. Low risk areas/procedures = administrative areas, non-TB lab, and TB cases surgery. Medium risk areas/procedures = emergency, maternity, nuclear medicine and outpatient clinic.
bHigh and very high-risk areas/procedures = area/procedure in health facility that has high/very high likelihood of TB exposure. These settings may also have high concentrated numbers of MDR/XDR-TB patients. High-risk areas/procedures = forensic medicine, inpatient ward and intensive care unit. Very high risk = isolation ward, TB lab, TB and MDR-TB clinic.
cFormal TB training which was held by National TB program.
dInformal TB training: symposium or conference by any institution.
*Estimate of the population proportions obtained using sampling weights that were calculated from the selection probabilities of the HCWs in each job category.
**Estimates adjusted for characteristics and exposures were obtained using modified backwards-stepwise logistic regression. Age and gender were retained in the model, and other variables were retained if p < 0.2. Age, body mass index and years of work were modelled and presented as continuous variables. For age and BMI, the quadratic term was not retained in the model.
In the multivariable analysis, number of service years in this hospital was the only factor significantly associated with TST positivity (AOR 1.07; 95% CI 1.03–1.11). Smoking was also associated with a higher risk (AOR 3.79; 95% CI 0.99–14.55) which was of borderline statistical significance, but noted due to the potential for clinical importance if a true association (Table 2).
In a secondary analysis, with history of TB disease excluded from the numerator, there was no change in the findings (Supplementary Table 2).
Discussion
In our study, 76.9% of HCWs had either a positive TST in our study or a history of TB disease after starting work in the hospital. Longer duration of work was the only independently associated factor with the odds increasing by 7% per year of work.
The prevalence of M. tuberculosis infection based on TST positivity in general hospital settings in other LMICs ranged from 15.4% to 71.8% (Belo and Naidoo, 2017; Borroto et al., 2011; De Oliveira et al., 2007; He et al., 2012; Li-fan et al., 2013; Munisamy et al., 2017; Ozdemir et al., 2006; Powell et al., 2011; Rutanga et al., 2015; Van Rie et al., 2013; Zhou et al., 2014). These results included all types of HCWs, as well as administrative personnel. The prevalence in our study was surprisingly higher than the upper range, although not as high as that found in a study in a specialist TB hospital and health centre in South Africa (83.7%) (Adams et al., 2015). The prevalence estimate was also higher than that reported in our systematic review (Apriani et al., 2019), which gave an average estimate of 49%, and 55% in high TB-incidence countries (irrespective of type of HCWs and type of settings). It was also higher than the prevalence estimate of 23.4% in a recent study of HCWs in primary health centres in Semarang, Indonesia that used interferon gamma release assay to test for M. tuberculosis infection (Erawati and Andriany, 2020). No data are available to make age-specific comparisons with Indonesia’s general population. However, when compared to a modelled prevalence estimate (46%), our result was higher (Houben and Dodd, 2016). It was also higher than in another population with a similar age structure in the South-East Asia region (approximately 40%) (Houben and Dodd, 2016). Interestingly, in our study, there was a rapid rise in prevalence up to 10 years of work and then a plateau at around 80% TST positivity, suggesting that either around 20% of individuals remain uninfected despite high M. tuberculosis exposure, or they are anergic to tuberculin. The TB case rate estimate of 4.7 per 1000 person per year was higher than the general population in Indonesia, which is around three per 1000 population (World Health Organization, 2018). The results from our study most likely reflect the high rates of M. tuberculosis exposure in the work environment in the presence of a high background incidence of TB in the community.
Longer working experience as a HCW as a risk factor for TST positivity is consistent with a number of international studies (He et al., 2012; Mirtskhulava et al., 2008; Rutanga et al., 2015; Zhou et al., 2014). We noted that smoking had an association with TST positivity which is consistent with other studies (De Oliveira et al., 2007; He et al., 2015). We also noted that the positivity rates were fairly high across all professional groups, which suggests that there is likely to be a high risk of transmission throughout the hospital. A similar result was reported in previous studies in LMICs (Belo and Naidoo, 2017; Li-fan et al., 2013; Mirtskhulava et al., 2008; Powell et al., 2011; Rutanga et al., 2015; Van Rie et al., 2013), although some of these studies compared HCWs providing direct patient care to those in management, administration and support. Two studies (Munisamy et al., 2017; Nasehi et al., 2017) reported that HCWs providing health services had a higher prevalence of M. tuberculosis infection than management, administration and support workers.
Assessing the implementation of TB infection control measures in this facility was outside the scope of the study. However, we noted that the Joint Commission International accredited the hospital as an academic medical centre hospital in 2016. For this, some improvements regarding the environmental control measures were undertaken in some of the buildings, including the rebuilding of some facilities for TB care and management, and policy for early detection or triage of patients with cough. While the use of personal respiratory protection was not formally assessed, we observed that HCWs in high and very high-risk areas and procedures were mostly using respirator masks when caring for their patients, whereas in other areas of the hospital they were rarely seen.
This is the first study of its type from Indonesia. Although we only included 455 of 3058 HCWs, we used stratified sampling to maximise the chance of obtaining a representative sample and achieved a high participation rate. Furthermore, we used statistical methods for survey data, calculated from the selection probabilities of the HCWs in each job category. This study has some limitations. We noted the potential for non-response bias due to the difference in response rates by job category, particularly in radiographers. We did not screen the HCWs with a positive TST using chest X-ray, as recommended by the WHO. Chest X-ray, of course, would be undertaken to rule out TB disease should TB preventive treatment be offered. Our study was conducted in one tertiary hospital in West Java province, which is a large facility in an area of a high number of annual reported TB cases, and is one of the 13 facilities designated for MDR-TB patients in Indonesia. Therefore, our results may be different to other hospitals throughout the country, limiting their generalisability. We did not include non-HCWs as a control group, and we did not have general population estimates to compare with.
In conclusion, the high rate of M. tuberculosis infection and of TB cases in this hospital is alarming and serves as a convincing reason to do operational research to improve TB infection control to protect HCWs, especially in this era of escalating MDR-TB. A package of TB infection control measures, including personal respiratory protection and education, will need to be implemented, not only in specific high-risk areas but also right across the hospital and for all HCWs. HCWs in this setting might also gain benefit from TB preventive treatment against the development of TB disease, and this requires further investigation.
Supplemental Material
sj-pdf-1-bji-10.1177_17571774211046887 – Supplemental Material for Mycobacterium tuberculosis infection and disease in healthcare workers in a tertiary referral hospital in Bandung, Indonesia
Supplemental Material, sj-pdf-1-bji-10.1177_17571774211046887 for Mycobacterium tuberculosis infection and disease in healthcare workers in a tertiary referral hospital in Bandung, Indonesia by Lika Apriani and Susan McAllister in Journal of Infection Prevention
Supplemental Material
sj-pdf-2-bji-10.1177_17571774211046887 – Supplemental Material for Mycobacterium tuberculosis infection and disease in healthcare workers in a tertiary referral hospital in Bandung, Indonesia
Supplemental Material, sj-pdf-2-bji-10.1177_17571774211046887 for Mycobacterium tuberculosis infection and disease in healthcare workers in a tertiary referral hospital in Bandung, Indonesia by Lika Apriani and Susan McAllister in Journal of Infection Prevention
Footnotes
Acknowledgements
The authors thank the Director of Hasan Sadikin General Hospital for facilitating the study, the HCWs for participating in the study and the research staff (Ita Aprianti) for collecting data.
Author contributions
LA, DM, KS, SM and PCH designed the study. LA, IA and HN undertook the data collection. LA undertook the statistical analysis on advice from KS. LA, SM, KS, RR, BA, DM and PCH interpreted the data. LA drafted the manuscript. All authors read, revised and approved the final manuscript.
Declaration of conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LA was supported by a University of Otago Doctoral Scholarship. Research costs were sponsored by the Centre for International Health with donations from Mercy Hospital Charitable Trust, Dunedin, New Zealand. The funders had no role in the study design, data collection, analysis or preparation of the manuscript
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

