Abstract
Continent urinary diversion is the preferred method of bladder substitution for many patients who undergo cystectomy for benign conditions. Additional interventions are often necessitated after a continent diversion to achieve continence. One such procedure is the injection of botulinum toxin into the neobladder. Previous injections into the pouch bladder have been performed percutaneously. Here we present a less invasive endoscopic injection technique where flexible equipment has been used successfully to inject botulinum toxin into the pouch bladder via the pouch stoma. We demonstrate the feasibility of the injection and its efficacy in reducing pouch contractions and the patient’s symptoms. A 36-year-old female presented with persistent urinary incontinence symptoms 1 year after Indiana pouch diversion. Despite previous conservative measures and ileocecal valve reinforcement surgery, incontinence persisted. The endoscopic injection, facilitated through the pouch stoma, proved effective in decreasing muscle contractions and alleviating spasm-like sensations without observed side effects. This case shows the feasibility, safety and efficacy of botulinum toxin injection into Indiana pouch endoscopically.
Keywords
Introduction
Bladder pain syndrome/interstitial cystitis (BPS/IC) is a debilitating pain syndrome of unknown aetiology that predominantly affects young women. 1 When all conservative measures fail to control pain, cystectomy may be indicated including a urethrectomy for patients with urethrally centralised pain after which a continent catheterisable stoma may be formed. Amongst other means of constructing a continent catheterisable pouch, the Indiana pouch is a well-defined surgical technique where the ileoceacal segment is reconstructed to form a reservoir and a conduit.2,3 Long-term results of Indiana pouches are satisfactory with good continence rates.4,5 Urinary incontinence after Indiana pouch construction could be due to incompetence of the reinforced ileoceacal valve, high pressures in the reservoir or contracted bladder.
Injection of botulinum toxin into the pouch could help in reducing pouch contractions however there is limited evidence for the efficacy and safety of such a treatment. There have been two case reports in the literature where incontinence and/or abdominal pain due to pouch contractions was successfully treated with botulinum toxin injections. In both of these cases, a percutaneous approach was used probably due to a lack of flexible endoscopic instruments. Therefore, the efficacy and safety of endoscopic injection of botulinum toxin into the Indiana pouch have not been reported so far.
In this report, we present a case of endoscopic injection of botulinum toxin into the pouch in a patient with pouch bladder overactivity and urinary incontinence.
Case presentation
A 36-year-old female patient with a diagnosis of persistent BPS/IC underwent a simple cystectomy, urethrectomy, and an Indiana pouch urinary diversion due to persistent perineal, urethral, and suprapubic pain and cramping sensations. In the Indiana pouch surgery, a 15–18 cm segment from the proximal ileocecal valve and an approximately 20 cm segment from the right descending colon and proximal transverse colon were isolated. The cecum and right colon were split at the antimesenteric border and closed transversely to form the pouch. Both ureters were reimplanted through incisions along the tenia. The ileal segment was plicated and the stoma was formed at the umbilicus. The patient started self-catheterisation via the stoma 3 weeks after surgery. The surgical outcome was very satisfactory with a resolution of the pain; however, 6 months after the operation, the patient complained of spasms and incontinence. The average volume expelled during each catheterisation throughout the day ranged from 200 to 400 cc. Leakage volumes were more than 500 ml daily necessitating the use of a urostomy bag. Kidney function was within normal limits, as confirmed by blood tests and a CT scan.
She was put on anticholinergic and antispasmodic medications for 3 months without any major benefit. Then she underwent an open ileoceacal valve reinforcement surgery. Although she had some improvement in her pouch capacity, urinary incontinence persisted. A video urodynamic study was performed showing phasic pouch spasms starting from a pouch volume of 150 ml and continuing throughout the filling cystometry. Each pouch contraction was felt by the patient as a fullness and pressure sensation at the right lower abdomen. At a pouch volume of 250 ml, the sensation of fullness/spasm and a feeling of nausea and retching was so severe that the infusion was stopped. The filling pouchometry was continued after a period of rest until a volume of 550 ml where the patient reported an unbearable sensation of pouch fullness/pressure (Figure 1). In the end, she also had a projectile leakage of urine from the pouch.

Video urodynamic study before and after botulinum toxin injection into the Indiana pouch. Phasic pouch contractions (red arrows) can be observed early in the filling pouchometry before injection. Also, pressure/fullness sensations (blue dotted lines) are observed starting from a pouch volume of 150 ml accompanying the pouch contractions. Both the pouch contractions and pressure/fullness sensations disappear after injection until a capacity of 450 ml.
To control the pouch contractions and the spasm/pressure sensations, an injection of 200 units of botulinum toxin into the pouch was performed under general anaesthesia (Figure 2). The botulinum toxin was diluted in 10 cc of saline solution and a 17 Fr flexible cystoscope and a 27 Gauge endoscopic injection needle was used for injection, targeting 20 different points within the pouch. Postoperatively, during the 1-year follow-up, she did not report any side effects in the early or late postoperative period. She continued to have repeat botox injections into her pouch bladder, a total of three injections were performed in a year, the last being 300 IU.
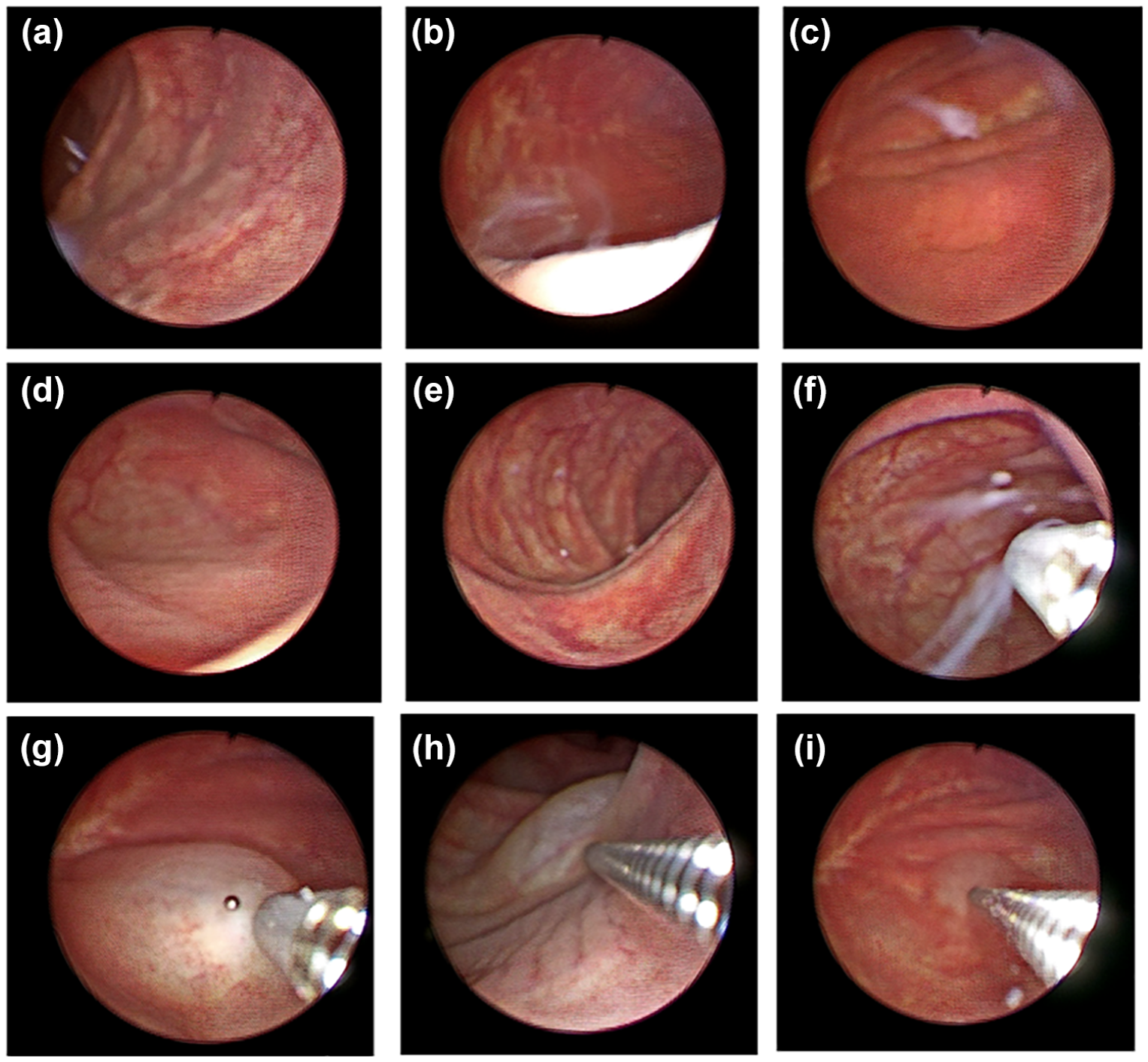
Botulinum toxin injection into the Indiana pouch. Endoscopic examination of the pouch has shown normal bowel mucosa without any signs of chronic pouchitis and with minimal mucous inside the pouch (a–e). Botulinum neurotoxin was effectively injected submucosally into the pouch using a flexible cystoscope and flexible endoscopic injection needle (f–i). The use of flexible instruments enables access to all corners of the pouch.
After the injection, the patient’s cramping sensation improved. Her functional bladder capacity, as reported by a frequency volume chart, was around 500 ml postoperatively, with a variable amount of urine leakage during the day (the maximum amount of leakage being 550 ml during the day). Nevertheless, she stopped wearing a stoma bag during the day. A follow-up video on urodynamics showed that the previously observed activity had disappeared, the capacity was around 600 cc and she had no leakage during cystometry. There was no impairment in kidney function, and there was no significant change or deterioration in the structure of the kidneys and the upper urinary system (Figure 3).

Both kidneys exhibit normal contours, size, localisation, parenchymal thickness, echogenicity and echo pattern. Sinus echoes are normal. No dilation is observed in the renal collecting systems.
Discussion
This report shows, for the first time, the feasibility of endoscopic injection of botulinum toxin (BoNT) into the Indiana pouch to prevent pouch contractions in a patient complaining of urinary incontinence and pouch spasms. There have been two previous case reports describing intrapouch injections of BoNT through a subcutaneous 6 and a percutaneous 7 route. The availability of flexible endoscopic instruments (both the endoscope and the needle) has made it possible to engage the endoscope through the cutaneous stoma into the pouch and reach out to all the margins of the pouch effectively. This technical availability has possibly had an influence on the perceived efficacy and tolerability of the procedure.
BoNT exerts its effects in the Lower Urinary Tract (LUT) by inhibiting vesicular neurotransmitter release at the neuromuscular and neuroglandular junctions. This action of BoNT-A involves inhibition of neurotransmitter release from not only efferent but also possibly from afferent nerve terminals and the urothelium. 8 The effects of BoNT on muscle contraction in the urinary bladder involve inhibition of the release of acetylcholine from both preganglionic and postganglionic nerves. BoNT is also thought to inhibit bladder sensations by decreasing purinergic (P2X3) and transient receptor potential vanilloid subfamily-1 (TRPV1) immunoreactivity in sensory nerve fibres.9,10 The use of BoNT in the treatment of BPS/IC and Chronic prostatitis is making use of its effects on modulating sensory function.
Although the same mechanism of action of BoNT on colonic muscularis and submucosa can be proposed as the basis of the observed effect in the presented case, the organisation of the epithelium and submucosal and mucosal layers are significantly different in the bladder and colon. Therefore, the safety and efficacy of injection of BoNT in the colon could be different. In the presented case, we have both observed a decrease in muscle contractions and spasm-like sensations after the injection of BoNT. Nevertheless, despite having no leakages in the video urodynamic study, she reported a persistent urine leakage from the stoma site. This could be due to several reasons including an overflow incontinence when she missed self-catheterisation intervals, a degree of sphincteric insufficiency during daily activities, persisting pouch contractions that have not been observed during the postoperative urodynamic test and our inability to demonstrate urine leakage during the urodynamic test. It is recognised that urodynamic studies may fail to diagnose existing incontinence in up to 50% of patients with stress urinary incontinence.11,12 The definitive diagnosis would require the use of advanced urodynamic techniques that can assess intraluminal pressures such as urethral pressure profile and ambulatory urodynamics. However, these are not standardised for use in this context and the expertise may not be available in all institutions.
With regards to the safety of BoNT injection into the colonic mucosa, one previous study reported side effects such as general muscle weakness and malaise a few weeks after injection; however, in our case, we did not observe any side effects at the early and late postoperative periods. 7
