Abstract
Objectives:
The purpose of this study is to determine the therapeutic value of surgery in individuals with urinary bladder signet ring cell carcinoma (SRCC). Surgery has not been examined as a prognostic factor for urinary bladder cancer (SRCC).
Materials and Methods:
Using the Surveillance, Epidemiology, and End Results program (SEER), patients with urinary bladder SRCC who presented from 1975 to 2018 were included in a retrospective study. The effect of surgical therapy on cause-specific survival (CSS) and overall survival (OS) was examined using univariate and multivariate Cox regression models. We subdivided 595 patients with SRCC into 2 groups, as follows: 496 who underwent surgery; and 99 who did not undergo surgery.
Results:
Males had high predominance in all cases in both groups (p = 0.04). Moderate and poor differentiation (III–IV) were observed in the majority of patients who underwent surgery (77.2 vs 58.6, p ⩽ 0.001) and had no insurance (p ⩽ 0.001). By using KM, the OS and CSS of the surgery group were found to be significantly better than those of the non-surgery group (p = 0.001,%) after adjusting for the variables of age, race, sex, primary site, grade, stage, lymph node removal, chemotherapy record, radiotherapy record, insurance, and marital status in the multivariate Cox proportional hazard model (hazard ratio [HR]= 0. 592; 95% confidence interval [CI] = 0.449–0.782; p = 0.0001). In comparison with chemotherapy and radiation, which resulted in poorer survival rates, surgery considerably improved survival outcomes in urinary bladder SRCC. The nomogram prediction model was built with C-index values of 0.70 and 73 for OS and CSS prediction, respectively. AUC in OS values were 0.77, 0.76, and 0.74, whereas AUC in CSS were 0.83, 0.80, and 0.79 for the 1-, 3-, and 5-year survival nomograms, respectively.
Conclusion:
Surgery was a significant independent predictor of bladder SRCC survival. Patients who underwent surgery had higher CSS and OS than people who did not undergo surgery. Surgery also led to better survival than the combination of the different treatment modalities.
Introduction
Bladder cancer (BCa) is the 11th most common type of cancer worldwide.1,2 According to the National Cancer Institute, the number of new BCa cases and deaths in the United States of America are predicted to reach 74,690 and 15,580, respectively, in 2014.3,4 Urothelial carcinoma (UC) is the most prevalent pathological type of bladder cancer, whereas primary signet ring cell carcinoma (SRCC) is a relatively uncommon kind of adenocarcinoma, accounting for only 0.24% of all bladder malignancies. Based on earlier classification, SRCC is the subtype with the worst prognosis. 5 Histologically, these tumors resemble signet rings. Considering their flat top and stamped or embossed mark, signet rings were used to seal correspondence. The accumulation of cytoplasmic mucin causes the nuclei to migrate to the periphery, giving them their signet cell form. 6 Based on observations of SRCC biological activity in gastric cancer, theories on how advanced disease pathology in SRCCs is inherited at the time of presentation have been established. 7 The diffuse-type tumors do not produce macroscopic or fungating lesions and are characterized by poor cell adhesion and lack of gland development; these tumors originated from signet ring cell, according to the Lauren classification developed in 1965. 8 In addition to their impairment of cell–cell adhesion, which results in their diffuse pattern of dissemination, the two most essential pathogenic aspects of SRCCs are mucin accumulation and cell–cell adhesion defects. 9 During the epithelial-to-mesenchymal transition (EMT) process of metastasis, the loss of E-cadherin expression is one of the first phases during which the damage of cell–cell connections promotes a more plastic cytoskeleton and anchorage-independent growth and survival during lymphatic and hematogenous cell spread. The mutations in the CDH1 gene likely contribute to the aggressiveness of SRCC in gastric. It plays a role in the onset of early-stage tumors. The protein called E-cadherin, which is determined by the CDH1, was discovered to exhibit truncations in diffuse gastric cancers for the first time in 1994. 10 Despite the fact that bladder SRCC is exceedingly malignant and commonly presents with a high-grade and bad prognosis, it is often overlooked because of its rarity; bladder cancer treatments are predominantly focused on UC.11,12 Large randomized controlled studies are difficult to conduct. Thus, consensus on treatment protocols is lacking. SRCC is believed to be resistant to chemotherapy and radiotherapy and has been linked to a bad prognosis. 13 Surgical resection remains the first consideration for managing colorectal SRCC patients. 14
However, few studies focused on the impact of treatment modalities on both CSS and OS of bladder SRCC. The Surveillance, Epidemiology, and End Results (SEER) is a comprehensive program that monitors incidence, prognosis, and cancer surveillance in the United States (US). SEER is funded by the National Cancer Institute’s (NCI) Division of Cancer Control and Population Sciences’ Surveillance Research Program (SRP) (DCCPS). Cancer data are collected through a coordinated system in 18 registries located throughout the United States. These regional registries cover around 35% of the United States’ population, representing the entire country’s demographics. In 1973, data gathering began with a few registrations and quickly expanded to encompass different places throughout the United States.15,16 We proposed studying treatment modalities for bladder SRCC and comparing surgery and non-surgery cases in 1975–2018 in 18 registries of the United States based on the SEER database.
Methods
Data source
Anonymised data were retrieved from the latest SEER registry 2018 (www.seer.cancer.gov) with additional treatment fields through SEER* Stat software (version 8.3.9), which was released on April 2021, based on the November 2018 submission, which almost covers 35% of the United States population. The SEER program orderly assembles the demographic, clinical, and follow-up information from 18 cancer registries. We applied for access to the SEER database after we applied for a signed agreement. Then, we made another application for the additional treatment fields, which we gained successfully.
Study population
We used a case listing session in the SEER database to extract patients if the histology was SRCC (ICD-O-3 8490/3). The diagnosis was confirmed by positive histology and was their first or only cancer diagnosis (a first positive indicator of malignancy). We included all microscopically confirmed (positive histology, exfoliative cytology, immunophenotyping and/or post genetic studies, positive microscopic confirm method non-specified) cases diagnosed with SRCC, aged more than 20 with only active follow up. We have measured the prediction ability of our study, and report the study based on the STROBE check-list for cohort studies (Supplementary Table).
Study primary measurement
This study’s primary measurement is to identify the impact of surgery on the CSS and OS of SRCC in the bladder and to conduct a detailed study of the survival rates between variables of these two groups. The study also aimed to construct a nomogram to predict the variable that affects CSS and OS of patients with a primary bladder SRCC.
Prognostic variables
The following variables were selected as the prognostic factors in our study: age at diagnosis, race, sex, primary site, marital status, grade, SEER historic stage, type of surgery, the reason for non-cancer-directed surgery, radiation, the sequence of radiation with surgery, information on chemotherapy treatment, SEER classification of other cause of death, lymph node removal, insurance status, marital status, and vital status record. The study period was calculated until the last update of the SEER database. The age groups were designated according to age at diagnosis (20–69 and ⩾ 70 years old). The cases were categorized according to marital status (married, unmarried, and others) and race (white, black, and others). The reason for non-cancer-directed surgery was used as an indicator of surgical history, and cases were grouped according to whether surgery was performed or not (not recommended, not performed, and unknown). The cases were also categorized depending on surgery status, as follows: no surgery, TUR-BT (transurethral resection for bladder tumors), partial cystectomy, radical cystectomy, pelvic exenteration, and other types of surgery. Local tumor destruction fulguration includes the use of hot forceps for tumor destruction. The cases were also categorized depending on radiation treatment, as follows: beam radiation, implants, and radiation not otherwise specified; no radiation; and other. Information on chemotherapy treatment, the cause of death by the same cancer, or other causes was also obtained. The demographic and tumor characteristics of patients in both groups were compared using χ2. OS based on the primary site was estimated by KM, and comparisons among groups were carried out by log-rank test. Univariate and multivariate Cox proportional hazards models were used to determine the effect of a prognostic variable on OS. The nomogram was constructed in the following manner. To begin, a univariate Cox proportional hazards model was used to determine the predictive ability of each parameter. Second, using a backward model selection approach, factors with a p-value of less than 0.05 in univariate analysis were further evaluated in a multivariate Cox proportional hazards model. The final model’s factors were used to construct the nomogram and risk classification system. A calibration plot was produced to evaluate the nomogram’s potential for clinical utility by comparing the observed and nomogram-predicted survival/recurrence. In addition, the nonadherence nomogram was confirmed by adjusting a comparatively corrected Harrell’s concordance index C-index using bootstrapping (1000 bootstrap resamples). The C-index was obtained using bootstrapping to determine the likelihood that the prognostic nomogram models are similar to the actual observed outcomes. The receiver operating characteristic (ROC) curves are used to compare the nomogram’s capacity to estimate patient mortality with that of the scoring criteria for OS and CSS in bladder SRCC. SPSS version 26.0 (SPSS Inc., Chicago, IL, USA) was used to determine statistical significance at p < 0.05 and had a limit of 0.0001. The predictive concert of the nomogram was measured by concordance index (C-index) performed in R version 3.3.3 (http://www.r-project.org/). The key outcome measures in this study were cancer-specific survival (CSS) and overall survival (OS). CSS was defined as the amount of time between the diagnosis and death due to bladder SRCC. The term ‘OS’ refers to the period defined as the interval between the time of diagnosis and the time of death from any cause.
Results
General patient’s characteristics
A total of 595 patients of bladder SRCC (496 surgery cases; and 99 non-surgery cases) were included. For the demographics, the therapeutic and tumor characteristics of both groups are summarized in Table 1. Of those who had cancer-directed surgery, 192 (38.6%) underwent TUR-BT alone, and 193 (32.3%) underwent cystectomy (partial or radical). A total of 106 (17.8%) patients underwent radiation therapy, and 150 (25.1%) had sequencing between surgery and radiation. Except for the age at diagnosis, race, radiotherapy, and chemotherapy, many variables were significantly different between the two groups. Males showed high predominance in all cases of both groups (p = 0.04). Moderate and poor differentiation (III-IV) were shown by the majority of patients who underwent surgery (77.2 vs 58.6, p ⩽ 0.001), had no lymph node removal (71.5% vs 66.9%, p < 0.001), had distant-stage non surgery vs surgery (53.0% vs 24.3%, p < 0.001), and had no insurance (65. 5% vs 67.9% p < 0.001). Cancer-directed surgery was conducted on 496 individuals (83.94%). However, SRCC demonstrated an independent factor for survival. The SRCC exhibited lower OS and CSS than the UC (HR = 1.51, 95% CI = 1.39–1.64, p = 0.0001) and CSS (HR = 1.40, 95% CI = 1.18–1.65, p = 0.001), as shown in Figure 1.
Demographic characteristics of bladder SRCC surgery and no surgery in the Unites State based on SEER database.
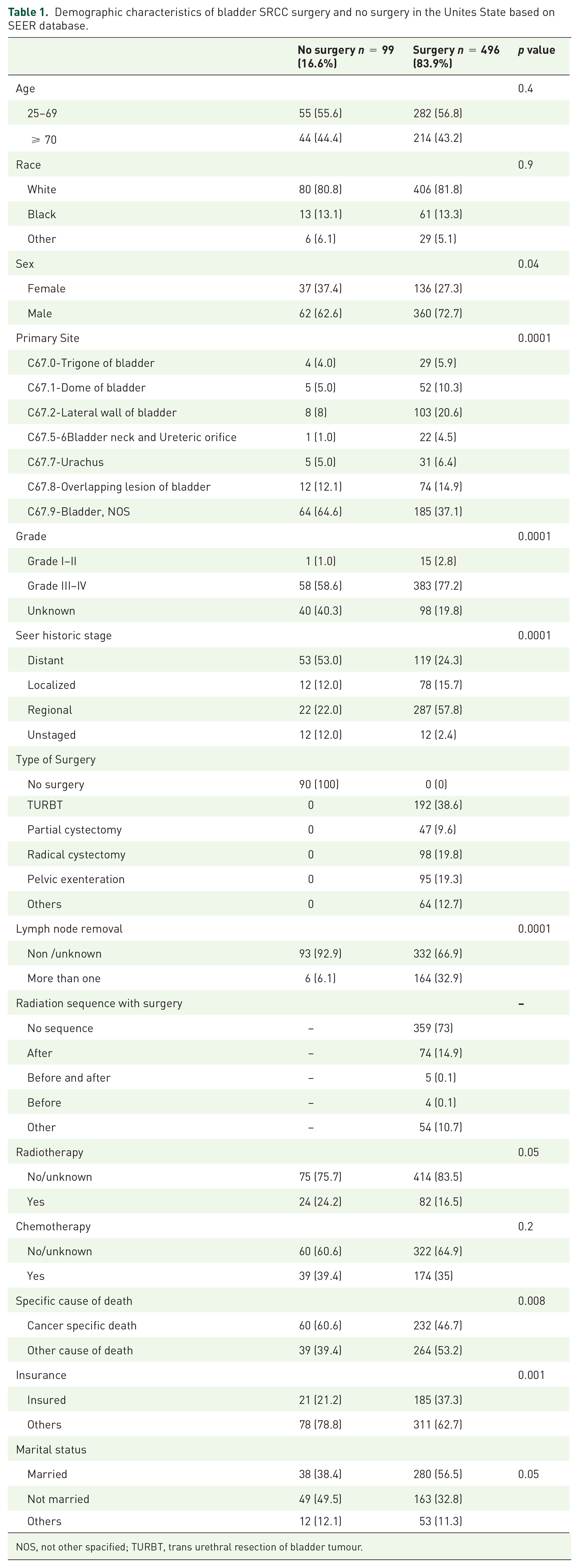
NOS, not other spacified; TURBT, trans urethral resection of bladder tumour.

(a) Cancer-specific survival and (b) overall survival of patients with SRCC and UC, respectively.
Survival analysis
In the study of bladder SRCC, the K–M curves revealed a significant difference in CSS and OS between surgery yes and surgery no. Patients who underwent surgery had superior OS and CSS. The median months CSS in surgery vs non-surgery were 20-median month CSS = 29.7% vs 5.0%, 60-median month = 4.4% vs 1.7%, and 100-median month 0.9% vs 0.0%. The median month OS values in surgery vs non-surgery were 20-median month = 37.6% vs 11.2%, 60-median month = 10% vs 2.1%, and 100-median month 3.4 vs 0% Figure 2. Even after the variables were adjusted for age, race, sex, primary site, grade, stage, lymph node removal, chemotherapy record, radiotherapy record, insurance, and marital status in multivariate Cox proportional hazard model (HR = 0. 592; 95% CI = 0.449–0.782; p = 0.0001), as shown in Figure 3(a). In addition, difference was detected in most of the other factors. Survival advantages were restricted to individuals who underwent surgery (e.g. male, white, black, chemotherapy, and radiotherapy status), as shown in Figure 3 and Table 2. The survival advantages were recorded in the non-surgery group in the following variables (female and site-trigon of bladder), as shown in Figures 3(b) and (f). In the univariate analysis of CSS and OS, we included all variables, such as age, race, sex, primary site, grade, stage, type of surgery, lymph node removal, radiation sequenced with surgery, surgery performed, radiotherapy record, chemotherapy record, insurance, and marital status. The variables found to be significant were included in the multivariate analysis. In the OS group, we found that age, primary site, stage, type of surgery, lymph node removal, and surgery performance were significant predictors of OS. In the CSS group, age, primary site, stage, type of surgery, chemotherapy, radiation sequence with surgery, surgery, radiation, insurance, and marital status were significant predictors of OS (p = 0.001). As a result, the surgery was a significantly protective factor for survival (CSS: hazard ratio (HR) = 0.285, 95% CI: 0.138–0.592, p = 0.001, OS: hazard ratio (HR): 0.572, 95% CI: 0.307–1.065, p ⩽ 0.001), as shown in Table 3.
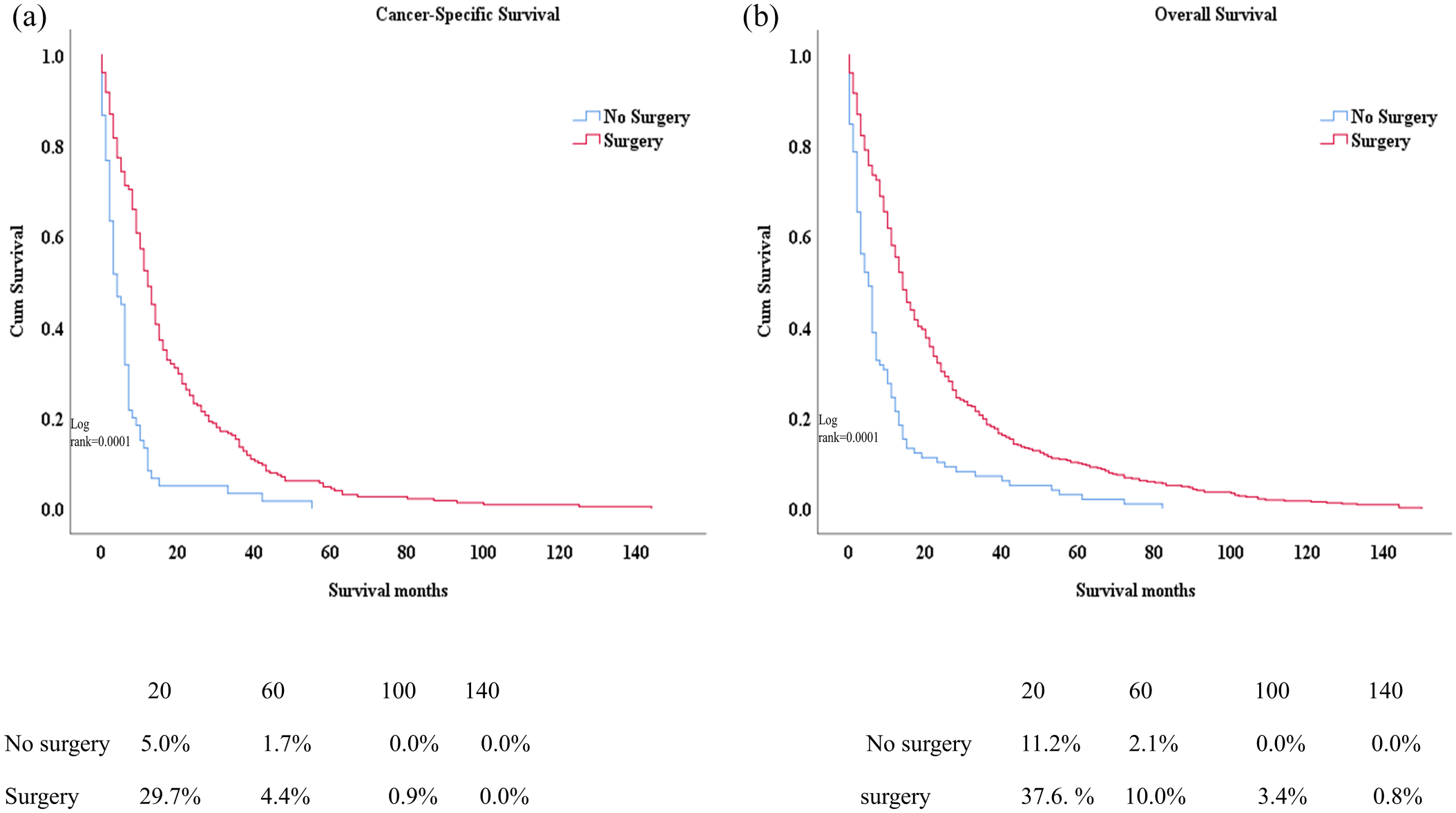
(a) Cancer-specific survival and (b) overall survival of patients underwent surgery vs non- surgery among SRCC patients.

(a) The surgery remain significant after the variables were adjusted for age, race, sex, primary site, grade, stage, lymph node removal, chemotherapy record, radiotherapy record, insurance, marital status in multivariate Cox proportional hazard model (HR = 0. 592; 95% CI = 0.449–0.782; p = 0.0001). (b) and (f) The survival advantages were recorded in the non-surgery group in the following variables (female, site-trigon of bladder) p = 0.0002,0.0005 respectively. (c–e), (g and h) A difference was detected in the majority of other factors, with survival advantages being restricted to individuals who underwent surgery (e.g. male, white, black, chemotherapy, and radiotherapy status) p = 0.0001.
Overall survival study using Kaplan-Meier analysis between the variables in the Unites State based on SEER database.

NOS, not other spacified; TURBT, trans urethral resection of bladder tumour.
Univariate and multivariate analyses of cancer special survival (CSS) and overall survival (OS) for 594 patients with bladder SRCC treated with surgery in the Unites State based on SEER database.

CI, confidence interval; CSS, cause-specific survival; NI, not indicated; NOS, not other spacified; HR, hazard ratio; OS, overall survival; TURBT, trans urethral resection of bladder tumour.
Treatment modalities
From 1975 to 2018, SEER retrieved data on SRCC treatment, which included surgery, radiation, and chemotherapy methods. The majority of patients with SRCC of bladder reported in the SEER had surgery; they accounted for about 83.4% of the total number of patients. Regarding chemotherapy treatments for patients with SRCC, no significant difference was found between patients who were administered adjuvant chemotherapy and those who were not treated. At the same time, radiotherapy variables revealed a higher number of cases who did not receive any radiation radiotherapy (approximately 71.2%) than those who received treatment (approximately 17.8%), as shown in Table 1. In terms of treatment modalities, no significant differences were found in the survival between chemotherapy and radiotherapy (p = 0.1, Figure 4(a)). Moreover, in the combination therapy, chemo-radio yes vs surgery yes revealed better survival in the surgery group. These results indicate that surgery was the most effective treatment modality for SRCC in the urinary bladder, as shown in Figure 4(b).

K-M In terms of treatment modalities, (a) there were no significant differences in the survival between chemotherapy and radiotherapy p = 0.1. (b) Moreover, in combination therapy, chemo-radio yes vs surgery yes revealed better survival in the Surgery yes group.
Subgroup analysis
Several subgroups were studied further to determine the effect of surgery on survival. Subgroup analysis found that the surgical population outperformed the non-surgical population in terms of OS in virtually all subgroups. Specifically, in no/unknown chemotherapy and marital status subgroups, patients could significantly benefit from surgery in terms of CSS (all p ⩽ 0.05), as shown in Figure 5.

Subgroup analysis demonstrated superior OS populations underwent surgery than the non-surgery group in nearly all subgroups. Specifically, in no/unknown chemotherapy and marital status subgroups, patients could significantly benefit from surgery in terms of CSS (all p < 0.05).
Building the novel nomogram for the overall survival and cancer-specific survival
A nomogram model was built based on the prognostic factors as significantly positive predictors of survival in OS and CSS multivariable cox regression. Each prognostic parameter was assigned a score based on its prognostic value, and the sum total of the scores was used to predict 1-, 3-, and 5-year survival. The sum of the scores for all factors was turned into an estimate of the likelihood of death. In the case of SRCC of the bladder, the C-indexes of the prognostic nomogram for overall survival and the cancer-specific survival prediction were 0.70 and 0.73, respectively (Figure 6(a) and (b)). The prediction model revealed that stage and surgery type were the most critical factors influencing prognosis in the OS. In CSS, the radiation sequence with surgery was followed by surgery and type of surgery. For each variable, a grade was assigned based on its level of complexity. Individuals’ total scores were calculated in accordance with the scores assigned to different values of each variable, and the 1-, 3-, and 5-year survival rates of individuals were calculated. To evaluate the nomogram’s performance, 1-, 3-, and 5-year receiver operating characteristic (ROC) curves were created. Figure 6(c) and (d) illustrate our innovative nomogram model. The assessment revealed significantly improved prediction accuracy. AUC in OS values of 0.77, 0.76, and 0.74 and AUC in CSS values of 0.83, 0.80, and 0.79 were obtained for the 1-, 3-, and 5-year survival nomograms, respectively.

After summing the points corresponding to each factor, including patient-related variables (a)
Discussion
SRCC has a poor prognosis and is one of the most aggressive malignancies. Signet ring cells are typically seen in the colon, rectum, stomach, prostate, and bladder with several metastatic sites. 17 Certain adenocarcinomas of the urinary bladder produce mucin in large amounts and are referred to as mucinous adenocarcinomas or gelatinous or colloid carcinomas depending on their location. 18 However, the Signet ring variation was previously classified in the adenocarcinoma group of variations in the earlier classification in 2004. It has recently been grouped with the plasmacytoid variety as a distinct entity in the 2016 WHO classification of urothelial carcinoma; the most significant differential diagnoses for SRCC is plasmacytoid urothelial carcinoma.19,20
The CDH1 (Cadherin-1) gene and its protein E-cadherin are responsible for the aggressiveness of gastric SRCC; Cadherin-1 mutations contribute to tumor initiation early in the disease course. 21 Given the rarity of CDH1 mutations, it is challenging to associate them with SRCC locations. Colorectal SRCC, for example, has more BRAF and KRAS gene aggregation than predicted tumors, but it has been linked to a history of inflammatory colitis. Methylation of the CDH1 promoter is found in 93% of dysplastic biopsies, compared with 6% of non-dysplastic biopsies in ulcerative colitis patients. 22 Given the rarity of SRCC, previous research has focused on case studies and singular institutional experiences, which may not adequately reflect the natural history of ‘real world’ patients. Large cancer referral centers may have a higher number of advanced and recurring cancers than smaller centers, but they also have a larger healthy population that can attend these facilities.23,24 We used the latest update of the SEER program. This study examined the largest collection of bladder SRCC reported to date, and this investigation was verified by the unfinished results previously through case series and limited database studies.25,26 In the study of prognosis of SRCC, variables such as age, primary site, stage, type of surgery, surgery performance, radiotherapy, and lymph node removal were significantly positive prognostic factors. However, when multivariate variables, such as age, surgery, and distant stage, were included, highly significant survival predictions were obtained. In addition, in comparison with UC, SRCC had a significantly lower OS. The findings revealed that SRCC might be an independent prognostic factor in patients with urinary bladder. In comparison with the study by Jin et al., 26 our study included more patients, for example, females with SRCC (29%, 172/595 vs 27.4%, 86/318). Regarding treatment, our study population provided extensive descriptions of chemotherapy and surgery, which was one of the previous study’s limitations. 26 It is vital to use a multidisciplinary approach in the therapy of bladder SRCC to establish the most effective treatment choices based on the natural history and the tumor’s prognostic characteristics. As of this moment, an effective treatment for SRCC of the urinary bladder is lacking. 25 SRCC of the urinary bladder is resistant to typical chemotherapy regimens used to treat advanced urothelial carcinoma. Investigations evaluating various chemotherapy regimens for this uncommon neoplasm are lacking due to the small number of cases. In our study, chemotherapy enhanced the OS for the SRCC, p = 0.001, but it has a negative prediction for CSS survival (1.125; 0.965, 1.312; p = 0.1), which could be explained by the high metastatic rate of this kind of tumor. Similar to our study, Luzzago et al. 27 found that chemotherapy had a negligible effect on survival in patients with no distant cancer. However, chemotherapeutic regimens, including cisplatin and gemcitabine or methotrexate, vinblastine, cisplatin, and doxorubicin, which are being used in the treatment of advanced urothelial carcinoma, are examples of conventional treatments. SRCC has demonstrated resistance to therapy. 28 Moreover, some individuals did not react to standard platinum-based chemotherapy regimens, including capecitabine or 5-fluorouracil-based regimens, despite that colon cancer type regimens have high therapeutic benefit. We found that chemotherapy is a negative predictor for both OS and CSS. 29 Interestingly, we found that the combination of radiotherapy and chemotherapy treatment modalities showed lower survival than surgery (p < 0.001); this may be explained by the radiosensitivity of urinary bladder SRCC.30,31 In our study, patients with urinary bladder SRCC who underwent surgery showed significantly better OS and CSS compared with those that did not undergo surgery. Even after we adjusted all the variables in the multivariable test, the group that underwent surgery preserved the high significance. Most of the patients with bladder SRCC underwent surgery (81.1%). 25 TUR-BT is the most common procedure, followed by partial and radical cystectomy. However, patients who received TURBT had a median survival of 11 months, whereas those who underwent partial and total cystectomy had better survival (median, 43 months). This raises the possibility that cystectomy is underutilized in this population. Considering that the cystectomy recommendation is based on expert opinion rather than data from randomized clinical studies, the urological community may seriously doubt the most appropriate clinical therapy for patients with SRCC due to the lack of adequate evidence to support the recommendation. Some scholars proposed that we should follow the treatment procedures for gastric SRCC, in which surgery is the first choice of treatment; such proposal is based on the studies that bladder and gastric SRCCs are comparable.32,33
Our study showed that the average C-index values of the existing models of both OS and CSS were 0.70 and 0.73, respectively. This is considered a unique model, because to the author’s knowledge, this is the first study that discusses the prediction of the OS and CSS of bladder SRCC. A nomogram model of OS and CSS was built using two independent prognostic predictors in association with age, primary site, stage, surgery type, lymph node removal, and surgery in OS, whereas age, primary site, stage surgery type, chemotherapy, radiation sequence with surgery, radiation, surgery, insurance, and marital status in CSS were used to predict the 1-, 3-, and 5-year events. We confirmed that the novel nomogram model had a much greater capability of predicting events after 1, 3, and 5 years. To explain CSS, the case of a 70-year-old patient who was single and had a TUR-BT was considered. By calculating the patient’s nomogram, the patient’s 1-year risk of death (AUC of 0.83) was nearly 20%, the 3-year risk of death was nearly 45%, and the 5-year risk of death was nearly 60%.
Numerous limitations were inherent in our research. First, there was selection bias in this study, because it was a retrospective analysis. 34 The effects of checkpoint inhibitors and genetic research provide a reason for optimism in the treatment of bladder cancer, and these have not yet been thoroughly investigated. 35 Extramural vascular invasion and obstruction/occlusion status, as well as microsatellite stability/instability (MSS/MSI) status, are all missing from the SEER database. Although further details would be preferable, we assumed that the currently available SEER database data would be adequate for our research. Future research should address the aforementioned concerns. Our research had some benefits. Prior to doing any multiform analysis, we collected enough samples from 594 patients with SRCC of the urinary bladder who presented between 1975 and 2018. Subgroup, univariate, and multivariate analyses were performed to investigate potential confounding factors. Moreover, our findings demonstrated that surgery had a significant impact on bladder SRCC survival, thereby highlighting the significant and consistent effect of surgery, especially when the majority of the patients had advanced stages of the disease.
Conclusion
Our study revealed that surgery was an independent predictive factor of bladder SRCC patients. Patients who underwent surgery had higher CSS and OS than those who did not undergo surgery. An alternative approach was taken. We developed and validated a new nomogram model to predict the overall survival and cancer-specific survival of patients with SRCC urinary bladder tumors. Using this model in clinical practice can allow health professionals to assess patients’ long-term survival status more objectively and to develop more individualized therapeutic approaches for specific patients. Our study revealed that surgery is an independent predictive factor in bladder SRCC patients. Surgery patients had higher CSS and OS than those who did not undergo surgery. Chemo-radio yes vs surgery yes in combo therapy demonstrated improved survival in the surgery yes group. These findings suggested that surgery was the most effective treatment strategy for SRCC in the urinary bladder. Our findings on subtype-related disparities in survival have important implications for customized cancer therapy and call for more research to provide this group of patients with better treatment alternatives.
Supplemental Material
sj-docx-1-tau-10.1177_17562872221079473 – Supplemental material for Surgery improves survival in bladder signet-ring cell carcinoma-a population-based study
Supplemental material, sj-docx-1-tau-10.1177_17562872221079473 for Surgery improves survival in bladder signet-ring cell carcinoma-a population-based study by Mohammed Alradhi, Mohammed Safi, Shenghua Tao, Abdullah Al-danakh, Marwan Almoiliqy, Salem Baldi and Xiancheng Li in Therapeutic Advances in Urology
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81972831).
Data availability
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
