Abstract
Introduction:
DNA damage repair genes are altered in 20–35% of metastatic castration-resistant prostate cancer (mCRPC). Poly-ADP (Adénosine Diphosphate)-ribose polymerase inhibitors (PARPi) showed significant activity for these selected tumors, especially with homologous recombination repair (HRR) deficiency. These alterations could also predict platinum sensitivity. Although carboplatin was inconclusive in unselected mCRPC, the literature suggests an anti-tumoral activity in mCRPC with HHR gene alterations. We aimed to assess the efficacy of carboplatin monotherapy in mCRPC patients with HRR deficiency.
Methods:
This prospective multicenter single-arm two-stage phase II addressed mCRPC men with HRR somatic and/or germline alterations, pretreated with ⩾2 taxane chemotherapy regimens and one androgen receptor pathway inhibitor. Prior PARPi treatment was allowed. Enrolled patients received intravenous carboplatin (AUC5) every 21 days for 6–9 cycles. The primary endpoint was the best response rate according to adapted PCWG3 guidelines: radiological response (RECIST 1.1 criteria) and/or biological response [⩾50% prostate-specific antigen (PSA) decline].
Results:
A total of 15 out of 16 enrolled patients started carboplatin treatment. Genomic alterations were identified for BRCA2 (n = 5), CDK12 (n = 3), ATM (n = 3) CHEK2 (n = 2), CHEK1 (n = 1), and BRCA1 (n = 1) genes. Objective response (partial biological response + stable radiological response) was achieved in one patient (6.7%), carrying a BRCA2 mutation and not pre-treated with PARPi; stable disease was observed for five patients (33.5%). Among seven patients (46.7%) with previous PARPi treatment, four patients (57.1%) had a stable disease. The median progression-free and overall survivals were 1.9 [95% confidence interval (95% CI), 1.8–9.5] and 8.6 months (95% CI, 4.3–19.5), respectively. The most common severe (grade 3–4) treatment-related toxicities were thrombocytopenia (66.7%), anemia (66.7%), and nausea (60%). Overall, 8 (53.3%) patients experienced a severe hematological event.
Conclusion:
The study was prematurely stopped as pre-planned considering the limited activity of carboplatin monotherapy in heavily pre-treated, HHR-deficient mCRPC patients. Larger experience is needed in mCRPC with BRCA alterations.
Trial registration:
NCT03652493, EudraCT ID number 2017-004764-35.
Introduction
In the area of personalized medicine, studies of metastatic prostate cancer (mPC) identified potentially actionable genomic aberrations. The most frequently altered and targetable pathway is loss-of-function alterations in DNA damage repair (DDR) genes, occurring in 20–25% of cases. 1 Among these, BRCA2 plays a critical role in the homologous recombination repair (HRR) of double-strand DNA breaks and its alteration is the most commonly observed in mPC, accounting for 6–12% of cases across studies. 1 Based on the concept of synthetic lethality, the poly-ADP-ribose polymerase inhibitors (PARPi) were successfully evaluated in these biomarker-selected metastatic castration-resistant prostate cancer (mCRPC) patients: in the phase III trial PROfound, olaparib significantly prolonged radiographic progression-free survival (rPFS) and overall survival (OS) compared with physician’s choice of enzalutamide or abiraterone (control) in mCRPC patients with alterations in HRR genes.2,3 These results led to the market approval of olaparib by the U.S. Food and Drug Administration for men with deleterious germline or somatic HRR gene-mutated mCRPC who have progressed following previous treatment with enzalutamide or abiraterone. Taking into account the variability of results among the altered genes, the European Medicines Agency (EMA) restricted the indications of olaparib to mCRPC patients with BRCA1/2 gene alterations.
Evidence suggests HRR pathway status is not only a predictive factor of PARPi efficacy but also predicts sensitivity to platinum-based chemotherapy in several tumoral types.4,5 Carboplatin efficacy has been already assessed in biomarker unselected mCRPC patients with low efficacy; similarly, a phase III trial with satraplatin, an oral platinum compound, failed to demonstrate a survival benefit.6,7 Thus, platinum-based chemotherapy is not a standard of care for mCRPC. Nevertheless, some case reports reported impressive responses in BRCA-mutated mCRPC and retrospective studies suggested clinical benefit in HRR-altered mCRPC compared with all comers mCRPC. Yet, to date, there have been no prospective dedicated studies.8–12
The PRO-CARBO study aimed to prospectively assess the efficacy of carboplatin chemotherapy in a selected population of mCRPC patients who presented HRR gene alterations.
Materials and methods
Study design
The PRO-CARBO trial was a prospective, multicenter single-arm, two-stage study assessing the efficacy of carboplatin as a single-agent therapy in heavily pretreated mCRPC patients with HRR gene alterations. It was conducted in three French cancer centers. The local ethics committee (Ref. 2018-53, Comité de protection des personnes Sud-Est I) and the French Health Authority (Ref. MEDAECNAT-2018-06-00011) approved the study protocol. All patients gave written informed consent before inclusion. This trial is registered as EUDRACT 2017-004764-35, Clinical trial NCT03652493. It was conducted in accordance with the Declaration of Helsinki and the International Conference on Harmonization/WHO Good Clinical Practice standards.
Patients
Eligible patients were men aged ⩾18 years with progressive mCRPC with adenocarcinoma histology and somatic pathogenic variants in a pre-specified list of 32 genes involved directly or indirectly in the HRR pathway (available upon request). The variants were interpreted according to the French Genetics and Cancer Group guidelines (https://recherche.unicancer.fr/fr/les-groupes-d-experts/groupe-genetique-et-cancer/). Only pathogenic or likely pathogenic variants were reported.
Progressive disease was defined as two consecutive increases of prostate-specific antigen (PSA) values within an interval of at least 1 week, and/or soft-tissue disease progression assessed by Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1), and/or bone disease progression assessed by apparition of two or more bone metastatic lesions on bone scans according to Prostate Working Group 3 criteria (PCWG3). Disease should have progressed during or after treatment by docetaxel for metastatic or non-metastatic CRPC and cabazitaxel and one novel hormone therapy (enzalutamide or abiraterone) for mCRPC, with no limited number of treatment lines. Prior treatment with PARPi was allowed. Other inclusion criteria were a performance status of 0–2; bilateral orchiectomy or ongoing androgen deprivation therapy with a gonadotropin-releasing hormone agonist or antagonist with a serum testosterone of 50 ng/dl or less at screening and adequate organ and bone marrow function, in particular hemoglobin >9 g/dl without blood transfusion in the 28 days before inclusion, and platelets >100,000/mm3.
Study treatment
Enrolled patients received monotherapy intravenous carboplatin area under the curve (AUC) 5 every 21 days for 6–9 cycles at the discretion of the investigator. 13 G-CSF, erythropoietin, anti-emetic, and bisphosphonates treatments may be used according to local practice guidelines. Other systemic anticancer treatments, including bone palliative radiation, were not allowed. Treatment should be discontinued in case of disease progression, unacceptable toxicities, investigator, or patient decision.
Assessments
Radiographic tumoral assessments by thoraco-abdomino-pelvic CT scan and whole-body radionuclide bone scan were performed at inclusion, thereafter every three cycles. Biological tests (including PSA measurement) and clinical assessments of safety were performed at screening and before each carboplatin cycle. Adverse events were reported using the National Cancer Institute Common Toxicity Criteria version 5.0.
Study objectives
The primary objective of this trial was to evaluate the antitumor activity of carboplatin in patients with mCRPC with alterations in HRR genes. As a primary endpoint, the carboplatin antitumor activity was assessed through the tumor response rate using adapted PCGW3 criteria, defined as the proportion of patients achieving a PSA decline of ⩾50% confirmed by a second PSA value ⩾3weeks later or/and a radiological objective response rate according to RECIST v1.1 criteria on CT scan evaluation, with no evidence of bone progression on bone scan per PCWG3 criteria. 14 Key secondary endpoints included duration of response, radiological progression-free survival (rPFS), OS, and treatment tolerance profile.
Statistical considerations
We used an optimal Simon’s two-stage phase II design to determine the sample size and decision rules. To assess the antitumor activity of carboplatin, we assumed a tumor response rate p < 20% as insufficient, and a p > 40% as demonstrating efficacy in this setting. With an alpha level of 5% and statistical power of 80%, 43 assessable patients were required, including 13 in the first stage. At interim analysis planned after the inclusion of 13 assessable patients, at least four patients with tumor response were required to pursue the inclusion of 30 additional assessable patients in the second stage. At final analysis, a minimum of 13 tumor responses in 43 assessable patients would allow us to conclude efficacy. The study was planned not to stop enrolment during the interim analysis.
The Kaplan–Meier method was used to estimate rPFS and OS distributions.
Results
Patient characteristics
From October 2018 to February 2021, a total of 16 patients were enrolled in the PRO-CARBO study, after which the planned interim analysis based on Simon’s two-stage design was performed. One patient dropped out of the study before starting treatment due to rapid deterioration of general condition (Figure 1). Baseline characteristics of the 15 assessable patients are presented in Table 1. Briefly, patients were heavily pretreated: all patients received both docetaxel (among them 26.7% in hormone-sensitive stage) and cabazitaxel chemotherapy, 11 (73.3%) received two or more previous androgen signaling inhibitor therapies, and 7 (46.7%) were already pre-treated with a PARPi. Two patients were treated with other DNA-damaging agents: one patient received mitoxanthrone and one patient received PSMA-lutetium. Five (33.3%) patients received palliative bone radiotherapy. Dealing with genomic alterations (Table 2), 5 (33.3%) tumors displayed a BRCA2 variant, 1 (6.7%) had a BRCA1 variant, and 3 (20%) had an ATM or CDK12 or CHEK1/2 variant. Three (20%) tumors (two tumors with BRCA2 variant and one tumor with ATM variant) had co-occurring DDR gene alterations.

CONSORT diagram of the PRO-CARBO study.
Baseline characteristics of patients (N = 15).
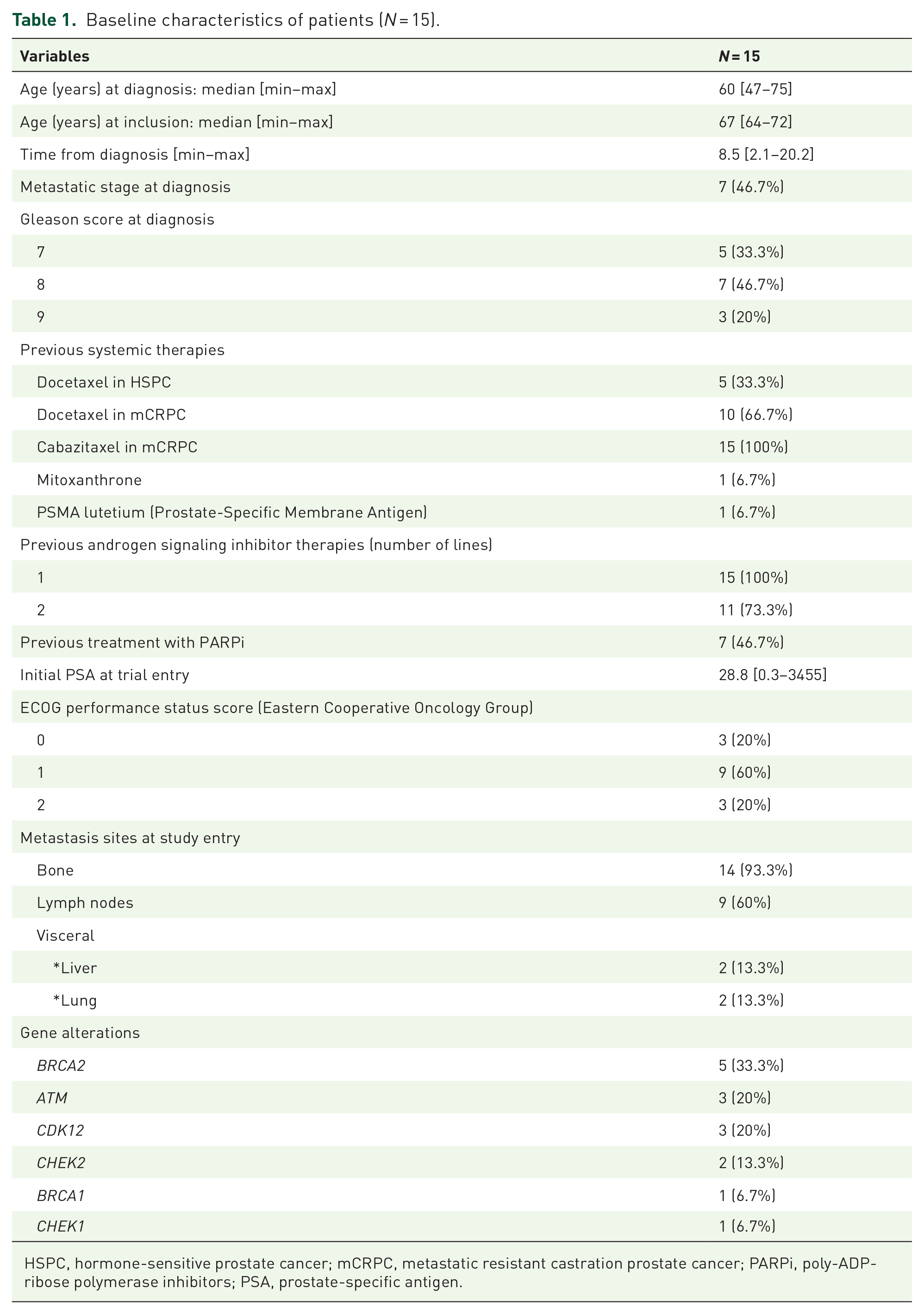
HSPC, hormone-sensitive prostate cancer; mCRPC, metastatic resistant castration prostate cancer; PARPi, poly-ADP-ribose polymerase inhibitors; PSA, prostate-specific antigen.
Genomic alterations of patients and best response.
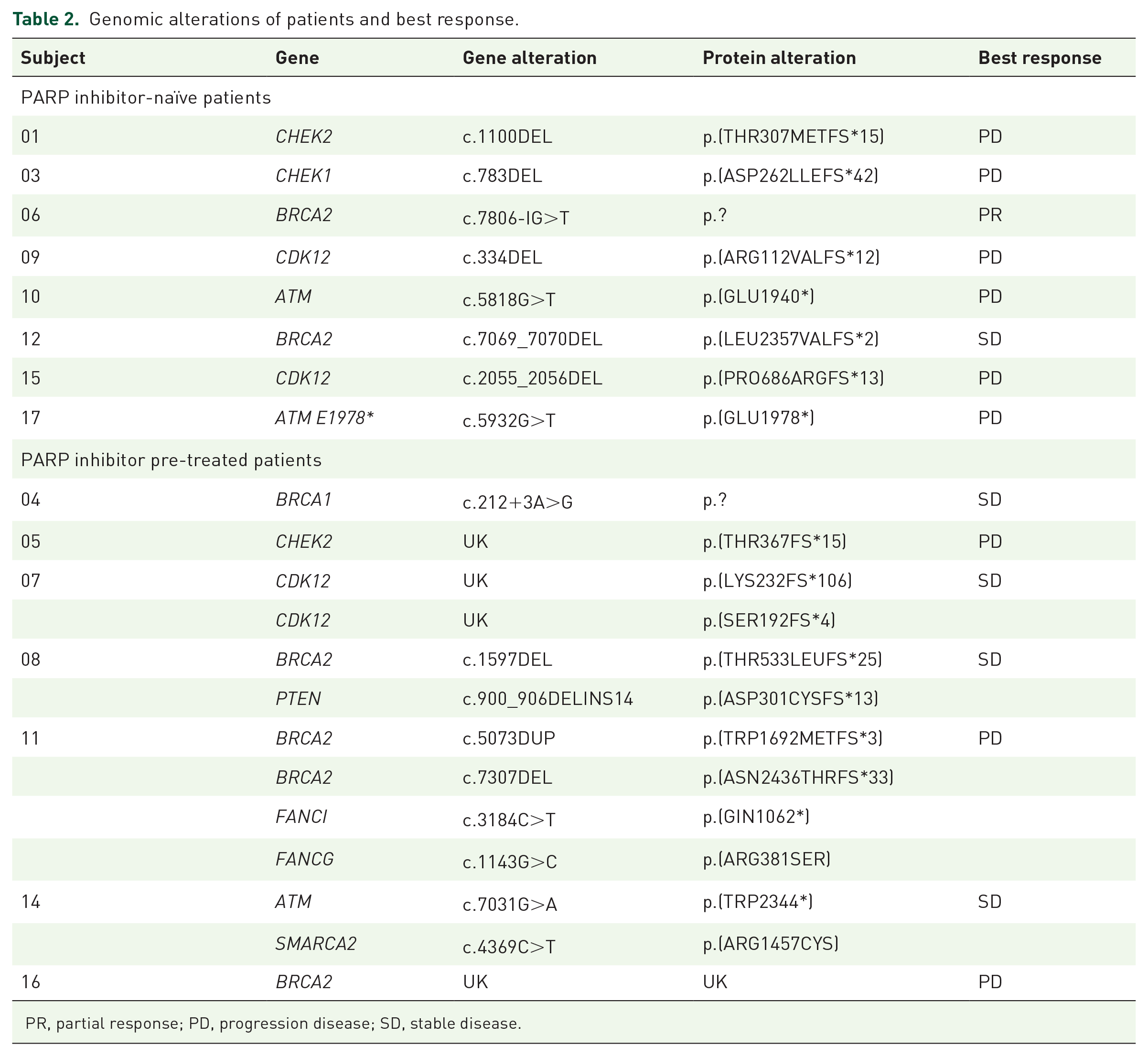
PR, partial response; PD, progression disease; SD, stable disease.
Treatment administration
All 15 patients received at least one cycle of carboplatin AUC 5, with a median of three delivered cycles [min 1–max 9]. Toxicities induced delays in carboplatin administration for 7 (46.7%) patients, and dose reduction for 4 (26.7%) patients. One (6.7%) patient stopped carboplatin for degradation of a general condition not related to carboplatin. The main reasons for permanent discontinuation were disease progression for 11 patients (73.3%), protocol ending for 2 patients (13.3%), toxicity for 1 patient (6.7%), and death for 1 patient (6.7%).
Antitumor activity
Among the first 13 assessable patients, only one observed partial tumor response (decrease PSA ⩾ 50% with stable disease on radiological exams), which was lower than the minimum number of responses required to pursue at second stage. The trial was therefore closed to inclusion after the results of the interim analysis for insufficient efficacy. However, during the interim analysis and up to the availability of the results, recruitment was ongoing and two additional patients were included with no further response. In total, one patient had a tumoral response, with a duration of response of 10 months, leading to a tumor response rate of 6.6%. Precisely, 5 (33.5%) patients observed stable disease, and 9 (60%) patients had progressive disease. Among the seven patients who received prior treatment with PARPi, carboplatin single-agent treatment led to stable disease for 4 (57.1%) patients and to disease progression for 3 (42.9%) patients (Table 2). Among the eight patients who did not receive PARPi, two (25%) patients had a partial response or stable disease, and six (75%) patients had disease progression. Disease responses by gene alterations were reported in Table 2. Median progression-free survival was 1.9 months [95% confidence interval (CI) 1.8–9.5] and median OS was 8.6 months [95% CI 4.3–19.5].
Safety profile
Overall, carboplatin demonstrated a predictable safety profile in this population heavily pre-treated as described in Table 3. Eight (53%) patients experienced a treatment-related grade 3–4 hematological event. The most common grade 3–4 toxicities observed were thrombocytopenia (66.7%), asthenia (66.7%), and nausea (60%). Four patients required at least one dose reduction because of grade ⩾ 2 thrombocytopenia and 7 (46.7%) patients required a carboplatin delay mainly because of hematological toxicity. One patient died further to a fall, concomitantly with a grade 2 thrombocytopenia related to treatment and one patient died on degradation of general condition, not related to treatment.
All-cause adverse events.

Discussion
To our knowledge, the PRO-CARBO trial is the first phase II study aiming to evaluate prospectively carboplatin monotherapy in a biomarker-selected population of HRR-deficient pre-treated mCRPC. Using tumor tissue analysis, biomarker selection consisted of a panel of 32 genes involved in the HRR pathway that were likely to be sensitive to PARPi and platinum-based chemotherapy. Unfortunately, our study failed to demonstrate sufficient carboplatin efficacy in this population, since only one tumor response (6.7%) was observed among 15 treated patients. Interestingly, this tumor response occurred in a patient with a BRCA2 alteration and was not pre-treated with a PARPi. The disappointing results of our study differed from prior publications, relative to retrospective studies or case reports.8–10,15–17 Cheng et al. 18 reported in ASCO GU 2020 Congress preliminary results of a pilot prospective phase II study assessing the efficacy of docetaxel and carboplatin for the treatment of patients with mCRPC containing bi-allelic inactivation of genes in the DDR genes. Patients should have progressed after any prior treatment, including prior docetaxel and/or PARPi. Seven out of eight (88%) patients with BRCA1, BRCA2, and ATM alterations and 10 out of 13 (77%) patients with other DDR-related genes including CDK12, CHD1, MRE11A, and PALB2 variants achieved a PSA decline of ⩾50%. Final data remain warranted but these results in favor of carboplatin efficacy in HRR defect mCRPC differ from our findings.
Several explanations could be discussed. First, we reported a high level of carboplatin dose reduction or delay due to toxicity. Toxicity was expected, notably hematological adverse events in relation to the patient’s frail profile: heavy pre-treatment with hematological toxicity, the presence of bone metastasis, and advanced age. Even if the risk toxicity was well known and manageable for physicians, the dose intensity of the treatment was not optimal.
Second, in our study, all patients had already received at least two lines of taxane-based chemotherapy and half of the patients were pre-treated with a PARPi. Recent data in ovarian cancer show that overlapping mechanisms of resistance exist between PARPi and platinum chemotherapy, leading to decreased efficacy of platinum chemotherapy after PARPi treatment.19,20 This phenomenon of cross-resistance is well known in mCRPC between taxanes or between different chemotherapy regimens and documented in preclinical and clinical studies.21–23 In our study, seven (46.7%) patients were pre-treated with PARPi mostly in clinical trials, of whom four (57.1%) patients had a BRCA1/2 mutation. We observed four (57.1%) patients with stable disease and three (42.9%) patients with disease progression. Therefore, the optimal treatment sequence is still a matter of debate. To answer to this question, the ongoing COBRA trial compares the efficacy of the sequence of carboplatin and olaparib in mCRPC presenting BRCA1, BRCA2, or PALB2 alterations (NCT04038502).
Finally, when the trial was designed, it was not completely understood which DDR alterations were useful predictive biomarkers for sensitivity to PARP inhibition. Recent results from trials with PARPi demonstrated that all proteins involved in the DDR pathways did not have the same predictive value. In the PROfound trial, patients were enrolled into two separate cohorts based on tumor genomics: those with alterations in the genes BRCA2, BRCA1, or ATM were eligible for cohort A and those with alterations in 12 other DDR-related genes (CDK12, CHEK1, CHEK2, FANCL, PALB2, PPP2R2A, RAD51B, RAD51C, RAD51D, RAD54C, BARD1, and BRIP1) were enrolled in cohort B. Biomarkers subgroups analyses showed that olaparib was less effective in cohort B as well as in ATM-deleted tumors, 24 explaining the decision of the EMA to restrict the approval of olaparib to patients with BRCA1/2-mutated mCRPC. Similar discordant results among DDR gene alterations in mCRPC were obtained with other PARPi.25–27 Fan et al. conducted a retrospective study on 23 patients with mCRPC with DDR alterations who received platinum-based chemotherapy. If the patients have better outcomes than the patients with all-comers tumors, results showed distinct responses to platinum chemotherapy according to the gene alterations. In particular, tumors with BRCA2 and/or ATM alterations showed superior outcomes compared to those with CDK12 defects. 9 Since a better comprehension of the biological process of DNA repair, other treatments were currently developed to target specifically other DDR alterations, such as CDK12 alterations and immune checkpoint inhibitors, or ATM alterations and ATR inhibitors.28,29 In our study, we selected patients with a large panel of genes involved directly or indirectly in the HRR pathway, and the unique tumor response was obtained in a BRCA2-mutated tumor. These results move to assess the efficacy of carboplatin in a more restrictive list of HRR biomarkers known to be sensitive to PARPi and/or in tumors presenting a functional defect of this pathway with functional tests or signatures as developed in ovarian cancer. 30 Final results of Cheng et al.’s 18 study that included an enriched population of mCRPC with BRCA1/2 alterations to evaluate the combination of docetaxel plus carboplatin are warranted (NCT02598895).
To conclude, the negative results of the PRO-CARBO study put in question the best therapeutic sequence between PARPi, taxanes, and platinum-based chemotherapy, and to better select patients with restricted biomarkers known to be sensitive to PARPi. We await the conclusions of ongoing trials regarding the optimal combinations and sequence of treatments.
Footnotes
Acknowledgements
We wish to thank the patients who participated in this study, their families, their referring physicians, and the clinical research teams. The Northwest Data Center (CTD-CNO) is acknowledged for managing the data. It is supported by grants from the French National League Against Cancer (LNC) and the French National Cancer Institute (INCa).
