Abstract

Introduction
Urolithiasis is an increasingly prevalent condition affecting an estimated 10% of the population, while recurrence rates are high and roughly one out of four patients will undergo an invasive form of treatment. 1 Stone burden represents one of the most significant predictive factors of stone-free rate (SFR) after invasive treatment for urolithiasis. The vast majority of published studies, along with existing nomograms for the prediction of clinical outcomes after stone disease treatment, rely on patient subcategorization according to single-plane stone metrics, namely maximal stone diameter. Consequently, clinical guidelines, such as those drafted by the European Association of Urology (EAU) 2 and the American Urological Association (AUA), 3 suggest the most proper treatment according to the maximal single-axis measurement of the target stone. However, there is an obvious pitfall when using single-plane measurements, since most stones are irregular in shape; thus, their volume may differ significantly even when the longest diameter is similar; De Coninck and Traxer demonstrated that a 20*20*20 mm stone volume (4.189 mm3) is 16 larger than a stone 20*5*5 mm volume (268 mm3), 4 even if the largest diameter is the same in both cases.
Multidetector CT scanners were initially launched more than 25 years ago and permitted radiologists to measure volumetric data. Since then, advances in image quality, protocols, and software, simplified the calculation of volume using multiplanar measurements. More recently, low-dose and ultra-low-dose non-contrast CT (NCCT) protocols led to the reduction of radiation exposure by 73% and 92%, respectively,5,6 without reducing sensitivity (90%–100%) and specificity (86%–100%) for stones > 3 mm and patients with a BMI less than 30 kg/m2. 7 However, we still face limitations in applying these volumetric measurements into evidence-based clinical practice, and this is the reason why only the most recently published French guidelines on urolithiasis use stone volume to guide clinical decision-making. 8
In this editorial, we provide a step-by-step guide on how to use a freely available software for medical imaging using MacOS, the Horos Project (Horos™), to calculate stone volume for all types of stones.
Methods for measuring stone volume
The gold standard imaging modality used to calculate stone volume is NCCT, since it not only permits size measurements but also specifies detailed renal anatomy, skin-to-stone distance, and indicates stone composition. MRI and ultrasound scans are interchangeably used to avoid radiation exposure during patient follow-up or in sensitive patient populations (children and pregnant women), but there is no available evidence, to the best of our knowledge, where stone volume is calculated using these imaging modalities.
Taking into consideration stone shape, traditional approaches to estimate stone volume included formulas of specific geometric shapes, such as sphere, scalene spheroid/ellipsoid, oblate spheroid, or prolate spheroid, 9 with EAU guidelines suggesting the scalene formula (π/6 * l * w * d) 2 . However, this approach is not the most accurate, as shown by Finch et al., who utilized several formulas for various stone sizes and reported that for smaller stones (<9 mm) the prolate spheroid formula was the most accurate, while for stone diameters ranging between 9 mm and 15 mm, the oblate spheroid formula was the most accurate. 9 Interestingly, the scalene formula was the least accurate to use, especially when increasing stone size, potentially due to acquiring more asymmetrical shapes. 9
Due to the aforementioned limitations, technology filled the gap by providing automated or semi-automated calculation of stone volume. Specifically, several tools were created, either as stand-alone programs or integrated into existing medical image-viewing applications, to provide volumetric estimations of stones. Automated tools provide direct calculations, usually by recognizing regions of interest (ROI) of specific Hounsfield Units (HU) that separate stones from the surrounding tissues; the technology behind this automatic calculation is based on deep learning methods. 10 Semi-automated tools necessitate manual input by the user, usually in the form of selecting the ROI, with the most commonly reported threshold of HU ranging between 130 and 250 HU11,12 (Table 1). After segmentation, the applications create 3D models and calculate the incorporated voxels within them to calculate the stone volume. 13 Jain et al. 14 using ex vivo stones, compared measured volumes based on the ellipsoid formula with a semi-automated segmentation method, while using the gold standard method of water displacement as the standard comparator. Authors reported that a correlation coefficient r = 0.99 was calculated for the semi-automatic method, while r = 0.82 for the formula approach, indicating a superiority for the former; authors reported that when the formula was used, an overestimated stone volume, especially for larger stones, was observed. 14 Neubauer et al. 15 compared four different semi-automated segmentation methods to estimate the accuracy of stone volume calculation and reported very high correlation (r > 0.98) between the tested approaches.
Available software tools for automated and semi-automated measurement of stone volume.

Time required is an estimation according to reports/publications and uploaded online videos, and may vary according to stone location and size.
NA, not available.
Horos Project (Horos™) constitutes a freely available, open-source medical image viewer (https://horosproject.org/about/) for MacOS software, which is not available for Windows users. Using this free software, we describe an easy-to-use, semi-automated method to calculate stone volume. In the provided videos, the calculation of a renal/ureteral stone, a ureteral stone, and a staghorn stone is demonstrated. Initially, after uploading the Digital Imaging and Communications in Medicine (DICOM) files on the software, we select the NCCT images and estimate the whole length of the desired stone to be measured. Subsequently, we select the “closed polygon” type of metric and start from the bottom or top part of the stone and move toward the opposite end slice by slice (or in case of thin cuts, we can skip some in-between slices). The circumference of each depicted stone area must be drawn, and to proceed further, the final circle point must meet the initial one to close the polygon. After finishing the total number of slices, incorporating the whole length of the target stone, we select ROI from the top bar menu and then ROI Volume—Generate Missing Volumes. Next, we rename the created measurement, giving the desired file name and, finally, from the ROI choice we select ROI value and then Compute Volume to generate the total volume of the stone (Figures 1–3). In case of multiple stones, the relevant volume for each one of them is calculated, and then all volumes are added.
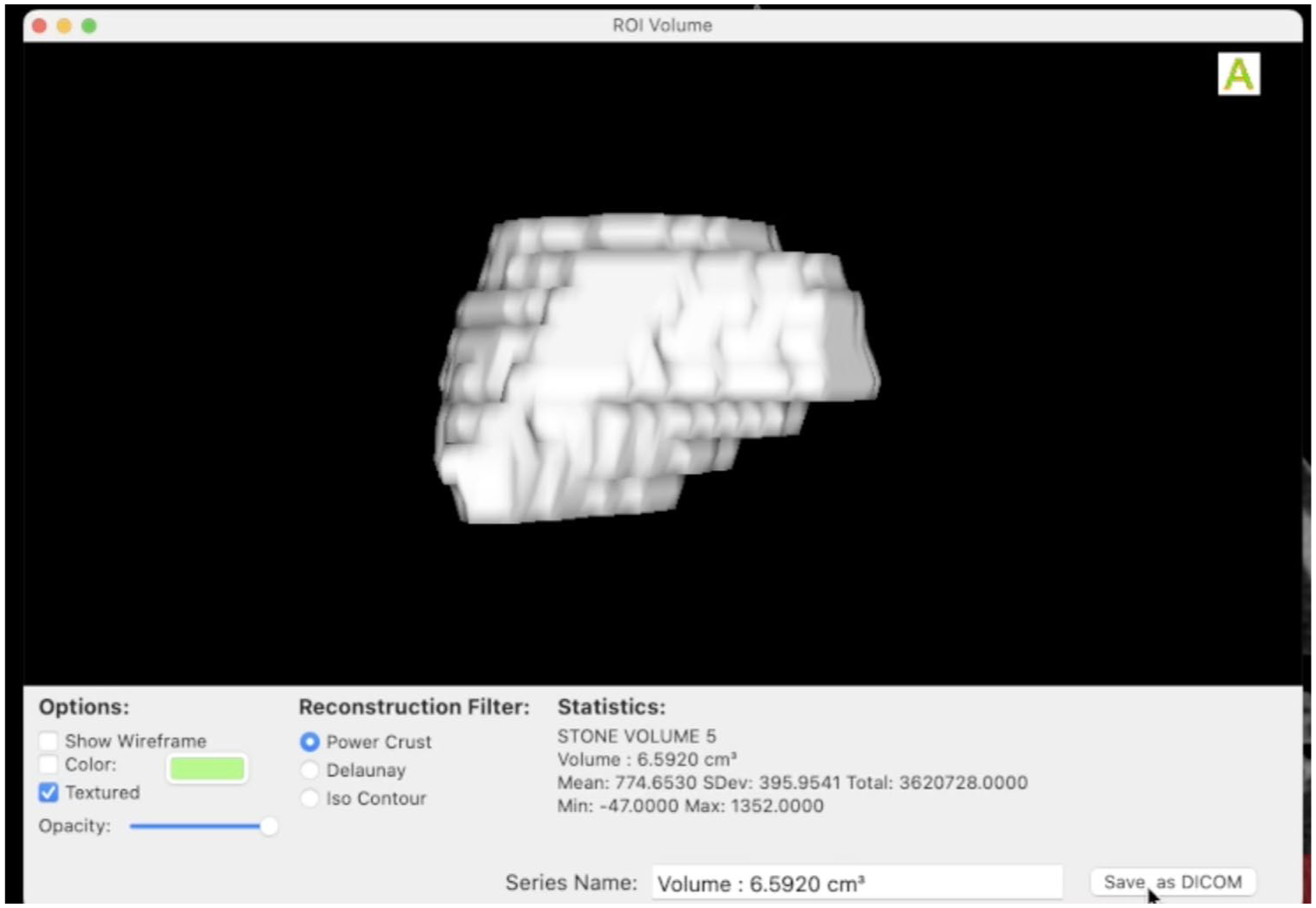
Measurement of renal stone volume.
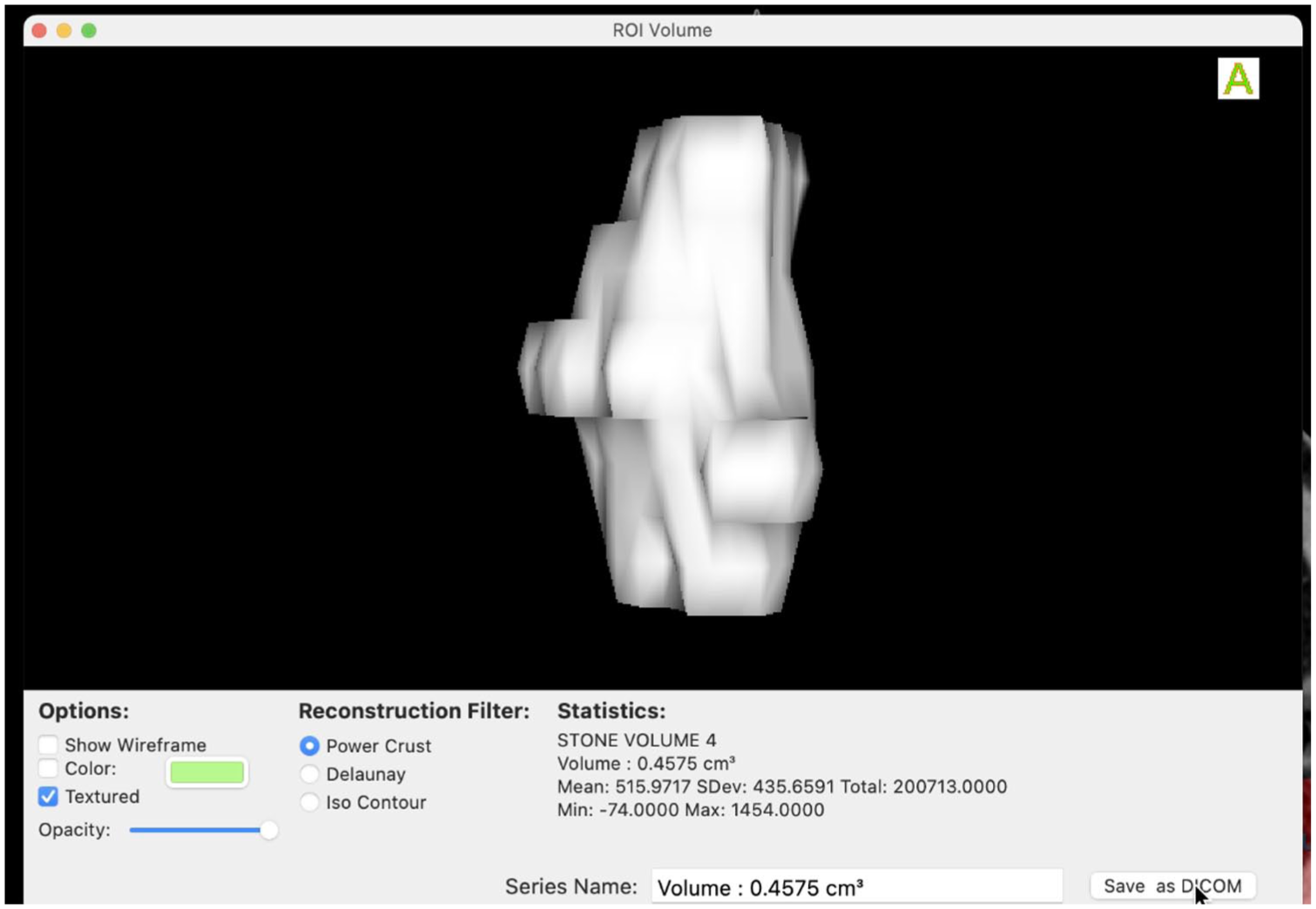
Measurement of ureteral stone volume.

Measurement of staghorn stone volume.
Supporting videos
(1) Kidney-ureteral stone
(2) Ureteral stone
(3) Staghorn stone
Measuring stone volume is a standard part of management in patients with kidney stones, so no ethical approval was needed. Written informed consent was obtained from all the patients for the procedure and for their anonymized data to be used for teaching and publication.
Clinical importance of measuring stone volume
The significance of stone burden in predicting clinical outcomes (stone-free rates, complications, operative time, and length of stay) is well debated and supported in the literature. The majority of clinical trials are conducted using single-plane metrics to measure stone burden, but there are also studies comparing volume to the conventional metric of maximal stone diameter. Chiou et al. 16 focused their interest on conservative management of ureteral stones and detected that the receiver operator curve (ROC) for predicting spontaneous stone expulsion using volume was superior compared to the largest stone diameter (0.93 vs 0.86, p < 0.001 at 4 weeks, 0.92 vs 0.85, p < 0.003 at 6 weeks). Stone volume was proved to be a significant predictor also for the occurrence of symptomatic events during a long-term (4.7 years) follow-up in patients with kidney stones (hazard ratio = 1.35 per quartile, p = 0.01). 17 Importantly, Geraghty et al. 18 recently conducted a systematic review and meta-analysis of existing literature to evaluate the significance of stone volume in predicting clinical outcomes (stone-free rates, complications, operative time) after extracorporeal shockwave lithotripsy (ESWL), percutaneous nephrolithotripsy (PCNL), or ureterorenoscopy (URS). The pooled meta-analysis of study arms revealed that prognostic accuracy regarding SFR was significantly higher for stone volume compared to stone diameter (AUC = 0.67 vs 0.71, p < 0.001) overall, for ESWL (AUC = 0.66 vs 0.69, p = 0.05) and for URS (AUC = 0.75 vs 0.79, p < 0.001). 18 For PCNL, no significant differences were detected, while existing data were very limited to conclude on complication rate and operative time; authors concluded that these findings provide a clue that stone volume may be superior in terms of predicting clinical outcomes, but the scarcity and heterogeneity of data necessitate further trials. 18
Conclusion
Stone volume impacts clinical outcomes after active treatment for urolithiasis significantly. The apparent advances in imaging technology (especially NCCT) permit the calculation of stone volume using several automated or semi-automated tools and software. In this editorial, we describe step-by-step by providing supporting videos, the method for calculating stone volume in MacOS using Horos Project software (Horos™).
