Abstract
Background:
Treatment outcomes in intermediate-risk prostate cancer (PCa) may be impaired by adverse pathology misclassification including tumor upgrading and upstaging. Clinical predictors of disease progression need to be improved in this category of patients.
Objectives:
To identify PCa prognostic factors to define prognostic groups in intermediate-risk patients treated with robot-assisted radical prostatectomy (RARP).
Design:
Data from 1143 patients undergoing RARP from January 2013 to October 2020 were collected: 901 subjects had available follow-up, of whom 479 were at intermediate risk.
Methods:
PCa progression was defined as biochemical recurrence and/or local recurrence and/or distant metastases. Study endpoints were evaluated by statistical methods including Cox’s proportional hazards, Kaplan–Meyer survival curves, and binomial and multinomial logistic regression models.
Results:
After a median (interquartile range) of 35 months (15–57 months), 84 patients (17.5%) had disease progression, which was independently predicted by the percentage of biopsy-positive cores ⩾ 50% and the International Society of Urological Pathology (ISUP) grade group 3 for clinical factors and by ISUP > 2, positive surgical margins and pelvic lymph node invasion for pathological features. Patients were classified into clinical and pathological groups as favorable, unfavorable (one prognostic factor), and adverse (more than one prognostic factor). The risk of PCa progression increased with worsening prognosis through groups. A significant positive association was found between the two groups; consequently, as clinical prognosis worsened, the risk of detecting unfavorable and adverse pathological prognostic clusters increased in both unadjusted and adjusted models.
Conclusion:
The study identified factors predicting disease progression that allowed the computation of highly correlated prognostic groups. As the prognosis worsened, the risk of PCa progression increased. Intermediate-risk PCa needs more prognostic stratification for appropriate management.
Plain language summary
Prostate cancer is a serious health concern in men, and those with intermediate-risk prostate cancer may experience disease progression. Urologists use various methods to predict the risk of progression in these patients. However, sometimes the predictions are not accurate. Therefore, researchers conducted a study to identify factors that could help predict disease progression in patients with intermediate-risk prostate cancer who underwent robot-assisted surgery. This study on 479 patients found that a percentage of biopsy-positive cores ⩾ 50% and the International Society of Urological Pathology (ISUP) grade group 3 were predictive factors of disease progression. Additionally, factors like ISUP > 2, positive surgical margins, and pelvic lymph node invasion also predicted disease progression. Patients were classified into three groups based on their clinical and pathological features: favorable, unfavorable (one negative prognostic factor), and adverse (more than one negative prognostic factor). The risk of prostate cancer progression increased as the prognosis worsened through these groups. The study concluded that a more accurate stratification of intermediate-risk prostate cancer patients is needed to manage the disease effectively.
Keywords
Introduction
Prostate cancer (PCa) diagnosis has become an epidemic issue for the extensive opportunistic prostate-specific antigen (PSA) screening in the aging male and its prognosis is assessed by classification systems that aim at grouping homogenous sets of patients.1–3 The European Association of Urology (EAU) and the National Cancer Comprehensive National Network (NCCN) are the two main reference systems, which are not equivalent, although referring to D’Amico’s risk classes.1,2 As an example, palpable tumors involving both lobes of the prostate (cT2c) are included in the intermediate risk class for NCCN but not for EAU that considers such cancers as belonging to the high-risk class; nevertheless, both systems consider PSA < 10/ng/mL, percentage of biopsy positive cores (BPC) less than 50% and tumor grade group <3, according to the International Society of Urologic Pathology (ISUP) classification, as features indicating a favorable prognostic subgroup of the intermediate risk class, which is largely non-homogenous.1,2 Clinical treatment decisions on intermediate-risk patients include options that may vary from delayed management, which include active surveillance (AS) and watchful waiting, to active treatments, which consider radical prostatectomy and radiation therapy (RT) with the former being more frequently performed by the robot-assisted radical prostatectomy (RARP) in tertiary high-volume centers.1–3 However, treatment outcomes may be impaired by adverse pathology misclassification including tumor upgrading and upstaging with positive surgical margins, as well as by pelvic lymph node invasion (PLNI).1–5 Nevertheless, while awaiting the introduction of prognostic molecular markers, standard clinical predictors of disease progression need to be improved in intermediate-risk PCa to reduce treatment-related regret, which is becoming an issue in everyday clinical practice for the urologist as well as for radiation oncologists. 6 The primary endpoint of the study was to identify clinical and pathological prognostic factors of disease progression after RARP to identify relative prognostic groups and assess their impact on PCa progression. The secondary endpoint was to evaluate associations between the two groups.
Materials and methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement (Supplemental Material 1). 7
Data collection, patient selection, and evaluation of parameters
The Institutional Review Board approved the study, and written informed consent was obtained from all subjects. Data were collected prospectively but evaluated retrospectively. Patients were classified into risk classes, according to EAU guidelines. 1 In a period ranging from January 2013 to October 2020, RARP was performed in 1143 patients but follow-up was available in 901 subjects of whom 479 were at intermediate risk. In each case, PSA; ng/mL, age (years), body mass index (BMI; kg/m2), prostate volume (PV; mL), percentage of BPC, and PSA density (PSAD; ng/mL/cc) were evaluated. PV (mL) was calculated by transrectal ultrasound (TRUS) standard methods. Biopsies performed elsewhere were assessed for the number of cores taken, tumor grade, and PV, which was measured by the trans-rectal approach. Clinical staging was assessed by the 2017 version of the tumor, node, metastasis (TNM) system (eighth edition) with the clinical T stage only referring to digital rectal exam findings. Preoperative physical status was evaluated by the American Society of Anesthesiologists (ASA) system. 8 RARP was performed by experienced surgeons. Extended pelvic lymph dissection (ePLND) was performed according to guidelines.1,2 Dissected lymph nodes were submitted in separate packages according to a standard anatomical template including external iliac, internal iliac plus obturator, Marcille’s common iliac, and Cloquet’s nodal stations, bilaterally. 9 Specimens including prostate and eventually dissected lymph nodes were placed into formalin and evaluated by the dedicated pathologists. Prostates were weighted and tumors were graded according to the ISUP system.1,2 Tumor quantitation was assessed as tumor load (TL), which was defined as the percentage of prostate involved by cancer; specifically, our dedicated pathologists assessed tumor quantitation by visual estimation of all the glass slides after all microscopically identifiable foci of carcinoma have been circled with a marked pen, according to ISUP recommendations. 10 Surgical margins (SM) were stated as positive when cancer invaded the inked surface of the specimen; furthermore, they were classified as focal and non-focal according to the linear extent, accordingly. 1 Removed lymph nodes were counted and assessed for cancer invasion. Surgical specimens were staged by the 2017 version of the TNM system (eighth edition), as well.1,2 Perioperative outcomes were evaluated for operating time, estimated intraoperative blood loss, nerve-sparing surgery, high- and low-volume surgeons, length of hospital stay, and hospital readmission after discharge. Postoperative complications, which were monitored for a period of at least 3 months, were coded according to the Clavien-Dindo system.1,2,9 Although patients were followed up according to guidelines, decisions of further treatments after surgery or at disease progression were taken in a multidisciplinary setting including urologists, radiation oncologists, and oncologists to optimize recommendations with patients’ issues.1–3
Evaluation of clinical and pathological prognostic factors for primary and secondary endpoints
To verify our hypotheses, we investigated clinical and pathological factors predicting PCa progression. After selecting independent factors, we classified patients into clinical and pathological prognostic groups. In each group, the prognosis was codified at two and three levels with the former including the absence or presence of one or more prognostic factors (favorable and unfavorable/adverse prognostic group, respectively) and the latter including no factors (favorable prognostic group), one factor (unfavorable prognostic group), and more than one factor (adverse prognostic group). The primary endpoint was to assess the impact of prognostic groups on PCa progression, which was defined as biochemical recurrence and/or local recurrence and/or distant metastases. The secondary endpoint was to assess associations between the two groups. Accordingly, clinical and pathological parameters were evaluated for study endpoints but not perioperative factors.
Statistical methods
Continuous variables were measured for medians and interquartile ranges (IQRs). Categorical factors were assessed for frequencies and rates (percentages). The length of time between surgery and the clinical outcome of interest (PCa progression) or the last follow-up was measured as time to event occurrence. Univariate and multivariate Cox proportional hazards models estimated the association of clinical and pathological factors with the risk of PCa progression; furthermore, hazard ratios and relative 95% confidence intervals (CIs) were evaluated. Clinical factors were evaluated as age, BMI, PV, PSA 10–20 versus PSA < 10 ng/mL, PSAD ⩾ 0.15 versus < 0.15 ng/mL/mL, BPC ⩾ 50% versus < 50%, ISUP 3 versus ISUP 1–2, while pathological factors as prostate weight, TL, ISUP 2 and > 2 versus ISUP 1, pT3a and pT3b versus pT2, R1 versus R0, and pN1 versus pNx/0. The impact of prognostic groups on the median time to PCa progression was assessed by the Kaplan–Meyer method (univariate analysis) and differences between groups by the Mantel-Cox log-rank test; thus, survival curves of PCa progression stratified by groups were generated. Associations with clinical and pathological prognostic groups were assessed by the binomial logistic regression method (univariate and multivariate analyses) for the two-level evaluated model and by the multinomial logistic method (univariate and multivariate analyses) for the three-level coded model. Odds ratios (OR) and relative 95% CIs were computed. The software used to run the analysis was IBM-SPSS version 26 (Armonk, NY:IBM Corp). All tests were two-sided with p < 0.05 considered to indicate statistical significance.
Results
Demography of the EAU intermediate PCa risk population
Population demography including 479 patients is reported in Table 1. Clinical disease factors were classified according to EAU guidelines. Patients were more frequently classified as ASA score 2 (82.5%) and less frequently as ASA score 1 (8.6%). In the surgical specimen, unfavorable tumor grade (ISUP > 2) was detected in 230 cases (48%) and adverse tumor stage including extracapsular extension (49 subjects) or seminal vesicle invasion (SVI) (45 cases) in 19.6% of patients. Overall, 105 patients (24.9%) had SM involved by cancer. Extended pelvic lymph node dissection was performed in 328 cases (68.5%) with PLNI in 22 (6.7%). The median (IQR) follow-up was 35 months (15–57 months). Adjuvant androgen deprivation therapy was given in 65 cases (13.6%) and RT was delivered in 75 (15.7%) subjects with salvage finality in 41 (8.6%). Deaths occurred in seven patients (1.5%) of whom two related to PCa progression (2.4%).
Demography of 479 PCa patients classified as intermediate-risk class according to the EAU prognostic system.

Continuous variables are reported as medians (IQR, interquartile ranges) and categorical factors as frequencies (percentages).
ASA, American Society of Anesthesiologists; BMI, body mass index; BPC, biopsy positive cores; EAU, European Association of Urology; ISUP, International Society of Urological Pathology; PCa, prostate cancer; PSA, prostate-specific antigen; PSAD, prostate-specific antigen density; PV, prostate volume.
Assessing clinical and pathological prognostic factors of PCa progression
As shown in Table 2, disease progression occurred in 84 patients (17.5%) who were more likely to present clinically with high tumor load and ISUP grade group 3 as well as to have unfavorable pathology features including adverse tumor grade (ISUP > 2) and stage (non-organ confined disease), positive SM and PLNI in the surgical specimens. On multivariate analysis, PCa progression was predicted by BPC ⩾ 50% and ISUP grade group 3 for clinical factors and by adverse tumor grade (ISUP > 2), positive SM, and PLNI for pathological features. Notably, PSA 10–20 ng/mL, clinical and pathological stage, and ISUP grade group 2 did not show any independent association.
Risk of PCa progression by clinical and pathological models in 479 EAU intermediate-risk patients treated with RARP.

Continuous variables are reported as medians (IQR, interquartile ranges) and categorical factors as frequencies (percentages); see materials and methods for abbreviations.
Evaluated by Wald’s forward method.
ASA, American Society of Anesthesiologists; BMI, body mass index; BPC, biopsy positive cores; CI, confidence interval; ECE, Extracapsular extension; EAU, European Association of Urology; HR, hazard ratio; ISUP, International Society of Urological Pathology; PCa, prostate cancer; PSA, prostate-specific antigen; PSAD, prostate-specific antigen density; PV, prostate volume; PW, prostate weight; RARP, robot-assisted radical prostatectomy; SVI, Seminal vesicle invasion; TL, tumor load.
Impact of clinical and pathological prognostic groups for predicting disease progression
Patients were then classified according to the absence or presence of clinical (BPC ⩾ 50%, ISUP grade group 3) and pathological (ISUP grade group > 2, R1, pN1) prognostic factors. Consequently, prognostic risk groups were coded as favorable versus unfavorable/adverse and favorable versus unfavorable versus adverse. Favorable cases did not show any adverse prognostic factor, unfavorable cases had at least one prognostic factor, and adverse subjects presented with more than one prognostic factor. The impact of clinical and pathological prognostic groups on PCa progression is detailed in Table 3, which shows that both unadjusted and adjusted stratified models predicted disease progression. Accordingly, the risk of PCa progression increased with worsening prognosis across groups. As a result, median (95% CI) PCa survival curves were progressively negative across groups, as shown in Figures 1 to 4, which provide detailed information on Kaplan–Meyer survival curves.
Unadjusted and adjusted models predicting PCa progression by clinical and pathological prognostic groups in 479 intermediate EAU risk patients treated with robot-assisted radical prostatectomy.

CI, confidence interval; EAU, European Association of Urology; HR, hazard ratio; PCa, prostate cancer; (*), models adjusted for remaining clinical factors; (**), models adjusted for remaining pathological factors; see also Table 2.
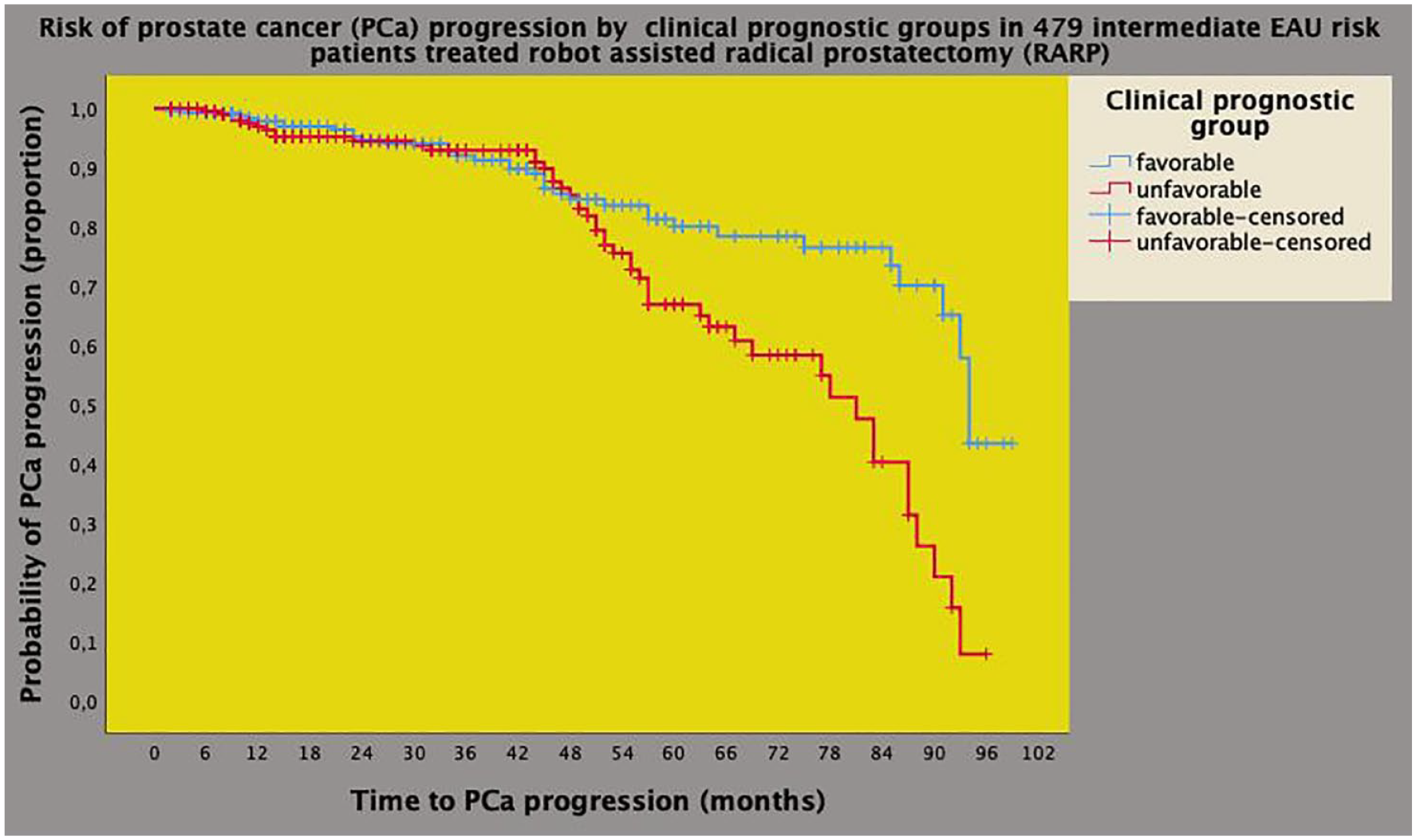
Kaplan–Meyer survival curves of PCa progression stratified by clinical prognostic groups in 479 intermediate-risk patients classified according to the EAU system. The median survival time of PCa progression was longer for the favorable prognostic group (94 months; 95% CI: 92.4–95.5 months) when compared with the unfavorable/adverse prognostic one (81 months; 95% CI: 69.9–92.3) and the difference was significant (Mantel-Cox log-rank test: p = 0.001). See also materials, methods, and results.
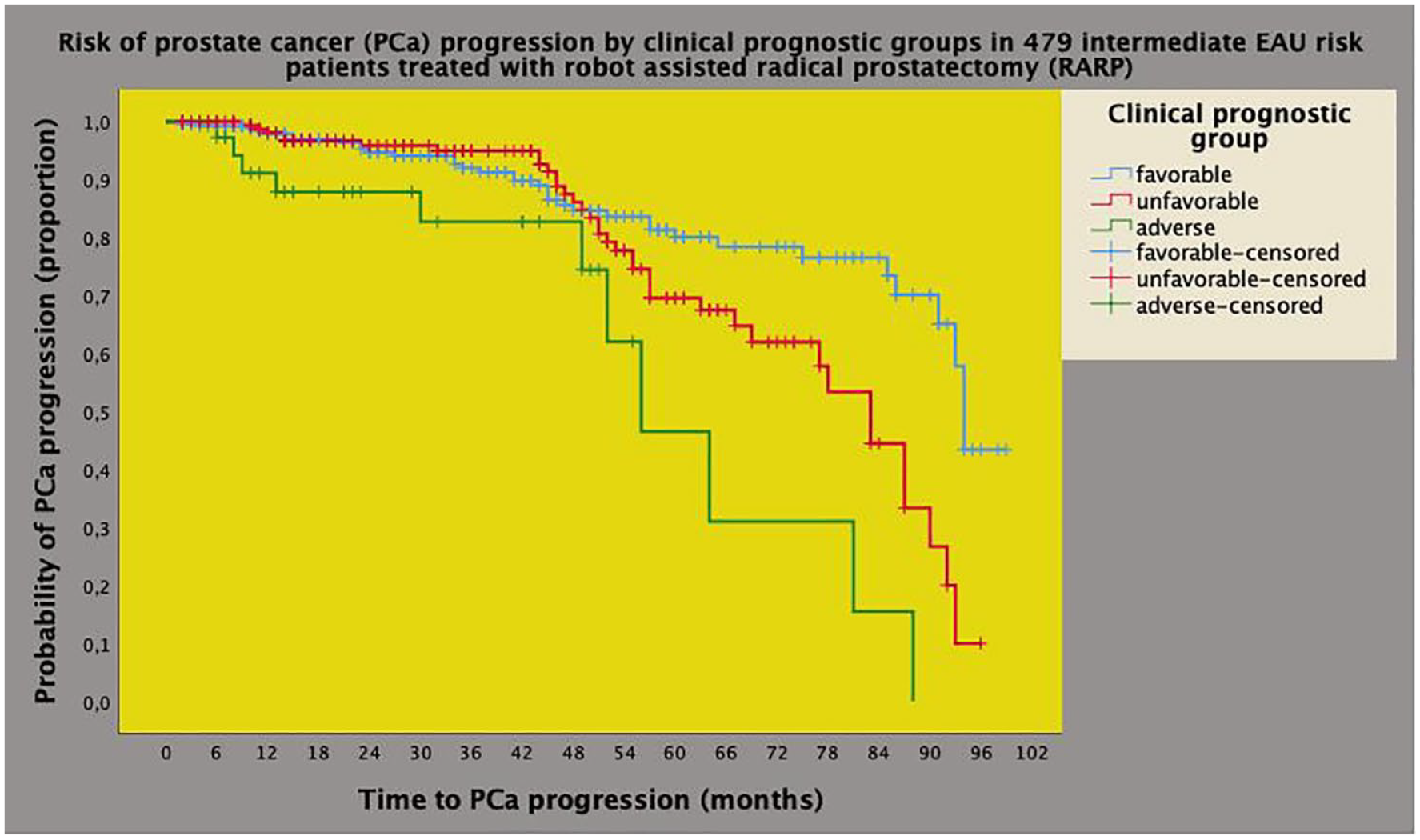
Univariate analysis of Kaplan–Meyer survival curves of PCa progression by clinical prognostic groups in 479 intermediate-risk cases classified according to the EAU system. The median survival time of PCa progression was longer for the favorable prognostic group (94 months; 95% CI: 92.4–95.5 months) when compared with the unfavorable group (83.4 months; 95% CI: 75.3–90.6 months) and with the adverse group (56 months; 95% CI: 43.0–68.9 months), and the difference was significant (Mantel-Cox log-rank test: p < 0.001). See also materials, methods, and results.

Univariate analysis of Kaplan–Meyer survival curves of PCa progression stratified by pathological prognostic groups in 479 intermediate-risk patients classified according to the EAU system. The median survival time of PCa progression was longer for the favorable prognostic group (94 months; 95% CI: 92.6–95.3 months) when compared with the unfavorable/adverse prognostic one (81 months; 95% CI: 69.9–92.3) and the difference was significant (Mantel-Cox log-rank test: p = 0.001). See also materials, methods, and results.
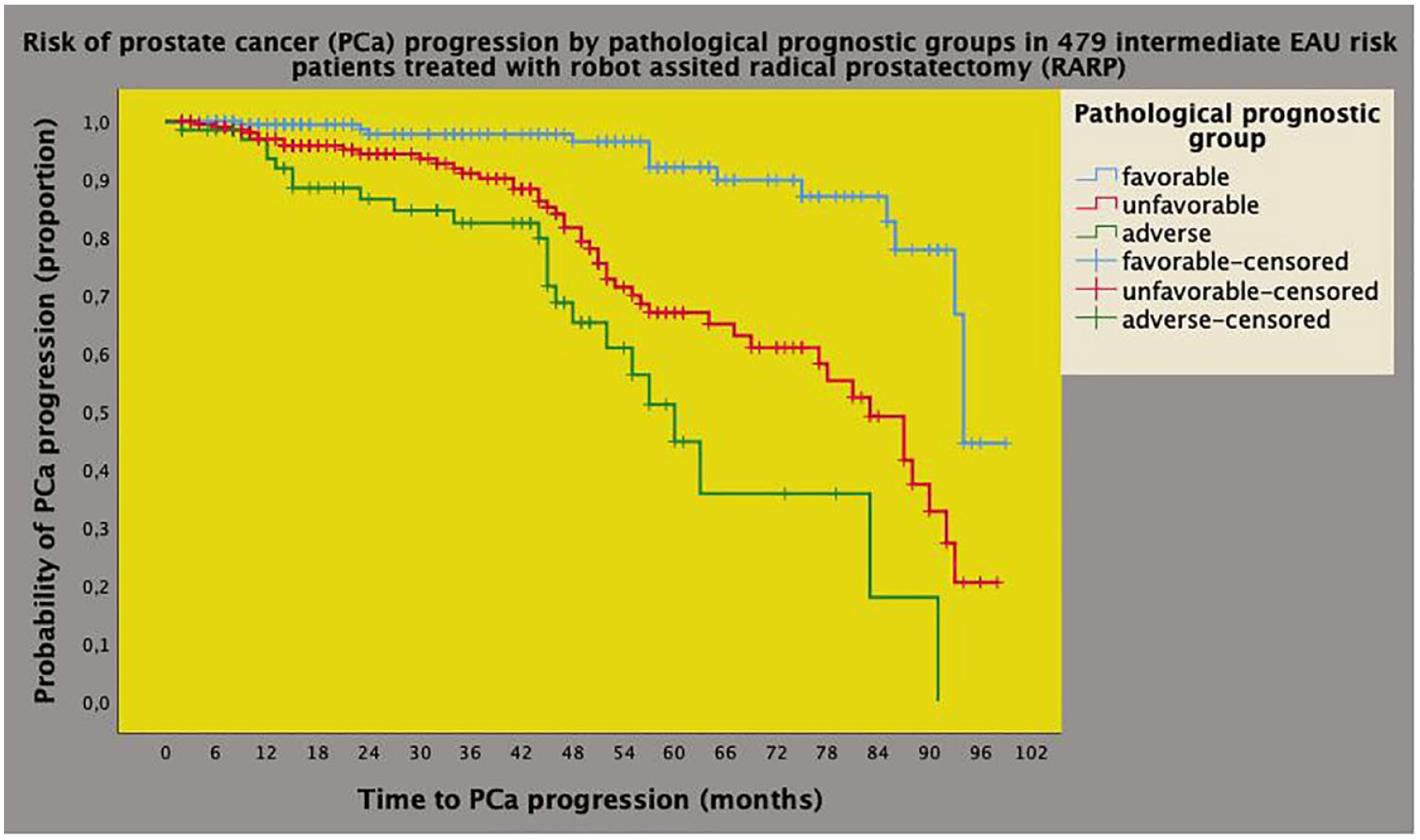
Univariate analysis of Kaplan–Meyer survival curves of PCa progression by clinical prognostic groups in 479 intermediate-risk cases classified according to the EAU system. The median survival time of PCa progression was longer for the favorable prognostic group (94 months; 95% CI: 92.6–95.3 months) when compared with the unfavorable group (83 months; 95% CI: 73.9–92.0 months) and with the adverse group (60 months; 95% CI: 51.3–68.6 months), with the difference being significant (Mantel-Cox log-rank test: p < 0.001). See also materials, methods, and results.
The positive association between clinical and pathological prognostic groups
As shown in Tables 4 and 5, a positive association was found between clinical and pathological prognostic groups that were predicted by the former in both unadjusted and adjusted models. Therefore, as clinical prognosis worsened, the risk of detecting unfavorable and adverse pathologic prognostic clusters increased in both unadjusted and adjusted models, as shown in the reference table.
Unadjusted and adjusted models predicting unfavorable/adverse PCa pathology by clinical prognostic groups in 479 intermediate EAU risk patients treated with robot-assisted radical prostatectomy.
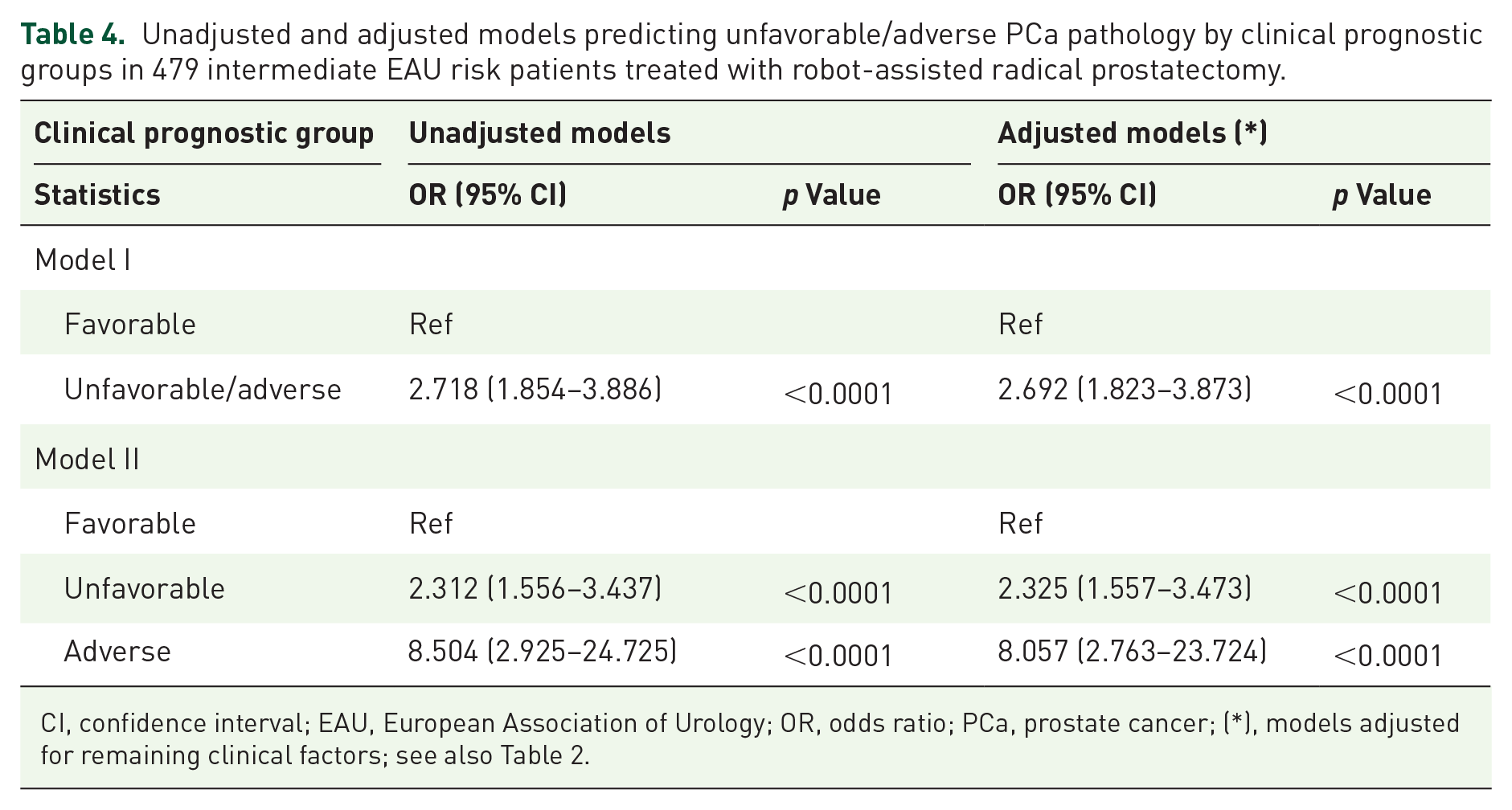
CI, confidence interval; EAU, European Association of Urology; OR, odds ratio; PCa, prostate cancer; (*), models adjusted for remaining clinical factors; see also Table 2.
Unadjusted and adjusted models predicting unfavorable and adverse PCa compared with favorable pathology by clinical prognostic groups in 479 intermediate EAU risk patients treated with robot-assisted radical prostatectomy.

Models adjusted for remaining clinical factors; see also Table 2.
CI, confidence interval; EAU, European Association of Urology; OR, odds ratio; PCa, prostate cancer.
Discussion
In PCa’s natural history, disease recurrence and progression after primary surgery are closely related to adverse pathological outcomes; therefore, tumor misclassification will finally impact both PCa overall and specific mortality.1–3 In particular, unfavorable tumor grade in the surgical specimen is the main pathological factor affecting PCa prognosis; consequently, prognosis worsens as the tumor grade group increases; interestingly, although ISUP grade group 2 out of 5 has a very good prognosis with rare metastases, it has not been assessed within clinical risk groups.1–3,11,12 Although adverse tumor pathology indicates a negative prognosis, its impact is not the same along risk groups for being more favorable for the low-risk category when compared with the intermediate- and high-risk class. 13 According to the Cambridge Prognostic Group (CPG) system, the 10-year risk of dying for PCa is 1.2% for low-risk disease (CPG 1) and 2.3% for favorable intermediate-risk PCa (CPG 2), which includes ISUP grade group 2 or PSA 10–20 ng/mL and stages T1–T2; moreover, in untreated patients, these rates increase up to 4.2% for the former and up to 4.7 for the latter. 14 A study has evaluated oncological outcomes after surgery comparing low and favorable intermediate-risk patients showing that adverse pathology was observed at a threefold higher rate in the latter when compared with the former which showed better overall survival. 14 Interestingly, an extensive literature review on the subject has recently confirmed that unfavorable pathology is an issue involving up to 30% of cases and for a negative impact on disease progression, which may be predicted by short PSA doubling time (PSA-DT) and a high tumor grade after surgery. 15 Furthermore, a large European study has shown that PSA-DT less than 2 years and pathology ISUP greater than 3 were factors identifying a high-risk group for metastatic progression and disease-specific mortality; however, it included ISUP grade group 3 in the low-risk category and EAU BCR groups need more assessments. 16 A multicenter study identified 10.6% of operated intermediate-risk patients as associating with favorable pathology including ISUP grade group 1 and organ-confined disease with better oncological results than unfavorable cases; however, it suffered several limitations including short follow-up and lack of central pathology revision. 17 In our study, we identified clinical and pathological factors of disease progression, which were grouped to compute relative risk groups stratified at two and three levels, respectively. As the number of prognostic factors increased, the risk of PCa progression increased; therefore, the prognosis worsened along unfavorable and adverse groups. Kaplan–Meyer curves diverge visibly after a period of 36–48 months; this may be due to a latency time of action of multiple prognostic factors; however, this hypothesis needs to be verified by controlled studies. Surprisingly, PSA levels at clinical and pathological tumor stages did not have any independent association for being subordinate to predictive factors. Interestingly, pathology ISUP grade group 2 showed no association with PCa progression, thus confirming non-aggressive biology as the ISUP reference group.
In operated low- and intermediate-risk PCa, a large European multi-center study evaluated the impact of clinical criteria in predicting the risk of adverse pathology including ISUP > 2, extra-prostatic extension (ECE, SVI), and PLNI. It demonstrated that clinical factors, including PSA, PSAD, ISUP, and cT, were independent predictors of unfavorable pathology; however, it was limited by inclusion criteria and lack of centralized pathology review. 4 Interestingly, a retrospective study demonstrated that 25% of specimens had adverse pathology in favorable intermediate-risk cases presenting as ISUP grade 2, PSA < 20 ng/mL, and no more than two biopsy-positive cores; however, it suffered with low-risk cases with low-risk cases. 18 The results of our study demonstrated significant associations between clinical and pathological prognostic factors. Therefore, with a worsening clinical prognosis, the risk of detecting unfavorable and adverse prognostic groups in the surgical specimen increased. So far, in EAU intermediate-risk PCa, BPC < 50% and ISUP grade group 1/2 indicate tumors that are less likely to have adverse pathology, including unfavorable tumor grade and stage, positive SM, and PLNI; furthermore, these cancers are less likely to undergo disease progression. In the surgical specimen, the detection of positive SM is related to PCa biology and less likely to the surgeon volume.19–22
There are several areas of controversy in the management of intermediate-risk patients when they are counseled for staging and treatment options; accordingly, AS is one of the most critical for increasing rates in North America, particularly for elderly patients with favorable intermediate PCa risk, with no difference in oncological and survival outcomes between those classified as favorable intermediate-risk either because of the PSA or because of Gleason Score 23 ; moreover, it has been demonstrated to be safer for the favorable class than the unfavorable group, with the latter showing significant higher PCa-specific deaths than other active treatments, including surgery and RT. 24 However, adverse tumor misclassification is an issue when monitoring low and favorable intermediate-risk patients for the risk of delayed treatment.1–3,14 Therefore, although actual rates of AS for patients with favorable intermediate-risk disease are increasing, more follow-up and research studies are needed. 24 Further controversy is fueled by inclusive criteria, which are more restrictive for EAU than for NCCN.1,2 Thus, additional clinical classification parameters are needed to optimize the monitoring management of low- and intermediate-risk patients. From this perspective, our results give important information for clinicians (urologists and radiation oncologists) when counseling intermediate-risk patients. We identified clinical and pathological prognostic groups, which allowed us to stratify patients before and after surgery. Interestingly, pathological prognostic groups were predicted by the clinical ones. As such, unfavorable/adverse clinical prognostic group patients need extensive counseling and further decisions on clinical staging, such as PET-PSMA, to provide appropriate optional treatments. For example, the radiation oncologist may decide to extend radiation to the pelvis, while the urologist can plan an extended template for PLND to produce a more accurate anatomical staging.
Some explanations are needed to interpret the results of our study showing that adverse tumor features included in the prognostic groups result in an unfavorable outcome on PCa progression. Theoretically, patients with clinical negative prognostic features are more likely to have undifferentiated cancers with a high-density growth pattern associated with non-organ confined disease and spread to the pelvic lymph nodes, due to the long exposure to genetic mutation dynamics and immune system impairment. As a result, patients with these features are more likely to have aggressive cancers, which are closely related to high-risk prognosis compared with intermediate-risk disease; however, these hypotheses need to be verified by controlled studies.
Our study has limitations. It was retrospective and single-center; mpMRI and molecular and/or genetic tests were not evaluated they were not available in all cases. We did not evaluate overall and cancer-specific survival because of the limited number of such events, and we did not evaluate the percentage of cancer involving each biopsy core for not being available in all cases. Our population was made of white Caucasian men, which is a limit as it did not permit us to consider race as a testable variable in a setting of active treatment; the role of race has been in fact already an object of debate for conservative management. 25 Finally, the percentage of Gleason pattern 4 was not evaluated in biopsy-positive cores. However, our study also has several strengths. The primary outcome was assessed at disease progression, which is a stronger endpoint than biochemical recurrence alone. Procedures were performed by both low- and high-volume surgeons who did not bias staging results, thus reflecting real-world practice in tertiary referral centers. The length of follow-up was appropriate for evaluating the primary endpoint. Finally, all surgical specimens were evaluated by our dedicated pathologists.
Conclusion
In EAU intermediate-risk PCa, the study identified predictive factors for disease progression that allowed the computation of stratified and highly correlated clinical and pathological prognostic groups. As the prognosis worsened, the risk of PCa progression increased. As a result, favorable groups were less likely to have an adverse pathological stage and to experience disease progression. EAU intermediate-risk PCa needs more prognostic stratification to plan more appropriate management.
Supplemental Material
sj-docx-1-tau-10.1177_17562872241229260 – Supplemental material for The impact of prognostic group classification on prostate cancer progression in intermediate-risk patients according to the European Association of Urology system: results in 479 patients treated with robot-assisted radical prostatectomy at a single tertiary referral center
Supplemental material, sj-docx-1-tau-10.1177_17562872241229260 for The impact of prognostic group classification on prostate cancer progression in intermediate-risk patients according to the European Association of Urology system: results in 479 patients treated with robot-assisted radical prostatectomy at a single tertiary referral center by Antonio Benito Porcaro, Alberto Bianchi, Andrea Panunzio, Sebastian Gallina, Alessandro Tafuri, Emanuele Serafin, Rossella Orlando, Giovanni Mazzucato, Stefano Vidiri, Damiano D’Aietti, Francesca Montanaro, Giulia Marafioti Patuzzo, Francesco Artoni, Alberto Baielli, Francesco Ditonno, Riccardo Rizzetto, Alessandro Veccia, Alessandra Gozzo, Vincenzo De Marco, Matteo Brunelli, Maria Angela Cerruto and Alessandro Antonelli in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
