Abstract
Nanotechnology represents an expanding area of research and innovation in almost every field of science, including Medicine, where nanomaterial-based products have been developed for diagnostic and therapeutic applications. Because of their small, nanoscale size, these materials exhibit unique physical and chemical properties that differ from those of each component when considered in bulk. In Nanomedicine, there is an increasing interest in harnessing these unique properties to engineer nanocarriers for the delivery of therapeutic agents. Nano-based drug delivery platforms have many advantages over conventional drug administration routes as this technology allows for local and transdermal applications of therapeutics that can bypass the first-pass metabolism, improves drug efficacy through encapsulation of hydrophobic drugs, and allows for a sustained and controlled release of encapsulated agents. In Urology, nano-based drug delivery platforms have been extensively investigated and implemented for cancer treatment. However, there is also great potential for use of nanotechnology to treat non-oncologic urogenital diseases. We provide an update on research that is paving the way for clinical translation of nanotechnology in the areas of erectile dysfunction (ED), overactive bladder (OAB), interstitial cystitis/bladder pain syndrome (IC/BPS), and catheter-associated urinary tract infections (CAUTIs). Overall, preclinical and clinical studies have proven the utility of nanomaterials both as vehicles for transdermal and intravesical delivery of therapeutic agents and for urinary catheter formulation with antimicrobial agents to treat non-oncologic urogenital diseases. Although clinical translation will be dependent on overcoming regulatory challenges, it is inevitable before there is universal adoption of this technology to treat non-oncologic urogenital diseases.
Introduction
The development of new technology often facilitates breakthroughs in scientific discovery, with concomitant advances in therapeutic modalities. This has been the case with nanotechnology applied to biomedical research, where a major focus for clinical translation has been toward optimizing its use for targeted drug delivery, imaging, diagnosis, and treatment.
Nanotechnology involves the understanding and control of matter and processes at the nanoscale dimension, a scale at which unique properties of materials emerge that usually enables novel applications. 1 By definition, nanomaterials have sizes falling between 1 and 100 nm in at least one dimension, and when used as transport vehicles for another substance, they are referred to as nanocarriers. There are several types of nanocarriers with different compositions (organic, inorganic or hybrid) and structures that give them unique physicochemical properties and cargo capabilities.2–5 Examples of commonly used nanocarriers include liposomes, micelles, nanoparticles, carbon nanotubes and carbon dots, dendrimers, and nanogels. The great interest in using nanocarriers in biomedical research and therapeutics stems from their ability to surmount limitations faced with delivering drugs and biologicals using conventional administration routes, increasing the cargo’s bioavailability, efficacy, and safety, particularly of those with low solubility, stability, tissue penetration, or that can cause off-target effects (Figure 1).
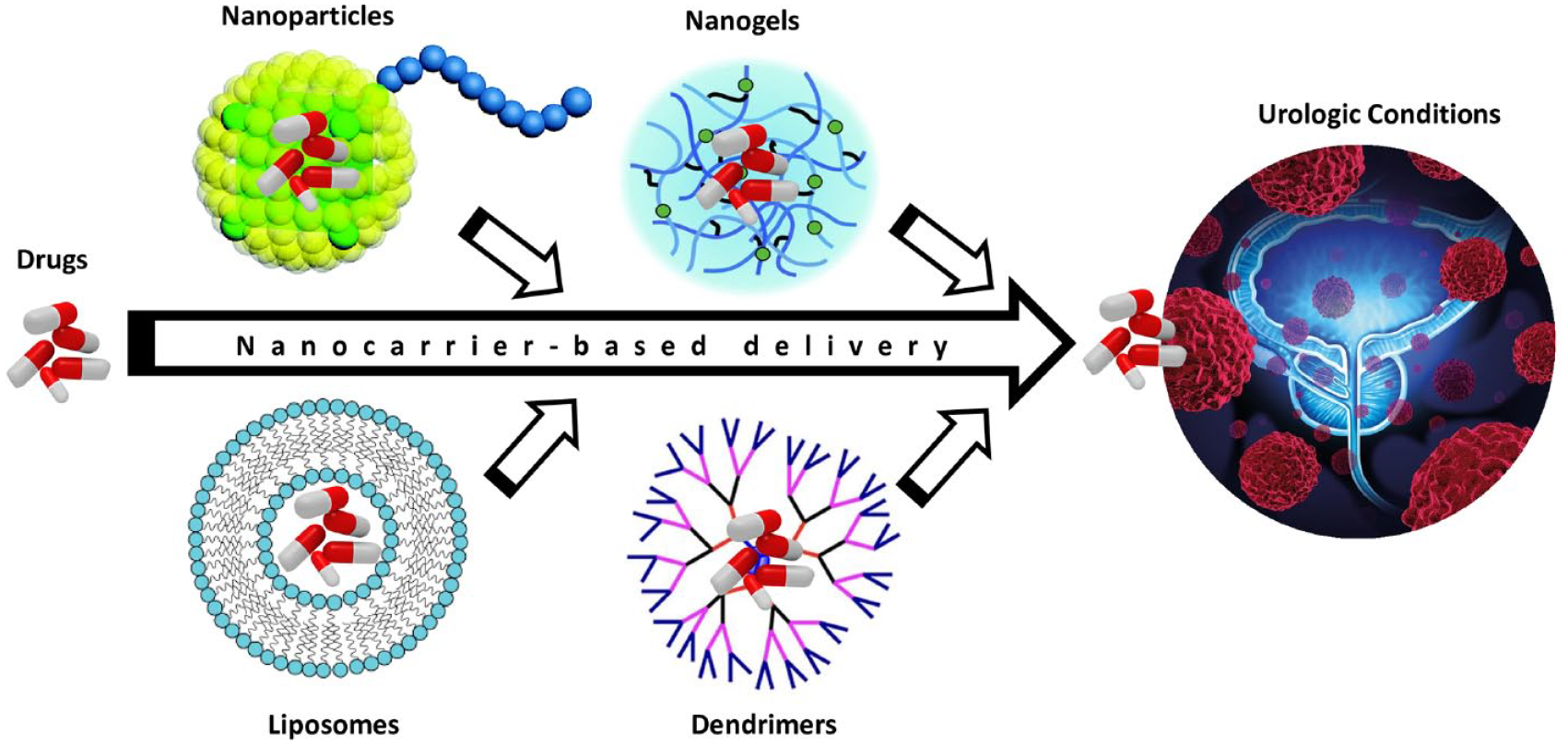
Nanocarrier-based delivery systems for targeted drug delivery.
In the field of Urology, most of the research involving the use and development of novel nano-based drug delivery platforms has focused on cancer treatment. However, the potential application of nanotechnology in non-oncologic urogenital research and therapeutics is immense. Current management of erectile dysfunction (ED) and urinary bladder diseases relies mainly on oral medications that are not effective for all patients and can be accompanied by systemic side effects that reduce the patients’ adherence to treatment. However, because the urogenital organs are easily accessible, they are highly amenable to topical delivery of therapeutics, as, for example, directly to the penile dermis or into the bladder by transurethral cannulation. These alternative routes have been explored and intravesical therapies are currently used to manage some bladder conditions. Nonetheless, delivery of therapeutics through these routes is also limited by natural obstacles. These include the remarkable barrier properties of the skin and the urothelium (the inner lining of the bladder) that make hard the penetration and absorption of dermally applied therapeutics, and dilution in the urine and washout with micturition of intravesically administered therapeutics.6–8 However, these natural barriers can be overcome using nanocarrier-based delivery systems.8–10
The use of nanotechnology in urologic cancers (kidney, prostate, bladder) has been well-described and reviewed. 11 In comparison, its use in research, diagnosis, and treatment of benign urological conditions, which affects a large proportion of the population, has been limited. Therefore, the goal of this article is to provide an update on how nanotechnology, and in particular the development and use of nano-based delivery systems, is paving the way to advance basic and clinical research, and treatment of non-oncologic urogenital diseases. In this review, we will highlight the background, investigational approaches, and clinical status of nanocarrier-based therapeutics for ED, overactive bladder (OAB), interstitial cystitis/bladder pain syndrome (IC/BPS), and catheter-associated urinary tract infections (CAUTIs) (Table 1).
Overview of current and nanotechnology-driven therapeutics for non-oncologic urogenital diseases.

BoNT-A, botulinum neurotoxin A; DHA, docosahexaenoic acid; DMSO, dimethyl sulfoxide; HA, hyaluronic acid; IND, Investigational New Drug Application; LP, liposomes; MBN, Mirabegron; NGF, nerve growth factor; NO, nitric oxide; PA, peptide amphiphile; PDE5, phosphodiesterase-5; PEG, polyethylene glycol; PGE1, prostaglandin E1; PGE1-EE, a PGE1 ethyl ester; UTI, urinary tract infection.
Materials and methods
PubMed/MEDLINE and Google Scholar databases were searched for relevant articles in the English language through 1 July 2021. Search strategy included the following terms: ‘nanotechnology in urology’ OR ‘nanotechnology erectile dysfunction’ OR ‘nanotechnology overactive bladder’ OR ‘nanotechnology interstitial cystitis bladder pain syndrome’ OR ‘nanotechnology urinary tract infection’. The term ‘nanotechnology’ was also replaced with ‘nanoparticle’ and ‘nanocarrier’ for each search term. Articles were considered eligible if they were written in the English language, were accessible via our university library services, and if the title was concordant with the content of our review. Reference lists of included articles and reviews were manually reviewed to identify additional relevant articles.
Erectile dysfunction
Background
ED is the inability to achieve or maintain an erection that is sufficient for satisfactory sexual performance. It affects at least 12 million men in the United States, with 52% of men aged 40–70 years experiencing some form of mild to moderate ED.12,13 A variety of factors (i.e. vascular, neurologic, psychological, hormonal) and conditions (i.e. diabetes mellitus, hypertension, hyperlipidemia, obesity, testosterone deficiency, prostate cancer treatment) can contribute to the development of ED. 12 The application of nanotechnology to organic ED has been the subject of several recent reviews.14–17
Current therapeutics
The NO (nitric oxide)/cyclic guanosine monophosphate (cGMP) pathway is central to the development of an erectile response and is the target of phosphodiesterase-5 inhibitors (PDE5i), which represent the first-line therapy in the treatment of ED. 18 Although these medications are typically safe and well-tolerated, a significant number of patients either cannot tolerate the side effects associated with PDE5i, such as headache, nasal congestion, and dyspepsia, or have difficulty swallowing medications (dysphagia). 19 Also, given that the efficacy of PDE5i is dependent on the active production of NO from the endings of the cavernous nerve (CN), patients who do not generate sufficient NO because of CN damage resulting from surgical trauma (that accompanies radical prostatectomy) or chronic vascular inflammation (that occurs in diabetic patients) are often refractory to this treatment.
A more invasive yet efficacious therapy for ED is intracorporal injections. Prostaglandin E1 (PGE1, known commercially as alprostadil) is a potent vasodilator agent used to treat ED administered by direct intracorporal injection. It causes corporal smooth muscle relaxation by binding to PGE receptors, resulting in the activation of adenylate cyclase and the subsequent accumulation of 3′5′-cAMP. Intracorporal injections have also been shown to commonly cause pain at the injection site, pain during erection, and hematomas/ecchymoses. 20 As a result, although intracorporal injections are efficacious, patients often prefer less invasive therapeutics, such as oral medications. 21
Nanotechnology-driven therapeutics
Nanotechnology, and in particular the engineering of nanomaterials for target delivery of therapeutics, holds great potential to advance treatment for ED.
Targeting the NO/cGMP pathway
One of the first studies that explored the potential of a nanocarrier-based delivery system to treat ED demonstrated the feasibility of topical penile delivery of NO and a PDE5i (tadalafil) to improve erectile function outcomes in a rat model of aging.22,23 A hydrogel-derived nanoparticle was generated for these studies using a silane-based sol-gel technology synthesized from tetramethoxysilane (TMOS).23–31 Both the NO- and tadalafil-nanoparticles were applied as a gel to the glans and shaft of the rat penis, representing approximately 10 nmol NO (steady-state delivery) or 1 mg tadalafil, respectively. 23 An erectile response was observed in treated animals approximately 5 min after the administration of NO-nanoparticles and was followed by several other erections of diminishing intensity. It is important to note that over the 2-h time course of the experiments, there was no significant effect on systemic blood pressure, suggesting that the NO derived from the nanoparticles was acting locally. Furthermore, tadalafil is a PDE5i which maintains intracellular levels of cGMP after neuronal stimulation to induce an erection. Therefore, it was reasoned that after application of the tadalafil-nanoparticles, stimulation of the CN would be necessary to obtain an erectile response. Sixty minutes after topical application of tadalafil-nanoparticles to the glans of the penis, there was a significant improvement in the erectile response at a 4 mA level of stimulation compared with animals treated with ‘empty’ nanoparticles (which served as negative controls). More recently, sildenafil-nanoparticles were also tested in the aging rat model and were shown to improve erectile function after local topical application to the penile shaft. 32
The NO-nanoparticles were subsequently shown to also be effective at improving erectile function outcomes in a rat model mimicking the nerve damage that occurs following radical prostatectomy.33,34 In one study, the NO-nanoparticles were applied topically to the penile shaft of a rat that had 2 weeks prior undergone bilateral CN transection. Following application, spontaneous erections were observed with a time of onset ranging from 5 to 37 minutes (with an average of 15 ± 11 minutes). 33 In another study, the topical application of NO-nanoparticles was combined with an oral PDE5i (sildenafil). Oral treatment with sildenafil resulted in no visible erectile response, but a combination of orally administered sildenafil with topical application of NO-nanoparticles produced significantly more spontaneous erections when compared with the NO-nanoparticles by themselves. 34
Modulate smooth muscle relaxation
Several studies have investigated the potential for transdermal delivery of a prodrug of PGE1 (a PGE1 ethyl ester, PGE1-EE) using 5% SEPA (soft enhancer of percutaneous absorption) in the formulation.35–37 The prodrug is hydrolyzed through the action of esterases present in the skin. However, these formulations are generally less efficacious than intracorporal injection. As an alternative, alcoholic nanoparticle hydrogels have been formulated, which could potentially act as efficient topical delivery vehicles of PGE1-EE. 38 In pharmacodynamic studies where the PGE1-EE hydrogels were applied to the penile dermis of cats, there was increased intracavernosal pressure compared with control nanoparticle hydrogels. However, the efficacy compared with direct injection has not been evaluated.
Papaverine hydrochloride is a direct-acting smooth muscle relaxant. Although its pharmacological mechanism of action is unclear, it is known to inhibit phosphodiesterases and may have direct actions on calcium channels. The normal route of administration is by intracorporal injection. Although several studies have described less invasive, transdermal approaches for its administration, the low bioavailability of these transdermal formulations has limited their efficacy and clinical translation. 39 Nevertheless, a recent study using a papaverine-nanoparticle formulation demonstrated improved transdermal delivery and therefore a potentially greater efficacy. 40 Specifically, the formula released 73% of its initial drug content within 2 h and the clinical evaluation showed an increase in the cavernous artery diameter and an increase in the peak systolic flow velocity. Although an erection was not reported, the increased basal blood flow observed in these studies suggests it may be useful as a tool in penile rehabilitation following radical prostatectomy, where a primary goal is to maintain basal blood flow to prevent fibrosis and penile atrophy. 41
Opiorphins are a family of peptides that act as neutral endopeptidase inhibitors and have been shown to play a role in relaxation of corporal smooth muscle tissue. 42 The rat opiorphin homologue (sialorphin) encapsulated in silane-hydrogel nanoparticles when applied to the penile dermis of an aging rat model of ED resulted in a prolonged erectile response (increased ICP/BP ratio in the absence of CN stimulation that lasted for 8 min) with an average onset time of 4.5 min after application. 23 The opiorphin family of peptides have been associated with priapism, 43 and the prolonged erectile response observed in rats may raise concerns that a treatment based on opiorphin-nanoparticles may elicit priapism in patients if the release of opiorphin from nanoparticles is not carefully regulated.
Promote nerve regeneration following radical prostatectomy
One of the first reports exploring the application of nanomaterials to enhance nerve repair involved the delivery of sonic hedgehog (SHH) applied to the site of CN injury in a rat animal model. The rationale for focusing on SHH was provided by findings demonstrating that intracorporal injection of the SHH protein at the time of CN injury decreased injury-induced apoptosis in a dose-dependent manner. 44 SHH was incorporated into aligned peptide amphiphile (PA) nanofibers, which consist of a hydrophilic peptide with attached lipid chains that self-assemble into structures with a high aspect ratio of nanofibers.45–47 The SHH PA-nanofibers were applied to the site of CN injury in a rat model, and 6 weeks later, the animals’ erectile function was evaluated by cavernosometry.48,49 Animals treated with SHH PA-nanofibers had improved erectile function (as determined by increased ICP/BP ratio following electrical stimulation of the CN) accompanied by improved penile architecture. 48 Subsequent reports using the same animal model reported that treatment with the SHH PA-nanofiber reduces penile apoptosis and fibrosis50,51 and promotes regeneration of CN fibers.48,52
Investigations on the potential of nerve growth factor (NGF) to improve erectile function following nerve injury have a long history. 53 However, if applied at the time of injury, or through intracorporal injection, NGF has a short half-life at the target site. To circumvent this limitation, an extended-release NGF formulation was developed where NGF was encapsulated into a hyaluronic acid (HA) hydrogel nanoparticle. In one such formulation, 20% of the loaded NGF is released over 35 days. Another potential advantage of this formulation is that HA binds to specific proteins on the cell surface which may tether the nanoparticles to the site of application. 54 In a rat model of CN injury, animals treated with the NGF-HA-nanoparticle at the time of injury showed approximately 40% better erectile function (determined by ICP/BP) than untreated controls. Improved erectile function was associated with reduced smooth muscle atrophy and increased endothelial nitric oxide synthase (eNOS) expression in penile tissue. Interestingly, there was a synergistic effect when treatment of the NGF-HA-nanoparticles was combined with human adipose-derived stem cells (hADSC). It was speculated that the HA component of the nanoparticle may anchor hADSCs at the site of application and provide a microenvironment for neuronal differentiation of the hADSC.
The ability of nanoparticles to anchor or target stem cells to the site of CN injury has been more directly investigated in two studies.55,56 Rat ADSC (rADSC) were magnetized with NanoShuttle, a commercially available biocompatible magnetic nanoparticle assembly consisting of gold, iron oxide, and poly-
Docosahexaenoic acid (DHA) has well-documented anti-inflammatory and antioxidant activities as well as neuroprotective properties. In a rat CN crush model, animals were administered a single intraperitoneal dose of either vehicle or a DHA-nanoemulsion at 10, 50, or 250 μg/kg. 57 At 28 days post-injury, erectile function (determined by ICP/BP) was significantly higher in all groups receiving the DHA-nanoemulsion than in the vehicle group, though was most pronounced in animals receiving 50 μg/kg. The improved erectile function following DHA-nanoemulsion treatment was associated with increased expression of neuronal nitric oxide synthase (nNOS), axon numbers, and smooth muscle cell content.
Studies have also investigated the potential of a microtubule regulator, termed Fidgetin-like 2 (FL2), to promote CN regrowth following injury. Targeted FL2 depletion via topical application of FL2-siRNA encapsulated in silane-hydrogel nanoparticle formulations (FL2-siRNA-nanoparticle) has been shown to promote the closure and repair of cutaneous wounds in a mouse model. 58 More recently, in a rat model of CN crush or transection injury, it was reported that when FL2-siRNA-nanoparticle was applied at the time and site of injury, there was an improved erectile response (determined by ICP/BP) compared with controls beginning at 2 weeks post-treatment. 59 Potentially, from a translation point, application of this formulation at the time of prostatectomy could bring dual benefits by combining CN regeneration with an overall increase in the rate of wound healing.
Prevent radiation-induced ED following prostate cancer radiation therapy
More than half of the patients who receive radiation therapy (RT) for prostate cancer will develop radiation-induced erectile dysfunction (RiED) within 3–5 years after treatment. 60 One study used rat models to investigate whether BIO 300, a synthetic nanosuspension of genistein (naturally occurring soy isoflavone that serves as an estrogen receptor beta selective agonist), improves the therapeutic index in prostate cancer treatment by preventing RiED without sacrificing tumor radiosensitivity. Investigators found that BIO 300 did not protect tumors from RT-induced cytotoxicity but prevented RiED when administered 3 days before RT and daily for up to 2 weeks post RT, and also reduced the development of RiED when administered 2 h after starting RT. 61 Therefore, if translated to clinical studies, this novel nanotechnology-based approach may offer an alternative therapeutic modality to preserve erectile function in men with prostate cancer undergoing RT while maintaining tumor response to RT.
Modulation of diabetic inflammatory response
Systemic inflammation is linked to the development of ED in those with diabetes. 62 Several studies in animal models have demonstrated the potential of oral administration of curcumin (a naturally occurring compound, which has anti-inflammatory, anti-oxidative stress, and anti-catabolic activity) to treat ED associated with diabetes.63–66 However, clinical translation of curcumin-based therapies has been limited because of its low oral bioavailability, poor aqueous solubility, and rapid degradation. Some of these shortcomings could be mitigated when curcumin is encapsulated into a silane-hydrogel nanoparticle (curcumin-nanoparticle) which allows slow and sustained transdermal delivery of curcumin, thereby avoiding first-pass metabolism. 67 In studies with a rat model of type II diabetes, it was shown that curcumin-nanoparticles penetrate the abdominal epidermis and persist in hair follicles for at least 24 h. 67 Diabetic animals treated with curcumin-nanoparticles, compared with animals treated with blank nanoparticles, exhibited higher average ICP/BP following CN stimulation, reaching statistical significance at 0.75 mA. This study showed that there was also an effect of curcumin-nanoparticle treatment on inflammatory markers in the diabetic rat corporal tissue. For example, NF-activating protein (NFKAP) expression was decreased by 60% and heme oxygenase-1 (HO-1) expression was increased by 60% in curcumin- compared with blank-nanoparticle-treated animals. 67 The mechanism underlying the improvement in erectile function following curcumin-nanoparticle treatment was not determined in these experiments. However, the fact that ICP/BP values were inversely correlated with NFKAP, and directly correlated with HO-1 expression in corporal tissue, suggests a reduction in local inflammation may be one of the mechanisms by which curcumin-nanoparticles improve erectile function in diabetic rats.
Benign bladder dysfunction
Patients with non-oncologic bladder conditions, such as OAB and IC/BPS, are treated with oral and intravesical medications, and ultimately surgery if they do not respond to or cannot tolerate the standard treatment options. Oral medications often produce off-target effects and intravesical uptake of therapeutics is often limited by the tight permeability barrier formed by the umbrella cells that line the luminal surface of bladder and by the reduced dwelling time of drugs in the bladder lumen. 68 Nanotechnology provides a platform for development of more efficient and bladder-targeted drug delivery systems that can circumvent these limitations and significantly advance management of these bladder conditions. Unlike studies in ED, in which the potential of nanocarrier-based therapeutics has only been investigated in animals, use of this technology for pathologies related to benign bladder dysfunction has been investigated at both preclinical and clinical levels.
Overactive bladder
Background
OAB is defined by the International Continence Society as a storage symptom syndrome of urgency, with or without urgency urinary incontinence, which is usually accompanied with increased daytime frequency and nocturia. 69 Approximately 3–46% of the population is affected by OAB, with a higher prevalence among women and the elderly. It is associated with a significant decrease in a patient’s quality of life. 70
Current oral and intravesical therapeutics
The mainstay treatment for patients with OAB is anticholinergic pharmacotherapy. However, oral use of these drugs is associated with considerable adverse side effects such as dry mouth and constipation that often result in poor patient compliance. 71 Conversely, intravesical administration of antimuscarinics has been reported to result in less frequent and less intense side effects.72–78 However, despite its efficacy and safety, intravesical treatment with antimuscarinics also has shortcomings. An important one is the unavoidable drug dilution and low retention in the bladder caused by urine buildup and voiding, entailing the need for frequent catheterizations. In this regard, this route of treatment would only be ideal for patients who already perform clean intermittent catheterization (CIC).
The development of intravesical treatment with botulinum neurotoxin A (BoNT-A) circumvented some of the limitations faced with use of anticholinergics. BoNT-A is a neurotoxin that binds with high affinity to the synaptic vesicle proteins of peripheral cholinergic nerve endings and inhibits neuromuscular and parasympathetic signaling by preventing release of acetylcholine into the synapse. Like antimuscarinics, BoNT-A inhibits detrusor contractions by targeting the bladder cholinergic system. However, because BoNT-A is directly injected into the detrusor muscle, it has the advantage of not only acting locally but also lasting longer, for about 6–8 months. 79 However, BoNT-A intravesical treatment also requires reinjections to maintain treatment efficacy and have been associated with considerable rates of urinary retention, UTI, hematuria, and pain at the injection site.80,81
Nanotechnology-driven therapeutics
Improving intravesical BoNT-A and capsaicin drug delivery
Use of liposomes (LP) to encapsulate and deliver BoNT-A through intravesical instillation has been tested in rats and shown to provide an efficient and alternative approach for BoNT-A delivery without bladder wall injection. Liposomes are self-assembled vesicles formed by synthetic or natural phospholipid bilayers that enclose an interior aqueous space. This unique structure allows encapsulation and transport of lipophilic cargos within the lipid bilayer and/or hydrophilic cargos in the aqueous compartment. Since the lipidic bilayer structure of LPs is similar to that of biological membranes, they can easily adhere to the cell surface and deliver their cargos by endocytosis. The liposomal BoNT-A (lipo-BoNT) thereby displayed high affinity for the urothelium without causing significant irritation. 82 A subsequent study investigated the use of lipo-BoNT in 24 OAB patients in a single-center, double-blind, randomized, parallel controlled trial. At 1 month, the lipo-BoNT group had a significant improvement in urinary frequency and reduction in urgency episodes when compared with the control group (treated with normal saline). Importantly, the treatment was well-tolerated with no significant adverse events related to lipo-BoNT or the instillation procedure. 83 Moreover, a multicenter, double-blind, randomized, placebo-controlled study enrolled 62 patients with OAB, who were refractory to anticholinergic oral medications, and found that at 1 month, the lipo-BoNT group had significant reductions in urinary urgency when compared with the control group. Similar to the previous study, lipo-BoNT was concluded to be safe as there was no increased risk of urinary retention and UTI. 84 Therefore, these studies have further supported lipo-BoNT to not only be efficacious in reducing OAB symptoms, but also to be safer than BoNT-A intravesical injections.
Intravesical administration of capsaicin, the pungent ingredient in red pepper, has been studied in rats as an approach to manage OAB by desensitizing the afferent C-fibers, thereby resulting in a decrease in the micturition reflex and bladder overactivity. Because capsaicin is hydrophobic, intravesical administration of capsaicin involves normal saline solution with 30% ethanol. However, the high ethanol concentration has been shown to induce histological epithelium thinning and submucosal edema. 85 In order to avoid solubilizing capsaicin through ethanol, one study encapsulated capsaicin within the hydrophobic core of LPs prior to intravesical instillation. Findings from this study demonstrated that intravesical treatment of rats with LP-encapsulated capsaicin resulted in complete blockage of the micturition reflex without any adverse histological changes. 86
Using antisense oligonucleotides to downregulate NGF expression
Overexpression of NGF in the rat bladder has been shown to cause hyperexcitability of C-fiber sensory pathways, subsequently contributing to OAB symptoms. 87 Currently, downregulation of NGF is encouraged through intravenous administration of monoclonal human NGF antibodies. Although these antibodies have been shown to be effective in relieving lower urinary tract symptoms, off-target effects resulted in considerable rates of paresthesia, hypoesthesia, and arthralgia. 88 One study investigated local downregulation of NGF in the rat bladder through intravesical delivery of antisense oligonucleotides. 89 Since the oligonucleotides were not able to penetrate the urothelium barrier and therefore to not exert their effects, the investigators encapsulated the oligonucleotides into LPs. As a result, the LP-encapsulated oligonucleotides were not only able to penetrate the urothelial cells, but also to significantly decrease NGF expression and bladder overactivity while avoiding off-target effects. 89
Targeting the NO and RhoA/Rho-associated kinase pathway
The prevalence of OAB in patients with sickle cell disease (SCD), an inherited disorder characterized by an abnormal structure of one of the globin chains of the Hb molecules, can be as high as 40%. 90 Physiologically, SCD patients are susceptible to experiencing OAB symptoms as SCD creates a state of chronic low bioavailability of NO that is accompanied by dysregulation not only of NO signaling but also of the RhoA/ROCK-signaling pathways, which have been proposed to contribute to detrusor overactivity and urethral dysfunction.91,92 A recent study explored the potential use of intravesical delivery of NO-releasing nanoparticles (NO-NPs) to improve bladder function in transgenic SCD mice. 93 This study demonstrated that intravesical NO essentially reversed the OAB phenotype in SCD mice. The frequency of voiding and non-voiding bladder contractions was reduced in NO-NP-treated SCD mice and the molecular imbalance observed in the NO and RhoA/ROCK signaling pathways was corrected when compared with wild-type mice. 93
Optimizing oral drug delivery
Mirabegron (MBN) is a sympathomimetic β-3 adrenergic receptor agonist used to orally treat OAB. However, the first-pass metabolism effect, low water solubility, and MBN high protein binding diminish its bioavailability. A study recently sought to increase MBN’s oral bioavailability by encapsulating MBN into polyethylene glycol nanoparticles (MBN-PEG-NP). 94 The MBN-PEG-NP were prepared through the pre-emulsion ultrasonication method and then evaluated for particle size, zeta potential, entrapment efficiency, and in vitro release. After optimization, MBN-PEG-NP were given orally to rats. Investigators found that the bioavailability of MBN-PEG-NP was twofold greater than the bioavailability of MBN without PEG-NP. These findings suggest that the nanoparticles were absorbed through intestinal lymphatics since they resembled chylomicrons in the gut, which would reduce the MBN first-pass metabolism effect. 94 Although novel and exciting, additional studies are warranted to investigate the drug’s toxicity since its clearance and distribution have been altered once encapsulated in PEG-NP.
Interstitial cystitis/bladder pain syndrome (IC/BPS)
Background
The Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction characterizes IC/BPS as an unpleasant sensation in the bladder consisting of pain, pressure, or discomfort that is longer than 6 weeks and does not have a clear identifiable cause. 95 Like OAB, IC/BPS can significantly decrease quality of life and is more prevalent in women than in men, affecting 2.7–6.5% of women versus 2.9–4.2% of men.96,97 There are no objective diagnostic tests and cystoscopic findings are nonspecific. Therefore, diagnosis of IC/BPS is based on subjective patient-reported symptoms.96,98
Current therapeutics
The etiology of IC/BPS is not entirely known. Among possible triggers are infection, autoimmune response, allergic reaction, neurogenic inflammation, urothelial dysfunction, and genetic predisposition. 98 Because of its multifactorial nature, various therapeutic approaches are recommended to manage IC/BPS, with the main goal of maximizing symptom control and patient quality of life while minimizing adverse events and patient burden.99,100 Unfortunately, there is no single therapeutic agent for IC/BPS and patients often require conservative therapeutic approaches that include behavioral modifications (e.g. avoidance of foods and fluids known to be bladder irritants), use of anti-allergic and anti-inflammatory drugs, pain modulators, anticholinergics, botulinum toxin, and drugs that protect the bladder mucosa. Bladder distension, neuromodulation, and surgical interventions are recommended when all other treatments fail to control symptoms within an acceptable level.
Oral medications commonly used to manage IC/BPS symptoms, including amitriptyline, cimetidine, hydroxyzine, and pentosan polysulfate [only one that is Food and Drug Administration (FDA)-approved], have shown limited efficacy and conflicting evidence supporting their therapeutic effect. In addition, these medications have well-documented undesirable side effects such as nausea, constipation, dry mouth, weight gain, diarrhea, vomiting, and lightheadedness that are in some cases seen in about 80% of patients. 101 Intravesical drug instillation is another route of drug administration and is generally more effective than oral medications. Some intravesical drugs, such as heparin and hyaluronic acid, aim at reconstituting the glycosaminoglycan (GAG) layer of urothelium, which is proposed to be impaired in patients with IC/BPS.102–104 Due to its anti-inflammatory and analgesic effects, dimethyl sulfoxide (DMSO) is the most common and only FDA-approved intravesical medication. However, unclear optimal dose and frequency of dosing combined with pain upon instillation makes DMSO an unattractive therapeutic agent. 101
Nanotechnology-driven therapeutics
Protecting and repairing the urothelial layer with empty liposomes
The first studies exploring the application of nanotherapeutics in IC/BPS focused on investigating the effects of intravesical treatment with LPs composed only by phospholipids (‘empty’ LPs). 105 These studies were based on the ability of LPs to form a molecular film on cell surfaces and were further motivated by reports describing the healing and regeneration properties of topically applied empty LPs in the treatment of ophthalmic conditions.105,106 One of these early preclinical studies revealed that intravesical instillation of empty LPs protected against irritants (including potassium chloride, protamine sulfate, and acetic acid), indicating an increase in the urothelial barrier properties. 102 When tested in humans, a study with 14 patients with IC/BPS showed that intravesical treatment with empty LPs composed of sphingomyelin resulted in significant improvements in pain, urgency, and overall symptoms without treatment-related adverse events. 107 A subsequent clinical study with 24 patients with IC/BPS evaluated the safety and efficacy of empty LPs when compared with oral pentosan polysulfate. 106 Both treatments were reported to result in comparable efficacy in reducing the IC/BPS symptoms of urinary frequency, nocturia, pain, and urgency. The mechanism of action of empty LPs in the bladder still remains to be fully elucidated, but their beneficial effect likely results from the combined protective action of the physical coating of the urothelium and the anti-inflammatory properties of sphingomyelin that would promote repair and regeneration of the bladder lining. 105
Improving intravesical BoNT-A and tacrolimus drug delivery
As with OAB, BoNT-A is also used for treating IC/BPS.99,108 The mechanism underlying the reported effects of BoNT-A in ameliorating IC/BPS symptoms seems to involve not only the inhibition of synaptic release of acetylcholine that reduces detrusor overactivity but also the blockade of release of pro-inflammatory neurotransmitters, such as substance P and calcitonin gene–related peptide (CGRP). A recent preclinical study evaluated the use of lipo-BoNT for the treatment of Ketamine-induced cystitis in rat models. 109 The investigators observed that intravesical lipo-BoNT improved suburothelial hemorrhage, recovered urothelial tight junction and adhesion proteins, decreased substance P release in the urothelium, and inhibited the overexpression of inflammatory mediators. 109 Although findings from this study using lipo-BoNT to treat Ketamine-induced cystitis in a rat model were promising, using lipo-BoNT to treat refractory IC/BPS in humans revealed conflicting results. A multicenter, double-blind, randomized control trial used lipo-BoNT to treat 96 patients with refractory IC/BPS. Although lipo-BoNT was well-tolerated and improved symptoms of IC/BPS, there was no difference in outcomes between the treatment and control groups. The investigators hypothesized that this may be due to lipo-BoNT not being able to penetrate the detrusor deep enough as it would through injection. 110
Hemorrhagic cystitis is commonly observed as a side effect in patients undergoing chemotherapy with cyclophosphamide/ifosfamide or pelvic radiation therapy. Tacrolimus, a potent hydrophobic immunosuppressant, helps treat hemorrhagic cystitis by interfering with interleukin-2-dependent T-cell activation through the inhibition of calcineurin phosphatase. 111 However, treatment with this drug is accompanied by serious systemic side effects, including neurotoxicity, nephrotoxicity, and hypertension, and intravesical administration is made difficult by its high hydrophobicity. To circumvent these problems, the potential use of liposomes to deliver tacrolimus through intravesical instillation was explored in a study conducted with an animal model of cyclophosphamide-induced cystitis. 112 Findings from this study demonstrated that intravesical instillation of liposome-encapsulated tacrolimus (lipo-tacrolimus) reduced bladder overactivity and suppressed cyclophosphamide-induced bladder inflammation in rats, reducing the expression of prostaglandin E receptor 4 (EP4) and normalizing the levels of interleukin 2 and PGE2 in the bladder tissue and urine. 112 A subsequent study evaluated the pharmacokinetics of lipo-tacrolimus in rats and reported that a single intravesical lipo-tacrolimus administration of 200 μg/ml can produce active drug levels in the bladder for 24 h, therefore increasing its residence time while also significantly decreasing systemic exposure. 113 Furthermore, a more recent study examined using lipo-tacrolimus to treat radiation-induced cystitis in rats. 114 Four weeks after receiving radiation, the rats were treated with a lipo-tacrolimus instillation and the investigators observed that the inter-micturition interval, which significantly decreased after radiation, returned to baseline after treatment. In addition, histology of the lipo-tacrolimus-treated bladder was identical to a normal bladder, whereas the control group showed changes associated with cystitis. 114
Using antisense oligonucleotides to downregulate NGF expression
Several studies have reported elevated urinary levels of NGF in patients with IC/BPS that would originate from its upregulated expression and release from bladder urothelial and detrusor smooth muscle cells.115–118 Urinary NGF levels have been shown to correlate with the severity of pain and urgency symptoms and proposed to play a role in IC/BPS pathophysiology by sensitizing the bladder afferents, causing bladder overactivity and nociceptive responses.119–122 One study examined the potential of using liposomes for intravesical delivery of NGF antisense oligonucleotides (LP-OND) in a rat model of IC/BPS induced by intravesical instillation of hydrogen peroxide. 123 In this animal model, IC/BPS-like symptoms are evident as increases in micturition, freezing behavior (motionless head turning toward lower abdomen), bladder weight, infiltration of inflammatory cells, submucosal bladder bleeding, and NGF protein overexpression. After intravesical administration of LP-OND, all the above-mentioned findings significantly improved. 123 The study also observed that LPs delivered the NGF antisense OND into the urothelium and not the detrusor, suggesting that urothelium NGF alone may be contributing to IC/BPS. 123 Therefore, if translated to clinical studies, use of nanocarriers to modulate the expression of urothelial NGF may prove to be highly beneficial to manage not only OAB but also IC/BPS symptoms.
Catheter-associated UTIs
Background
Urinary catheterization (UC) is a common healthcare procedure. Up to 25% of hospitalized patients have an indwelling urinary catheter placed sometime during their hospital stay, and about 10% of all residents receiving care in nursing homes live with an indwelling catheter.124,125 UC is also the treatment of choice for individuals with difficulty in emptying their bladders due to neurogenic and non-neurogenic causes. For those patients, intermittent self-catheterization is the optimal procedure, but many individuals also require short- or long-term use of indwelling catheters. The major concern with UC is the associated risk of UTIs. UTIs are the most common type of healthcare-associated infection reported to the National Healthcare Safety Network (NHSN). UTIs account for about 40% of nosocomial infections, and about 80% of these are CAUTIs.126,127 The pathogenesis of CAUTIs relates mainly to the susceptibility of the catheter materials to bacterial colonization and biofilm formation. 128 The presence of bacterial biofilm also complicates the treatment of CAUTIs. The biofilm protects the bacteria, and minimal concentrations of antibiotics required to combat the bacteria embedded in the biofilm may be 101–104 times higher than against the planktonic bacteria. 129 The emergence and spread of multidrug resistant bacteria are thus constant concerns with the treatment of CAUTIs.
Current therapeutics
Current approaches to reduce the risk of CAUTIs are limited and are based mainly on recommendations to reduce urinary catheter use and duration of use in all patients, which is not always possible, particularly in individuals who require chronic catheterization. Research efforts have thus focused on identifying antimicrobial agents that can be used other than antibiotics, and novel catheter materials and coatings that can deter the formation of bacterial biofilms. Silver ions are the most extensively studied catheter coatings. By the release of silver ions into the bladder after catheter placement, oxidative stress is induced, which disrupts bacteria’s cell membrane and proteins. 130 However, animal studies and clinical trials showed mixed results in terms of efficacy and were not cost-effective when compared with nitrofurazone-coated catheters, silicone catheters, and other standard catheters.131–136 Translation to the clinic falls short due to several factors, but for the most part, due to the lack of testing in animal models of CAUTIs. Catheterization changes the bladder environment generating ideal conditions for bacterial colonization.137–139 Antimicrobial agents may also indirectly alter the bladder environment, the bladder function, and the bladder responses to infection. Unfortunately, in vitro studies cannot simulate these changes or indirect effects of treatment, neither can they account for the complexity of the host–catheter–microbe interactions.
Nanotechnology-driven therapeutics
Decreasing antimicrobial activity through catheters with metal-coated nanoparticles
Coating catheters with silver, copper, and zinc-doped copper oxide (CuO) as nanoparticles have been investigated. Silver nanoparticles have the same mechanism of action as mentioned above, and although studies have shown catheters coated with silver nanoparticles to inhibit biofilm formation and growth of several bacteria when compared with uncoated catheters, inflammation and toxicity assessment are limited due to the lack of testing in animal models of CAUTIs.140,141 Copper nanoparticles decrease antimicrobial activity by crossing bacterial cell membranes and then damaging bacterial anti-apoptotic enzymes. 142 An in vitro study investigated the antimicrobial activity of copper nanoparticles against Escherichia coli and found that by 2 min after the bacteria incubation with the copper nanoparticle–coated catheter, no viable bacteria was recovered. 143 Furthermore, zinc-doped CuO nanoparticles, whose antimicrobial mechanism is currently under investigation, have been investigated in one study with rabbit models of CAUTIs. 144 The study observed that after rabbits were catheterized with uncoated and coated catheters, and infected for 7 days, there was a significant reduction in bacterial biofilm formation. In addition, urine samples, which were collected throughout catheterization, revealed lower bacteria formation in animals catheterized with the Zn-doped CuO-coated catheter. 144 Although the results from these early studies are quite promising, clinical trials are needed to assess toxicity.
Delivery of NO through nanoparticles
NO is well-known for its broad-spectrum antifungal and antibacterial activity.32,33 Increased inducible NOS (iNOS) activity and nitrite levels have been documented in the bladder and urine of patients suffering from UTI. NO has therefore been suggested to play an important role in development and host response to UTIs.145,146 Intravesical use of NO, instead of conventional antibiotics to treat and prevent CAUTI, would be advantageous given its broad-spectrum bactericidal activity without risk of developing multidrug resistant (MDR) bacteria. NO, however, is a highly reactive gas proven to be difficult to deliver conveniently, and this has largely precluded its clinical use, even in hospital settings. NO-delivering systems, such as NO nanoparticles, have proven ideal in circumventing this limitation. Studies have shown that NO nanoparticles interfere with or prevent adhesion and biofilm formation of Staphylococcus aureus and Candida albicans on intravenous catheters using rodent models of implanted catheters.147,148 Moreover, moieties that can be included in the nanoparticle composition, like chitosan, can further disrupt biofilms formed on intravenous catheters.149,150 However, testing of their therapeutic potential in preclinical animal models of CAUTI has not been performed and represents a major barrier to clinical translation. Nevertheless, there are currently ongoing studies to evaluate nanoparticles for sustained and controlled intravesical NO release in animal models that mimic the conditions encountered by catheterized patients.
Additional potential usage of nanoparticles for benign urogenital pathologies
There have been significant advancements in the use of nanocarrier therapeutics for ED, OAB, IC/BPS, and CAUTIs because of the accessibility of the organs involved in these pathologies. Topical and intravesical application of nanocarrier-based therapies allows for a minimally invasive approach that is difficult to achieve for other benign urogenital pathologies, such as benign prostatic hyperplasia (BPH), due to anatomical barriers. As a result, a more invasive modality would have to be implemented to evaluate the potential benefits of using nanocarriers for such anatomically constrained conditions.
Nevertheless, two recent animal studies have investigated the use of nanoparticles in treating BPH. One study evaluated the effect of intraperitoneal injection of 50 and 20 nm gold nanoparticles (AuNps) in a rat model of testosterone-induced BPH. 151 Interestingly, findings from this study show size-dependent effects of AuNps treatment on BPH progression. While the 20-nm AuNps treatment inhibited prostatic cell proliferation, inflammation, and angiogenesis and ameliorated BPH, treatment with 50 nm AuNps enhanced prostatic inflammation and hyperplasia. 151 The other recent animal study used the ablative properties of laser nanoparticles (hyperthermia technique) to successfully irradiate and induce apoptosis of prostatic tissue in the rat model of testosterone-induced BPH. 152 However, since both studies failed to extensively evaluate the toxicity of nanoparticles on prostatic tissue, additional preclinical validation is needed before beginning clinical studies.
Discussion
There is increasing awareness and utilization of nanotechnology as a tool to advance basic and clinical research in the field of non-oncologic urogenital disease that is paving the way for development of novel therapeutic approaches (Figure 1, Table 1). Indeed, compared with diseases afflicting internal tissues and organs, urogenital diseases are often more amenable to nanocarrier-based therapeutics given the easy and direct access of urogenital organs by intravesical or transdermal administration. However, some specific barriers in applying this technology to urogenital disease arise from the dilution and washout of intravesically delivered therapeutics and transdermal penetration depth of topically applied nanocarriers. These drawbacks can be addressed by modifying the physicochemical composition of the nanocarriers and tailoring them for their intended target. For example, nanocarriers can be decorated with ligands that can recognize the target cell surface or include paramagnetic nanomaterials in its composition to target and hold the nanocarriers in place through externally applied magnets.153–155
In general, the use of nanocarriers to deliver therapeutic agents has several advantages over conventional drug administration routes. They allow local and transdermal applications of therapeutics that can bypass the first-pass metabolism associated with an oral route and improve drug efficacy through encapsulation of hydrophobic drugs, thereby lowering the effective therapeutic dose and reducing systemic toxicity. Furthermore, nanocarriers can allow for a sustained and controlled release of encapsulated agents. Most nanomaterial-based delivery vehicles are also highly malleable, such that they can deliver a great variety of therapeutic agents (including nucleic acids, naturally occurring and synthetic organic and inorganic therapeutics, and even gaseous compounds, such as NO), and formulations are relatively easily modified to change the release kinetic of therapeutic agents (Table 1).
In any disease and therapeutic focus area of investigation, studies with animal models of human diseases have been an instrumental and essential step in the research process before conducting clinical trials and translating findings to patients. However, it has become increasing evident that conclusions made based on preclinical studies cannot be directly transferred to human studies, as inherent species differences make extrapolation from animals to humans unreliable.156,157 Given that most preclinical research in the use of nanocarrier-based therapeutics has been conducted on small rodent models of disease, concerns can be raised related to significant differences between these small animals and humans in terms of skin and bladder wall thickness (in research where nanocarriers are investigated as transdermal or intravesical delivery systems) and organ/body size (in research related to externally placed magnets used in targeting paramagnetic nanoparticles). In addition, toxicity arising from changes in physicochemical properties of nanomaterials needs to be considered, even when the formulation of individual chemical components of the nanocarriers and therapeutic agents is considered to be safe.158,159
Conclusion
In conclusion, despite a lag in the application of nanotechnology in non-oncologic compared with oncologic disease, there is a growing body of literature supporting its use in research and in novel therapeutics. As a delivery system, nanocarriers have several advantages over more conventional delivery systems, and in many ways, urogenital diseases are more amenable to its application than other diseases. Although there remain several obstacles and unknowns to clinical translation, many of these are being researched and overcome in other fields, and it is only a matter of time before there is widespread application of this technology to advance treatment of non-oncologic urogenital diseases.
