Abstract
Background:
Penile shortening, frequently resulting from end-stage Peyronie’s disease (PD), has a negative impact on patients’ sexual activity and overall quality of life, especially when accompanied by Erectile dysfunction (ED). Various surgical techniques have been described to manage concomitant ED and penile shortening through penile prosthesis (PP) implantation.
Objectives:
To evaluate the benefits and risks of different penile length preservation techniques during PP implantation.
Design:
A systematic review of the available literature on the use of penile length preservation maneuvers in conjunction with PP implantation was conducted.
Data sources and methods:
For this systematic review, three databases (Medline, Embase and Cochrane) and clinical trial.gov were queried for relevant publications from 1 January 1990 to 1 September 2022. The review process followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses guidelines.
Results:
The qualitative analysis included 15 relevant articles involving 1186 adult patients who underwent penile length preservation techniques during PP implantation. Penile lengthening of 1–7 cm was reported. Overall, postoperative complications were described in up to 21.7% of cases. Only five studies reported functional outcomes, showing a significant improvement in postoperative period based on the administered questionnaire (e.g. IIEF – International Index of Erectile Function, EDITS – Erectile Dysfunction Inventory of Treatment Satisfaction).
Conclusion:
Penile length preservation procedures appear to offer a viable option for managing acquired penile shortening, particularly in cases of PD. However, they are associated with a significant risk of complications. Proper patient selection, thorough discussion of risks and benefits, and referral to high-volume centers are mandatory to achieve optimal outcomes and minimizing complications.
Trial registration:
PROSPERO database registration CRD42022360758.
Keywords
Introduction
Erectile dysfunction (ED), especially if unresponsive to conservative treatments, either pharmacological or physical, can be treated with the implantation of a penile prosthesis (PP). 1 Indeed, the primary purpose of the device is to provide sufficient rigidity and a long-lasting erection to achieve satisfactory sexual intercourse. It is important to note that the implantation of a PP alone does not result in an increase in penile length. In fact, recent evidence suggested that some patients may experience perceived penile shortening of up to 1 cm after PP insertion. 2
ED may frequently be associated with significant penile shortening, particularly in cases of end-stage Peyronie’s disease (PD) where ED and penile shortening occur together. In the management of penile shortening with or without associated curvature, various surgical procedures that do not involve tunica albuginea incisions have been described. Techniques such as the Perito scratch technique, Wilson maneuver, or tunica albuginea plications can be performed to correct penile curvature during PP implantation. However, these techniques do not restore penile length but on the contrary can contribute to further penile shortening. 3 In case of ED and penile shortening, additional penile length preservation maneuvers may be necessary during PP implantation. A wide range of surgical techniques have been described to manage concomitant ED and penile shortening. These procedures vary depending on the type of tunica albuginea incisions used: circumferential, sliding, multiple sliding, or mesh-like incisions. 3
To date, there is currently a lack of evidence guiding the use of penile length preservation maneuvers associated with PP implantation to optimize penile length.
Our objectives were to systematically review the available literature on this topic, assess the benefits and risks of various surgical techniques used in this context, conduct pairwise comparisons, if feasible, and explore sources of heterogeneity for treatment efficacy and harms through relevant subgroup and/or sensitivity analysis.
Materials and methods
Protocol and registration
This systematic review was registered in advance with PROSPERO database (CRD42022360758). We conducted the review process following the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines (Supplemental File 1). 4
Study eligibility
A specific population (P), intervention (I), comparator (C), outcome (O) and study design (S) (PICOS) framework was used to define study eligibility. Studies were considered eligible for inclusion if they met the following criteria:
- (P): cisgender men aged >18 years old, with a diagnosis of ED with or without concomitant PD associated with a short penis, who were naïve to penile surgery, and had symptomatic conditions (ED associated with sexual difficulties related to a short penis)
- (I): PP implantation associated with tunica albuginea lengthening incisions: ■ Sliding technique (ST) ■ Modified sliding technique (MoST) ■ Multiple sliding technique (MuST) ■ Tunica mesh expansion procedure (TMEP) ■ Circumferential incision (CI)
Studies adopting straightening procedures to address penile curvature (e.g. scratch technique, Wilson’s maneuver, plaque excision/incision or PICS) were excluded.
Studies adopting visual penile lengthening procedures (e.g. scrotoplasty, suprapubic lipectomy, ventral phalloplasty, suspensory ligament release) were excluded.
- (C): any of the aforementioned surgical treatments or no treatments (pairwise comparisons allowed). Subgroups comparisons of interest were inquired if available (CI versus ST; ST versus MuST; ST versus TMEP).
- (O): The primary benefit outcome was the gain in penile length. The primary adverse outcome was early postoperative complications (<90 days) such as PP infection, PP malfunction, glans necrosis and wound infection.
The secondary outcomes included:
Functional outcomes, assessed using validated questionnaires (e.g. International Index of Erectile Function – IIEF; Sexual Encounter Profile – SEP; International Prostate Symptom Score – IPSS; Erectile Dysfunction Inventory of Treatment Satisfaction – EDITS) or as defined by the trialists.
Cosmetic outcomes (as defined by the trialists).
Impact on patients’ quality of life (as defined by the trialists).
All outcomes were reported at 12, 18, 24, and 36 months follow-up time points, except for the primary adverse outcome, which was reported at 90 days. For studies where outcomes were not reported at the pre-specified time points, a descriptive text was provided.
- (S): Randomized controlled trials (RCTs), nonrandomized controlled studies (NRS), and retrospective and prospective cohort studies including ⩾5 men were included. Case reports, conference abstracts, reviews, letters, and editorials were excluded. Additionally, animal studies, studies with a sample size of <5 patients, papers published before 1990 and articles in languages other than English were excluded.
Search and study selection
The Medline, Embase, and Cochrane controlled trials databases and clinicaltrial.gov were searched for relevant publications on 1 September 2022. The search terms used are presented in Supplemental File 2. The search was filtered for male and human. Studies published between 1 January 1990 and 1 September 2022 were included.
Following de-duplication, four review authors (AM, GR, MP, and IS) independently screened the titles and abstracts of the identified records for eligibility, working in pairs. The full text of all potentially eligible records was retrieved and screened independently by four review authors using a standardized form, linking together multiple records of the same study. Any disagreements were resolved through discussion or by consulting a fifth review author (MF).
Data extraction and analysis
The four review authors (AM, GR, MP, and IS) independently extracted outcome data, working in pairs. One review author extracted study characteristics, which were then checked for accuracy by a second review author. Any disagreements were resolved through discussion or by consulting a fifth review author (MF). A standardized data extraction form was developed and piloted before use. In case of incomplete data reporting, study authors were contacted.
Data to be extracted and included in the ‘characteristics of included studies’ table were as follows: study design; countries and institutions where the data were collected; dates defining start and end of patient recruitment and follow-up; methods used to form intervention and comparator groups; whether an a priori protocol or analysis plan was present; participant demographic and clinical characteristics (similar to the pre-specified confounder variables shown in the ‘risk of bias’ section below); eligibility criteria for participants; the numbers of participants included in the study, assigned to each intervention or comparator group, received the intended treatment, and analyzed; losses and exclusions of participants, with reasons; description of interventions; study funding sources; ethical approval; power calculation.
Assessment of risk of bias in included studies
The ‘risk of bias’ for each included study was assessed by the four review authors (AM, GR, MP, and IS), working in pairs. Any disagreements were resolved through discussion or by consulting a third review author (MF). The risk of bias in RCTs was assessed using the recommended tool in the Cochrane Handbook for Systematic Reviews of Interventions.5,6 This includes the assessment of: random sequence generation; allocation concealment; blinding of participants and personnel; blinding of outcome assessment; incomplete outcome data; selective reporting; and other sources of bias.
The risk of bias in non-randomized comparative studies was assessed using all domains above, and an extra item to assess the risk of findings being explained by confounding. Sequence generation and allocation concealment were retained as domains but were assessed by default as ‘high risk of bias’ given the non-randomized nature of these studies. An extra item to assess the risk that the findings may be explained by confounding was included. This was a pragmatic approach informed by methodological literature pertaining to assessing Risk of Bias in NRS. 7
Risk of bias in non-comparative studies was extended to addressing external validity (applicability of results to different people, places or time) of non-comparative studies. This was done by assessing:
Was there an a priori protocol? (yes/no)? If ‘no’, the study was at ‘high’ risk of bias.
Was the total population included or were study participants selected consecutively? If ‘no’, the study was at ‘high’ risk of bias.
Was outcome data complete for all participants and any missing data adequately explained/unlikely to be related to the outcome? If ‘no’, the study was at ‘high’ risk of bias.
Were all pre-specified outcomes of interest and expected outcomes reported? If ‘no’, the study was at ‘high’ risk of bias.
Were primary benefit and harm outcomes appropriately measured? If ‘no’, the study was at a ‘high’ risk of bias.
If the answer to all five questions was ‘yes’, then the study was at ‘low’ risk of bias.
Results
Evidence acquisition
The PRISMA flowchart summarizing literature research and article selection is reported in Figure 1. After screening 120 records, 15 full-text studies published from 1995 to 2020 were included in the qualitative analysis. All of them were single-center retrospective case series.

PRISMA flow diagram.
Study and patients’ characteristics
Overall, we detected a high-risk of bias in all retrieved publications (Figure 2).
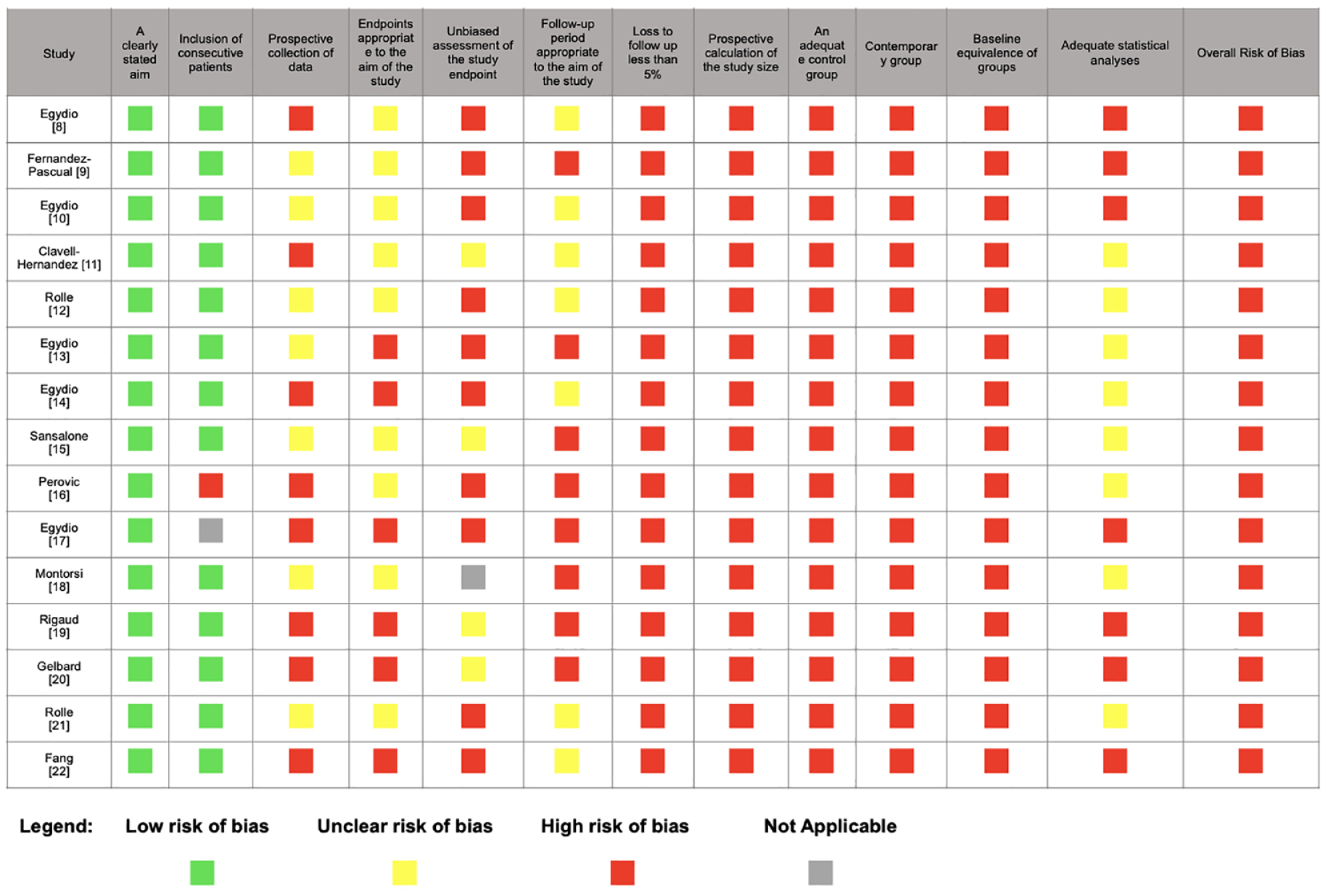
Risk of bias.
Study and patients’ basic characteristics are resumed in Table 1. Overall, 1186 adult patients underwent penile length preservation techniques during PP implantation were included. The pooled mean follow-up was 17.7 month. Range of mean age was 21–84 year. The most common comorbidities associated were diabetes (18–52%) and smoking habit (68%). The prevalent causes requiring PP implantation and penile length preservation maneuvers were organic ED, pelvic surgery/radiotherapy, and PD in (15.6–24.6%), (13.7–21.7%), and (60.1–100%), respectively.
Baseline characteristics for studies on corporal lengthening techniques during PP implantation.
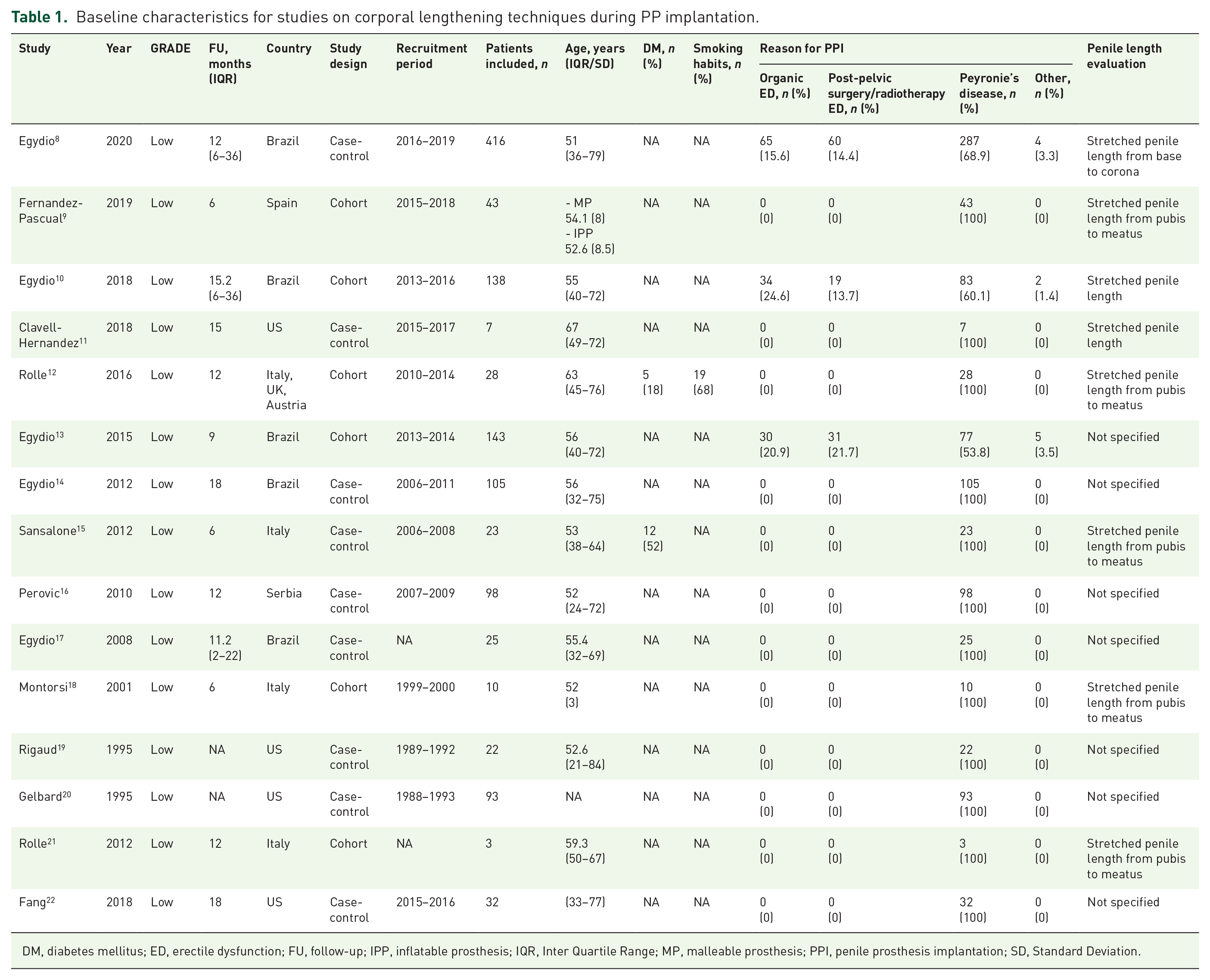
DM, diabetes mellitus; ED, erectile dysfunction; FU, follow-up; IPP, inflatable prosthesis; IQR, Inter Quartile Range; MP, malleable prosthesis; PPI, penile prosthesis implantation; SD, Standard Deviation.
Surgical features are summarized in Table 2. Preoperative stretched penile length ranged 6–18.2 cm. In most studies, penile length measurement was performed in-office measuring with a ruler the distance from the pubis to the tip of the glans on the dorsal aspect of the stretched penis. An inflatable PP was considered in 13.2–100% of cases, whereas a malleable PP was inserted in 0–93% of patients. A sub-coronal access alone or in combination with a penoscrotal incision was performed in most cases (up to 100%). Among the various technique applied for penile length preservation ST, MOST, MUST, TMEP, and CI were used in 3.5%, 23.7%, 3.8%, 35%, and 25.7% of cases, respectively. The Buck’s fascia only was the preferred option used to cover the albugineal defect in 742/1186 (62.5%) patients. Secondary options were the application of bovine pericardium patch in 135/1186 (11.4%) and the collagen fleece in 50/1186 (4.2%) of patients.
Intraoperative characteristics for studies on corporal lengthening techniques during PP implantation.

AMS, American Medical System (Boston Scientific); CI, circumferential incision; IPP, inflatable prosthesis; MOST, modified sliding technique; MP, malleable prosthesis; MUST, multiple sliding technique; OT, other techniques; SPL, penile stretched length; ST, sliding technique; TMEP, tunical mesh expansion procedure.
Surgical outcomes
Surgical outcomes are summarized in Table 3. A penile lengthening ranging from 1 to 7 cm was achieved. No intraoperative complications were reported. Overall, a significant incidence of postoperative complications was described in up to 21.7% of cases. In particular, PP infection and glans necrosis were reported in up to 9.1% and 4.5%, respectively.
Surgical and functional outcomes for studies on corporal lengthening techniques during PP implantation.

AMS, American Medical System (Boston Scientific); EDITS, Erectile Dysfunction Inventory Treatment Satisfaction; IC, intraoperative complications; IIEF, International Index Erectile Function; mo, months; nr, Not reported; Preop., Preoperative; PC, postoperative complications; PL, penile length; PP, penile prosthesis; SEP, sexual encounter profile.
Functional outcomes
Table 3 summarizes the functional outcomes. A minority of studies (5 out of 15) assessed functional outcomes using validated questionnaires in both preoperative and postoperative settings. The results showed a significant increase in IIEF-15 values, ranging from 9 to 51 in the preoperative setting to 48–74 at 6 and 12 months follow-up. EDITS scores ranged from 30 to 55 at 6 months follow-up and 27–55 at 12 months follow-up.
Subgroup analysis
Due to inconsistent data, it was not possible to provide satisfaction rates based upon subgroup analysis.
Discussion
During the last three decades adjuvant penile length preservation maneuvers during PP implantation have been used to restore penile length and girth, an issue that often afflicts patients with end-stage PD and ED. 6 The review of the 15 included articles revealed a heterogeneous group of case series, published by key opinion leaders in the surgical treatment of PD. Different surgical approaches were reported, but no dominant technique, tunical incision, or implant type was identified. It is important to note that most of the included populations consisted exclusively of patients with intrinsic penile shortening due to PD which may limit the generalization of our findings to other causes of penile shortening.8,9,11,12,15,17–22 Furthermore, most of these case-series report had a short- and/or medium-term follow-up8–18,21,22 and all studies were classified as having a high-risk of bias.
Concerning penile lengthening, adjuvant penile length preservation maneuvers were found to increase penile length by a median of 2.5–3.8 cm. Moreover, if the lower end of the range is taken into account at least a 1 cm increase in penile length is to be expected, with the vast majority of studies reporting at least a 2 cm increase in penile length.8–19,21 However, it is unclear how much of an increase in penile length is needed for a significant clinical effect (also referred by some as clinically important difference) to be noticeable by the patient; second, it is also unclear if penile length increase is homogenous among patients or if some patient or surgical characteristics can limit corporal length restoration and, thus, satisfaction.
Patient-reported satisfaction was assessed in only five studies, with three using the EDITS validated questionnaire,8,10,12,14,21 which was originally intended and validated for ED treatment, not PD.
The usefulness of EDITS as a tool to assess the added value of penile length preservation maneuvers in PP implantation for ED is still unknown
Although EDITS was not reported in its intended form (a scale from 0 to 100), the results suggested that most patients were satisfied, as the lower quartiles reported were still above what is usually considered a satisfactory outcome using EDITS.8,10,12 Due to the design of the included studies, it is currently difficult to determine whether patients satisfaction is primarily due to surgical treatment for ED and/or penile curvature correction, or if there is an additional value in restoring penile length.
It is noteworthy that increasing penile length is not without complications. While no intra-operative complications were reported, post-operative complications were fairly common, with an overall incidence of 21.7% out of 1161 cases, as shown in Table 3.8–14,16,18–22 The most commonly reported complications were genital hematoma, occurring in 13.6% of patients,8–16,18–22 and glans numbness, reported in 4.05% of patients. It is worth mentioning that glans numbness can be transient, and most studies did not provide details on how it was defined and assessed.8–15,18–22 Glans necrosis, a severe and devastating complication, was extremely rare, with only two cases reported among the 1161 patients included in our systematic review.10,19 Although not an ideal historical control, tunical lengthening procedures for moderate-to-severe penile curvatures do pose a similar rate of genital hematoma and glans numbness, which may allow us to conclude that performing adjuvant penile length preservation maneuvers during a PP implantation may be a safe procedure.
Regarding PP-specific complications, the risk of infection does not appear to be increased, as only 0.7% of cases reported PP infections, which is below the accepted risk of 1–2% for stand-alone PP insertion.8,9,12,14,16,18,19,23 Mechanical failure was reported in only one case, but the follow-up period may have been too short for this complication to emerge. 20
Our current systematic review has some limitations that need to be acknowledged. First, most of the included case-series are from renowned experts in PD and ED, and their results may not be as applicable to other surgeons, as the learning curve for these maneuvers is unknown. Furthermore, one author alone accounted for two-thirds of the included population, which may have influenced our results and conclusion.8,10,13,14 After a funnel plot analysis of the complications rate, there may be a publication bias. Some case-series did not report any complications which is unusual given the known morbidity of these procedure, and it may be a tell-tale sign of a self-report bias.15,17,21,22
While the potential benefit of a larger penis may be enticing, it is still unclear whether an increase in length and/or girth has an impact on patient-reported satisfaction. However, it is clear that performing an adjuvant penile length preservation procedure adds at least the morbidity associated with tunical lengthening procedures to a PP implantation. Patients need to be informed that while the majority will be satisfied with the outcome of their surgery, one in five patients may experience mild complications such as genital hematoma and glans numbness, and severe complications, like glans necrosis, although rare, can occur. Since complication rates depend on the surgeon’s experience, penile length preservation procedures should only be performed by experienced surgeons in high-volume centers with expertise in genital reconstructive techniques.
Conclusion
Penile length preservation procedures, within the described limits, offer a viable option for managing acquired penile shortening, particularly in cases of PD. However, they come with a significant rate of complications, including serious ones.
To confirm these results, multicenter randomized controlled studies are needed in order to overcome the limitations associated with the patients’ selection and outcomes/complications reporting biases connected with these procedures. In any case, the penile length preservation corporal lengthening procedures need to be performed in high-volume centers by surgeons skilled in genital reconstructive techniques.
Supplemental Material
sj-docx-2-tau-10.1177_17562872231215177 – Supplemental material for Risk and benefits of penile length preservation techniques during penile prosthesis implantation: a systematic review by the young academic urologists sexual and reproductive health working group
Supplemental material, sj-docx-2-tau-10.1177_17562872231215177 for Risk and benefits of penile length preservation techniques during penile prosthesis implantation: a systematic review by the young academic urologists sexual and reproductive health working group by Marco Falcone, Mirko Preto, Afonso Ribeiro Morgado, Ioannis Sokolakis, Selçuk Sarıkaya, Marco Capece, Paolo Capogrosso, Celeste Manfredi, Georgios Tsampoukas and Giorgio Russo in Therapeutic Advances in Urology
Supplemental Material
sj-pdf-1-tau-10.1177_17562872231215177 – Supplemental material for Risk and benefits of penile length preservation techniques during penile prosthesis implantation: a systematic review by the young academic urologists sexual and reproductive health working group
Supplemental material, sj-pdf-1-tau-10.1177_17562872231215177 for Risk and benefits of penile length preservation techniques during penile prosthesis implantation: a systematic review by the young academic urologists sexual and reproductive health working group by Marco Falcone, Mirko Preto, Afonso Ribeiro Morgado, Ioannis Sokolakis, Selçuk Sarıkaya, Marco Capece, Paolo Capogrosso, Celeste Manfredi, Georgios Tsampoukas and Giorgio Russo in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
