Abstract
Objective:
To study the effect of the COVID-19 pandemic on sacral neuromodulation (SNM) implanted patients and examine patient concerns.
Methodology:
A web-based survey was sent to all SNM patients, including those with implants and who had a cancelled operation because of the pandemic. The survey consisted of 15 questions in Arabic language, which sought to evaluate outcomes, as well as patient concerns and preferences during the COVID-19 pandemic.
Results:
A total of 66 patients were contacted, and of which, 62 replied. Most of the patients (n = 51; 82.3%) had the device implanted, and 11 (17.7%) patients had a postponed operation secondary to the pandemic. There were 20 males and 42 females. The mean age was 34 years ± SD 16.5 (9–62 years). Indications for sacral neuromodulation therapy were refractory overactive bladder OAB 35 (56.5%), retention 17 (27.4%), OAB + retention 3 (4.8%). When questioning the effect of the lockdown on patients, most reported no effect (43.5%), while 14.5% had some programming difficulties. The patients preferred telephone calls for device emergencies and clinic follow-up with 88.7% and 98.4%, respectively. Most patients had no concerns regarding their Interstim device during the pandemic and found it manageable; 8.1% had insurance concerns due to the economic changes.
Conclusion:
Patients with implanted SNM for lower urinary tract symptoms were mainly concerned with device programming. Telemedicine is a great solution for continuous care in this group.
Keywords
Introduction
The healthcare system has faced many challenges throughout the COVID-19 pandemic. COVID-19 was first reported as pneumonia from Wuhan, China.1,2 By March, the World Health Organization announced that COVID-19 was a pandemic with a recommendation that all countries take immediate action. 3
The pandemic led to many changes in healthcare, with treatment priority given to those with urgent medical conditions. Urology practice was affected as emergency and oncology patients were prioritized. 4 Patients with lower urinary tract symptoms were delayed management until the pandemic was under control. Treatment for patients with urinary incontinence, non-obstructive retention, frequency urgency, operative interventions were all postponed.
Sacral neuromodulation (SNM) therapy involves implantation of an electrode and battery. This is performed in two phases with 2 weeks between them. The first phase is electrode implantation under fluoroscopy. Patients need to complete a voiding diary prior to surgery and during the test period (after phase I). When patients show ⩾50% improvement, the second operation (phase II) is done for battery. This procedure needs further programming to initiate the benefits of therapy. Both procedures (phases I and II) are performed under general anesthesia in our center. However, programming sessions might require several repeated attempts until the best program for symptom control is defined. Complications of SNM include loss of efficacy, battery depletion, as well as electrode migration, breakage, and erosion. These require surgical intervention.
Sacral neuromodulation was introduced in Saudi Arabia in the last 10 years. As any new introduced therapy, close patients care, frequent programing, and good follow-up is needed. The COVID-19 pandemic caused Saudi Arabia to announce a lockdown, and elective operations were postponed to reduce the risk of patients being exposed to COVID-19. The International Neuromodulation Society has published several reports on postponed Interstim cases because of the COVID pandemic. We conducted our study to assess the impact of COVID-19 lockdown on sacral neuromodulation implant patients in our center.
Materials and methods
This study was approved by our hospitals ethics review board (Unit of Biomedical Ethics Research Committee of King Abdulaziz University,
Data was collected and exported as Excel sheets; it was then coded for statistical analysis using IBM SPSS 26 software. p-values below 0.05 were considered statistically significant. Data were reported as frequencies in each question and reported as a percentage.
Results
A total of 62 out of 66 patients completed the questionnaire. Most patients, 51 (82.3%), already had the device implanted before the pandemic, while 11 (17.7%) patients had the operation postponed secondary to the COVID-19 lockdown. Most patients were from the public sector, 44 (71%); there were 12 insurance cases (19.4%) and six cash patients (9.7%). There were 20 men and 42 women. The mean age was 34 years ± SD 16.5 (9–62 years). Indications for SNM therapy were refractory overactive bladder (OAB) 35 (56.5%), retention 17 (27.4%), OAB and retention three (4.8%), pelvic pain syndrome five (8.1%), others two (3.2% including erectile dysfunction). At the time of completing the questionnaire, only one patient was COVID-19-positive with mild symptoms.
When questioning the effect of the COVID-19 lockdown on patients, most reported no effect (43.5%) while 14.5% had some programming difficulties, and 17.7% cancelled their operation. Programming is usually done by company personnel, and most patients preferred to delay their programming sessions (32.3%); 25% agreed to programming while following COVID-19 precautions. Patients preferred telephone calls and virtual clinic for both emergency issues with the device and clinic visits for follow-up (88.7% and 98.4%, respectively). Patients preferred to delay their implantation and surgical intervention (88.7%) and preferred other less effective alternative therapies (intermittent catheterization in the retention group and using multiple anticholinergic medications in refractory OAB despite limited benefit). A total of 69.4% of cases there was a preference for general anesthesia (56.5%) over local anesthesia (43.5%) for surgical intervention. Most patients had no concerns regarding their implanted device during the pandemic; they found that it was manageable, but 8.1% had concerns regarding insurance coverage issues; 9.7% were concerned about the time delay until COVID-19 issues were resolved (Tables 1 and 2; and Figures 1 and 2). There were no reported complications by any patient during the 3-month lockdown.
Patient demographics, diagnosis, health sector, answers to effect of lockdown, programming and clinic preference and preference of using other medications.

OAB, overactive bladder; SNM, Sacral nerve stimulation.
The questionnaire sent to patients translated into English.

OAB, overactive bladder; SNM,Sacral nerve stimulation.
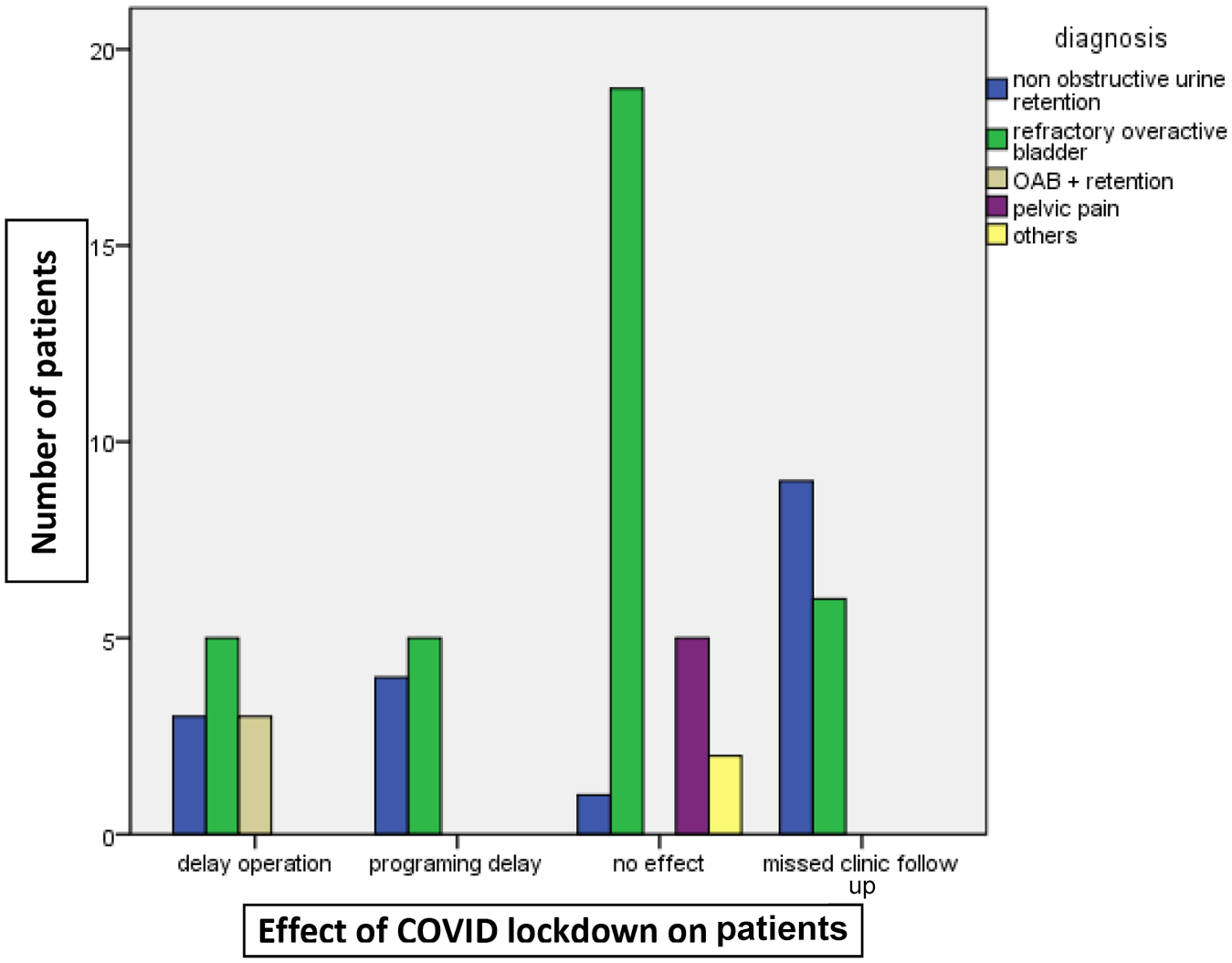
Bar chart showing the effect on SNM (sacral nerve Stimulation) implanted patients (delay operation, programming delay, no effect, missed clinic follow-up) categorized according to patient diagnosis.

Bar chart shows the patient preference of other treatment during COVID-19 lockdown (yes: prefer to use other treatment; no: don’t need) categorized according to diagnosis.
Discussion
This paper presents the results of a survey study examining the effect of the COVID-19 pandemic on patients who underwent SNM surgeries and programming at a single clinical center in Saudi Arabia. The survey specifically asked about care during a 3-month lockdown window from March to May 2020.
Most SNM implanted patients reported no effect on their condition (43.5%), but programming was a major problem in 14%. Programming requires direct contact with trained personnel, which poses an infection risk, and 32.3% of patients preferred to delay their programming session or used alternative therapy to cope with their condition (43%). During the lockdown period, no patients developed any erosion, infection, or electrode issues. Other concerns regarding clinical visit were managed as recommended in all health sectors, by telemedicine and virtual clinics, which was our patients’ preference (98.4%). Telemedicine or virtual clinics are good tools for healthcare follow-up without any personal contact, as reported by Miller et al. and Katharina et al.5–7
Sacral neuromodulation is an FDA approved therapy for refractory overactive bladder, 8 frequency urgency syndrome and non obstructive urinary retention.8–10 It showed to be effective in other off label uses as chronic Pelvic pain, 11 in neurogenic bladder. 12 Anne et al. and Mahran et al. reported its use in pediatric patients and in pregnancy13,14 Pediatric sacral neuromodulation is not FDA approved but showed favourable results in multiple studies. 14 In our study we included pediatric cases who were implanted for refractory overactive bladder and urinary retention, neurogenic bladder secondary to spina bifida.
Bekkers and Koopman 15 and Evenett et al. 16 predict the COVID-19 pandemic will have major effects on the world economy, which might affect patient decisions. As sacral neuromodulation can be an expensive option, our patients had concerns about private insurance coverage in the future.
Study limitations include the single center and small sample size, included in the study. It would be preferable to increase the sample size and perform a multicenter study, worldwide. The questionnaire is also not validated, and the authors want to consider this in the future. The age range of our patients is 9–62 years as the use of this therapy in children is relatively, which limits the generalizability of the results.
Conclusion
We found that patients with implanted SNM for lower urinary tract symptoms were facing major issues with device programming during the COVID-19 lockdown.
Footnotes
Consent
All patients gave written consent, parents of paediatric patients gave consent on behalf of children.
All consents were obtained electronically.
Author contributions
Mai Banakhar: data collection, design, writing; ethical approval request: statistical analysis; review.
Conflict of interest statement
The author(s) declared that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
