Abstract
The prognosis and optimal management of bladder cancer are closely dependent on accurate clinical staging. Traditional staging has relied on transurethral resection for T-staging in combination with cross-sectional imaging for N and M-staging. Computed tomography (CT) is the most accessible and common cross-sectional imaging modality to assess for local and distant spread of disease. However, CT alone lacks the contrast resolution to distinguish between non-muscle-invasive and muscle-invasive disease and has known difficulty distinguishing metastatic from normal lymph nodes (LNs). Magnetic resonance imaging (MRI) and positron emission tomography (PET) have been increasingly used in the workup of bladder cancer to address the shortcomings of CT imaging. MRI using the multiparametric combination of T2-weighted, dynamic contrast-enhanced, and diffusion-weighted imaging can more clearly delineate the extent of tumor involvement in the bladder. PET can use a variety of metabolically active radiotracers to visualize cancerous tissue, allowing the identification of metastases that may not be readily visible on CT. PET scans can also detect disease in normally sized LNs that do not meet conventional CT size thresholds. The combination of MRI and PET scans has not been explored in detail with respect to bladder cancer workup but may theoretically combine the strengths of both approaches to provide the most comprehensive, noninvasive tool for preoperative staging, with improved sensitivity and accuracy compared to other modalities. This narrative review uses a systematic search of the PubMed database from 2000 to 2025 to analyze MRI and PET, both separately and in synergy, for the local and distant staging of bladder cancer, as well as key technical advancements in these imaging technologies. Synergy of the imaging modalities potentially increases efficacy in bladder cancer staging.
Keywords
Introduction
Bladder cancer accounts for approximately 4% of all new cancers in the USA and 3.1% for all new cancers in the world.1,2 Patients undergo an initial workup of cystoscopy to identify the primary tumor with abdominopelvic cross-sectional imaging to search for regional/distant spread. 3 Subsequently, surgical resection via transurethral resection of bladder tumor (TURBT) is performed to assess the final diagnosis, grade, and local stage. For non-muscle-invasive bladder cancer (NMIBC), tumor grade is the most important predictor of survival, but the most important prognostic indicators of muscle-invasive bladder cancer (MIBC) survival remain the stage at which a patient is diagnosed.4,5 Lymph node (LN) involvement in particular worsens survival with only 20%—30% surviving 5 years after diagnosis. 6
Bladder cancer is ultimately staged based on pathologic analysis from TURBT and assessment of regional and distant spread on abdominopelvic imaging according to the TNM classification system. 7 Stages range from Ta, which is non-invasive papillary carcinoma, to T4, in which cancer has invaded surrounding structures such as the prostate or uterus. 7 Given that higher stage correlates with worse survival, accurate staging is critical.5,7 Understaging remains a critical concern with reports of 46%—48% of cT1 patients found to be understaged after cystectomy, even after re-staging TURBT. 8 Additionally, significant interobserver variability amongst pathologists has been reported resulting in a change of diagnosis on TURBT specimens for over a third of second opinion reviewed cases. 9 The regional staging of urothelial carcinoma with assessment of LN involvement, while critical to the proper prognostication of oncologic outcomes and derivation of treatment pathways, has also proved problematic. Pathologic evaluation and detection of micrometastatic deposits within LNs, for example, can vary based on the methodology of specimen processing and the different approaches to identifying suspicious nodes on preoperative imaging used. 10
The American Urological Association (AUA)/American Society of Clinical Oncology (ASCO)/American Society for Radiation Oncology (ASTRO)/Society of Urologic Oncology (SUO) guidelines on bladder cancer recommend staging evaluation of bladder cancer with chest and abdominopelvic imaging with contrast. 11 Computerized tomography (CT) scans have been useful in identifying stage T3b bladder tumors, which have invaded into the perivesical fat.12,13 Studies report that the sensitivity and specificity of CT in staging T3 bladder tumors are around 80% and 100%, respectively.13,14 Nevertheless, multiple limitations exist with CT, most notably an inability to distinguish between T1 and T2 disease, along with low sensitivity to detect LN invasion, and high false-positive rates for extravesical extension, among others.13,15 –17 These limitations warrant the investigation of alternate imaging modalities for staging bladder cancer patients. Two notable alternatives include positron emission tomography (PET) imaging and magnetic resonance imaging (MRI). MRI may have improved resolution for the local extent of bladder cancer and may more effectively identify suspicious LNs, while PET scans may have improved sensitivity and specificity for regional and distant metastases. Unfortunately, guidelines from societies like the AUA, ASCO, ASTRO, SUO, and European Association of Urology (EAU) lag in their inclusion of MRI and PET. The purpose of this narrative review is to provide an overview of contemporary staging performance for MRI and PET in patients with bladder cancer, and to illustrate how these modalities could be used to mitigate the recognized challenges to accurate clinical staging.
Methods
A single author performed queries of the PubMED database using combinations and synonyms of the terms “multiparametric MRI,” “bladder cancer,” “positron emission tomography,” and “pelvic lymph node” up to July 1, 2025. Studies were screened to only include articles pertaining to the use of multiparametric triphasic MRI (mpMRI) or PET scans to perform local or nodal staging of bladder cancer. The references of the identified papers were also screened to expand the scope of the literature search.
Duplicated studies, non-English studies, case reports, and case series were excluded. Studies published before the year 2000 were also excluded. Studies solely examining the staging of bladder cancer after resection or chemotherapy were excluded from systemic review of the efficacy of PET and MRI but were included in the discussion of imaging in the broader clinical context. Review articles and a non-systematic search of the literature were included only to provide a descriptive summary of the underlying mechanisms of each imaging modality. When studies with overlapping patient populations were assessed, we used the most recent study.
Each study included in systemic review was analyzed for data regarding sensitivity, specificity, positive predictive value, negative predictive value, overall accuracy, and area under the receiver operating characteristic curve. Data regarding the study size and study type was also recorded. Studies were organized into tables based on imaging modality.
MRI in bladder cancer
MRI presents a promising alternative to CT scans with respect to pre-treatment staging due to the higher soft tissue contrast. 12 Within the current guidelines, MR urography (MRU) is recommended for upper tract evaluation in cases where there are contraindications to CT urography or when multiple scans are needed to minimize radiation. 15 A typical MRU protocol combines T2 weighting for a static fluid evaluation alongside gadolinium-enhanced T1 weighting for a multiphase dynamic evaluation, including excretory phase imaging to highlight the upper tracts. 18 MRU has high sensitivity in detecting ureteral obstruction secondary to malignancy at around 90% and is equivalent to CT urography with respect to diagnosing bladder cancer.19,20 An additional important advantage of MRU over CT is the high sensitivity and specificity of diffusion-weighted imaging (DWI) for urothelial carcinoma in both the bladder and upper tracts. This unique contrast mechanism, highlighting malignant lesions via lower apparent diffusion coefficient (ADC) values, may also predict the invasiveness of urothelial tumors. 21 Data directly comparing CT to MRI for bladder cancer staging is sparse, although one recent study did demonstrate a slightly higher accuracy for MR in detecting muscle invasion, with one reader improving from 66.7% to 75.6% and the other improving from 75.6% to 78.9%. 22
The ideal MRI sequences for complete evaluation of bladder cancer are a multiparametric combination of DWI, T2-weighted imaging (T2WI), and dynamic contrast-enhanced (DCE) imaging. 18 The combination of anatomic and functional evaluation provided by mpMRI results in improved sensitivity and specificity of cancer staging. 23 A study by Takeuchi et al., 24 for example, showed that combining T2WI and DWI has been shown to improve T staging accuracy from 62% to 88%, with the addition of DCE further improving accuracy to 92%. 24
T2 is capable of differentiating tumors, which have intermediate to high signal intensity, from normal detrusor, which appears as a hypointense line, and can identify MIBC with moderate accuracy.24,25 It is recommended to acquire multiplanar fast spin echo 2D images in at least 2 planes with a slice thickness of 3–4 mm and a phase matrix of at least 256 for optimal resolution. 12 Ta and T1 stage disease do not show any interruption in detrusor signal intensity, while T2 disease would. 18 T3b disease is identified as the T2 signal intensity of the tumor extending into the perivesical fat, leading to poor definition of the bladder wall. 26 It should be noted that T2 disease cannot be differentiated from T3a disease by definition, as T3a disease is microscopic, though both stages have the same management. 15 T4 disease can be identified on T2WI as an extravesical tumoral signal that replaces the normal signal of an adjacent structure. 27
DWI evaluates the Brownian movement of water molecules, which is reduced (“restricted”) by the high cellularity of tumors. 12 Bladder tumors have increased signal intensity on DWI with accompanying reduced ADC value that represents the restricted diffusion of movement. 28 A classic imaging sign in lower-stage disease is the “inch worm” sign, in which the stalk of a papillary tumor has low DWI signal while the rest of the tumor has a high signal. 24 Kazan et al. 29 found that the inchworm sign had a negative predictive value of 94.6% for MIBC, and assert that its absence can be interpreted by urologists. A thickened submucosal signal in a sessile tumor on DWI is another sign of low stage, whereas T2 disease demonstrates elevated smooth signal intensity without submucosal components or extension into the perivesical space. 26 T3 disease typically demonstrates an irregular contour of high DWI signal in muscular invasion or perivesical extension, and T4 disease involves high DWI signal in adjacent organs. 26 DWI has better accuracy than T2WI for differentiating invasive and noninvasive cancer, and may even help differentiate high-grade T1 disease from less severe phenotypes.30,31 However, there is no universally accepted ADC threshold value to distinguish tumor from surrounding tissue, resulting in possible under-staging of some infiltrative tumors.32,33 Furthermore, the low spatial resolution may impede identification of small tumors, and nonmalignant material such as a blood clot may also have a high DWI signal, highlighting the importance of combining data from other sequences for multiparametric interpretation. 13
Finally, DCE involves 3D T1-weighted fat-suppressed gradient echo imaging obtained prior to, and at multiple timepoints after, the intravenous administration of gadolinium-based contrast media, which allows the dynamic assessment of tumoral enhancement features due to its abnormal vascularity. 12 As opposed to CT, multiple timepoints can be evaluated without concern for mounting radiation dose. Urothelial carcinomas enhance earlier than the muscle layer, so that contrast between the two tissues is maximized at this timepoint. In late timepoints, the bladder lumen is filled with excreted contrast media, highlighting the lesion as a low-signal filling defect. 34 It has been suggested that DCE offers enhanced resolution, allowing for clearer definition of the individual bladder layers and for artifacts like perivesical fat stranding to be differentiated from the tumor. 35 T1 tumors have no early enhancement of the muscle, T2a lesions show interruption of muscle signal without clear enhancement, T2b lesions have interruption of the muscle signal with early enhancement, and T3b and T4 lesions show early enhancement involving the perivesical fat and surrounding structures, respectively. 36
MRI for local staging of bladder cancer
mpMRI is an effective tool for the local staging of bladder cancer, owing to the complementary aspect of its component sequences. A retrospective study by Yamada et al. 37 demonstrated an overall staging accuracy of 80% across a 100-patient subset that incorporated DWI, which improved to 83% when assessing for muscle invasion. Additional studies showed improved accuracy for T staging, ranging from 87% to as high as 95%.23,24,38 –42 These studies showed similar accuracy for muscle invasion, ranging from 68% to 98%. Three of these studies reported the area under the receiver operating curves for detection of MIBC, ranging from 0.99 to 1. A summary of this data can be found in Table 1. Though each of these studies is limited by small sample sizes, mpMRI staging nonetheless appears to correlate strongly with pathological staging. This is improved from a reported accuracy of 56% for T staging in a recent meta-analysis by Cornelissen et al., 43 which included studies that did not use all three mpMRI sequences to evaluate T stage. With respect to identification of muscle invasion, a meta-analysis by Woo et al. 44 found that mpMRI had a sensitivity of 94% and specificity of 95%, which was noted to be improved compared to studies examining conventional MRI and concordant with the studies identified in our review.
Studies using mpMRI to stage bladder cancer without VIRADS.

mpMRI, multiparametric triphasic Magnetic Resonance Imaging; VIRADS, Vesical Imaging Reporting and Data System.
Using a 5-point Likert score, the VI-RADS (Vesical Imaging Reporting and Data System) classification system has been proposed to standardize the reporting of mpMRI findings corresponding to the likelihood of MIBC, allowing for more generalizable clinical applications. 45 Each mpMRI parameter (T2, DWI, DCE) is scored separately. A score of 1 or 2 predicts muscularis invasion to be unlikely; a score of 4 or 5 predicts muscularis invasion to be likely; a score of 3 is considered equivocal for invasion. 46 With respect to imaging findings, VI-RADS 1 and VI-RADS 2 lesions both show uninterrupted low signal intensity corresponding to an intact muscularis, although a VI-RADS 2 lesion is >1 cm and may have a thickened inner layer or stalk. VI-RADS 4 lesions demonstrate an interruption of the low signal intensity muscularis, and VI-RADS 5 lesions additionally show the tumor’s signal intensity reaching the perivesical fat. A VI-RADS score of 3 covers all lesions with no clear disruption of the muscularis signal intensity, but no specific thickened inner layer or stalk. 46 T2WI is primarily used to score the structure of the tumor, while DWI and DCE are used to score invasion, with DWI being the dominant score when T2WI and DCE are discordant. 46
Since its introduction in 2018, VIRADS has been verified to have high inter-observer reproducibility and accuracy in multiple studies.47 –54 Ueno et al. 55 performed the first such study examining 74 patients and found an intraclass correlation coefficient of 0.85, showing excellent inter-reader agreement, and an area under the receiver operating curve of 0.9, showing excellent accuracy. A later study by the same group found similar results even for less experienced radiologists. 56 There is some debate on the optimal VIRADS cutoff to measure muscle invasiveness, with both VIRADS 3 and VIRADS 4 being proposed. Based on our review, either cutoff may be used effectively (Table 2). Studies using VIRADS 3 as a cutoff had sensitivities ranging from 70% to 100%, and specificity ranging from 54% to 100%.22,48,50,52 –54,56 –72 Accuracy for muscle invasion ranged from 76% to 95%, and the area under the receiver operating curve ranged from 0.81 to 0.96. For VIRADS 4 as a cutoff, sensitivity ranged from 64% to 100%, and specificity ranged from 76% to 100%, which reflects the higher threshold for diagnosing muscle invasion.47,49,51,53,54,56,66,73 –85 Regardless, ranges for accuracy and areas under the receiver operating curve were similar, from 75% to 98%, and from 0.83 to 0.99, respectively. On meta-analysis, the cutoff of VIRADS 3 appears to be slightly superior. Del Giudice et al. 86 found an area under the curve of 0.93 for VIRADS 3 and 0.91 for VIRADS 4 across 20 studies, though both correspond to effective discrimination of muscle invasion. Al-Qudimat et al. 87 found that a VIRADS cutoff of 3 was optimal in a large meta-analysis of 31 studies, finding that it corresponded to a pooled sensitivity and specificity of 89%. Though the VIRADS system appears to be reproducible and effective to interpret even for less experienced radiologists, it continues to be assessed for improvement. Further work is underway for integrating artificial intelligence (AI) in the arena of radiomics to automate and further standardize the interpretation of MRI for bladder tumor delineation and local staging. 88
Studies using mpMRI to identify MIBC with VIRADS.

MIBC, muscle invasive bladder cancer; mpMRI, multiparametric triphasic magnetic resonance imaging; VIRADS, Vesical Imaging Reporting and Data System.
Multiparametric MRI presents a potentially effective screening and prognostic tool for bladder cancer. Bryan et al. 89 performed a randomized control study in which they compared time to correct treatment in patients initially staged with cystoscopy and mpMRI to patients initially staged with TURBT, finding that mpMRI-directed staging significantly decreased the time to correct treatment of MIBC without having a detrimental effect on patients with NMIBC. A VIRADS cutoff of >2 in mpMRI has also been shown to be effective in predicting MIBC on restaging TURBT in patients with high-risk NMIBC, with a sensitivity of 85% and specificity of 94%, leading to an area under the ROC of 0.93 in a study by Del Giudice et al. 86 A cutoff of VIRADS <3 after the first TURBT was associated with a negative subsequent TURBT in over 97% of patients in another study. 69 Furthermore, in a study of bladder cancer patients before and after neoadjuvant chemotherapy, the pre-treatment and change in both the ADC and tumor size on mpMRI were found to be significantly associated with clinical response to chemotherapy. 90 Though these studies show that staging by mpMRI is promising, the sample sizes of patients with MIBC in each study are small, and the results must be interpreted with caution.
MRI for regional staging of bladder cancer
Nodal disease in bladder cancer typically spreads through the pelvic chain, with one regional metastasis classified as N1, >1 regional metastasis classified as N2, and common iliac involvement classified as N3. 91 Increased LN involvement is closely correlated with reduced cancer specific survival after radical cystectomy, as well as higher T stages, with 30% of T2 and 60% of T3 disease involving the LNs.92,93 LNs are classically evaluated by CT or MRI to identify characteristic morphologic features, while accounting for the fact that disease tends to spread along the standard pelvic lymphatic drainage pattern without skipping sites.18,94 Conventional CT and MRI, utilizing nodal size and shape, are generally regarded to have similar accuracy, with detecting metastases. 34
The prediction of LN metastasis has always been somewhat problematic with conventional CT or MRI, particularly regarding the ability to strike a reasonable balance between sensitivity and specificity for a given threshold value or combination of values. The assessment of LN metastasis is traditionally stratified by size, with a >8 mm cutoff being most commonly used. 15 Wollin et al. 95 found that having a short axis >5 mm was the most accurate predictor of metastasis with 88% sensitivity and a more modest specificity of 75%. Notably and problematically, it is not uncommon to have metastases in normal sized LNs, with Thoeny et al. 96 finding that over 90% of micrometastases in LN dissection were found in nodes 5 mm or less. Many investigators have therefore examined additional MR imaging features beyond size to improve metastatic nodal detection. 18
Mir et al. 97 found that adding DWI to conventional T2WI improved detection of pelvic LNs, but did not improve distinction of metastatic from normal nodes. Thoeny et al. 96 found that the use of DWI was able to identify metastasis in normal sized pelvic nodes of patients with bladder cancer with an accuracy of 64%—79%. Papalia et al. 98 found a significant difference in mean ADC values between positive and negative nodes in bladder cancer patients with normal CT node diameters, with a sensitivity of 76.4% and specificity of 89.4%. However, derived ADC values may be affected by the relatively small size of nodes compared to the slice thickness of DWI, and ADC may be falsely lowered by other conditions such as inflammation, reactive hyperplasia, and lipomatosis. 18 MR lymphangiography with ultrasmall superparamagnetic iron oxide (USPIO) agents has recently become the most promising avenue to identify malignant LNs, being associated with higher sensitivity. 99 This technique reduces the T2 signal of normal nodes via accumulating iron oxide and has demonstrated impressive accuracy of up to 92% and a 65%—75% sensitivity with 93%—96% specificity.100,101 However, MR lymphangiography currently requires specialized expertise and involves the off-label use of USPIO agents, hampering its widespread use.
Regarding the overall diagnostic accuracy of MR for the detection of metastatic LNs, limited data from meta-analyses is available. Woo et al. 99 evaluated 24 studies including 2928 patients with bladder or prostate cancer, finding a poor pooled per-patient sensitivity of 57% but a high specificity of 96%. A subgroup analysis did reveal a significantly higher sensitivity of 86% for studies utilizing USPIO agents compared to 46% for studies that did not use USPIO agents. Another meta-analysis by Crozier et al. 102 evaluated 10 studies, with a similarly modest summary sensitivity point estimate of 60% and high pooled specificity point estimate of 91%. In our review of the literature, we found eight studies that evaluated the efficacy of MRI in LN staging for bladder cancer only. Sensitivity ranged from 41% to 96%, excluding a study without positive nodes detected, whereas specificity ranged from 64% to 95% and accuracy ranged from 67% to 95% (Table 3).95,98,103 –108 Applying the results of these studies, however, is problematic in that many different node assessment criteria were used to define suspicious nodes.
Studies using mpMRI for regional lymph node staging.

ADC, apparent diffusion coefficient; mpMRI, multiparametric triphasic MRI.
PET scans in bladder cancer
PET imaging in bladder cancer most commonly employs 18F-fluorodeoxyglucose ( 18 F-FDG), a radiolabeled glucose molecule taken up by GLUT-1, which is overexpressed in bladder cancer cells due to increased glycolytic activity.12,109 The radiotracer is subsequently trapped in the cell by rapid phosphorylation and then released after dephosphorylation into the urinary system. 110 This metabolic imaging provides functional information that is not attainable with anatomic imaging alone, allowing for the identification of hypermetabolic lesions that may not yet be morphologically apparent on CT or MRI. PET is frequently paired with CT imaging in order to give a proper anatomical reference frame. 110 The main drawback of 18F-FDG PET/CT scan in bladder cancer staging is the urinary excretion of 18F-FDG, which severely limits its sensitivity to detect intravesical tumors and local invasion. However, 18F-FDG PET serves as an alternative tool to perform accurate staging thanks to its use in detecting nodal or distant metastatic disease. Unlike MRI, there is no clear consensus on the role of PET scans in the staging of bladder cancer, with the NCCN guidelines recommending PET/CT scans with level 2b evidence in the workup of select patients at risk for having metastases. 111
Several alternative tracers to 18F-FDG that are not excreted in urine have been proposed to improve local staging for PET, such as carbon-11 ( 11 C) tracers, which can include choline and acetate. 11C-choline is taken up by the rapidly proliferating tumor cells, and has almost no or negligible urinary clearance, offering improved visualization of the bladder wall. 112 11 C-choline is normally incorporated into the cell wall and thus is preferentially incorporated into rapidly dividing cells like tumors. 11C-choline uptake in tumor cells is dependent on the rate of lipid synthesis, as it is incorporated into the lipid pool. A similar process occurs with 11C-acetate, as both are eventually metabolized into phosphatidylcholine. 113 However, the half-life of 11C is relatively short, at 20 min compared to 110 min for FDG, which may limit the effectiveness of 11C-labeled studies if there is no cyclotron on site. 110
Most recently, 18F and 68Ga radiolabeled FAPI agents show promising preliminary results. FAPI-targeted molecular imaging utilizes cancer-associated fibroblasts, which are abundant in most solid tumor microenvironments. 114 These fibroblasts overexpress FAP receptors, which then can be targeted via FAPI PET to assess the extent of disease. However, urinary excretion of FAPI tracers faces similar problems to 18F-FDG PET at local staging due to intense background uptake within the bladder. Nevertheless, approval of FAPI PET is eagerly awaited as this method allows for earlier detection of micro metastases thanks to improved specificity of molecular PET imaging.
PET/CT in local staging of bladder cancer
Data is variable on the use of PET/CT for staging bladder cancer, and few guidelines recommend or endorse it for local staging of bladder cancer. Still, there are instances where it is useful in the T staging of bladder cancer patients. The EAU and NCCN recommend CT or MRI to stage MIBC, and the EAU guidelines specifically state that “the additional value of fluorodeoxyglucose PET/CT still needs to be determined.”15,111
18 F-FDG PET is able to identify bladder cancer relatively accurately, though data regarding its efficacy for evaluating muscle invasion is sparse. Nayak et al. 115 performed a prospective study with 25 patients with suspicion of bladder cancer who underwent intravenous contrast-enhanced CT along with PET/CT. In this study, the sensitivity to detect primary bladder tumors was greater in PET/CT (96% vs 92%), so the authors concluded that 18F-FDG PET/CT was superior to contrast-enhanced CT for local bladder cancer staging. A prospective study by Lodde et al. 116 compared PET/CT to CT in detecting residual cancer after TURBT, also finding that PET/CT had better sensitivity than CT (85% vs 77%) but a poor specificity of 25%, making it a poor option for excluding residual disease. PET/CT was found to have 89% concordance with final clinicopathologic TNM staging, and correctly staged all stage 2 cases in a retrospective study. 117 Though the study found that PET/CT had better concordance with pathology than CT, this improvement was primarily centered around detecting distant metastasis, and the study was limited by a small population of patients with localized disease. Bacchiani et al. 118 performed a narrative review, including 23 studies involving PET/CT for staging bladder cancer, and concluded that the limited diagnostic accuracy due to intense background 18F-FDG excretion in the bladder makes its use uncertain for identifying bladder lesions. A meta-analysis by Zhang et al., 119 which examined both primary and recurrent/residual bladder cancer, found the sensitivity and specificity of PET/CT to identify bladder lesions to be 82% and 92%, respectively. Overall, there is a paucity of level 1 evidence on PET/CT’s local staging of bladder cancer, especially prior to intervention, which warrants alternative approaches in initial staging and detection.
The most notable limitation in PET/CT’s ability to locally stage bladder cancer is 18F-FDG’s natural excretion into the bladder. 120 Intense 18F-FDG clearance into the bladder obscures tracer-avid bladder wall lesions, which demonstrate less uptake than the urinary activity. 115 Nayak et al. 115 have proposed the use of diuretics and retrograde bladder irrigation to overcome these limitations. Anjos et al. 121 employed a similar protocol where furosemide was used and washout imaging was obtained with PET/CT in 17 patients with either a prior cystectomy (N = 11) or no cystectomy (N = 6). They found significant improvement with the diuretic washout protocol in the detection of bladder lesions and in bladder tumor recurrence. Kamel et al. 122 ’s study of 32 bladder cancer patients (14 which were undergoing initial staging) who received PET scans with a furosemide administration protocol found that diuresis and hydration removed significant 18F-FDG activity from the bladder/ureters in 97% of patients. Higashiyama et al. 123 investigated the role of oral hydration in improving tumor detection by prospectively enrolling 267 bladder cancer patients who underwent 18F-FDG PET/CT scans before and after fluid intake. Patients held their urine for 1 h for delayed imaging after initial image acquisition to increase bladder volume, decrease urinary 18F-FDG activity in the bladder, and maximize tumor-to-background ratio. The sensitivity for detecting residual MIBC on early and delayed imaging was 24% and 92%, respectively, which was statistically significant. Likewise, Mertens et al. 124 published a prospective analysis of 18F-FDG PET/CT imaging in bladder cancer patients with four different filling/draining protocols. The authors found that bladder flushing with retrograde filling resulted in the best bladder tumor visualization on PET/CT. These studies clarify that if PET/CT is to be used in locally staging bladder cancer, some type of bladder washout needs to be employed to optimize tumor identification.
Carbon-11 labeled tracers are often proposed as an alternative to 18F-FDG due to their lower renal clearance, which provides certain advantages, particularly in cases of local recurrence. Schöder et al. 125 performed a small study examining 11C-acetate PET/CT, finding a 80% accuracy for detecting residual bladder cancers, though false positives were noted after BCG installation or cystitis. Vargas et al. 104 found that 11C-acetate PET/CT had similar accuracy to CT and MRI in staging bladder cancer as a whole. Graziani et al. 126 examined the efficacy of 11C-choline PET/CT and found a sensitivity of 66.7%, specificity of 84.6%, and accuracy of 76% in detecting local recurrence. Picchio et al. 127 found that 11C-choline PET/CT was similarly effective in detecting residual bladder cancer in a 27-patient cohort undergoing cystectomy, and found that choline PET/CT had a slightly higher accuracy of 96% compared to 84% for CT for residual tumors after TURBT on histopathology. 11C-acetate is a radiotracer that utilizes lipid metabolism, and is less commonly used in clinical practice for bladder cancer. Early reports showed that 11C-choline may have better resolution and uptake compared to 11C-acetate, although the data is limited. 128 Overall, the role of 11C-labeled radiotracers in the detection of bladder tumors remains elusive, and the issues with production and short half-life of 11C (20 min) limit further investigations.
PET/CT in regional staging of bladder cancer
The primary benefit for PET/CT is hypothesized to be more accurate staging of LNs and distant metastases compared to CT alone. The accuracy of 18F-FDG PET in detecting positive LNs is generally reported to be between 80% and 90%. 120 However, 18F-FDG-PET/CT tends to have low sensitivity and high specificity, much like conventional CT imaging, indicating that it is most beneficial for ruling out nodal disease. 129 In our review of articles using 18F-FDG PET/CT to identify nodal metastases, the accuracy ranged from 62% to 92% with areas under the receiver operating curve ranging from 0.64 to 0.97, indicating moderate to good discrimination for positive nodes (Table 4).103,116,130 –154 Soubra et al. 133 performed the first meta-analysis examining 18F-FDG PET/CT for regional nodal staging in bladder cancer, finding a 57% pooled sensitivity and 95% pooled specificity for positive nodes. However, the authors noted that there was great variation in methodologies, inclusion criteria, interpretation criteria, and the use of diuresis, contrast, and neoadjuvant chemotherapy. In a subsequent meta-analysis, Ha et al. 155 found a similar sensitivity of 57% and specificity of 92% on a per-patient basis, and Aydh et al. 156 found similar sensitivity and specificity with an overall accuracy of 65%—89%.
Studies using 18F-FDG PET for regional lymph node staging of bladder cancer.
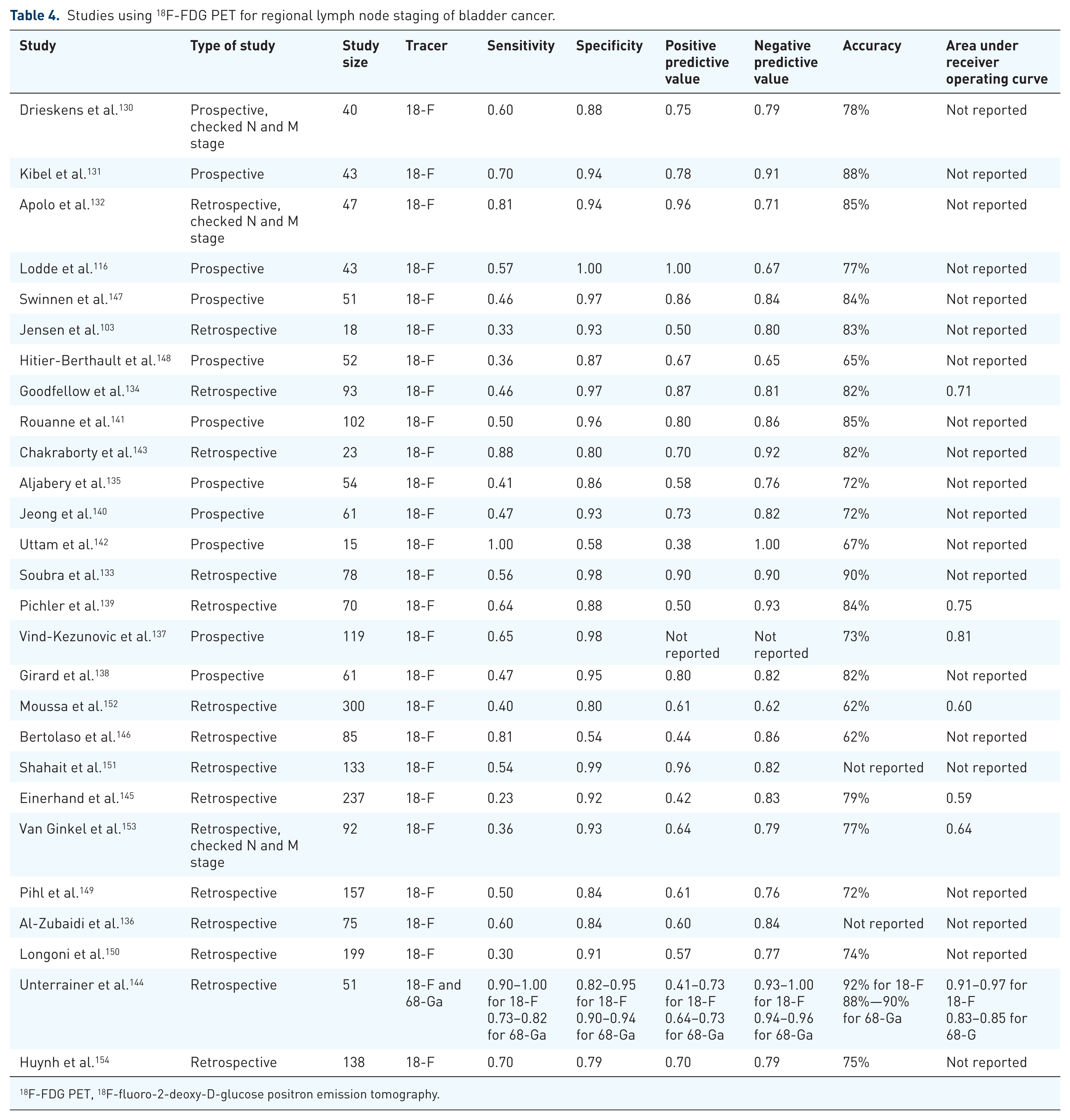
18 F-FDG PET, 18F-fluoro-2-deoxy-D-glucose positron emission tomography.
PET/CT has been advocated to be particularly helpful in detecting metastatic LNs that are not large enough to be detected by conventional imaging. A prospective study by Kibel et al. 131 performed 18F-FDG PET/CT imaging to assess N stage in 43 MIBC patients with negative CT scans prior to cystectomy and showed that PET/CT had a sensitivity of 70% and a specificity of 94% for detecting metastases. On the other hand, Pichler et al. 139 found that 18F-FDG PET/CT resulted in significant improvement of sensitivity over CT when the LN size cutoff was >10 mm, but not when the cutoff was >8 mm, suggesting some limitations in using 18F-FDG PET/CT to detect cancer in smaller nodes. However, a later study by Huynh et al. 154 was able to find significantly improved sensitivity for 18F-FDG PET/CT compared to CT alone using the >8 mm cutoff. Recently, Girard et al. 138 found that by combining LN size criteria and standard uptake values in the interpretation of 18F-FDG PET/CT, accuracy was improved by 6% on a per-area analysis and 8% on a per-patient analysis. The most recent meta-analyses comparing PET/CT to CT reflect the improved ability of PET/CT to detect positive nodes. Kim et al. 120 found a weighted average sensitivity of 51.3% for PET/CT versus 38.8% for CT alone, while Girard et al. 129 found a pooled sensitivity of 52.4% for PET/CT versus 37.5% for CT alone. Jensen et al. 103 performed the only known trial directly comparing PET/CT to MRI for regional LN staging, and found an 80% specificity for MRI and a 93.3% specificity for PET/CT, a difference that was not statistically significant but may be clinically relevant. The authors indicated that limited conclusions could be drawn from the small sample size in the study, and felt that the data overall favored PET.
Positive 18F-FDG PET/CT scans have shown both prognostic significance and provide information to change patient care. The presence of 18F-FDG avid disease is associated with higher recurrence rates and worse overall survival, reflecting the aggressive biology of metabolically active tumors. 157 Extravesical lesions on PET/CT have been shown to be an independent predictor of mortality on multivariate analysis, with a hazard ratio of 3. 158 Kibel et al. 131 found that positive PET imaging was associated with worse overall survival, disease-specific survival, and recurrence-free survival over a median follow-up of 14.9 months. Huynh et al. 154 found that positive nodes on 18F-FDG PET/CT were associated with poorer 5 year survival, but that positive nodes on CT were not, indicating that 18F-FDG PET/CT may provide a more accurate assessment of regionally advanced disease. A retrospective study by Coskun et al. 159 showed that MIBC was significantly more likely to have increased nodal staging identified on PET/CT compared to CT alone. This finding was supported by a population based study, which found that 18F-FDG PET/CT found nodal metastases in 21% of MIBC patients with no nodal metastases on CT, and that patients with nodal metastases were more likely to be treated with chemotherapy. 160 A retrospective single institution study by Shahait et al. 151 found that 18F-FDG PET/CT changed management in 26.3% of the patients compared to if CT alone was used in work up. This change in management was evenly split between MIBC and NMIBC, as well as from palliative to curative and vice versa.
11 C-choline has emerged as a possible alternative to 18F-FDG as a PET tracer for nodal staging. The initial proof of concept study by de Jong et al. 161 showed a sensitivity of 67% and specificity of 100% in detecting involved LNs. A more recent and larger study by Ceci et al. 162 showed similar results with a sensitivity of 59% and specificity of 90% as well as an overall accuracy of 80%. However, the data on whether 11C-choline PET/CT is superior to conventional CT for regional staging is divided. Picchio et al. 127 found that 11C-choline PET/CT had a 62% accuracy for detecting positive nodes compared to 50% for CT in a prospective study, with an absence of false positive nodes for 11C-choline PET/CT. Brunocilla et al. 163 showed improved sensitivity for 11C-choline PET/CT over conventional CT in patient, field, and lesion based analyses, though the two modalities had similar specificities. Meanwhile, Maurer et al. 164 reported no significant improvement in accuracy between 11C-choline PET/CT and conventional CT, as patient-based analysis showed 64% accuracy for 11C-choline PET/CT and 61% for conventional CT, while field-based analysis showed 91% accuracy for 11C-choline PET/CT and 90% for conventional CT. Because 11C-choline, unlike 18F-FDG, is not excreted by urine, 11C-choline PET/CT may be able to provide effective local staging as well as regional staging. However, positive 11C-choline PET/CT has not demonstrated an association with poorer survival, unlike 18F-FDG PET, and the 11C-choline radiotracer is more expensive and has a shorter half-life than 18F-FDG, as discussed above.120,165 The sole study comparing 11C-choline and 18F-FDG PET/CT for detecting metastases found no significant difference in accuracy between the two radiotracers. 166 A meta analysis by Kim et al. 167 found a moderate pooled sensitivity of 66% and a pooled specificity of 89% for 11C based studies, which is similar to the reported sensitivity and specificity of 18F-FDG PET. This was roughly concordant to our review, which found that sensitivity for 11C based studies ranged from 33% to 100%, specificity from 66% to 100%, and accuracy from 64% to 89% (Table 5).104,125 –127,161 –164,166
Studies using 11C PET for regional lymph node staging of bladder cancer.

PET, positron emission tomography.
Given challenges in 11C-labeled radioligand availability in the US, 18F or 68Ga radiolabeled FAPI tracers are highly anticipated. Early studies showed 30% increased detection rate compared to 18F-FDG PET/CT, especially in small (less than 1 cm) regional LNs.168,169 Unterrainer et al. 144 analyzed 51 patient in their retrospective study and found that FAPI PET/CT with either 18F- or 68Ga was superior to CT by providing 55.5% to 63.6% sensitivity for nodal metastatic disease, when compared to 30% sensitivity for CT. The authors also noted multiple distant metastases in this group of patients including peritoneal deposits and a patient with bone metastasis.
PET/MRI in bladder cancer
Low-dose CT with limited anatomical resolution underlying the conventional PET scans is another challenge in incorporating PET/CT imaging in bladder cancer staging. The integration of 18F-FDG PET with MRI represents a promising new approach to bladder cancer staging, as it combines the superior soft‑tissue resolution of MRI with the metabolic and functional insights provided by PET. While excessive urinary clearance of 18F-FDG remains as a pitfall, this synergistic effect between PET and MRI may improve the overall sensitivity and specificity, especially for locoregional staging and distinguishing muscle invasion, which are both paramount for treatment selection. This enhanced resolution of PET/MRI is especially useful within the pelvic malignancies since most PET/CTs lack oral or IV contrast depending on the institutional protocol, severely limiting the evaluation of the pelvic organs, and cause various challenges to distinguish small bowel loops or the ureters from possible sites of malignancies. PET/MRI is an excellent tool to overcome these hurdles, and it has been increasingly incorporated in the clinical practice whilst the medical community is aiming to increase its accessibility.
Catalano et al. 170 performed a retrospective comparative study of 18F-FDG PET/MRI with 18F-FDG PET/CT across a variety of cancers types, including pelvic malignancies, and found that PET/MRI findings would lead to a change in management over PET/CT in 17.9% of patients, with the opposite being true in only 1.5% of patients. These impacts were attributed to PET‑MRI’s superior sensitivity in detecting lymphadenopathy and local tumor extent in pelvic malignancies, suggesting a distinct advantage for its use in bladder cancer imaging. A later meta-analysis by Abdlkadir et al. 171 demonstrated the superiority of PET/MRI to PET/CT with respect to local staging of bladder cancer, with PET/MRI finding lesions 90% of the time versus 67% for PET/CT with the added benefit of reduced radiation exposure.
Rosenkrantz et al. 172 performed the first pilot study examining the ability of 18F-FDG PET/MRI to detect bladder cancer specifically. The accuracy of 18F-FDG PET/MRI was found to be 86% for bladder cancer and 95% for malignant pelvic LNs, leading to a change in suspicion for 36% of patients regarding local staging (75% correct) and 52% of patients for regional staging (95% correct). 172 In the most recent retrospective study by Li et al., 173 18 F-FDG PET/MRI showed high diagnostic performance for both local and regional staging of bladder cancer, with a sensitivity of 96.7%, specificity of 88.9%, and accuracy of 94.9% in detecting muscle-invasive disease, along with a sensitivity of 80%, specificity of 89.5%, and accuracy of 87.5% in detecting positive LNs. The authors also found that the change mean standardized uptake value (SUV) in 18F-FDG PET/MRI after neoadjuvant chemotherapy correlated significantly with tumor downstaging to NMIBC (⩽pT1), although these metrics are difficult to measure due to adjacent urinary activity. Patients whose tumors exhibited larger declines in mean SUV were more likely to have a complete pathological response, suggesting that 18F-FDG PET/MRI not only accurately stages disease but may also serve as a noninvasive biomarker for predicting and monitoring response to treatment. Salminen et al. 174 performed the sole prospective study exploring other tracers in PET/MRI, namely 11C-acetate. The sensitivity, specificity, and accuracy of detecting muscle invasion were 100%, 69%, and 73%, respectively, while the sensitivity, specificity, and accuracy of detecting LN involvement was 20%, 96%, and 80%, respectively. 174 Though the study size was small, the authors concluded that 11C-acetate was a viable radiotracer for use in PET/MRI, and may be more advantageous for local staging as 11C-acetate is not excreted in urine.
Eulitt et al. 175 then performed a small pilot study comparing 18F-FDG PET/MRI to conventional CT, analyzing 18 patients prior to cystectomy, most of whom received neoadjuvant chemotherapy. 18F-FDG PET/MRI had a sensitivity of 80% and specificity of 56% for detecting primary tumors with a sensitivity of 0% and specificity of 100% when detecting positive regional LNs. These findings were essentially equivalent to CT, which had a sensitivity of 91% and specificity of 46% for local staging, and a sensitivity of 0% and specificity of 93% for LN involvement. However, the authors noted that LN staging was limited due to the use of neoadjuvant chemotherapy, and that the CT scans were analyzed with access to the medical record, which may have improved accuracy compared to historical values. A more recent comparative study by Civelek et al. 176 evaluated 18F-FDG PET/MRI against conventional CT in patients with muscle‑invasive, locally advanced, or metastatic bladder cancer and reported a 36% higher lesion detection rate with PET/MRI, including metastases in LNs, liver, lung, soft tissue, and bone. This improvement led to clinically meaningful changes in treatment planning, including at least one patient whose management was altered based on these findings. However, the authors cautioned that PET/MRI is significantly more costly (~50% higher per examination) than PET/CT, and that PET/MRI scan times are substantially longer as well. While a typical PET/CT study takes around 20–25 min, PET/MRI studies commonly require 45–60 min or more, depending on the number of MRI sequences performed, which can introduce logistical challenges and reduced throughput.
Synergistic role of PET/MRI in bladder cancer
The synergistic roles of PET and MRI are exponentially utilized in various cancer types. Of these, bladder cancer is a new frontier following PET/MRI’s adoption in prostate cancer and neurodegenerative diseases in the past decade as each modality complements each other’s weaknesses. Coupled with molecular imaging, PET tracers have been adopted rapidly into the various national and international guidelines thanks to their improved detection rates, 177 including the prostate specific membrane antigen and the above mentioned FAPI tracers. Yet high soft-tissue contrast of MRI is still the mainstay in surgical planning for most pelvic malignancies as it provides excellent visualization of the local invasion and it provides information regarding involvement of the neurovascular and other structures in vicinity.
PET imaging without either CT or MRI provides fast and reliable staging for bladder cancer patients but lacks soft tissue resolution for the primary tumor. The complementary role of MRI strengthens the anatomic imaging in the pelvis, aids in differentiating local T staging (i.e., T2 vs T3a or T3b), and assesses the perivesical fat invasion better than PET, while the PET portion can assess the whole-body for metastases allowing for better patient selection prior to surgery. Simultaneous acquisition of PET/MRI allows co-registration of two cutting edge modalities to create near-perfect spatio-temporal image resolution.
Identification of muscle invasion is crucial in bladder cancer management. T2WI can distinguish NMIBC from MIBC by evaluating the low signal intensity muscle layer, although various conditions such as underlying inflammation or intermediate signal intensity can hinder accurate detection. 26 18 F-FDG FDG PET can confirm whether increased metabolic activity is present in this region, adding confidence to the designation of muscle invasion. Moreover, assessment of high b-value DWI (800–1000 s/mm2) creates a sharp contrast from the low signal intensity from the bladder lumen due to urinary activity, which helps distinguish urinary excretion from actual tumor FDG activity in PET. 178
Low ADC values in MRI are often correlated with greater metabolic activities, which may in turn be correlated with more aggressive tumors.41,179 Through a combination of ADC and SUVmax, PET/MRI yields a powerful, non-invasive, and quantitative tool for tumor characterization and understanding tumor biology that can further tailor the next best treatment.
Most FDG PET scans are followed by non-IV or oral contrast, low dose CT imaging for attenuation correction, and for anatomical correlation of the PET signal. This is the Achilles’ heel for most PET scans as they do not provide optimal visualization of the anatomical structures, most prominently in the lower pelvis. Contrast-enhanced CT can also alter the attenuation correction for PET due to overestimation of tissue density, which can lead to spurious increase in SUV. 180 DCE MRI images, however, do not alter PET attenuation correction as the PET quantification is obtained through other MRI data without over-correction from the contrast agent. 181 This allows artifact-free and improved resolution without compromising anatomical detail thanks to synergistic use of PET and MRI. This feature may clarify treatment response in patients undergoing neoadjuvant chemotherapy by accurately evaluating the change in SUVmax as well as the characteristic “wash-in and wash-out” pattern of contrast, similar to how residual tumors are differentiated from non-enhancing fibrotic tissue.
Drawbacks and cost implications of PET/MRI
One of the main drawbacks of PET/MRI is its capital cost compared to relatively cheaper PET/CT. PET/MRIs combine two complex and high-end imaging modalities and may require either a fully integrated or a separate MRI scanner along with PET. Each scanner needs shielding, advanced cooling technologies for conducting magnets, and software to avoid interference between two modalities. 182 This increases the cost of fully integrated PET/MRI scanners, as they need more space, highly skilled ancillary staff to operate both PET and MRI and maintenance costs while producing a lower patient throughput due to prolonged imaging time, inherently increasing per-patient cost. All of these factors led to a slower adaptation of PET/MRI, which are mainly available at certain academic and research centers despite clear advantages discussed above. 183
It is critically important to recognize the cost of PET and MRI scans in their relation to bladder cancer. Bladder cancer costs over 6.5 billion yearly in the United States alone. 184 Moreover, depending on the stage, diagnosis and treatment costs can range from ~$19,000 per year to upward of $169,000 per year for metastatic disease. 184 Huo et al. 185 compared PET/CT to MRI CT scans in imaging bladder cancer patients. The authors used Surveillance, Epidemiology, and End Results data between 2004 and 2011, finding increased utilization of PET/CT over time for all clinical stages of bladder cancer, while MRI decreased and no change was recorded for CT. Shockingly, they concluded that due to the rapid implementation of PET/CT, there was an estimated 11.6 million dollars of excess spending in bladder cancer alone. This is important given the data presented here, and questionable sensitivity in preoperative staging of PET/CT, with some estimates as low as 46%. 147 In a prospective trial by Mayerhoefer et al. 183 bladder cancer patients undergoing staging/follow-up underwent PET/CT and PET/MRI, with cost effectiveness compared. They noted that PET/MRI is 50% more expensive than PET/CT for bladder cancer imaging. However, they found PET/MRI to have greater accuracy and a greater likelihood to change patient management strategies, which exceeded the cost difference.
In addition to cost, time to obtain imaging studies is a consideration. PET/CT often requires patients to fast before undergoing an exam, and limit activity such as strenuous exercise. 186 While not specifically focused on bladder cancer, Virarkar et al. 187 compared PET/CT and PET/MRI in staging gynecologic malignancies. PET/MRI had a significantly quicker acquisition time in the study compared to combined PET/CT and MRI combination imaging. Muehe et al. 188 were also able to demonstrate that streamlined protocols can help reduce the time to acquire PET/MRI scans in patients with an additional reduction in radiation dosing relative to standard imaging for malignancies like PET/CT. Regarding PET/CT, some evidence exists that prolonged time from injection of FDG to PET improves visual interpretation of the study, however this is less practical in busy clinical settings relative to standard protocols.189,190
When considering PET/CT and/or MRI to stage a bladder cancer patient, clinicians need to be aware of the cost and time that comes with each modality, and weigh this against the potential benefit they may garner from image acquisition when deciding on an imaging strategy.
AI and future considerations
The role that AI will play in cancer management is still unclear, though PET/MRI technology is highly dependent on AI or deep-learning algorithms to tackle problems with image processing. Conventional PET/CT scanners utilize low-dose CTs to reconstruct the images for attenuation correction since each annihilation photon in PET scanners needs to travel through various tissues, including dense tissues such as bones or soft tissue. 191 This leads to underestimation of the counts as denser tissues block more photons, leading to a lower count PET study with suboptimal resolution. Evaluation of the bladder and pelvic organs is very challenging since pelvic bones and the bladder block a substantial number of photons, requiring attenuation correction more than other parts of the body. 192 While PET/CT scanners can overcome this by scaling the PET data with the underlying low-dose CT derived electron density information, PET/MRIs lack proper attenuation correction as MR signal does not provide any information regarding electron density. Thus, obtaining attenuation maps mainly relies on deep-learning algorithms or special sequences such as ultrashort echo time to predict the attenuation coefficients to process the image and improve resolution. 193
There are also ongoing efforts to improve the detection of bladder cancer with AI. Ye et al. 194 have published on the use of an AI deep learning model to differentiate NMIBC with MIBC on T2 weighted MRI, which was superior to a radiologist manually performing the task. Cao et al. 195 was able to generate an accurate diagnosis of muscle invasion using a machine learning paradigm called federated learning, which allowed data from four different centers to be used to train global model while preserving data privacy. Other models have also used radiomics from T2WI to predict likelihood of MIBC.196,197 AI’s ability to impact bladder cancer management is still in its infancy, but there is certainly promise in early publications. However, AI models need to be interpreted with caution and are not yet ready for routine clinical usage.
Conclusion
Although conventional CT remains the imaging modality used most for staging patients with bladder cancer, its limitations in detecting muscle invasion and micrometastatic LN involvement underscore the need for more advanced techniques to be explored in routine practice. Furthermore, T staging with pathology has its own limitations including inadequate sampling and incorrect interpretation due to surgical artifact. mpMRI provides effective local staging by using T2WI, DWI, and DCE sequences that all distinctly characterize tumor from normal bladder mucosa. PET improves nodal staging by identifying nodes with higher metabolic activity concordant with cancer, even if they are not enlarged. Combining PET and MRI may provide the optimal framework for bladder cancer staging, as synthesizing the quantitative characteristics of local and nodal disease on both imaging modalities improves accuracy and resolution. However, larger prospective trials are necessary to validate the accuracy of PET/MRI in bladder cancer staging, and the widespread implementation of PET/MRI is limited by its high overhead cost. Nonetheless, clinicians may consider PET and MRI as complementary tools in select patients where conventional imaging is inconclusive or when precise staging has direct therapeutic implications.
Footnotes
Acknowledgements
None.
