Abstract
Background:
The leakage of urine during sexual arousal, known as climacturia, is an under-recognized clinical condition often overshadowed by erectile dysfunction in men who have undergone radical prostatectomy.
Objectives:
This study aims to determine and evaluate the role of the Mini-Jupette technique and its alternatives in the treatment of climacturia.
Data sources and methods:
We conducted a systematic review following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines for systematic reviews. We searched Medline via PubMed, Scopus, and the Cochrane Library databases until October 2022.
Results:
We included seven studies involving 120 patients with climacturia. Different types of grafts were used, ranging from synthetic mesh to autologous grafts. In all seven studies, the use of the Adrianne Mini-Jupette (AMJ) and its alternatives showed a high percentage of improvement in climacturia, with reported complete resolution ranging from 65% to 93%. Regarding postoperative complications, one study reported the highest sling explantation rate at 11% (4/38), while other studies reported complications ranging from subjective symptoms such as dysuria and perineal pain to the need for subsequent artificial urinary sphincter placement.
Conclusion:
The AMJ sling and its variations are low-cost, time-efficient, and relatively safe procedures with high patient satisfaction rates among those treated for climacturia.
Keywords
Introduction
Prostate cancer (PCa), the second most common solid tumor in men and the fifth leading cause of cancer mortality, poses a significant burden on healthcare systems and patients worldwide. This burden is not only due to the need for radical treatment, either surgical or conservative, but also due to the relatively high rate of complications following different modalities. 1 Since 5-year survival rates can reach as high as 99% post-treatment, physicians should also focus on the functional status of their patients. Erectile dysfunction (ED) is one of the most well-known complications post-radical prostatectomy (RP), affecting varying percentages of PCa patients, ranging from 19% to 74%.2,3 Treatment options for ED vary from phosphodiesterase-5 inhibitors to penile prosthesis implantation.4,5
Although not as frequent, orgasm-associated incontinence, also known as climacturia, is a side effect of RP that leads to sexual dysfunction. Climacturia, classified as orgasmic dysfunction, involves involuntary urine leakage during ejaculation or sexual arousal. Koeman et al. first described this phenomenon in 9 out of 14 post-RP patients, and subsequent studies reported a prevalence ranging from 15.7% to 93%, making it a relatively common yet neglected complication. 6 To date, treatment options for climacturia are limited. Behavioral treatment, pelvic floor muscle training (PFMT), and the use of certain devices are considered conservative measures, while surgical intervention has played an important role in cases of conservative management failure.7–9 The ‘Mini-Jupette’ technique and its alternatives have shown promising results in resolving climacturia-related symptoms, especially when combined with surgical ED treatment. 10 This technique involves the use of a sling that extends across the urethra, ensuring proper tension while the penile prosthesis cylinders are inflated. This tension aims to minimize, and even better, eradicate possible urine leakage during sexual arousal or orgasm.
The aim of this article is to review all available studies reporting on the safety and efficacy of implementing the ‘Mini-Jupette’ technique in treating patients with persistent climacturia.
Materials and methods
Search strategy
This systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 11 Two independent reviewers (ES and GL) systematically searched Medline via PubMed, Scopus, and Cochrane Library databases for relevant studies until October 2022. The search terms included the following: [(prostatectomy) OR (radical prostatectomy) AND (orgasm-associated incontinence) OR (climacturia) OR (sexual incontinence) AND (mini jupette)], along with relevant synonyms, truncated words, and MeSH terms. In addition, sources of gray literature, including conference abstracts published in major urology journals, were hand-searched, and the reference lists of all eligible studies and relevant reviews were reviewed (see Figure 1).
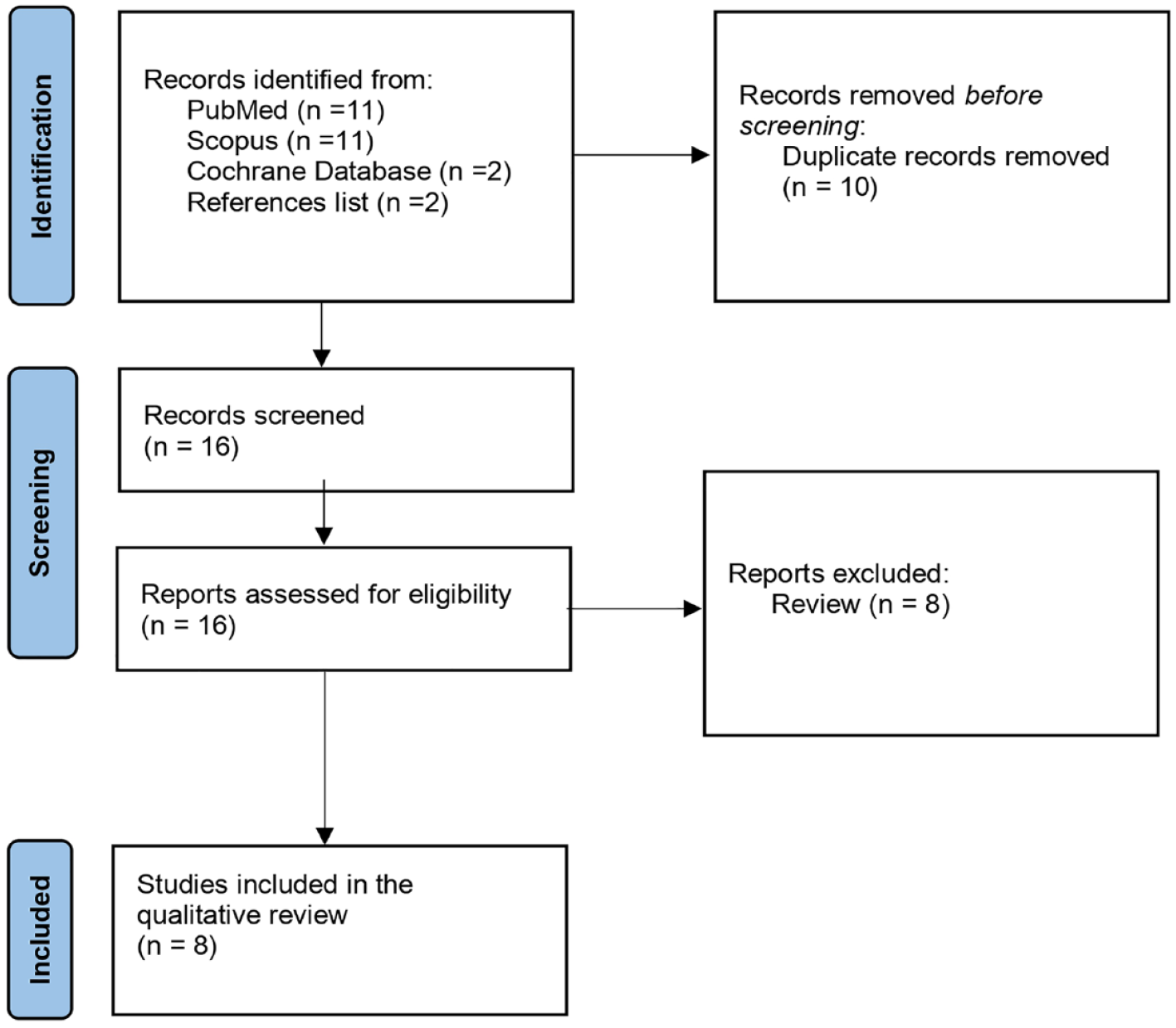
Flow diagram of the study selection process.
Study eligibility criteria
Our inclusion criteria included randomized controlled trials (RCTs), prospective or retrospective studies published in any language, which enrolled post-RP patients with climacturia and ED who underwent synchronous inflatable penile prosthesis (IPP) implantation and the ‘Mini-Jupette’ technique. In cases where records with overlapping patient populations were identified, we only considered the most recent study.
Data extraction
Two independent reviewers, who were blind to each other’s assessments (ES, ST), evaluated the titles and abstracts of all identified articles. The full texts of all eligible studies were then screened based on the predefined selection criteria. In consultation with all authors, a predefined Microsoft Excel spreadsheet was developed to extract relevant information about the study and patient characteristics (such as author, design, follow-up period, and patient age), surgical technique details (including the type of implants and operation time), and outcomes (including postoperative sexual function, postoperative PAD use, and complications).
Data synthesis
Due to the limited available literature and the heterogeneity among them, we conducted a qualitative analysis of the included studies.
Climacturia: Proposed pathophysiologic mechanisms
The pathophysiological mechanism of climacturia in patients after RP is not yet fully understood. Over the previous decades, many researchers have proposed potential mechanisms but the verdict is still pending. To comprehend the mechanisms behind it, a thorough understanding of the physiology of ejaculation is necessary.
Ejaculation consists of two phases: emission and expulsion. During emission, the internal sphincter-bladder neck-external sphincter functional unit contracts, and prostatic fluids are expelled into the prostatic urethra, creating a high-pressure zone. Expulsion follows emission, with semen being projected from the meatus due to rhythmic contractions of the pelvic striated muscles, along with the bulbospongiosus and ischiocavernosus muscles. To prevent retrograde ejaculation, the internal sphincter remains closed while the external urethral sphincter is open. Simultaneously, even though a reflex bladder contraction occurs, the contraction of the internal sphincter functional unit proves effective in preventing involuntary urination. 12
Koeman et al. were the first to propose that climacturia is a direct side effect of internal sphincter removal during RP. 13 This hypothesis was weakened as further studies suggested a possible correlation between external sphincter injury and internal sphincter dysregulation, eventually leading to climacturia. 14 The aforementioned mechanism could also explain the notably lower climacturia rates in patients who undergo transurethral resection of the prostate (TURP) compared to those after RP, as the external sphincter typically remains intact during TURP. 5
On the other hand, an alternative, indirect mechanism of sphincter damage was proposed by O’Neil et al., who reported climacturia in 5.2% of patients treated with radiation as radical therapy for PCa. 15 This outcome supports the hypothesis that radiation-induced nerve damage can potentially play a key role in the development of climacturia. In addition, adding to the theory of nerve damage, studies have reported that during surgery, the indirect stretching of the pudendal nerve can disrupt the physiological mechanism of ejaculation, thus leading to functional dyssynergia. 16
Loss of functional urethral length in post-RP patients is an independent risk factor for climacturia, as indicated by various studies.14,17,18
According to Mendez et al., a combination of the aforementioned mechanisms eventually leads to climacturia. The authors propose that anatomic damage to the pelvic floor after RP or stretch injury causes weakening of the pelvic floor and disruption of urethral support, while pudendal nerve damage leads to a deficit in bladder neck closure and weakened external sphincter. 19
Is climacturia a lifelong diagnosis?
Reduction in the prevalence of climacturia over time has been reported in many studies.14,20,21 Notably, Mitchell et al. reported that only half of the patients suffered from climacturia 24 months post-RP, compared to 3 months post-RP (12.1% and 22.4%, respectively). 20 Choi et al., who evaluated the prevalence of climacturia before and after 12 months post-RP, concluded that the time since RP was a predictor of climacturia, as men interviewed within <12 months postoperatively reported higher rates of climacturia compared to those interviewed >12 months post-RP. 14 Capogrosso et al., who studied recovery rates after an initial diagnosis of climacturia in a specific timeline of 12, 24, and 84 months post-RP, outlined recovery rates of 2.3%, 5.5%, and 24%, respectively. Interestingly, robotic-assisted RP was correlated with faster climacturia recovery compared to open RP (4%, 8.5%, and 48% versus 2.5%, 5%, and 15%). 21
Sociosexual impact of climacturia
Reduced sexual satisfaction, depression, and an overall decline in quality of life are the impacts of climacturia in post-RP patients. More precisely, Nilsson et al. reported that 27% of patients avoided sexual intercourse due to the fear of failure, resulting in concomitant low partner satisfaction and low orgasm satisfaction. 17 When accompanied by the detrimental effects of ED, it imposes a significant toll on patients’ sexual lives.
Is there a ‘gold-standard’ therapeutic treatment?
As previously mentioned, there is a significant possibility of spontaneous remission of climacturia symptoms. Nevertheless, when the ‘leak of pleasure’ persists, several conservative and invasive treatments have been proposed. In terms of conservative treatment, various modalities have been suggested, including PFMT, behavioral treatment (such as using a condom during intercourse and emptying the bladder before sexual arousal), and the use of external occlusion loops. These conservative approaches have all shown comparable long-term results.7–9 On the other hand, for post-RP patients experiencing persistent and bothersome climacturia, the surgical intervention appears to be the last but not least therapeutic option. 22 Furthermore, when combined with ED that does not respond to conservative treatment, a simultaneous surgical approach seems to be a ‘lucrative evolution’. 23 So far, the ‘Mini-Jupette’ technique has played a significant role in safely and effectively treating climacturia.
Mini-Jupette surgical technique
Back in 2005, Professor Robert Andrianne first described the Mini-Jupette, a surgical approach he developed for post-RP patients with concurrent ED and climacturia. 23 The primary concept behind this technique was to create a high-pressure zone preventing urine leakage during IPP inflation, particularly during sexual intercourse, through a combination of passive and active compression of the urethra alongside IPP implantation (see Figure 2). This approach was named ‘Mini-Jupette’, which translates to ‘miniskirt’ in French due to its suspension across the two corporal bodies (legs).
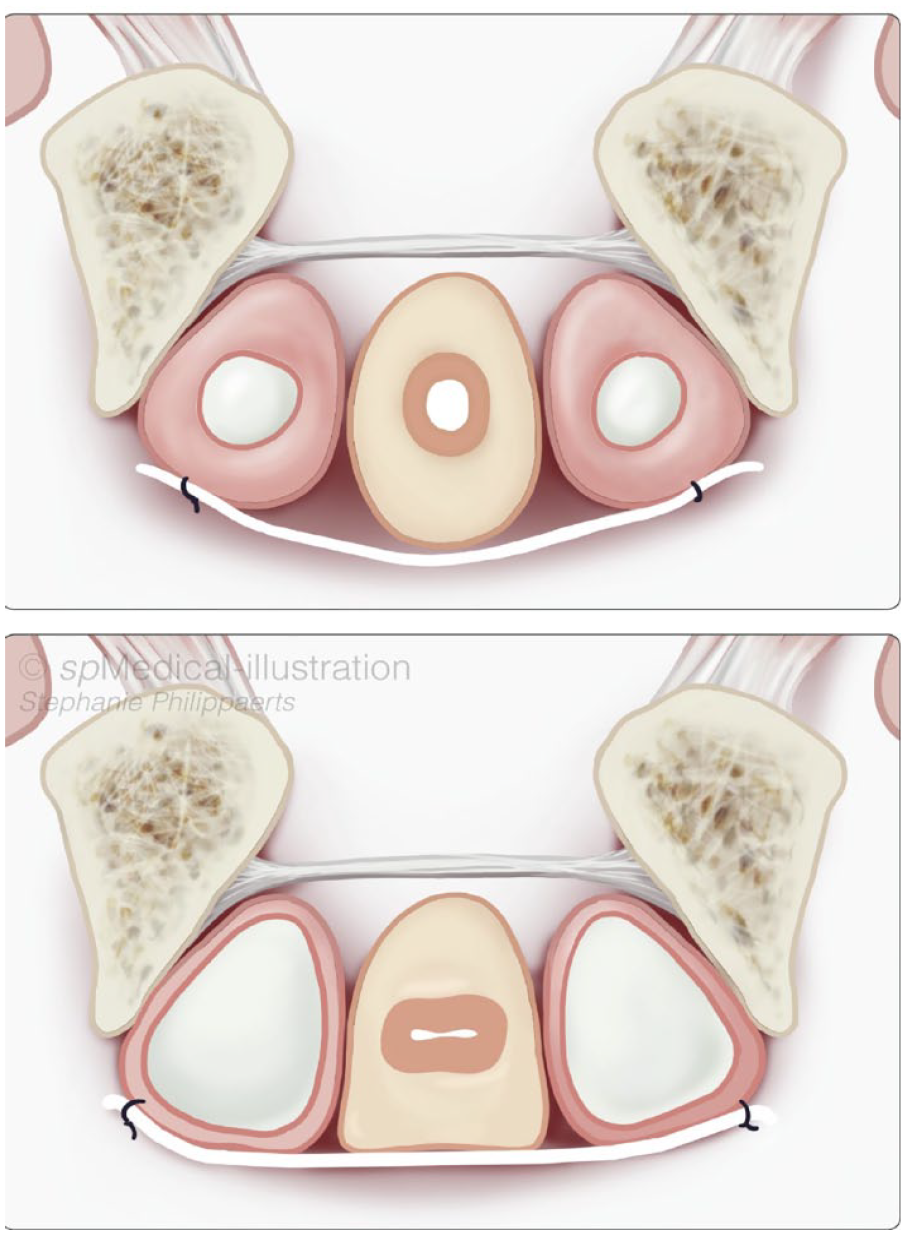
Urethral compression after inflation of the IPP.
The procedure involves proximal dissection to expose the corpora at the point of their separation, followed by corporotomies (CC) of at least 2 cm in length, performed 1–1.5 cm laterally from the urethra, and near the insertion of the cavernous bodies on the ischiopubic bone. After measuring synthetic or cadaveric grafts, distal and proximal dilatations of the CC are carried out between the inner borders of cavernotomies according to the chosen implant type. Subsequently, the mesh is affixed to the inner borders of the two cavernotomies, cylinders are introduced into the two cavernous bodies, and the cavernotomies are closed (see Figure 3).
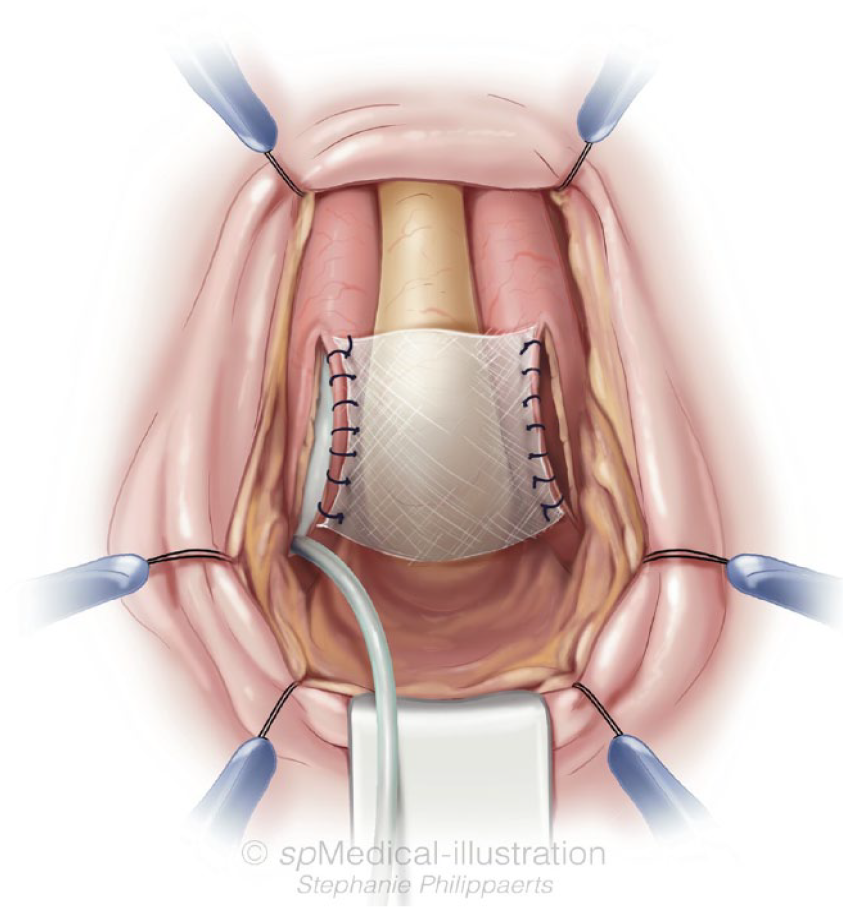
Graft placement and fixation on the inner borders of the corporotomies.
In his study, Professor Andrianne published data on 15 patients who underwent ‘Mini-Jupette’ surgery for ED and mild incontinence [1–2 pads per day (ppd)] with or without climacturia over a 9-year period. The mean time post-RP was 32 months, and the average patient’s age was 65.9 years. The IPPs of choice were AMS 700 LGX in four patients (26.7%), AMS CX in nine (60%), and Coloplast Titan in two patients (13.3%). The operative time averaged 72 min (SD = 19.8), with no reported intraoperative complications. Postoperative complications were reported in three patients, consisting of dysuria in two patients and urinary retention in one patient. These patients received a brief reinsertion of a 16 Fr urinary catheter and anti-inflammatory treatment along with complete deflation of the implant. Importantly, during follow-up assessments at months 1, 3, and 6, no significant instances of dysuria or de novo urgency were observed among the patients.
At the 6-month follow-up, there was a mean improvement of 13.6 points in IIEF-5 scores. Regarding incontinence and climacturia, 80% and 83% of patients reported the resolution of their incontinence and climacturia, respectively.
To date, the largest study to evaluate the Mini-Jupette surgical technique was conducted by Yafi et al. 10 This prospective multicenter pilot study included 38 post-RP patients with ED, climacturia, and/or mild urinary incontinence over a 19-month period. Of these patients, 36 underwent RP for PCa while the remaining two underwent transurethral resection of the prostate for benign prostate enlargement. The mean time post-RP was 63.6 months, and the patients’ mean age was 65.3 years. Incontinence (mean 1.3 ppd) was reported by 30 patients, while 32 patients had climacturia. Penile implants (IPP) included AMS 700 LGX, AMS 700 GX, and Coloplast Titan in 4, 3, and 31 patients, respectively. The mean length and width of grafts were 3.4 and 3.2 cm, respectively, while the mean corporotomy length was measured at 2.9 cm. Cadaveric fascia and synthetic grafts were used in this study.
Compared to a mean preoperative International Index of Erectile Function (IIEF-5) score of 7.9, postoperative follow-up results showed a statistically significant increase in the IIEF-5 score, with a mean value of 24.6 (p < 0.0001). The mean follow-up time span was 5.1 months. Interestingly, 89.3% of 28 patients reported a reduction in the use of ppd, with 21 of them reporting complete resolution of this symptom. In terms of climacturia, improvement was reported in 22 out of 28 patients with follow-up, with 19 of them reporting no symptomatology at all. Considering these findings, the authors suggest that the results of the Andrianne Mini-Jupette, regarding both urinary incontinence and climacturia, are comparable to other treatment algorithms, such as pelvic floor muscle therapy.
Complications were reported in five (13.2%) of the patients, with results similar to those of male suburethral slings. Among these, one patient reported persistent postoperative pain without further treatment. Epigastric vessel bleeding and subsequent hematoma formation at the reservoir site were noted in one patient, leading to device infection and explantation. Another device was explanted due to an ongoing psychiatric condition while the third patient reported persistent penile pain and urethral discharge. A diagnosis of urethra-corporal fistula was made, so the IPP was removed, and prolonged urinary catheterization along with urethroplasty was proposed. The fistula was believed to be a result of suturing the graft onto the urethra. One patient required IPP explantation and subsequent urethroplasty due to device infection and urethral erosion, but unfortunately, this patient passed away due to a massive pulmonary embolism despite anticoagulation therapy.
In conclusion, the authors suggest that a proximal corporotomy along with graft fixation over the urethra provides optimal results. They also highlight the importance of proper graft selection for each patient, given the greater experience and promising results with synthetic grafts.
In 2022, Brady et al. reported the experience of a single surgeon with the concomitant Andrianne Mini-Jupette sling and IPP placement. 24 A total of 32 patients underwent this combined procedure, with all surgeries involving the use of a cadaveric pericardium graft (Tutoplast®IOP Ophthalmics, Costa Mesa, CA, USA). Three parameters – foreplay incontinence, the presence of climacturia, and stress urinary incontinence (SUI) rates (graded on a scale from ‘0, dry’ to ‘4, squirt of urine’) – were measured and compared before and after the combined treatment. The respective preoperative rates were 27 (84%), 26 (81%), and 27 (84%).
Interestingly, all three parameters showed significant improvement after a mean follow-up of 17.9 months. Resolution rates varied, with 47.9% for foreplay incontinence, 64.7% for climacturia, and 57.1% for SUI. A significant improvement in Sexual Health Inventory for Men (SHIM) scores was also noted, increasing from 5.0 to 23.8. In this scientific study, a subset of patients (9.4%) required the utilization of an artificial urethral sphincter (AUS) to manage persistent incontinence following the Mini-Jupette procedure. Moreover, a single patient required a revision of the IPP due to foreskin irritation while another patient encountered issues related to the IPP’s pump mechanism. The etiology of these side effects is likely multifactorial, with some patients exhibiting more severe stress urinary incontinence beyond the capacity of the Mini-Jupette procedure to be fully addressed. Individual anatomical variabilities and technical aspects during the surgical intervention may have contributed to suboptimal treatment outcomes. In addition, improper sizing or positioning of the IPP and the potential impact of the Mini-Jupette procedure on local anatomy might have contributed to the observed side effects.
It is essential to underscore that the occurrence of these side effects does not necessarily suggest inherent flaws in the Mini-Jupette procedure itself. Surgical outcomes are influenced by a combination of patient-specific factors, surgical techniques, and the quality of medical devices employed. Effective mitigation of such side effects and the enhancement of patient outcomes in the management of climacturia and stress urinary incontinence can be achieved through a meticulous approach to patient selection, precise surgical execution, and thorough postoperative monitoring. As the first study reporting results in foreplay incontinence, the authors highlight the contribution and safety of this combined approach to all three aforementioned parameters.
Saavedra-Belaunde et al. reported their results in 17 patients with climacturia and SUI, primarily following RP. 25 All patients underwent treatment with IPP and Mini-Jupette cadaveric pericardium sling (Tutoplast®) placement. Due to the persistence of reported SUI, two patients also underwent AUS placement. The authors did not specify the timeframe of the second intervention in these patients.
The first follow-up at 6 weeks showed climacturia resolution in 76.6% of the patients. The mean International Consultation on Incontinence Questionnaire Short Form (ICIQ-SF) score was 7.4, compared to a preoperative score of 14. Interestingly, compared to the preoperative average of 2.4 ppd due to urinary incontinence, the patients reported an average of 0.8 ppd at 6 weeks. Among the 11 patients who presented at the 6-month follow-up, only one did not report any gradual improvement in climacturia, SUI, and the number of ppd used.
Recently, Hammad et al. became the first to conduct a comprehensive evaluation of incontinence outcomes and the safety profile associated with the Mini-Jupette procedure over an extended period of 5 years. 26 Of the initially 38 patients with a history of prostate procedures, 23 were able to adhere to the 5-year follow-up plan. All patients underwent IPP insertion with simultaneous placement of an AMJ sling. Graft selection was based on surgeons’ criteria. The mean age was 69 years (SD = 6.8) with a follow-up time of 59 months (SD = 8.8). A significant improvement of 0.4 ppd was noted compared to a mean of 1.4 ppd preoperatively. Of the 23 patients, 91.3% reported subjective improvement of SUI, and 73.9% reported improvement in climacturia. Regarding complications, only one patient underwent uncomplicated AUS placement due to persistent irritating SUI while another patient is contemplating a repeat incontinence procedure because of mild, persistent leakage.
The findings presented in this study strongly support the safety and feasibility of the AMJ technique as a promising approach for addressing climacturia and SUI. The long-term assessment of incontinence outcomes demonstrated significant improvements in patients’ quality of life. Moreover, the absence of major late complications, which may be associated with alternative procedures, underscores the safety profile of this technique. The authors could not arrive at a definitive recommendation concerning the most suitable graft material. Nevertheless, their consistently positive outcomes indicate that the technique employed might hold greater significance than the particular choice of graft material.
Male urethra mini sling
In another approach published by Valenzuela et al., a modified Virtue Coloplast mesh was used. 27 Their modification to the Andrianne Mini-Jupette technique was based on the proximal placement of the Virtue mesh. This approach proposes the fixation of the mesh medially to the lateral corpora and separation from the planned corporotomy, over the bulbar urethra, to minimize the potential risk of damaging the inflatable prosthesis. Avoiding a major corporotomy and subsequent suturing ensures the integrity and safety of the prosthesis. In this alternative, there are two incision options: penoscrotal or subcoronal. The penoscrotal incision is preferable when there is no consideration for Peyronie’s grafting or other penile interventions.
After exposing the urethra and both corpora, the length between the midline of the two corpora is measured laterally. Customarily, a 3 × 5.5 cm mesh, tailored without the limbs, is used. The mesh is placed and secured with Ethibond Polyester 2-0 interrupted sutures, and its resilience is ensured without compressing the urethra. The following steps and IPP placement are similar to the aforementioned AMJ procedure. The elongation of the procedure for male urethra mini sling (MUMS) placement is stated to be 15–20 min. In light of these findings, the authors conducted a retrospective review and reported their results on 36 patients who underwent IPP with MUMS placement after RP alone (83%) and prostatectomy plus radiation treatment (17%). The mean follow-up was 5.9 months, and the mean patient age was 68 years. The prevalence of climacturia in this patient group was 83% (30/36), with 58% (21/36) of them reporting both climacturia and SUI. The results regarding climacturia and SUI complete resolution were 93% (28/30) and 59% (16/27), respectively, while 85% (23/27) of patients reported subjective improvement in SUI symptoms with a ppd decrease from 1.4 preoperative to 0.4 postoperative (p = 0.02). Ephemeral urinary retention was observed in two patients (6%), one patient reported persistent scrotal pain after 5 months, which improved with anti-inflammatory drugs, and one patient encountered urethral erosion with mesh removal. These occurrences underscore the existence of a slight yet tangible risk of postoperative complications.
Mayo Clinic Mini-Jupette sling
Similar to the MUMS (a graft located at a different level compared to the corporotomy site), Kohler et al. reported their experience with six patients with climacturia who underwent a modified version in which the cylinders were placed after the insertion of eight sutures in total. 28 More precisely, four vertical Ethibond stay sutures were placed inferior to four vertical corporotomy Vicryl stay sutures. After the IPP and catheter insertion, a Coloplast Virtue MeSH sling is then placed and stabilized with the use of the four Ethibond sutures, with the cylinders not inflated and the corporotomies closed. The next step involves inflation of the cylinders to ensure proper tensioning over the urethra and coaptation. All six patients reported no climacturia-related symptomatology, as well as significant improvement in SUI. Concerning complications, no major adverse effects were reported, apart from persistent perineal pain.
Autologous Mini-Jupette sling
The decision regarding the optimal mesh or graft is not an easy one. This challenge arises from the recently reported high rates of mesh-associated complications in urological procedures in women. 29 In a multi-institutional collaborative study by Rac et al. to evaluate the possible impact of the Food and Drug Administration Public Health Notifications of 2008 and 2011 on surgical trends for SUI surgeries, a decreasing trend in mesh usage and increased utilization of autologous fascia slings were noticed. 30 The rate of mesh revision surgeries almost tripled, including urethrolysis and mesh removals.30,31
In the context of autologous fascia utilization, Towe et al. published a case series of two patients who underwent robotic-assisted RP and presented with concomitant ED, SUI, and climacturia. 32 Both patients were treated with IPP and Mini-Jupette graft placement. For the Mini-Jupette graft construction, autologous rectus fascia was harvested during the procedure. A 5 cm midline incision was performed just below the umbilicus and carried down to the pubic bone. A 3 × 4 cm fascial graft was harvested and placed across the urethra. The graft was sutured medially to the corporotomies alongside, as in the standard Mini-Jupette. They reported complete resolution of climacturia and SUI at 2 and 4 months of follow-up, respectively. No complications were stated by the authors, and the patients proved to be completely satisfied with the results.
All studies assessing the efficacy and safety of Mini-Jupette and its alterations as treatment options for climacturia and/or SUI are included in Table 1.
Studies assessing efficacy and safety of Mini-Jupette and its alterations for climacturia and SUI management.

Not published.
AUS, artificial urethral sphincter; IPP, inflatable penile prosthesis; ppd, pads per day; SUI, stress urinary incontinence.
Conclusion
Although PCa presents significantly higher survival rates, patients’ quality of life is compromised due to ED and climacturia. This finding underscores the necessity for a treatment algorithm that can simultaneously address these two parameters. Our review suggests that the Mini-Jupette technique and its variations constitute a relatively safe and cost-effective addition to IPP placement. This combined approach offers the potential to treat ED and urine incontinence during sexual arousal in a single operation. However, larger cohorts and RCTs, along with long-term follow-up, are required to validate these results and provide robust data regarding optimal graft selection.
