Abstract
Urinary diversions are performed for a wide variety of indications, including bladder removal for cancer treatment, post-cancer treatment complications, trauma, or bladder pain. The robotic approach has been increasingly used in performing urinary diversions since the introduction of the surgical robot. A PubMed keyword search was performed on September 14, 2023 with the terms: robotic and urinary diversion. A narrative review of the literature was then conducted, with a focus on outcomes and complications following urinary diversion. Studies demonstrated that the robotic approach to cystectomy with urinary diversion was safe and productive, whether the diversion was performed intracorporeally or extracorporeally, and that outcomes are comparable to the open approach. Despite reports of successes and technique improvements, the complication rate following robotic cystectomy with urinary diversion is over 50%. Common complications associated with urinary diversion include bowel complications, ureteroenteric anastomotic strictures, urine leak, urinary tract infections, internal hernias, and parastomal hernias. Many strategies have been developed to improve the complication rate following robotic urinary diversion, including specialized training programs and enhanced recovery after surgery protocols. In conclusion, with the increasing adoption of the robotic approach for urinary diversions, it is important to continue to develop strategies to mitigate surgical risk. Future research should focus on further refinement of training and surgical approaches to prevent and treat complications following robotic urinary diversions.
Plain language summary
Urinary diversion, or the use of the intestine to form a reservoir for urine, can be performed during cancer treatment or due to bladder trauma, pain, or neurologic conditions causing impaired bladder function. Urinary diversion strategies vary, including open surgical approaches or minimally invasive approaches using the surgical robot. Removing the bladder and diverting the urine has a high complication rate and the urinary diversion portion of the case can cause complications involving the intestine or the urinary tract. Specialized training in minimally invasive robotic urinary diversions and specific surgical recovery pathways may improve the complication rates for this newer strategy for creating urinary diversions.
Keywords
Introduction
Urinary diversions are frequently performed following cystectomy for bladder cancer or with or without cystectomy for benign conditions such as bladder pain syndrome or refractory radiation cystitis. Urinary diversions with and without cystectomy have been performed via an open approach for decades; however, with the introduction and increasing acceptance of the surgical robot for various surgeries, there has been interest in performing cystectomy and urinary diversion via the robotic approach.1,2 A study published in 2022 using data from the United States-based National Cancer Database demonstrated increasing adoption of robotic-assisted laparoscopic cystectomy with 22.9% of cystectomies performed robotically in 2010 compared with 40.6% in 2015. 1 Similarly, another group noted an approximately 30% rate of robotic cystectomy among their patient population with increasing utilization beginning in 2008. 2
The robotic approach to cystectomy with urinary diversion has emerged as a safe and effective alternative to open cystectomy. Compared to the open approach, robotic cystectomy with urinary diversion is not associated with an increase in complications, readmission, or a difference in quality of life. 3 The robot also has significant advantages, such as the minimally invasive approach, access to the pelvis, excellent visualization, the ability to assess perfusion with indocyanine green (ICG), and surgeon ergonomics. 4 Given these advantages, coupled with increasing surgeon training on the robotic platform, it is likely that robotic urinary diversion rates will continue to increase. Additionally, the techniques available for robotic-assisted urinary diversion, documentation of complications and their mitigating strategies, and examination of outcomes continue to be published as the field matures. These novel publications warrant further study in the setting of a narrative review to aggregate outcomes and improve strategies for urinary diversion in the robotic surgery era. There are substantial gaps in our understanding of developing and managing complications after urinary diversion using the surgical robot. This narrative review discusses relevant literature regarding the performance of urinary diversions with the surgical robot, including diversion types, outcomes, complications, and strategies for improving outcomes.
Methods
A thorough search of PubMed was performed on September 14, 2023 with the terms “robotic” and “urinary diversion.” These papers were then reviewed for relevance by KMD. BJF evaluated the completeness of the literature review. Inclusion criteria were: referencing urinary diversion including operative strategies, complications, or outcomes specific to urinary diversions. We included any type of urinary diversion, including incontinent bowel conduits, orthotopic neobladder, continent cutaneous diversions, cutaneous ureterostomy, or other diversions for completeness. Papers specifically discussing oncologic outcomes or those with a focus on the cystectomy portion of a cystectomy with urinary diversion were excluded.
Literature review findings
Diversion types
Urinary diversions include incontinent diversions such as ileal conduit, orthotopic urinary diversions like the orthotopic ileal neobladder, and continent cutaneous diversions such as the Indiana pouch. 5
Ileal conduit
The ileal conduit is the most commonly performed bowel diversion and can be readily performed robotically. 6 To accomplish this diversion, a section of the intestine is separated from the remaining intestine and brought through the abdominal wall as an incontinent stoma. Compared to other more complex diversions, discussed later, the ileal conduit has the fewest steps. It requires the least amount of bowel, making it the preferred approach in less healthy patients and individuals with prior bowel resections. The ileum is an excellent bowel segment because it is well-tolerated and results in fewer metabolic abnormalities than other segments. 7 The reduced metabolic abnormalities are partly due to the incontinent stoma, which prevents the reabsorption of electrolytes from urine sitting in a reservoir made of the bowel. Because of this, the ileal conduit is suitable for individuals with poor kidney function and can be performed in the setting of a renal transplant. 8
Orthotopic neobladder and continent cutaneous urinary diversions
An orthotopic neobladder allows patients to continue to void via the urethra after urinary diversion with urine contained in a reservoir created from the bowel and anastomosed to the urethra. There are a variety of approaches to creating a neobladder, which are beyond the scope of this review.9–11 Robotic approaches to the orthotopic ileal neobladder have been extensively described.9,12 Advantages of the orthotopic neobladder include maintenance of patients’ ability to urinate via the urethra and lack of abdominal stoma. Neobladder formation is more complex to perform, resulting in longer operative times, and some studies have demonstrated a higher complication rate than the ileal conduit.13,14 However, other studies have noted no difference in complication rates between robotic ileal conduit and neobladder. 10 The variation in these findings may represent differences in patient population, surgeon experience, or selection strategies for each diversion, but warrant consideration when offering a neobladder to patients. While the orthotopic neobladder is a continent diversion, patients must be aware that they will be insensate of their new bladder and that they may experience urinary incontinence and urinary retention. 15 Some female and few male patients will need to self-catheterize to manage urinary retention, so they must be appropriately counseled ahead of undergoing a continent urinary diversion. 15
A variety of continent cutaneous diversions have been described. The most common of these is the Indiana Pouch, which can be performed robotically. 16 This diversion uses a segment of the right colon and the ileum to form a reservoir and a segment of the ileum as a catheterizable channel, through which a patient can pass a small catheter to drain urine. The advantages of this approach are that it allows patients to maintain continence, and that it results in a smaller stoma than the ileal conduit. However, this diversion strategy results in both an abdominal stoma and the need to catheterize, about which patients must be appropriately counseled. One study comparing the robotic Indiana Pouch to the neobladder and the ileal conduit demonstrated similar readmission rates across the diversion types. 17 However, compared with the ileal conduit, continent cutaneous diversions are more complex to perform and lead to a higher complication rate. 18 Additionally, many continent cutaneous diversions require the use of the colon, which results in higher pressures transmitted to the upper tract, resulting in renal damage.19,20 Despite these disadvantages, continent cutaneous diversions can be safely performed in appropriately selected and counseled patients.
Adoption of robotic approaches to continent diversions, including both orthotopic and cutaneous diversions, has been slow, despite increasing utilization of the robot to perform cystectomy and urinary diversion overall. 1 One study noted that subjects who underwent a robotic cystectomy at their institution were more likely to receive an ileal conduit while those who underwent open cystectomy were more likely to receive a neobladder, a difference that was statistically significant. 2 Diversion time is longer for robotic ileal neobladder than robotic ileal conduit, which may serve as a barrier to adoption. 12
As discussed throughout this section, there is a need for more data regarding the complication rates and other outcomes for different diversion types. Surgeon experience, patient comorbidities, and other confounding factors likely impact complication rates. However, even with evident data regarding complication rates, selection of bowel diversion type would involve shared decision making between a patient and their surgeon, taking into account patient comorbidities, patient preferences, and surgeon experience. Some patients are willing to accept a higher risk of complications to achieve continence, while others prioritize the shorter operative time of an incontinent diversion. Furthermore, patients with comorbidities, such as short bowel, may be steered toward an incontinent diversion as less bowel is required for this approach. Thus, studies examining patient and surgeon decision making surrounding the selection of urinary diversion strategy are warranted and might aid in improving outcomes for patients requiring these surgeries.
Extracorporeal versus intracorporeal approach
Following the introduction of robotic cystectomy, urinary diversion was performed extracorporeally, wherein a port site incision is widened, and the urinary diversion is performed via an open approach. 21 This diversion technique has the advantage of being similar to surgeons’ prior experience with open urinary diversions, allowing them to focus on the technical aspects of the cystectomy portion of the case and thus reducing cognitive load. The RAZOR study sought to compare open cystectomy to robotic cystectomy with extracorporeal diversion with a primary endpoint of progression-free survival, which was not significantly different between the two groups. 22 In this study, operative times in the robotic cohort were longer (median 428 vs 361 min) than the open cohort, but complications related to the diversion, such as ureteroenteric anastomotic leak, ureteral strictures, or gastrointestinal injuries, were rare and occurred at similar rates in each group. 22 One early study from 2014 compared open cystectomy to robotic cystectomy with extracorporeal urinary diversion, noting similar health-related quality of life outcomes. 23 This research group reported improved sexual function, as measured by the Bladder Cancer Index Questionnaire, in the open approach compared to the robotic approaches evaluated in this study. However, with further surgeon experience, this outcome has likely improved. 23 Furthermore, this study did not separately evaluate male and female subjects, information that could contribute to the improvement of technique and patient counseling.
As bowel stapling devices, surgical robots, and surgeon experience have progressed, more surgeons are performing robotic-assisted urinary diversions intracorporeally, including ileal conduit, continent cutaneous diversion, and orthotopic neobladders.6,16,24 In this approach, the entirety of the bowel diversion, including isolation of the bowel segment, and anastomosis of the ureters to the urinary diversion, is performed minimally invasively using the robot (Figure 1). Multiple studies have demonstrated increasing adoption of the intracorporeal technique. Among an international consortium of surgeons, 0% of cases were performed intracorporeally in the early 2000s compared with 95% of cases in 2018 being performed intracorporeally and almost 100% in 2020.25,26

Intracorporeal ileal conduit formation with the multiport robot. (a) The robotic arms are used to stabilize the bowel so an assistant can use a laparoscopic stapler to separate the ileal conduit. (b) The bowel reanastomosis is formed. (c) The ileal conduit is positioned. (d) The ureteral anastomosis is performed.
Numerous studies have compared intracorporeal and extracorporeal diversions with varied inclusion criteria, outcomes measurements, and findings. One large consortium retrospective study with propensity score matching comparing cystectomy with intracorporeal versus extracorporeal diversion demonstrated a slightly higher rate of overall complications and readmissions for the intracorporeal cohort but no difference in significant complications. 25 However, multiple meta-analyses have demonstrated reduced complication rates for subjects undergoing intracorporeal diversions compared to extracorporeal diversions at 30 and 90 days.3,27,28 Additionally, a propensity score matched study of robotic radical cystectomy with intracorporeal ileal conduit compared with open cystectomy demonstrated no difference in complications. 29 In cystectomies performed for the management of neurogenic bladder, no differences in complications were noted between the intracorporeal and extracorporeal approaches. 30 Thus, there are conflicting reports regarding complication rates in intracorporeal versus extracorporeal urinary diversion.
Another study of ileal neobladders demonstrated fewer reoperations at 30 days but more readmissions for subjects undergoing intracorporeal diversions than extracorporeal diversions. 26 For hospital length of stay, some studies demonstrated shorter lengths of stay for patients undergoing intracorporeal diversions, while others noted shorter lengths of stay in their extracorporeal population, and some showed no difference.26,31–34 Differences in findings between these studies may represent differences in local practice, diversion types, or other confounding variables.
Several studies have examined specific complications in the setting of intracorporeal urinary diversion. Multiple studies have demonstrated advantages for intracorporeal diversions compared to the extracorporeal approach, such as decreased estimated blood loss (EBL), lower transfusion rates, and shorter hospital lengths of stay without a difference in complications.27–29,34–36 Several studies noted a reduced rate of bowel complications, decreased venous thromboembolism, and faster return of bowel function in some studies with the intracorporeal compared with the extraperitoneal approach.31–33 Compared with open or extracorporeal diversions, intracorporeal diversion did not appear to impact health-related quality of life substantially. 23
One multivariable analysis demonstrated no difference in cancer specific-survival, recurrence-free survival, or overall survival between intracorporeal and extracorporeal urinary diversion. 37 However, in this study, intracorporeal diversions were associated with lower rates of overall recurrence and pelvic recurrence. 37 It is unclear what factors caused these differences in recurrence rate, but this parameter may improve with longer surgeon experience.
In the majority of studies, the extracorporeal approach is associated with shorter operative times.32,33,38 However, one study of subjects undergoing ileal conduit demonstrated shorter median operative times for intracorporeal compared with extracorporeal diversions, highlighting variability in operative times reported in these approaches. 28 Operative times may improve with more extended surgeon experience as well as further improvement and optimization of equipment in the performance of this step.
To examine which patients most benefit from intracorporeal diversions, one study stratified subjects based on comorbidities and found fewer complications with intracorporeal diversion in the high comorbidity population. In contrast, patients with fewer comorbidities exhibited no difference in complication rate between the approaches. 14 Thus, there may be some patients in whom an intracorporeal approach would be more advantageous and defining these patient populations warrants further investigation.
The outcomes of studies vary when comparing extracorporeal and intracorporeal bowel diversions. There are several potential reasons for these discrepancies. One possible explanation is that outcomes are better at whichever technique is more familiar to the performing surgeon. The extracorporeal approach is similar to open bowel diversion and, thus, surgeons who have extensive open experience likely have improved outcomes with this approach. In contrast, surgeons with more robotic experience may feel more comfortable performing the diversion intracorporeally. Additionally, robotic instruments have improved, which may have impacted intracorporeal outcomes for older manuscripts. Furthermore, intracorporeal diversions require less dissection of the abdominal fascia, which may be less morbid when performed by an experienced surgeon. More research is needed to reconcile these data with the modern robotic platform and demonstrate which patients most benefit from an extracorporeal versus an intracorporeal diversion.
Robotic platforms used for urinary diversion
Da Vinci Xi
The majority of studies evaluated discussed the performance of cystectomy with urinary diversion using the Da Vinci Xi robot (Intuitive Surgical, Inc., Sunnyvale, CA, USA). According to Intuitive Surgical, Inc., the manufacturer of the Da Vinci platform, there were over 6500 Da Vinci robots in 67 countries in 2021, most of which were Xi robots. 39 Given its broad availability and familiarity to surgeons, it is unsurprising that the Da Vinci Xi is broadly used in studies examining the performance of robotic-assisted urinary diversion.
Da Vinci single-port robot
The Da Vinci single-port robotic platform (Intuitive Surgical, Inc., Sunnyvale, CA, USA) can be used to perform both intracorporeal and extracorporeal urinary diversions with good success.40–44 The single-port platform is especially helpful in reducing the number of entry sites, which is useful when approaching the hostile abdomen and may reduce the rate of bowel injury. 45 While a majority of studies use a transabdominal approach to robotic cystectomy and urinary diversion, one group demonstrated the successful performance of a transperineal intracorporeal ileal conduit with the single-port robot. 46 Following a small case series demonstrating the success of the single port as a platform for performing transabdominal cystectomy and urinary diversion, a larger retrospective series was published, including cystectomy with ileal conduit or orthotopic neobladder in 41 subjects with a 17% rate of open conversion and few complications. 44 Comparing multiport robotic cystectomy to single-port cystectomy with either ileal conduit or neobladder, subjects receiving cystectomy and urinary diversion by the single-port approach were noted to have decreased narcotic use and faster return of bowel function with similar operative times and complication rates. 47
Other robotic platforms
One study examined the use of the Hugo RAS robot (Medtronic, Minneapolis, MN, USA) to perform two robotic-assisted radical cystectomies with intracorporeal ileal conduit creation, demonstrating its feasibility. 48 One abstract discussing the successful performance of radical cystectomy with ileal conduit or neobladder using the Mantra (SS Innovations International, Gurugram, Haryana, India), was presented at the American Urological Association Meeting in 2023, but thus far, these data has not been published elsewhere. 49 As other robotic platforms continue to develop, there may be future reports of robotic-assisted urinary diversions using these novel platforms.
Complications of robotic urinary diversion
Robotic cystectomy with urinary diversion is associated with an acceptable complication rate (Table 1). For experienced surgeons, the complication rates are relatively high and do not vary drastically between robotic and open surgeons. In a multi-institutional cohort study, 53% of 959 subjects undergoing robotic-assisted radical cystectomy with various urinary diversions experienced complications; however, the majority of these complications were low-grade. 50 Radical cystectomy has also been associated with a 3% reoperation rate and a 14% hospital readmission rate. 51 An early study, published in 2013, of 209 patients who underwent robotic cystectomy demonstrated that 77.5% experienced a complication within 30 days, including a 32% rate of significant complications. 18 A later study, published in 2023, comparing ileal conduit to ileal neobladder in the setting of robotic-assisted cystectomy for bladder malignancy demonstrated an approximately 58% complication rate for the overall cohort, again with the majority of the complications being low-grade. 5 A meta-analysis of radical cystectomy with various urinary diversions, including robotic-assisted, pure laparoscopic, and open approaches, noted a 50% rate of Clavien I–III grade complications and a 5% rate of Clavien IV–V grade complications. 51 In this study, differences in complications between the approaches were minimal, and the robotic approach was associated with a lower transfusion requirement but higher rates of infection, hospital readmission, urine leak, and fistula. 51 The differences between reported complication rates likely represent differences in experience gained over time by surgeons past their learning curves as well as differences in diversion and approach in each of these studies; however, complication rates remain a significant concern for patients undergoing urinary diversions. A comprehensive list of complications and their mitigating strategies are included in Table 2.
Complication rates reported in the literature for various approaches of urinary diversions.
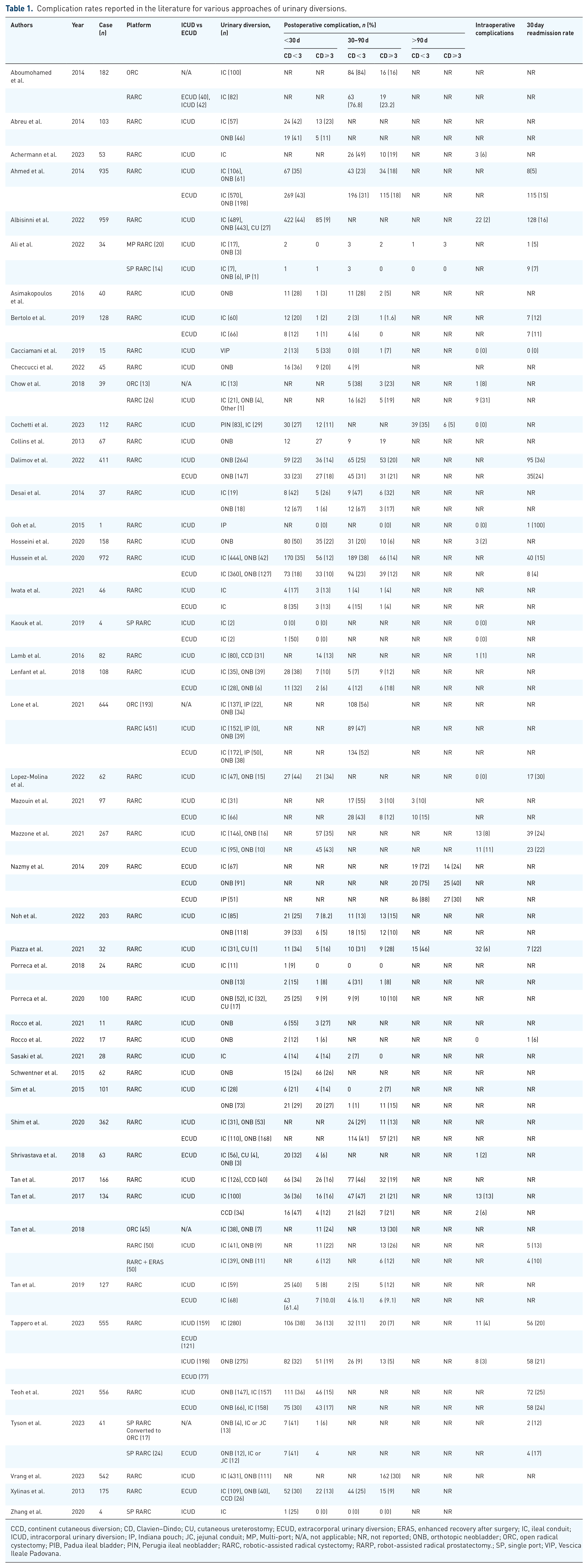
CCD, continent cutaneous diversion; CD, Clavien–Dindo; CU, cutaneous ureterostomy; ECUD, extracorporal urinary diversion; ERAS, enhanced recovery after surgery; IC, ileal conduit; ICUD, intracorporal urinary diversion; IP, Indiana pouch; JC, jejunal conduit; MP, Multi-port; N/A, not applicable; NR, not reported; ONB, orthotopic neobladder; ORC, open radical cystectomy; PIB, Padua ileal bladder; PIN, Perugia ileal neobladder; RARC, robotic-assisted radical cystectomy; RARP, robot-assisted radical prostatectomy.; SP, single port; VIP, Vescica Ileale Padovana.
Complications of robotic-assisted cystectomy and urinary diversion with risk factors and mitigating strategies.
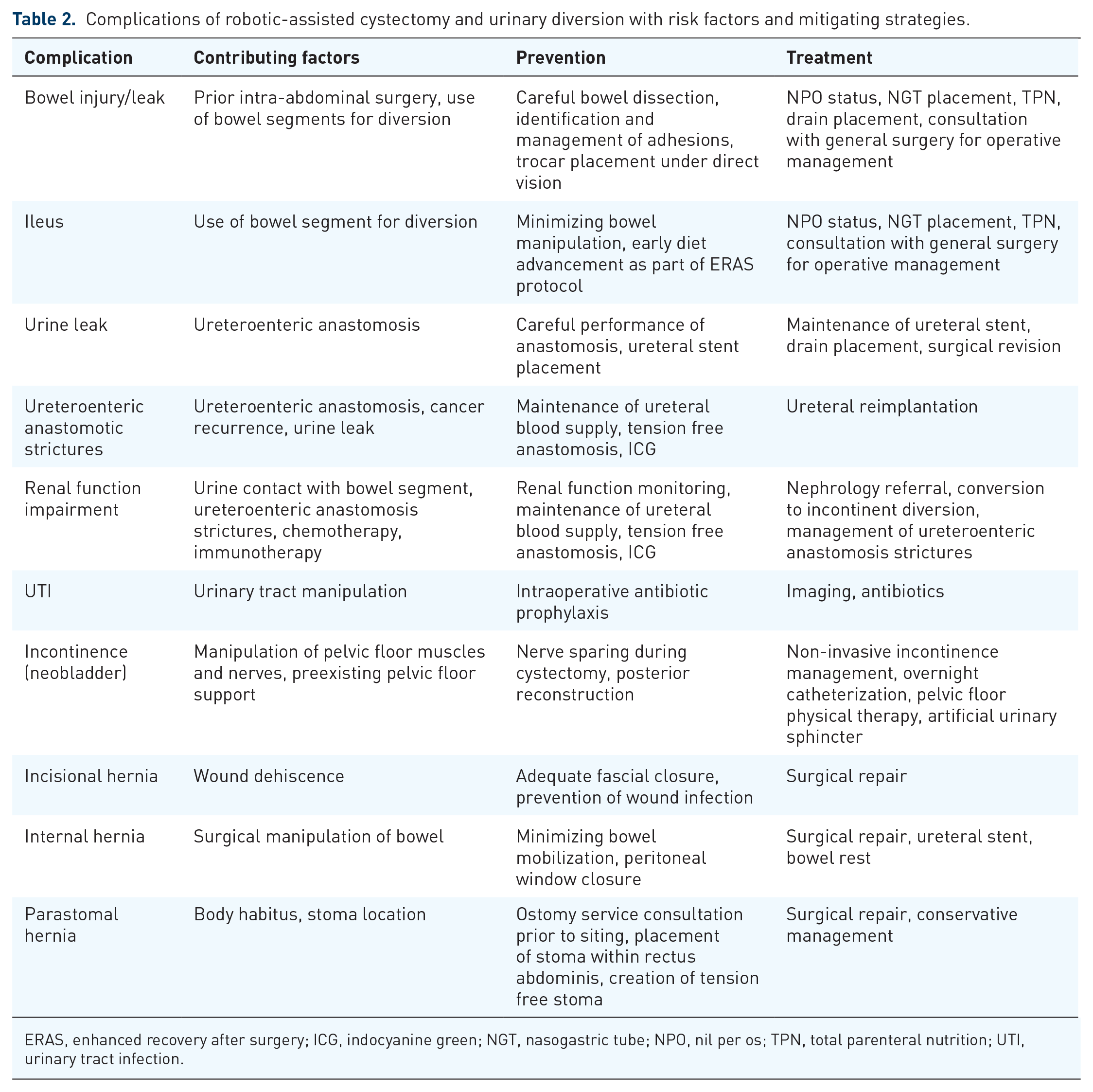
ERAS, enhanced recovery after surgery; ICG, indocyanine green; NGT, nasogastric tube; NPO, nil per os; TPN, total parenteral nutrition; UTI, urinary tract infection.
Risk factors for complications
Risk factors associated with complications have been extensively evaluated. One study noted an increased risk of complications for subjects based on the use of anticoagulation therapy, ureteroenteric anastomosis type (higher with Wallace), intracorporeal diversion, or history of tobacco consumption. 52 One case series of 32 subjects with a prior history of pelvic radiation undergoing robotic-assisted cystectomy with ileal conduit urinary diversion demonstrated eight instances of complications related to their diversion after 90 days, including new-onset chronic renal disease, parastomal hernia, and ureteroileal anastomotic strictures. 53 Due to the lack of a control group for this study, however, the impact of radiation on these complications cannot be determined and warrants further investigation. Subjects undergoing robotic cystectomy with intracorporeal diversion in one prospective cohort study were more likely to experience a complication if they required a blood transfusion either intraoperatively or postoperatively. 54 Preoperative anemia was associated with the need for a perioperative blood transfusion but not with complications, indicating that other factors likely impact both complications and the need for blood transfusion. 54 Cardiopulmonary exercise testing results were not predictive of adverse events or length of stay in a study of robotic cystectomy with intracorporeal urinary diversion. 55
One risk factor for complications, which has been noted in multiple studies, is the use of continent diversions over incontinent diversions.18,50,56,57 It is possible that the longer bowel segment required for an orthotopic neobladder drives the increased complication rate noted in these studies. However, a more recent meta-analysis from 2023 noted no increase in the risk of complications with an orthotopic neobladder compared with ileal conduit urinary diversion, which may indicate improvement of technique with time. 51 Thus, it is unclear whether the creation of a continent diversion truly impacts the complication rate and whether this finding is something that would improve with a surgeon’s learning curve or improved patient selection.
Bowel complications
Bowel injuries, ileus, and bowel leaks are a concern following urinary diversion due to the need for a bowel resection and reanastomosis required for use of the ileum or other bowel segments; however, reported rates of bowel complications vary based on definitions of these complications, study inclusion criteria, and diversion type. One multi-institutional series of robotic-assisted cystectomy with various urinary diversions noted a 15% rate of gastrointestinal complications such as anastomotic bowel leak, ileus, gastrointestinal bleeding, and perforation. 5 Another prospective case series noted a 2% rate of small bowel obstruction. 58 A separate study in 2022 demonstrated an approximately 25% rate of ileus after intracorporeal urinary diversion, while a recent meta-analysis from 2023 noted only a 14% ileus rate, which may reflect the introduction of enhanced recovery after surgery (ERAS) protocols, differing diversion techniques, or differences in reporting.51,59 Ileus has been associated with frailty and delayed time to mobilization, hinting that improving these modifiable risk factors may reduce the rate of this complication. 59 Management of bowel complications often involves continued nil per os (NPO) status or placement of a nasogastric tube. Prolonged NPO status can risk worsening nutritional status and thereby negatively impact recovery, so total parenteral nutrition may be required in this setting. Additionally, significant bowel complications can cause sepsis and acute abdomen, which may require drain placement or surgical management.
Urine leak
Urine leak is a significant concern because it can negatively impact renal function and healing of the ureteroenteric anastomosis. A multi-institutional retrospective series found the rate of urine leak after robot-assisted radical cystectomy was approximately 2%, with no difference between subjects undergoing an ileal conduit versus a neobladder. 5 Urine leaks can often be managed by maintaining ureteral stents that were placed during the index surgery longer than planned postoperatively. Refractory or symptomatic urine leaks may require percutaneous drain placement or, rarely, surgical revision of the affected ureteroenteric anastomosis.
Ureteroenteric anastomotic strictures
Ureteroenteric anastomotic strictures are a significant concern following urinary diversion. Strictures can lead to infection, renal function decline, and pain. 60 Stricture rate has been reported as 3%–21% following intracorporeal urinary diversion, including both ileal conduit and neobladder.5,61,62 In one study, the median time from urinary diversion to stricture development was 5 months. 60 Another study including robotic and open radical cystectomy with ileal conduit demonstrated an overall 13% ureteroenteric anastomotic stricture rate, with a 12% rate at 1 year and a 19% rate at 5 years, indicating that some strictures develop later in a patient’s postoperative course. 62
In subjects who underwent urinary diversion due to cancer, it is important to evaluate the strictured area for cancer recurrence. 60 However, once a stricture has been confirmed to be benign, ureteral reimplantation is a mainstay of management. The surgical robot is an excellent tool for addressing ureteral strictures and can be used to implement a variety of techniques to reimplant the ureters into the urinary diversion, including the Bricker, Nesbit, or Wallace reimplantation strategies. 63 A technique similar to a Boari flap is performed with a portion of the enteric segment used for the initial diversion if additional length is needed. 60 Management of ureteroenteric anastomotic stricture was associated with an approximately 15% complication rate in one study of 46 subjects with strictures, including an 8.7% rate of high-grade complications. 60 Thus, this complication is difficult to manage.
Several studies have attempted to determine the causes and correctable factors underlying ureteroenteric anastomotic strictures. One randomized clinical trial of open versus robotic cystectomy noted five strictures in 118 subjects, all of which occurred in the open surgery group, indicating that the robotic approach is not a substantial risk factor for the development of strictures and, in fact, might be advantageous. 64 Another retrospective study of 573 robotic and open cystectomies noted 47 ureteroenteric anastomotic strictures (8.2%) with a 2.6% stricture rate in the intracorporeal diversion group, a 9.6% rate in the extracorporeal division group, and an 8.0% rate in the open group, differences that were not statistically significant. 65 Risk factors for ureteroenteric anastomotic strictures noted in this study included higher BMI and urine leak, with other factors such as the presence of cancer, age, history of diabetes, anastomosis type (Wallace vs Bricker), or neoadjuvant chemotherapy appearing noncontributory in their analysis. 65 In another study, BMI, intracorporeal diversion, resected right ureteral length, estimated glomerular filtration rate at 30 days postoperatively, urinary tract infection (UTI), and urine leak were associated with strictures. 62 Another retrospective study of 968 subjects undergoing radical cystectomy, including open and robotic cystectomies with both intracorporeal and extracorporeal diversions, noted that intracorporeal diversion was associated with an increased rate of ureteroenteric anastomotic strictures. 66 In this study, the stricture rate appeared to improve over a surgeon’s learning curve, indicating that this difference may disappear with time. 66 Ileal conduit versus ileal neobladder did not appear to impact the ureteroenteric anastomotic stricture rate in one prospective series. 58
Some proposed strategies to prevent ureteroenteric anastomotic strictures include preserving blood supply by minimizing periureteral dissection, creating a tension-free anastomosis, and minimizing the handling of the ureter during implantation into the bowel segment. ICG can be used to evaluate tissue health and perfusion, allowing the surgeon to ensure high-quality tissue is used for a ureteroenteric anastomosis or identify ureters at increased risk of stenosis. One study retrospectively evaluated the use of ICG during ureteroenteric anastomosis, finding a statistically significant reduction in strictures following ICG use at a median follow-up of 12 months. 4 A study attempting to identify surgical characteristics associated with strictures during the review of surgical videos found that neither surgeons nor lay reviewers could identify subjects at risk for anastomotic strictures following robotic cystectomy with intracorporeal ileal urinary diversion. 67 Thus, surgical techniques may not entirely explain the development of these strictures. 67 Again, as surgeons overcome learning curves and improve their preservation of periureteral tissue, stricture rates likely decrease.
Renal function impairment
The decline in renal function is typical after radical cystectomy with urinary diversion regardless of approach, with approximately 4% of subjects in one meta-analysis experiencing renal failure.51,68 Thus, patients undergoing ileal urinary diversions should be monitored for declining renal function as well as electrolyte and metabolic abnormalities. 7 Worsening of renal function was seen in approximately 64% of radical cystectomy patients after 2.4 years of follow-up in one study. 68 The primary drivers of declining renal function following radical cystectomy were preoperative renal dysfunction and the development of ureteral strictures. 68 Renal function decline associated with urinary diversion is likely multifactorial and can be related to all of the following: chemotherapy, chronic obstruction with a delayed decrease in renal function, surgical technique, diversion type, and even immunotherapy.
Urinary tract infections
UTIs after urinary diversion are a significant clinical concern but are difficult to diagnose and manage. A recent meta-analysis noted an overall 8% rate of UTIs following cystectomy with urinary diversion. 51 However, the rate of postoperative UTIs was as high as 37% in one study of subjects undergoing urinary diversion, a majority of which occurred within the first year postoperatively. 58 Findings regarding the impact of urinary diversion type on UTI rate are conflicting, with one study demonstrating an increased rate of UTIs in subjects with continent diversions compared to incontinent diversions, while another study showed no difference.18,58 Complicating evaluation of UTIs, many individuals experience asymptomatic colonization of their diversion. Thus, routine monitoring with urine cultures in the absence of symptoms is not indicated. Antibiotic therapy is a mainstay of management for UTIs in the population of individuals who have undergone urinary diversion. For subjects experiencing recurrent UTIs, imaging should be performed to evaluate for causes, such as anatomic abnormalities and urolithiasis.
Incontinence after neobladder creation
Incontinence, while not a true complication, can be distressing for patients with an orthotopic neobladder. Incontinence can occur whether the neobladder is performed robotically or via an open approach, and patients must be appropriately counseled preoperatively that incontinence can occur. Nerve-sparing approaches during cystectomy have been proposed to improve continence outcomes. 69 In one study, 100% of subjects had a return of daytime continence after nerve-sparing cystectomy with intracorporeal neobladder with no instances of cancer recurrence. 69 Another strategy for improving continence during the creation of an orthotopic neobladder is the performance of a posterior reconstruction, wherein the rhabdosphincter and remnant Denonviller’s fascia are joined with suture to facilitate a tension-free anastomosis of the intestinal segment to the urethra.70,71 This strategy improves the strength of the tissue supporting the ileourethral anastomosis and has demonstrated a 100% daytime continence rate and a 44% nighttime continence rate in one study of 11 subjects. 71
Incisional hernia
In one study from 2018, the rate of incisional hernia after robotic or open cystectomy with urinary diversion was 14.3% and was not different between the robotic and open approaches. 57 The hernia rate in this study was predicted by rectus diastasis width and the use of a continent diversion compared with an ileal conduit. 57 Another study of 5646 individuals in the BladderBaSe database noted an incisional hernia rate of approximately 8% across the study during the period of 1997–2014, with hernia rates increasing over the course of the study. 72 This study did note an increased risk of incisional hernia in the robotic cohort compared with the open cohort, but this likely reflects the learning curve associated with the adoption of the robot as this study was performed early in its use. 72 Risk factors associated with incisional hernia in this study included the performance of a continent cutaneous pouch over other types of diversion and wound dehiscence. 72 In a separate study, the rate of incisional hernia was similar between individuals undergoing ileal conduits compared with neobladder, with an approximately 20% incisional hernia rate observed across the study subjects 58 Incisional hernias are frequently managed with surgical hernia repair, with or without mesh. Prevention of this complication requires careful identification and closure of the fascial layers of the abdominal wall.
Internal hernia
Rarely the bowel can herniate between the bilateral ureters and the urinary diversion, which can result in obstruction of the herniated bowel or the ureters.73–75 This complication can be prevented by minimizing bowel and ureteral mobilization during the performance of urinary diversion. 74 Additional maneuvers to prevent this complication include closing the peritoneal window through which the left ureter is passed and closing the mesentery at the site of the bowel reanastomosis. When evaluating patients experiencing ileus or bowel obstruction following cystectomy, surgeons must consider the possibility of a contributing internal hernia, which may be identified with abdominal imaging. This complication may require surgical repair or may be managed conservatively with ureteral stenting or bowel rest in less severe cases.
Parastomal hernia
A parastomal hernia, which is a fascial defect at the stoma site, can occur in any patient with a urostomy. Abdominal contents, including the bowel, may herniate through the fascial defect around the stoma. Thus, parastomal hernias can result in a symptomatic bulge that is bothersome to patients and can even lead to bowel obstruction. In one series of 383 subjects who underwent robotic cystectomy with an ileal conduit, 20% experienced a parastomal hernia, 31% of which were symptomatic and 11% of which required surgery to correct. 76 Stomas should be placed within the bounds of the rectus abdominis muscle to reduce parastomal hernia risk. Additionally, creating a tension-free stoma is imperative for stomal success. A consultation with the wound-ostomy service may reduce parastomal hernia by examining the patient’s habitus and providing siting recommendations. Parastomal hernias can be successfully repaired using either the multiport or the single-port surgical robot or by an open approach. 77 These repairs can be performed with or without the use of mesh at the site of the hernia. 77 In some individuals, especially those who are not bothered enough to elect surgery or poor surgical candidates, parastomal hernias are managed conservatively with a hernia belt.
Improving outcomes following robotic urinary diversion
Defining learning curve for robotic urinary diversion
Adoption of robotic assistance in the performance of cystectomy and urinary diversion is hampered by the length of the learning curve. Many studies have attempted to evaluate the impact of the learning curve on operative and patient outcomes. The learning curve following robotic-assisted cystectomy may impact complication rate, length of stay, operative time, and oncologic outcomes. However, the results of studies vary, likely due to surgical approach, robotic experience of surgeons prior to attempting cystectomy, and patient factors.
Several studies have examined patient outcomes over the learning curve for cystectomy and urinary diversion. One study of the first 63 robotic cystectomies with extracorporeal diversions performed at an institution noted longer hospital stays and operative times, slower return of bowel function, and higher EBL than those reported in the literature, which the authors felt was reflective of a learning curve. 78 Another study demonstrated that operative outcomes, such as 30- and 90-day complications, improved over the course of surgeons’ learning curves in the setting of intracorporeal urinary diversion. 79 EBL and risk of open conversion have been shown to decrease with further experience in the setting of cystectomy with urinary diversion.24,28,80 A separate study found that hospital length of stay was reduced over the course of the learning curve for the robotic performance of both ileal conduit and neobladder. 81 Multiple smaller studies have demonstrated no difference in complication rates over surgeons’ learning curves for urinary diversion.24,28,61
Operative times and urinary diversion times appear to improve over a surgeon’s learning curve in the setting of robotic-assisted cystectomy with urinary diversion.12,28,80 A manuscript evaluating the learning curve of a senior and two junior surgeons demonstrated that operative times improved over the learning curve, but there were no differences in other parameters, such as complications or hospital length of stay. 61 However, the cohorts analyzed in this study were only about 20 patients, which may not have been enough to thoroughly evaluate the learning curve. 61 A separate study of 28 subjects demonstrated decreased intracorporeal diversion time in the second 14 subjects (148-min median) compared with the first 14 subjects (201-min median) without any difference in complication rate. 82 However, these results are confounded by the fact that a majority of the subjects in the first half had a Bricker anastomosis and the majority in the second half underwent a Wallace anastomosis. 82 Another study of 46 subjects demonstrated a longer operative time for intracorporeal diversion compared to extracorporeal diversion for the first 10 intracorporeal patients, but by the last 10 patients of the cohort this difference had disappeared. 32 Thus, evidence suggests that operative time is one of the parameters that consistently improve over a surgeon’s learning curve in the setting of urinary diversion.
Several studies have attempted to evaluate the length of surgeons’ learning curve. A multicenter retrospective study modeled the time to reach a plateau in key outcome measures following cystectomy with intracorporeal urinary diversion. 83 The time to plateau for each outcome of interest, including major and overall complications at 90 days, operative time, EBL, and length of stay, varied between 75 and 198 cases, indicating that different outcome measures progress at different rates. 83 The fastest to improve was operative time, which plateaued at 75 cases, followed by EBL at 88 cases. 83 The slowest to improve was the length of stay at 198 cases. 83 Of note, the average plateau rate of major complications, defined as Clavien–Dindo Grade III or greater, in this study was 14%, highlighting that even experienced surgeons will have complications in this complex surgery. 83 A separate study of 203 cases of robotic cystectomy with ileal conduit or neobladder determined that proficiency was reached at about 140 cases based on console time, oncologic outcomes, complication rate, and development of ureteroenteric anastomotic strictures. 84 A study of the first 53 robotic cystectomies with intracorporeal ileal conduit performed by a single surgeon demonstrated a statistically significant reduction in operative time and minor complications rates over time, leading the authors to recommend that complex cases be performed by surgeons who have performed at least 40 cystectomies. 85
At our own institution, retrospective review of unpublished personal data from a single surgeon (JBK) who has completed over 250 ileal conduits as well as numerous orthotopic neobladders via intracorporeal robotic approach showed findings in line with previously published reports. Using the “My Intuitive” app, JBK prospectively gathered personal console times during robotic-assisted radical cystectomy with either ileal conduit or orthotopic neobladder. Review of trends showed significantly decreased console time for robotic-assisted laparoscopic cystectomy with ileal diversion after 10 cases, after which console time plateaued until it significantly decreased again after 100 cases. On the other hand, JBK’s console times for robotic neobladders saw a steady decrease over her first 100 cases (Figure 2).

Trend in robotic console time for JBK performing an intracorporeal neobladder.
Thus, surgeon experience appears to impact outcomes for robotic cystectomy with urinary diversion. Wijberg et al. demonstrated that the rate of major complications at 90 days plateaued at 14% after 137 cases and that overall complications at 90 days plateaued at roughly 50% at 75 cases. Other studies have also demonstrated reduction in complication rates with further surgeon experience.84,85 Thus, given that complication rates fall with surgeon experience, there is an argument to be made that cystectomy with urinary diversion may be best performed by high-volume centers. However, this strategy may limit access to care and may serve as a barrier to recruiting new surgeons to perform these complex surgeries. Thus, surgical training strategies to shorten the learning curve are desperately needed.
Training
Training programs have been established to reduce the length of the learning curve for robotic-assisted laparoscopic cystectomy. Many studies of training programs have focused on modular training, wherein the case is broken into discrete steps, which can be mastered separately. One training course included theoretical training, simulation-based training, and clinical training, which was established by expert consensus. 86 In this study, the cases were divided into 11 steps or 5 modules, which authors felt helped learners to focus on specific sub-skills rather than being overwhelmed by the whole case. 86 As the purpose of this manuscript was to describe the training program rather than to evaluate its success, it did not include a report of outcomes. 86 Another study evaluated outcomes for a single experienced robotic surgeon participating in a modular training program to learn to perform robotic cystectomy with intracorporeal diversions, including neobladder and ileal conduit. 87 This program included theoretical lessons, videos, and stepwise training on cystectomy cases. In this study, operative time and time to create the diversion decreased after the 30-day program and there were relatively few complications. 87 Another study demonstrated that operative times were reduced when trainees followed a modular training program for cystectomy, without changes in other outcomes such as complications and EBL. 88 Thus, structured training programs are promising to promote adoption and improve surgeon comfort with robotic urinary diversions.
Models can also be used to confirm the ability to perform a type of diversion. One group used a combination of cadaveric and silicone modeling to confirm that it was possible to perform a Vesica Ileale Padovana neobladder and that it would have adequate compliance. 89 The authors of this study noted that by practicing on a model and reviewing steps, they were able to identify areas of improvement and refine their technique. 89 Further research in this approach is warranted to establish its efficacy for other diversion types and establish its impact on the learning curve.
Enhanced recovery after surgery
There has been substantial interest in using ERAS protocols to reduce complications, readmissions, and hospital length of stay by optimizing patients’ performance status and standardizing management both in and out of the hospital for individuals undergoing cystectomy. ERAS protocols are now broadly used by surgeons performing robotic cystectomy with urinary diversion and often include preoperative counseling and patient optimization, guidance for the hospital course such as diet advancement and drain management, and standardized posthospitalization follow-up. The introduction of an ERAS protocol decreased hospital length of stay without increasing readmissions within 90 days following intracorporeal urinary diversion using the robotic platform in one study, highlighting the utility of ERAS protocols in this population. 90 These results were corroborated by another retrospective study of an ERAS protocol for robotic cystectomy with intracorporeal urinary diversion, with the added benefit of reducing transfusion rate in the group receiving the ERAS protocol. 91 Similar successes have also been reported in other studies, further corroborating the effectiveness of ERAS protocols.92,93 Thus, ERAS now plays a critical role in perioperative outcomes for patients undergoing robotic urinary diversions.
Female pelvic organ sparing
The sparing of pelvic organs, including the vagina, uterus, and ovaries, in female patients undergoing robotic-assisted radical cystectomy with urinary diversion, has been proposed as a strategy to preserve sexual function and, for those undergoing a neobladder, continence. 94 This technique has demonstrated good oncologic and sexual function outcomes. 94 Additionally, for neobladder patients, continence rates are high, with 80% of subjects dry overnight and 70% of subjects using one pad per day or less in one series. 94 As more surgeons learn this technique, this may become a strategy for improving outcomes in patients undergoing radical cystectomy and may especially benefit patients receiving a neobladder as their urinary diversion.
Limitations
There are several limitations to the available data. Many studies are retrospective in nature. Additionally, some studies are case series describing a new technique with limited data on outcomes. Furthermore, studies used different definitions of and timelines for reporting complications, making it difficult to reconcile the data or compare between studies. Finally, many of the studies reviewed focused on the cystectomy portion of the case and on oncologic outcomes rather than focusing on the diversion, which is the subject of this narrative review, making the data difficult to interpret. Furthermore, even when describing similar types of diversions, techniques varied widely, complicating comparisons between studies.
Implications of results
Given that there are conflicting data regarding success rates for various diversion strategies, it is unlikely that guidelines based on current data will strongly suggest any particular diversion type but rather leave it up to shared decision making between patients and surgeons. Future research could demonstrate the superiority of intracorporeal or extracorporeal diversions or help shed light on which patients will most benefit from each type of diversion, which would guide patient counseling. Furthermore, as more publications examine complication rates and types for different diversion strategies, surgeons will be better able to diagnose and manage these complications. Finally, there are numerous published strategies to improve outcomes, including ERAS protocols and training programs. Implementation of these strategies and publications describing their results will inform future patient management and improve surgeon training, and thereby improve results.
Conclusion
The robotic approach to urinary diversion, either with or without cystectomy, has been gaining traction among practicing urologists. Complication rates for this procedure are acceptable regardless of approach; the advantages of the surgical robot, such as improved visualization, mean that robotic urinary diversion will continue to be utilized. Thus, strategies to continue to reduce complications and improve outcomes, such as modular training and ERAS protocols, should continue to be employed to optimize results following robotic urinary diversions. As the field continues to develop, outcomes will improve and new strategies will develop to make robotic urinary diversion easier to perform and safer for patients.
