Abstract
Background:
Clean intermittent catheterization (CIC) is the golden standard in patients with lower urinary tract dysfunction, leading to bladder emptying problems, due to neurogenic or non-neurogenic causes. CIC affects patient Quality of Life (QoL) both positively and negatively.
Objectives:
The aim of this systematic review is to determine which measurements are used to report on the QoL of patients who are on CIC in the currently available literature, to determine the overall QoL of patients who are on CIC and lastly, to determine whether QoL in patients who are on CIC is dependent on the underlying cause (neurogenic vs non-neurogenic).
Design:
This systematic review was conducted following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement.
Data sources and methods:
The Embase, Medline, Web of Science Core Collection, CINAHL, Google Scholar, and the Cochrane CENTRAL register of trials databases were systematically searched for relevant publications until March 2023.
Results:
A total of 4430 abstracts were screened and 43 studies were included. Studies were published between 1993 and 2022 and consisted of only neurogenic patients in 22 studies, the others included a mixed population. The included patient populations and the used measurements/tools were heterogeneous. There were 21 measurements/tools used to measure QoL, of which 3 were not validated. One questionnaire was developed to measure QoL in patients on CIC (intermittent self-catheterization questionnaire). Other measurements were suitable for general health-related QoL, to evaluate neurogenic bladder symptoms or incontinence oriented.
Conclusion:
The 43 included studies showed a great variety of used tools to measure QoL in patients on CIC due to neurogenic and non-neurogenic causes. Because of lacking uniformity of the measured aspects of QoL, the different included studies could not be compared and subgroup analysis was not performed. Recommendations for future research and practice are provided.
Trial registration:
This systematic review was registered and published beforehand at Prospero (CRD42020181777; https://www.crd.york.ac.uk/prospero).
Keywords
Introduction
There are millions of people worldwide who suffer from lower urinary tract dysfunction (LUTD) due to various underlying conditions. Often, the cause is either an unknown (idiopathic) condition or a more evident neurological disease such as spinal cord injury (SCI) or multiple sclerosis (MS). LUTD can lead to problems with emptying the bladder, resulting in urinary retention or clinically significant post-void residual (PVR).1,2 If left untreated, this urinary retention or PVR could lead to infections, stone formation, and ultimately kidney damage and renal failure. One of the preferred treatments for bladder emptying is clean intermittent self-catheterization (clean intermittent catheterization (CIC)), or third-party catheterization if a patient is not able to perform CIC him/herself. Patients or their caregivers are recommended to administer CIC 4–6 times per 24 h, allowing the catheterized volume to stay below 400–500 mL to prevent the bladder from overstretching.3,4 Performing CIC can have a great impact on a patient’s quality of life (QoL) and has an impact on the activities of daily living of patients or their caregivers, social relationships, and cultural contexts. 5
There are various ways to measure and report QoL, which makes it difficult to interpret or compare results between different studies in patients on CIC. Standardized use of validated tools can help to improve outcome measures and improve the generalizability of those outcomes. This enables researchers and clinicians to uniformly interpret the measured QoL of patients on CIC. The first step is to make an overview of the used tools and overall measured QoL.
Therefore, the aim of this systematic review is to evaluate the currently available literature and to determine which outcome measures are used to report on QoL in patients who perform CIC and to establish whether they are validated for this use. In addition, we will assess if the overall QoL of patients who are on CIC can be determined. Our last aim is to determine whether QoL is dependent on the underlying cause (neurogenic vs non-neurogenic) for CIC.
Materials and methods
Study registration
This systematic review was performed according to the Cochrane Handbook for Systematic Reviews and intervention and the results are reported according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement. Before the literature search was started, the study protocol was registered and published on Prospero (CRD42020181777; https://www.crd.york.ac.uk/prospero).
Literature search
The Embase, Medline, Web of Science Core Collection, CINAHL, Google Scholar, and the Cochrane CENTRAL register of trials databases were systematically searched for relevant publications until March 2023. No date restrictions were applied and duplicates were removed. The complete search string can be found in the Attachment 1 (Supplemental Material). The selection process is described using the PRISMA flow diagram. 6
Eligibility criteria
Our aim was to include all publications of original studies concerning patients on CIC that use a QoL measurement. Studies that included patients ⩾12 years of age on chronic CIC (>3 months) due to a neurogenic or a non-neurogenic cause were found eligible. Studies on CIC for dilatation of the urethra, or studies that included patients with any form of urinary diversion were excluded. Other exclusion criteria were: the use of non-specified measurements for QoL, mixed populations included and the eligible population accounted for <90%, or if data on them were not reported separately, studies with less than 10 patients, case reports, review articles, non-English language studies, conference abstracts, or if the full-text article was not available.
Selection of studies
Titles and abstracts were screened in Endnote independently by two researchers (T.D. and J.G./R.L.C.). Studies that were found eligible for full-text retrieval were compared between the two reviewers, and discrepancies were discussed individually. Full-text screening of the potentially eligible publications was performed by the same reviewers using a standardized screening form. Any disagreements were resolved by a third reviewer (B.F.M.B.).
Data extraction
The predefined data were extracted from the included full-text publications by two reviewers (T.D. and J.G.) independently and documented in a standardized form. Again, any disagreements between the two reviewers were resolved by the third reviewer (B.F.M.B.). Data extracted from the articles included: study characteristics, number of included patients, sex, age, underlying cause for CIC, duration of CIC, mean age at the time of the start of CIC, frequency of CIC per 24 h, self or third-party catheterization, frequency of urinary tract infections (UTIs), comorbidities reported, whether or not patients with diversions were included, type of catheter, single-use/reusable catheter, the used tool/measurement for measuring QoL, whether or not the tool was validated, measured QoL, differences in QoL in case of follow-up, and whether or not any predictors of QoL were named.
Outcome measurements
The primary outcome consists of the used measurements for assessing QoL and establishing the validation for the specific patient population. Secondary outcome measurements are measured QoL, measured QoL related to type of catheter used, and QoL of neurogenic versus non-neurogenic patients.
Subgroup analyses
The following subgroups of interest were predefined: male versus female, pediatric versus adolescent patients, neurogenic versus non-neurogenic causes for catheterization, and urethral versus non-urethral (continent catheterizable tubes/stoma’s) catheterization. Analyses of subgroups were performed if the data available was sufficient to do so.
Risk of bias assessment
The risk of bias analysis of each included study was performed by using the Cochrane Risk of Bias Assessment tool. 7 The following items that might have led to bias were assessed: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), other bias, a priori protocol, and consecutive study participants. A list of predefined possible confounders was developed and agreed on by the authors of this systematic review: age, gender, underlying cause/disease for CIC, comorbidities, mobility/physical disability, accessibility/reimbursement of catheters, and duration of CIC. The mentioned possible points of bias or confounders were scored as “low,” “unclear,” or “high.” The risk of bias was scored high if that specific type of bias could have influenced the reported results. The confounders possibly leading to bias were classified as high if no adjustments were made in the analysis of the results or in the protocol, or if there was an imbalance between groups for that specific aspect. The analysis was performed by two researchers (T.D. an R.L.C.). Any disagreements were resolved by a third reviewer (B.F.M.B.). The Review Manager version 5.3 of Cochrane Collaboration was used to make the figure describing the risk of bias.
Results
Search results
The PRISMA flow diagram is shown in Figure 1, which shows the results of the literature search and study selection. The initial search resulted in 7184 articles and after duplicates were removed, 4430 articles were left to screen. Title and abstract selection resulted in 227 articles for full-text evaluation. A total of 43 studies5,8 –49 were included in this systematic review. Reasons for excluding studies after the full-text articles were screened, are named in the flow chart.
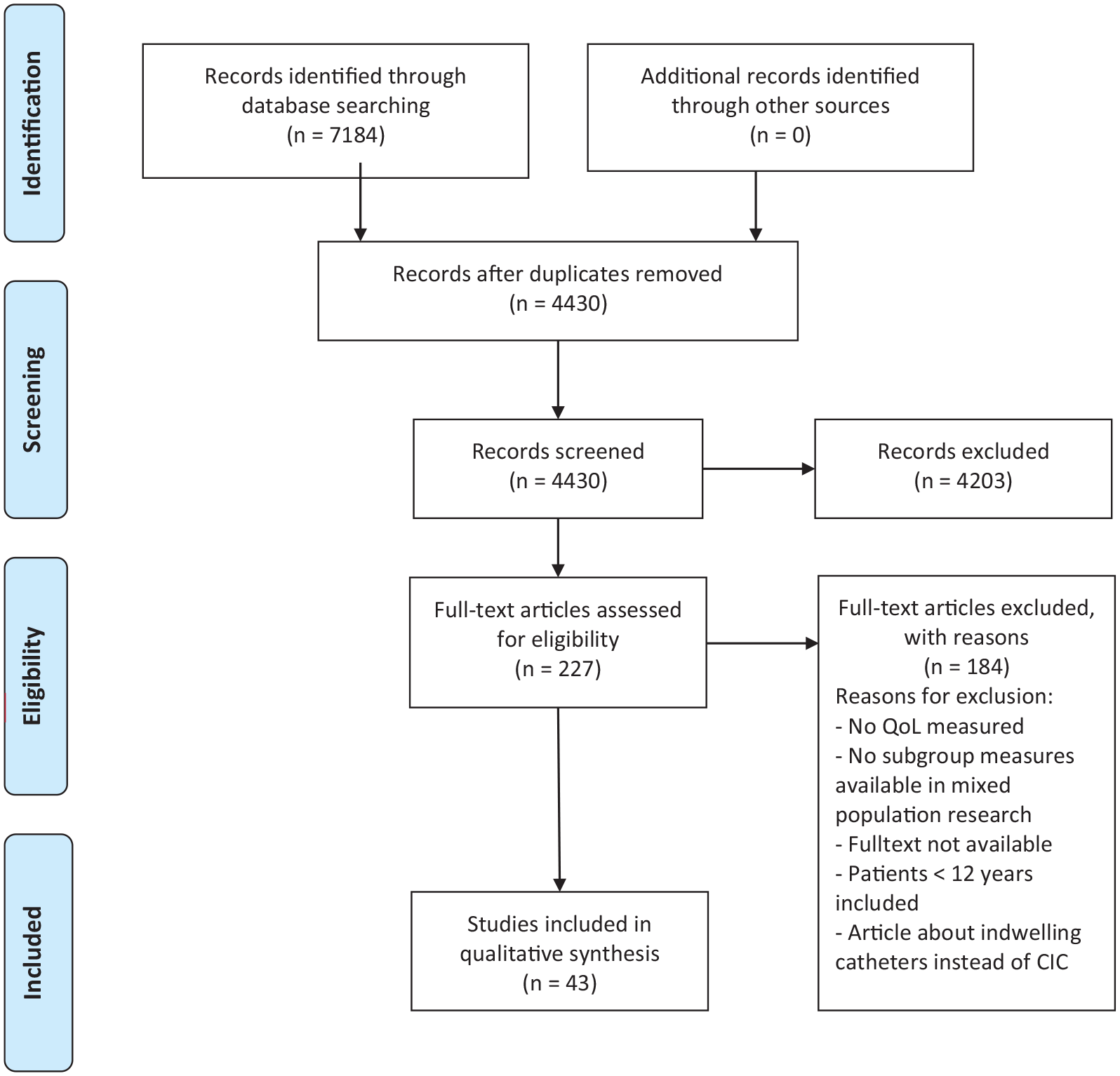
PRISMA 2009 flow diagram.
Characteristics of included studies and study population
Study characteristics
In Table 1, the characteristics of the 43 included studies can be found. The studies included were published between 1993 and 2022 and have QoL in patients on CIC as (one of their) outcome measures. Most studies were multicenter studies (n = 24) or mono-centric (n = 12) and seven were cohort studies where participants were derived from several databases. One randomized crossover study met the inclusion criteria of this systematic review. The follow-up, shown in Table 2, was reported in 17 studies and was not applicable in 26 studies due to the type of study procedure. The duration of follow-up varied between 1 month and 7 years. All studies used the same questionnaire/tool to measure QoL for follow-up and all but two reported on the change over time; these results can be found in Table 3.
Characteristics of included studies.

BPH, Benign prostate hyperplasia; CIC, clean intermittent catheterization; LUTD, lower urinary tract dysfunction; MMC, Meningomyocele (spina bifida); MS, multiple sclerosis; NLUTD, Neurogenic Lower Urinary Tract Dysfunction; NR, not reported; PVR, post-void residual; SCI, spinal cord injury.
Characteristics of included studies.

CIC, clean intermittent catheterization; COPD, Chronic Obstructive Pulmonary Disease; DM, Diabetes Mellitus; MS, multiple sclerosis; UTI, urinary tract infection.
Characteristics of included studies.

BP, bodily pain; CIC, clean intermittent catheterization; CISC, Clean Intermittent Self-Catheterization; EM, emotions; EQ-5D, EuroQol 5D; EQ-5D VAS, EuroQol 5D Visual Analog Scale; GH, general health; GHP, General Health Perceptions; GHQ-28, General Health Questionnaire-28; IC-A, intermittent catheterization by an attendant/caregiver; IC-P, intermittent catheterization by patient; ICIQ-FLUTS, International Consultation on Incontinence Questionnaire Female Lower Urinary Tract Symptoms Modules; ICIQ-LUTS, International Consultation on Incontinence Modular Questionnaire-Lower Urinary Incontinence Quality of Life Module; ICIQ-SF, International Consultation on Incontinence Questionnaire-Short Form; ICIQ-UI SF, International Consultation on Incontinence Questionnaire-Urinary Incontinence Short Form; II, incontinence impact; I-QoL, Incontinence Quality of Life; ISC-Q, intermittent self-catheterization questionnaire; MCS, Mental Component Summary; MH, mental health; mSF36, This is the RAND36– Item Health Survey, with or without modification of the Physical Functioning Scale for those with spinal cord injury; NBSS, The Neurogenic Bladder Symptom Score; PCS, Physical Component Summary; PF, physical functioning; PL, physical limitations; PR, personal relationships; QBB, bowel/bladder; QFI, family/independence; QoL, quality of life; QUALAS-T, QUAlity of Life Assessment in Spina bifida for Teenagers; RE, role emotional; RL, role limitations; RP, role physical; SCI, spinal cord injury; SCI-QoL, Spinal Cord Injury Quality of Life; S/E, sleep/energy; SF, social functioning; SF-12, 12-Item Short-Form Health Survey; SF-36, 36-Item Short-Form Health Survey questionnaire; SF-Qualiveen, Short form of Qualiveen 30; SL, social limitations; SM, Severity measures; SS, symptom severity; SWLS, satisfaction with life scale; UTI, urinary tract infection; VAS, visual analog scale; VT, vitality; WHOQoL, World Health Organization Quality of Life.
Characteristics of study population
Eighteen studies solely included patients with SCI as an underlying cause for CIC, three studies only included patients with MS and one study solely included patients with spina bifida. The remaining studies comprised participants with various underlying diseases or only described if participants had a neurogenic or non-neurogenic cause to perform CIC. Nineteen studies included patients with a non-neurological disease, like (postoperative) retention, bladder outlet obstruction, or an underactive bladder The experience on CIC was reported in 29 studies and varied from starting CIC at the time of inclusion to a median of 10.5 years. One study included female participants exclusively and one study included only male patients. All other studies included a mixed population. The mean age of the participants varied from 31.8 to 62.0 years, five studies did not report the age of the participants other than they were adults at the time of inclusion and one study only reported that the age of participants ranged from 14 to 20 years old. Only seven studies reported on the mean age when CIC was started, which varied from 35.2 (±20.5) to 62 (±13.4) years. Patients with diversions were excluded in five studies and were analyzed separately in five other studies. One study included patients with a diversion but did not analyze the QoL separately for this group. The majority of the studies (32) did not report whether or not patients with a diversion or non-urethral catheterization (continent catheterizable tubes/stomas) were included in their population.
Reported information on catheters and CIC
Thirteen studies reported which catheters were used in their included population, which can be found in Table 2. One of these studies described the used catheter as discreet, but no further explanation on which type of catheter was given. Twelve studies provided information about their participants using single-use or reusable catheters. In seven studies only single-use catheters were used, in the other five studies reusable catheters were also used. The frequency of catheterization per 24 h was reported in 12 studies and varied from ⩽1 times per 24 h to >6 times per 24 h. Six studies described a mean frequency, which varied between 3 and 6 times per 24 h.
Five studies explicitly described that CIC was performed by the included patients themselves, one study described that it was performed by a third party in all patients and 15 studies reported that both CIC by the patients themselves or a third party occurred. The remaining 25 studies did not provide information on this. UTIs in the study population were reported in various ways in 14 studies and the presence of comorbidities was reported in 7 studies (Table 2).
Results on outcome parameters
Used tools to measure QoL
A total of 21 different questionnaires/tools were used to measure QoL in patients on CIC in the included studies (Table 3). The characteristics of these tools and how often they were used in the included studies can be found in Table 4. The 36-Item Short-Form Health Survey questionnaire (SF-36) was the most frequently used tool, namely in nine of the included studies. The Intermittent self-catheterization questionnaire (ISC-Q) was used to measure QoL in eight of the included trials. Twelve included studies used two or more tools to measure QoL. Three studies used tools that are not validated, James et al. used a 7-point Likert scale questionnaire that was developed for the database specifically, 27 Kessler et al. used a questionnaire that was based on the validated 12-item Short-Form Health Survey (SF-12) 28 and Yilmaz et al. used a five-point Likert scale on the effect of CIC on QoL. 47
Used tools to measure QoL, abbreviations of the questionnaires, what is measured, their normal ranges, and how often they were used in the included studies in this systematic review.
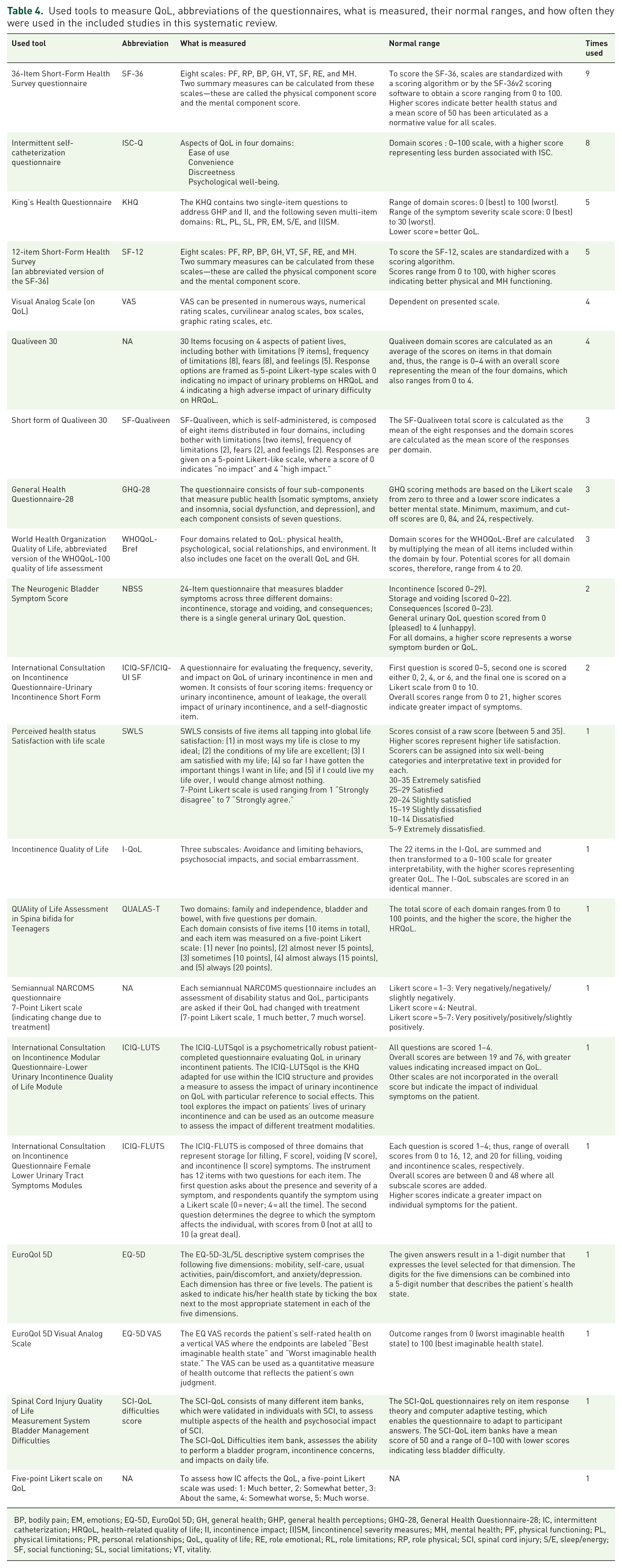
BP, bodily pain; EM, emotions; EQ-5D, EuroQol 5D; GH, general health; GHP, general health perceptions; GHQ-28, General Health Questionnaire-28; IC, intermittent catheterization; HRQoL, health-related quality of life; II, incontinence impact; (I)SM, (incontinence) severity measures; MH, mental health; PF, physical functioning; PL, physical limitations; PR, personal relationships; QoL, quality of life; RE, role emotional; RL, role limitations; RP, role physical; SCI, spinal cord injury; S/E, sleep/energy; SF, social functioning; SL, social limitations; VT, vitality.
One patient reported outcome measure (PROM) was developed to measure QoL in neurogenic patients on CIC (ISC-Q), others are suitable to measure general QoL (SF-36, King’s Health Questionnaire (KHQ), SF-12, Visual Analog Scale (VAS), General Health Questionnaire-28 (GHQ-28) World Health Organization Quality of Life (WHOQoL)-Bref, satisfaction with life scale (SWLS), EuroQol 5D (EQ-5D), EuroQol 5D Visual Analog Scale (EQ-5D VAS), and five-point Likert scale on QoL), developed for neurogenic bladder dysfunction (Qualiveen 30, SF-Qualiveen, The Neurogenic Bladder Symptom Score (NBSS), QUAlity of Life Assessment in Spina bifida for Teenagers (QUALAS-T), and Spinal Cord Injury Quality of Life (SCI-QoL) difficulties score) or were mainly incontinence oriented (International Consultation on Incontinence Questionnaire-Urinary Incontinence Short Form (ICIQ-SF/ICIQ-UI SF), Incontinence Quality of Life (I-QoL), International Consultation on Incontinence Modular Questionnaire-Lower Urinary Incontinence Quality of Life Module (ICIQ-LUTS), and International Consultation on Incontinence Questionnaire Female Lower Urinary Tract Symptoms Modules (ICIQ-FLUTS)).
Measured overall QoL and predictors of QoL
As shown in Tables 3 and 4 the way of reporting on QoL between the used tools differs tremendously. Therefore, no statement/qualification on the overall QoL in patients on CIC can be made or given.
Nine studies reported on possible predictors that contribute to the measured QoL in patients on CIC (Table 3). Some predictors that were mentioned are: UTIs, level of SCI, the occurrence of spontaneous micturition or incontinence, frequency of CIC, type of catheter, third-party CIC/independence, and pain during catheterization.
Measured QoL dependent on type of catheter
There was a lack of information on the relationship between the used type of catheter and QoL in most of the studies, so no conclusions on this could be drawn.
Subgroup analysis
Due to several reasons, it was not possible or contributive to perform a subgroup analysis for all predefined subgroups. First, a large heterogeneity of the studied population was seen in the included studies. This was the case for age, gender, cause for catheterization, and whether or not patients with non-urethral catheterization were included. Second, there was also a great variety seen in the used tools/questionnaires to measure QoL. The outcomes of these tools/questionnaires differed widely, which made comparison of QoL between the different measures, impossible.
Instead of performing an analysis of subgroups, the usage of different QoL measurements within these subgroups will be described.
Men and women
The majority of the studies included mixed-sex populations, one study only included female patients, and one study only included male patients. Therefore, no conclusions can be drawn about the used questionnaires in sex-specific populations.
Pediatric/adolescent patients
Only one study included patients of mostly adolescent age (14–20 years of age). 25 The authors used the QUALAS-T questionnaire, 50 which is validated in a population of 13–17 years old.
Neurogenic and non-neurogenic causes for catheterization
The studies that included a mixed population of patients that are on CIC due to neurogenic and non-neurogenic causes, did not report the QoL of these groups separately. Eighteen studies solely included patients with SCI; no uniformity was seen in the used questionnaires in this population, as 10 different tools to measure QoL were used (Table 3).
Diversion or non-urethral catheterization (continent catheterizable tubes/stoma’s)
The five studies14,16,22,23,32 that included patients with a diversion or with non-urethral catheterization all used different tools/measurements to report on QoL: SF-36, ICIQ-SF, NBSS, SF-12, and I-QoL. No validation of these measurements in this specific patient population could be found in the current literature.
Risk of bias analysis
The Cochrane Risk of Bias Assessment tool was used to assess the risk of bias in the included studies. Bias and the predefined possible confounding factors in these studies were classified as high (−), unclear (?), or low (+). Bias was assessed as high or unclear in most studies. The risk of confounding factors was scored as low or unclear in most studies. A summary of the findings of this assessment can be found in Figure 2.
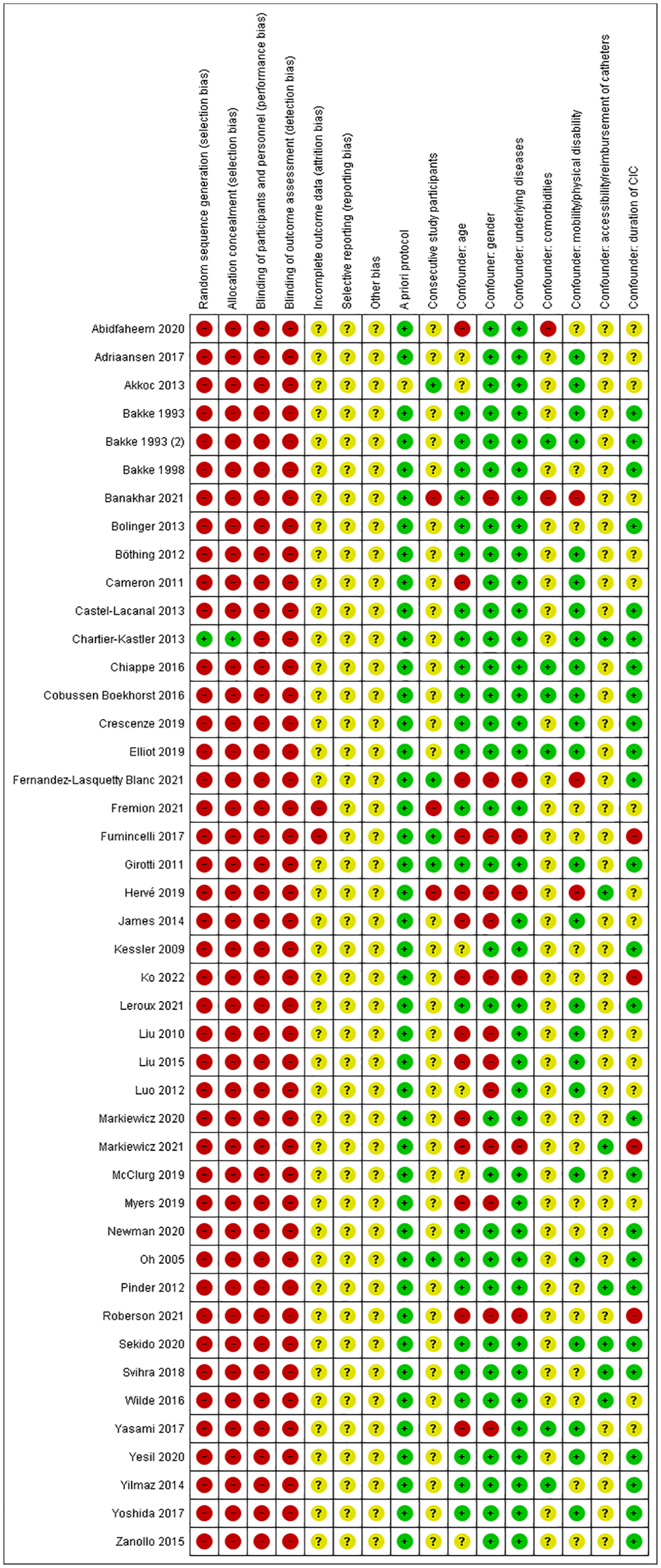
Risk of bias summary.
Discussion
CIC is the golden standard treatment for patients with neurogenic and non-neurogenic LUTD leading to bladder emptying difficulties.3,4 In the Netherlands alone there were 46,000 CIC users in 2018 51 and worldwide there are millions of people that self-catheterize. 1 Evaluation of their QoL and identifying possible challenges that these patients face can help to improve the standard of care and their QoL. Using patient reported outcome measures (PROMS) can also be helpful in initiating conversations between patients and caregivers about certain aspects, like QoL.
This systematic review showed a great variety in the used tools/measurements and reported QoL of patients on CIC. In the 43 included studies, 21 different tools were used. There are a lot of tools available to measure QoL, one was specifically developed and validated in patients who perform CIC (ISC-Q), others are suitable to measure general QoL (SF-36, KHQ, SF-12, VAS, GHQ-28 WHOQoL-Bref, SWLS, EQ-5D, EQ-5D VAS, and five-point Likert scale on QoL), some other tools were developed for neurogenic bladder dysfunction (Qualiveen 30, SF-Qualiveen, NBSS, QUALAS-T, and SCI-QoL difficulties score) or were more incontinence oriented (ICIQ-SF/ICIQ-UI SF, I-QoL, ICIQ-LUTS, and ICIQ-FLUTS). The SF-36 was the most frequently used questionnaire. This questionnaire was designed for use in clinical practice and research, health policy evaluations, and general population surveys. It consists of one multi-item scale that assesses eight health concepts and is a generic instrument to measure Health Related Quality of Life (HRQoL). 52 The SF-12, which was used five times in this review, is an abbreviated version of the SF-36 and measures the same concept. 53 The second most used questionnaire was the ISC-Q, which was developed to specifically evaluate QoL in patients who are on CIC. Validation was done in patients with urinary retention due to a neurogenic cause. 40 A systematic review of QoL performed by Pequeno et al. in 2020 in adult patients on CIC-assessed instruments in population-based studies around the world. The authors debate that heterogeneity of the used tools hinders comparison between similar populations and that further research is necessary to determine which QoL instrument is best suited in which situation. 51
As seen in earlier research performed by our group, the majority of the patients on CIC have a non-neurogenic underlying cause. 54 Most studies in this systematic review included a mixed neurogenic and non-neurogenic population or solely a neurogenic population. The measured QoL was not reported separately in the mixed populations. The distinction is important because a neurogenic population differs from a non-neurogenic population, with more comorbidities and other factors contributing to QoL like a possible dependency on caregivers. Thus, separate reporting of QoL of neurogenic and non-neurogenic patients is advisable.
It was challenging to identify the scoring methods and normal ranges of each questionnaire/measurement tool. These normal ranges and measurement properties differed, as shown in Table 4. It was easily confirmed in the literature if the used tools were validated, but it could not always be confirmed that the tools were validated for use in this specific patient population or to confirm that the tool was administered the correct way.
In the current literature, there are multiple comparable systematic reviews. In 2017 Fumincelli et al. 5 conducted a scoping review on the QoL of patients on intermittent catheterization and their caregivers. They included 13 studies with a neurogenic bladder patient population and compared the main results and conclusions of these studies, rather than looking at the measured outcome of the used tools. They found that patients on CIC had lower QoL compared to patients with normal bladder function. When good pain control during the procedure was encouraged, better QoL scores were observed, particularly in the social and psychological domains. Dependency on caregivers for performing CIC was the main factor negatively affecting the patient’s QoL. Wang et al. 55 performed a systematic review evaluating PROMS in CIC users. They found the ISC-Q as the most widely used questionnaire to measure HRQoL in patients on CIC but emphasized the need to perform translation and validation studies and cross-cultural adaptations to allow usage of the tool by professionals and patients in their own language and in different countries/societies/cultures. Clark and Welk 56 performed a systematic review regarding the used PROMS in neurogenic patients and found only a few tools that were specifically developed for this population. They underlined the need for understanding the principles of PROM development and systematic evaluation, so clinicians and researchers can select the most suitable PROM for their specific purpose. In our systematic review, we included all patients on CIC in whom QoL was measured, regardless of their underlying cause, and checked if the used tools were used in the correct population, which resulted in a complete and critical overview of all tools used.
This systematic review was conducted according to a premade and preregistered protocol that conforms to the Cochrane Handbook for Systematic Reviews of interventions 57 and PRISMA guidelines. 6 It gives an overview of the current literature available on the used measurements to evaluate QoL in patients on CIC. There are however some limitations. First, the predefined subgroup analysis could not be performed due to the heterogeneity of the patient populations, the used tools, and their outcome measures. Second, our search was limited to only English-written publications. Lastly, our research question was quite broad because we aimed to include both neurogenic and non-neurogenic patients, resulting in a high yield of articles. A more specified search strategy regarding a specific patient population might have led to a feasible overall QoL analysis in that population.
Conclusion
The 43 included studies showed the variety of used tools to measure QoL in patients on CIC. Because of the lacking uniformity of the measured QoL outcomes, various included studies could not be compared and subgroup analysis was impossible. Only one measurement tool was developed for evaluating QoL in neurogenic bladder patients on CIC. In a future perspective, it is advised to develop or validate an existing PROM that can measure QoL in patients on CIC explicitly and is validated in both a neurogenic and a non-neurogenic patient population and to perform translation and validation studies for cross-cultural adaptation. Furthermore, it is advised to report QoL of neurogenic and non-neurogenic patients separately.
Lastly, the scoring system must be transparent and easily used to enable smooth implementation in clinical practice and research as complicated tools are more challenging to carry out in daily practice. This enables clinicians and researchers to compare different treatment options and studies and leads to a better insight into QoL in patients on CIC and their challenges.
Supplemental Material
sj-docx-1-tau-10.1177_17562872241303447 – Supplemental material for Quality of life aspects of intermittent catheterization in neurogenic and non-neurogenic patients: a systematic review on heterogeneity in the measurements used
Supplemental material, sj-docx-1-tau-10.1177_17562872241303447 for Quality of life aspects of intermittent catheterization in neurogenic and non-neurogenic patients: a systematic review on heterogeneity in the measurements used by Tess van Doorn, Rosa L. Coolen, Jan Groen, Jeroen R. Scheepe and Bertil F. M. Blok in Therapeutic Advances in Urology
Supplemental Material
sj-docx-2-tau-10.1177_17562872241303447 – Supplemental material for Quality of life aspects of intermittent catheterization in neurogenic and non-neurogenic patients: a systematic review on heterogeneity in the measurements used
Supplemental material, sj-docx-2-tau-10.1177_17562872241303447 for Quality of life aspects of intermittent catheterization in neurogenic and non-neurogenic patients: a systematic review on heterogeneity in the measurements used by Tess van Doorn, Rosa L. Coolen, Jan Groen, Jeroen R. Scheepe and Bertil F. M. Blok in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
