Abstract
Bladder paraganglioma is a rare neuroendocrine neoplasm, either functional or non-functional, arising from the urinary bladder. Functional variants present with catecholamine-related symptoms, while non-functional variants pose diagnostic challenges, mimicking urothelial carcinoma. Misdiagnosis risks underscore the importance of accurate identification for appropriate patient management. In this case, a 52-year-old man, diagnosed incidentally with hypertension and reported occasional post-micturition tachycardia, underwent abdominal ultrasound for known hepatic cyst follow-up, revealing an oval hypoechoic bladder mass. Initial consideration of bladder urothelial carcinoma prompted further investigation with contrast-enhanced CT scan and cystoscopy that confirmed extrinsic mass nature, and subsequent robotic-assisted partial cystectomy was performed. Histologically, the removed mass exhibited characteristic features of bladder paraganglioma. Postoperative recovery was uneventful, with resolution of post-micturition tachycardia at 1 month. Follow-up includes endocrinological evaluation and a 6-month CT scan. In conclusion, bladder paraganglioma should be considered in para-vesical mass differentials. This case highlights the importance of meticulous history collection, even in asymptomatic patients, the need for a multidisciplinary approach for accurate diagnosis and management of this rare condition, and the robotic approach as a viable option.
Introduction
Bladder paraganglioma (BPG), whether functional or non-functional, is an uncommon neuroendocrine neoplasm arising from the urinary bladder. The first case of paraganglioma of the urinary bladder was reported and described by Zimmerman in 1953. 1 The bladder is the most affected organ of the urinary tract, originating from the chromaffin cells. Paraganglioma represents 0.06% of primary bladder tumors and about 6% of all paragangliomas. 2 While functional variants represent 61.3% of all reported cases 3 and they may exhibit catecholamine secretion with associated symptoms (hypertension, palpitations, headache, sweating, painless hematuria, low back pain), 4 not-functional variants pose diagnostic challenges as they can mimic urothelial carcinoma, with no specific presentation symptoms: sometimes painless hematuria, low back pain, or onset of hypertension during surgical resection.5,6
The rarity of this condition, especially the not-functional cases, is related to the risk of misdiagnosis and interferes with the right patient management.
Accurate diagnosis relies on the vigilance of pathologists recognizing characteristic histopathologic features and employing immunohistochemistry. Urologists play a crucial role in distinguishing BPGs from bladder cancer, considering their clinical, radiological, and pathological similarities and in the choice of treatment options that include transurethral resection and cystectomy.
In our study, we reported the case of a 52-year-old man who underwent the removal of a solid perivesical lesion that is a BPG.
Case presentation
In August 2023, a 52-year-old man underwent abdominal ultrasound for the follow-up evaluation of known hepatic cysts. The examination revealed an oval hypoechoic bladder mass along the right lateral wall, with dimensions of 4.6 × 4.2 × 4.9 cm. The bladder urothelial carcinoma was considered the first diagnosis. Further investigation with contrast-enhanced abdominal CT scan confirmed the abdominal mass, compressing the right lateral wall of the bladder, with dimensions substantially overlapping those of the ultrasounds. The formation exhibited non-homogeneous morpho-structural characteristics and clear and irregular margins with fluid content. It has thick walls that show marked and persistent contrast enhancement. The radiological differential diagnosis was Müllerian duct anomaly or ancient schwannoma [Figure 1(a) and (b))]
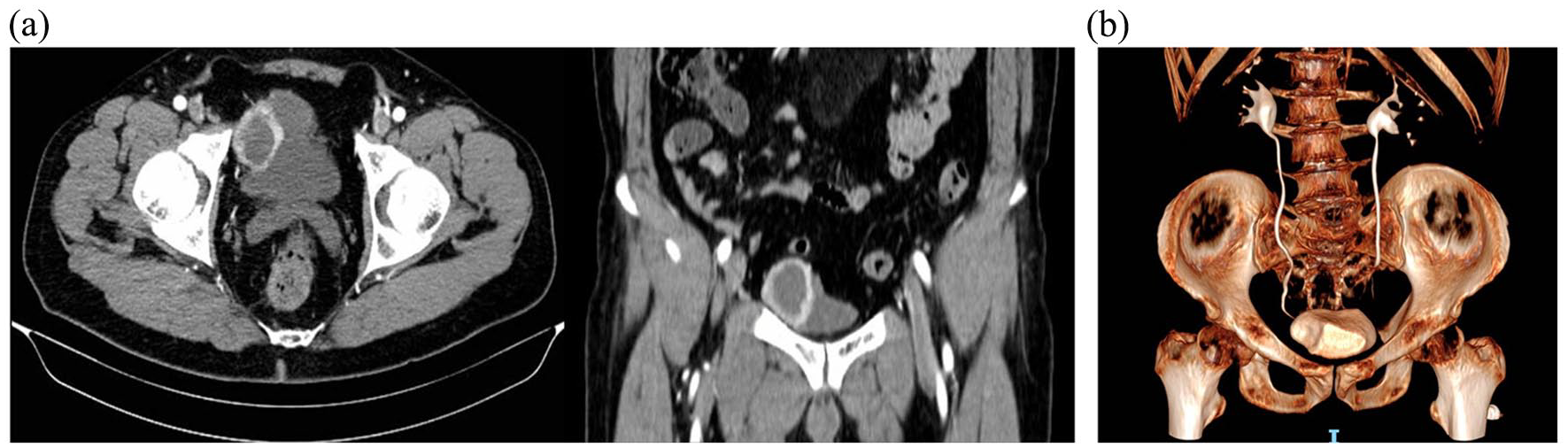
(a) In the CT images, evidence of a contrast-enhancing lesion along the right bladder wall is observed. (b) In the 3D reconstruction of the urinary tract, the compression of the right bladder wall by the noted mass becomes evident.
Subsequent cystoscopy confirmed the extrinsic nature of the mass without involvement of the bladder mucosa. In the evaluation of the current medical case, a decision has been made to abstain from requesting urinary cytology due to the nature of the extravesical lesion.
The patient had no significant medical history, except for hypertension discovered 1 year before the surgery, which led to the beginning of hypertension treatment. No urinary symptoms or hematuria were reported, but later, in a post-surgery interview, the patient referred to occasional post-micturition tachycardia episodes escalating in the last month before surgery. During pre-hospitalization, routine blood chemistry tests, without any specific endocrine tests (metanephrines and normetanephrine), were performed. The preoperative adrenergic blockade was not administered.
The surgery started with the patient in the 25° Trendelenburg position and docking of the Da Vinci Xi system. Five laparoscopic trocars were positioned with a 0° lens and three EndoWrist instruments: monopolar curved scissors, bipolar fenestrated forceps, and ProGrasp. During general anesthesia, a robot-assisted partial cystectomy was performed. Within the 30-min surgical timeframe, the known lesion on the right lateral wall of the bladder was identified, isolated, removed with careful dissection, and sent for histological examination. The bladder wall was sutured using a V-loc thread. The surgical procedure concluded with the placement of a 24-Ch tubular drainage and layered closure.
The postoperative hospitalization was regular. The drainage was removed on the second postoperative day and the patient was discharged on the third postoperative day, while the catheter was left in place and removed on the 12th postoperative day.
Macroscopically the removed mass has a rounded shape measuring 5 × 4.5 × 5 cm. Upon cutting, there is an internal cavity, surrounded by adipose tissue [Figure 2(a) and (b)]

(a) Macroscopically, the surgically removed mass has a rounded shape. On the right, the removed bladder wall is clearly visible and adhered to the mass itself. (b) Upon cutting, the lesion reveals a cavity with surrounding adipose tissue. The yellow arrow indicates the paraganglioma itself.
Histologically, it features principal cells, with rare mitotic figures, arranged in cords or nests, and enveloped by sustentacular cells and capillary networks. Positive immunohistochemical staining for neuroendocrine markers, including synaptophysin and chromogranin was observed [Figure 3(a)–(c)].

(a) Hematoxylin and eosin staining. The cells are arranged in cords with sustentacular cells and capillary networks. (b) At higher magnification, cells express chromogranin. (c) At higher magnification, cells express synaptophysin.
The pathological diagnosis was BPG.
Endocrinological evaluation has been referred to as part of the postoperative program, with dosage of metanephrines and normetanephrines. A total body CT scan with and without contrast has been scheduled for the 6-month follow-up.
The patient has maintained a good condition so far. At the 1-month postoperative visit, he reported no longer experiencing post-void tachycardia, while suspension of the anti-hypertensive drug was being evaluated.
Discussion
Given the rarity of BPGs, predicting prevalence, especially in locations like the genitourinary tract, is challenging and it can be easily misdiagnosed as bladder carcinomas. BPGs are found in various sites, including the kidney, renal pelvis, ureters, urethra, prostate gland, and bladder, with the bladder being the most common (79.2%). 7 Despite this, bladder BPGs are exceedingly rare, comprising less than 0.06% of all bladder neoplasms and 6% of BPGs. It may exhibit functionality or non-functionality (17%). 7 The functional one typically manifests with symptoms like hypertension, headaches, palpitations, and intermittent painless gross hematuria and it may lead to increased catecholamine levels, particularly during micturition, commonly known as ‘micturition attacks’.8,9 Conversely, not-functional BPG lacks readily apparent clinical symptoms. In our case, the patient reported the onset of hypertension 1 year before surgery and, in a postoperative interview, occasional post-micturition tachycardia episodes.
BPGs are detectable through ultrasound and CT imaging. 10 Ultrasound highlights hypoechoic lesions with increased blood flow in 60% of cases, while CT displays hyperdense, rounded lesions with arterial phase enhancement, peritumoral vessels, and occasional calcification. 11 magnetic resonance imaging (MRI), more sensitive than CT, provides excellent soft tissue contrast, depicting hyperintense tumors with diffusion restriction and a ‘salt and pepper’ appearance in larger cases. 12 In contrast to MRI, iodine131 metaiodobenzylguanidine scintigraphy has high specificity and could be used to help diagnose the tumor. 9 68Ga-DOTATATE PET showed a higher detection rate than 18F-FDG in the detection of paragangliomas. An integrated PET/MRI adds value in comparison to 18F-FDG PET but not much to 68Ga-DOTATATAE PET in identifying PPGL lesions. 13
The main treatment for localized BPG is surgery. It is recommended to undergo surgical resection, involving partial or complete cystectomy, along with pelvic lymph node dissection. 7
Partial cystectomy has been proposed as a favorable alternative in certain reports, 14 while others advocate for transurethral resection of the bladder tumor. 15 Beilan et al. reported that the primary treatment for patients with BPGs is partial cystectomy (68.9%), while other treatment alternatives for localized or locally advanced BPGs include transurethral resection of bladder tumor (19.8%) and radical cystectomy (11.3%).
Table 1 summarizes select studies referenced in our work. While surgical approaches vary, robotic removal of the lesion remains uncommon.
Different clinical presentations and surgical approaches may vary. Robotic removal of the lesion with concurrent partial cystectomy is not widely adopted.

In our case, we decided to proceed with a robotic-partial cystectomy due to the imaging and cystoscopic evidence of no compromise of the bladder mucosa and for all the advantages offered by the robotic surgeries. In the preoperative evaluation of the patient, a deliberate decision was made to forego a biopsy before surgery. This decision was influenced by the challenging nature of identifying the exact characteristics of the neoplasm, leading to uncertainty about the necessity of a biopsy. In addition, the patient expressed a strong preference for the complete removal of the lesion, given the uncertainty surrounding its nature. Irrespective of the surgical approach, it is advisable to use the preoperative adrenergic blockade to stabilize blood pressure when a functional lesion is going to be removed. Surgical removal resulted in a full recovery for the patient.
Following the surgical procedure, genetic investigations were conducted to assess the presence of alterations in genes commonly associated with similar medical conditions. 16 No genetic alterations were found on the NF1, RET, and VHL genes.
The postoperative program encompasses a 6-month CT scan and an endocrinological visit involving the assessment of metanephrines and normetanephrines levels.
After the endocrinological assessment, the patient was offered a PET scan with 68Ga-DOTATATE as an additional follow-up examination. It is worth mentioning that 68Ga-DOTATATE can be complemented by contrast-enhanced CT/MRI, as demonstrated by Carrasquillo et al. 17
Conclusion
BPG should be considered in the differential diagnosis of para-vesical masses. Even in the absence of symptoms, the physician collecting the medical history should meticulously investigate concurrent situations related to micturition. Robotic partial cystectomy may be the first-line surgical choice for removing abdominal masses without a clear cleavage plane with the bladder, providing significant advantages in surgical time, rapid recovery, and hospital discharge.
