Abstract
Treatment of urolithiasis depends on several important factors which include stone location, size, composition, and patient symptoms. Although significant advancements have been made in the surgical management of urolithiasis in the last decade, pharmacotherapy which can prevent the formation of new stones and decrease the recurrence of urolithiasis has not experienced the same level of success. Currently, urolithiasis is regarded as a complicated syndrome that is determined by numerous factors, and any treatment plan for urolithiasis should be individualized while considering any potential damage arising from stone-forming factors. This review introduces the most popular methods currently used to evaluate urolithiasis and the pharmacotherapy of urolithiasis based on patient-specific factors.
Introduction
Urolithiasis is one of the most common problems encountered by a urologist. Stone incidence is heavily influenced by geographical, climatic, ethnic, dietary, and genetic factors. The recurrence risk is determined by the underlying disease or disorder that is causing stones to form. The prevalence rates for urinary stones vary from 1% to 20% 1 and this value is continuously rising due to social, economic, nutritional, and environmental changes in both developing and developed countries, and from improvements in clinical-diagnostic procedures. 2 The optimal treatment of urolithiasis depends on several important factors, such as stone location, size, composition, and patient symptoms. Urinary stones can be classified according to size, location, X-ray characteristics, etiology of formation, composition, and risk of recurrence. 1 In the last two to three decades, great advancements have been made in the surgical treatment of kidney and ureter stones. Currently, the treatment options available include extracorporeal shock wave lithotripsy (ESWL), percutaneous nephrolithotomy (PCNL), retrograde intrarenal surgery, and laparoscopic ureterolithotomy. 3 Urolithiasis has a high rate of recurrence; the recurrence rates at 2, 5, 10, and 15 years were 11%, 20%, 31%, and 39%, respectively. 4 In randomized, controlled trials, patients with recurrent calcium stones who served as control subjects had newly formed stones in 43–80% of cases within 3 years. 5
While there has been significant success in the surgical treatment of urolithiasis, pharmacotherapy which can prevent the formation of new stones and decrease the recurrence of urolithiasis has not experienced the same level of success. This may be because urologists are typically more focused on the development of surgical skills and new instruments, but as increasing evidence suggests that pharmacotherapy decreases stone recurrence rates significantly, urologists should consider the importance of pharmacotherapy when they consider the best treatment for urolithiasis.
Urolithiasis should not be regarded as a simple disease with fixed characteristics; it is a complicated syndrome determined by numerous factors that can lead to urinary stone formation and even influence the stone’s characteristics. Such factors include age, sex, diet, geographical location, climate, and etiological factors like metabolic disorders within the kidney and intestinal tract. 6 These factors form a sophisticated network, and damage to this network will result in pathophysiological outcomes such as urine supersaturation and mineral deposition. Therefore, urologists should consider the whole picture as much as possible and avoid focusing only on the stones during the management of urolithiasis. The basic principle of urolithiasis management is to treat every patient individually by carefully evaluating the damage to the network. Identification of patient-specific factors provides key information to determine when pharmaceutical invention is necessary. Here we review the most popular methods currently used to evaluate urolithiasis during clinical practice and the pharmacotherapeutic strategies for the management of urolithiasis based on patient-specific factors.
Basic evaluation of urolithiasis
Careful medical history evaluation and routine blood tests, urine tests, and urine culture should be performed for every patient who is diagnosed with urolithiasis, as it provides key insights into the systemic diseases and reveals the most common conditions associated with urolithiasis. 1 Routine blood tests, urine tests, and urinary culture are generally inexpensive and essential, and should be used to screen for possible abnormalities associated with urolithiasis.
Radiological evaluation of urolithiasis
Although conventional imaging techniques such as abdominal radiography [kidney, ureter, bladder (KUB)] and intravenous pyelography are still commonly used in the evaluation of urolithiasis, helical computed tomography (CT) without enhancement is still considered the gold standard today. Unenhanced helical CT is highly sensitive (>95%) and specific (>96%) in the diagnosis of urolithiasis and is the first imaging choice for the initial assessment of patients with suspected urolithiasis. 7 A 2014 multicenter trial found no significant difference in both the medical intervention of choice or hospital admission rates between patients who received CT or ultrasonography. Furthermore, despite recommendations from the American College of Emergency Physicians, American College of Radiology, and the American Urology Association (AUA) for a reduced-radiation dose CT as the diagnostic tool of choice to reduce the patient’s radiation exposure, it is rarely used by clinicians. 8
Five parameters can be evaluated using CT imaging in vitro or in vivo: stone location, stone burden, obstruction, stone fragility, and the stone’s composition. 7
Stone location, stone burden, and any accompanied urinary tract obstructive signs provide crucial information in the management of urolithiasis, especially in patients that require surgical intervention. Small (<3 cm) and simple caliceal and pelvic stones may be treated with ESWL or flexible ureteroscopy; 9 larger and complex kidney stones are treated with PCNL. Proximal ureteral calculi can be treated with ESWL, flexible ureteroscopy, or PCNL. Most distal ureteral calculi can be treated by ureteroscopy or medical expulsive therapy.
Stone size is a simple metric for stone burden assessment and can be reliably obtained using CT. 7 The selection of urological treatment strategies, which include medical expulsive therapy or endoscopic/percutaneous interventions, relies strongly on stone size determination. 7
Stone fragility determines the efficacy of ESWL treatment. Studies have shown that the internal morphologic features of the stone had a higher correlation with stone fragility compared with its X-ray attenuation value. The internal structure of the stones can be either heterogeneous or homogeneous. Stones with internal homogeneity have a uniform internal structure and are therefore more rigid and difficult to break with lithotripsy, and stones with internal heterogeneity have areas of low attenuation or internal voids within the stone component. Therefore, internal heterogeneity is an indication of high stone fragility, as the internal irregularities within the stone structures facilitate easier disintegration of stones with ESWL. 7
An interesting advancement in recent years is the use of CT to analyze the stone’s composition in vitro and in vivo. 10 The attenuation values of urinary calculi at 120 kV usually fall within certain ranges: uric acid, 200–450 HU; struvite, 600–900 HU; cystine, 600–1100 HU; calcium phosphate, 1200–1600 HU; and calcium oxalate monohydrate and brushite, 1700–2800 HU,11–13 but because multiple factors can influence the CT attenuation value in vivo, it is not reliable to differentiate stone composition by CT attenuation in vivo. 14
Dual-energy CT shows great promise in the determination of stone composition in vivo. A dual-source CT system contains two X-ray tubes and two corresponding 64-detector arrays mounted on a gantry at a 90°angle. 12 Concurrent scanning at two different energies (80 and 140 kVp) can be performed by dual-energy CT, and the resulting data can be exploited to characterize tissue material. 15 Dual-energy post-processing software algorithms assume a mixture of water, calcium, and uric acid for every voxel and color-code voxels that show a dual-energy behavior similar to calcium in blue and ones that are similar to uric acid in red. 13 Using dual-energy CT, the differentiation of pure uric acid, mixed uric acid, and calcified stones is possible. Furthermore, the differentiation of struvite and cystine is possible by adapting the slope of the three-material decomposition algorithm. 13 As some stones (the hardness factors are as follows: calcium oxalate monohydrate, 1.3; calcium oxalate dehydrate, 1.0; hydroxyapatite, 1.1; brushite, 2.2; uric acid/urate, 1.0; cystine, 2.4 14 ) are difficult to fragment by ESWL, analysis of stone composition by dual-energy CT may contribute not only to the identification of the stones but also to the chemical characterization of the stones in the urinary tract, which could be useful when making surgical or other treatment decisions. 15
Stone composition analysis
Crystals (both inorganic and organic) and even bacteria can be found in stones, but only crystals have been well studied. In most situations, stone composition analysis is similar to the crystal composition analysis. Most stones are composed of a mixture of different chemical compositions. Pure single composition-containing stones only accounts for a small proportion of urolithiasis. Currently, urinary stones can be classified based on several specific chemical components, including oxalate, phosphate, apatite, struvite, uric acid, and cystine. Mixtures of these chemical compositions in a single stone are also common, resulting in a spectrum of different stone chemistries. 16 Calcium-containing stones, which include calcium oxalate monohydrate, calcium oxalate dihydrate, and calcium phosphate stones, account for around 70–80% of stones. Struvite (magnesium ammonium phosphate) stones account for 15% of urinary calculi and are typically associated with urease-producing urinary tract infections and carry significant morbidity. 17 Uric acid urolithiasis constitutes approximately 7–10% of all urinary stones. 18 Cystine stones account for 1% of stones. Other stones, such as xanthine and drug-induced calculi (e.g. triamterene, indinavir), account for less than 1% of stones (Table 1). There are variations in the composition of stones, which are influenced by the differences in geographical, economic, or sanitation conditions. For example, in China’s central underdeveloped areas, more infectious stones are found; however, in China’s east coast, where the economic and hygiene conditions are far better compared with China’s central area, the incidence of uric stones are higher. 19
Types of various common stones.

Analysis of stone composition can provide useful information for the management of urolithiasis. 20 Based on European Association of Urology (EAU) and AUA guidelines, stones that were either passed spontaneously, removed surgically, or excreted as fragments should be analyzed to determine their composition. The analysis should be repeated if any changes in factors such as medical treatment, dietary habits, environment, or disease which could influence the composition of the stone occurs.
Once the mineral composition is known, the potential metabolic disorders can potentially be identified. Valid analytical methods that are currently in use are infrared spectroscopy and X-ray diffraction. 1 Infrared (IR) spectroscopy is a modern physical-chemical method suitable for the investigation of urolithiasis. 6 A full representation of the principle and methods of IR spectroscopy is out of the range of this review. Here we will describe a brief overview of the procedures. First, stone debris is collected and dried, then the whole stone is ground up and scanned using IR spectroscopy. The acquired spectrum will then be compared with a computer library that contains the relevant normative spectrums from different chemical compositions. Different methods such as the artificial neural network have been successfully developed to compare the acquired spectrums with normative spectrums and to interpret the results.
As mentioned above, in vitro CT is also used in the analysis of stone composition in recent studies. Although CT is able to provide a quick and simple way for the detection of the main composition of stones in vitro, it cannot analyze the composition of the stone in vivo currently and is therefore not commonly used in routine clinical practice. However, dual-energy CT can provide useful information about stone composition in vivo and should be considered during clinical practice.
First-time estimate stone recurrence risk
After performing a basic and radiological evaluation of urolithiasis and a stone composition analysis, further metabolic evaluation and pharmacotherapeutic strategies should be considered. However, not all patients require a metabolic evaluation or undergo pharmacotherapy. Typically, the decision depends on the risk of recurrence, which should be estimated. Children, patients with recurrent stones, and several naive stone formers may require further extensive metabolic evaluation or pharmacotherapy. The following are the indications for an extensive metabolic evaluation to be performed due to a high risk of stone occurrence: children, recurring stones former, multiple stones former, bilateral stones former, uric acid stones, staghorn calculi, nephrocalcinosis, solitary kidney, renal insufficiency, intestinal disease or chronic diarrhea, osteoporosis. 21
Extensive evaluation of urine metabolic abnormalities
Currently, the metabolic evaluation of urine is regarded as an important method to determine the potential cause of urolithiasis. A 24-hour urine collection test is the mainstay in an extensive evaluation of urine metabolic abnormalities. The urinary parameters are typically assayed in a 24 h urine specimen which includes calcium, oxalate, citrate, uric acid, cystine, sodium, potassium, phosphate, magnesium, urinary volume, and pH. Table 2 shows the normal values of these parameters. 22
Normal values in the extensive evaluation of urine metabolic abnormalities.

Urinary volume and urinary pH
Volume and pH of urine are important factors used to determine the formation of different types of stones. 23 Many pathways cause increased urinary supersaturation which facilitates stone formation, and low urinary volume is one of them. 24 Uric acid stones and cystine stones are usually formed in acidic urine, while calcium phosphate stones are more common with basic urine pH >7.0. 25 Urine volume and pH are key factors in stone formation, but they can vary extensively. The relationship between calcium oxalate stones and pH requires a comprehensive meta-analysis; some studies have observed that calcium oxalate stones can form at any pH level, but other studies have observed that the highest calcium oxalate crystallization risk is between pH 3.5 and 5.5 and that alkalization is beneficial. 26
Hypercalciuria
Hypercalciuria is elevated calcium in the urine, exceeding 300 mg/dL in men and 250 mg/dL in women. Hypercalciuria is one of the most common metabolic abnormalities in urolithiasis. 27 Based on the presumed site of the primary abnormality, hypercalciuria has been categorized into three groups, absorptive hypercalciuria, renal hypercalciuria, and resorptive hypercalciuria. However, it should be noted that most hypercalciuria are idiopathic and have non-specific etiologies. 28 Differentiation of the presumed site of hypercalciuria is difficult to isolate and may have minimal clinical value.
Hyperoxaluria
Hyperoxaluria is an excess of oxalate in the urine, exceeding 45 mg/dL in women and 55 mg/dL in men per 24 h. 29 Based on its mechanism, it is also classified into three groups including enteric hyperoxaluria, increased ingestion, and primary hyperoxaluria. Most hyperoxaluria is caused by high dietary oxalate intake or increased enteric absorption of dietary oxalate. 30 Primary hyperoxaluria is a rare inherited disorder of oxalate metabolism, with urinary oxalate levels over 100 mg/dL, and two distinct types of primary hyperoxaluria have been reported.
Hyperuricosuria
Hyperuricosuria is defined as elevated urinary uric acid excretion exceeding 800 mg/dL in men and 750 mg/dL in women.31,32 Hyperuricosuria is not only associated with the formation of uric acid stones but also the formation of calcium stones. Low urine pH (pH <5.5) increases the possibility of uric acid stones in hyperuricosuria.33,34 Furthermore, hyperuricosuria occurs in 10% of patients with calcium stones because the increase in dissolved uric acid salts causes the formation of the nidus for deposition of calcium and oxalate. 35
Hypocitraturia
Hypocitraturia is low urinary citrate excretion <320 mg/dL. Urinary citrate excretion is mainly determined by tubular reabsorption. Urinary citrate is a major inhibitory factor for the prevention of stone formation. Low urine citrate levels are often seen in chronic metabolic acidosis. However, in most urolithiasis patients with hypocitraturia, the etiology is not apparent and cannot be determined.36,37
Cystinuria
Cystinuria is characterized by a high concentration of cystine in the urine, exceeding 250 mg/dL. 38 Cystinuria is an inherited autosomal recessive disorder where there is a tubular defect in the dibasic amino acid transport which results in an increased urinary excretion of cystine, ornithine, lysine, and arginine. 39 Among them, cystine is the most insoluble in acidic urine and thus precipitates into stones. 40
Genetic evaluation
The formation of stones may be caused by environmental, anatomical, metabolic, and genetic factors. These factors can act in conjunction or isolation and lead to stone formation.41,42 Though most reports focus on the environmental, anatomical, or metabolic factors, recent genomics studies could provide new clues into the mechanism of stone formation. An example is primary hyperoxaluria. Currently, it is recognized that primary hyperoxaluria can be divided into two types. Type I is caused by base changes (mutations or polymorphisms) in the gene that codes for the peroxisomal enzyme alanine-glyoxylate aminotransferase. Type II is caused by mutations that inactivate the genes that code for glyoxylate reductase and hydroxypyruvate reductase. Although monogenic diseases can cause urolithiasis, most clinical manifestations have polygenic determinants. 30 Genomic-associated research principles that were successfully established for the research of hypertension, diabetes, and cancer should be encouraged to search for potential genetic loci associated with urolithiasis.
Hyperparathyroidism, distal renal tubular acidosis, and gout
Hyperparathyroidism, distal renal tubular acidosis (dRTA), and gout are three disorders that are related to urolithiasis that may be frequently encountered during clinical practice. They should be considered as they are common etiologies of urolithiasis. 43
Approximately 4.9% of nephrolithiasis is caused by hyperparathyroidism, but only 17% to 24% of patients with hyperparathyroidism will form urinary stones.44,45 Hyperparathyroidism can be confirmed by determining the patient’s serum parathyroid hormone and serum phosphorous, but the diagnosis cannot be eliminated even when values are normal as hypercalcemia may be intermittent in this disorder and should be measured repeatedly. 46 Hyperparathyroidism usually forms calcium oxalate or calcium phosphate stones.
Renal tubular acidosis (RTA) is a form of metabolic acidosis caused by tubular disorders. Although several types of RTA may be discerned, nephrocalcinosis and nephrolithiasis frequently occur in dRTA. 47 RTA may be caused by autoimmune disease, drugs, toxins, or genetic defects. Clinical features of dRTA are hyperchloremic hypokalemic metabolic acidosis, alkaline urine, hypercalciuria, hypocitraturia, and nephrolithiasis. Patients with dRTA generally form calcium phosphate stones.48–50
The main characteristics of gout are hyperuricemia, gouty arthritis, gouty tophi, and uric acid kidney stones. The incidence of urolithiasis ranges from 10% to 20% in patients with gout. 49 Although patients with gout will commonly form stones composed of uric acid, in rare situations calcium oxalate stones may also form. 51
Second-time estimate stone recurrence risk
Although extensive metabolic evaluation may disclose the underlying causes of urolithiasis for specific patients, a second-round evaluation of recurrence risk is recommended at this time. After a careful review of all information acquired from the patient before and after metabolic evaluation, pharmacotherapy should be considered to adjust risk-related factors that can stimulate mineral disposition and stone recurrence in high-risk patients.
Special consideration with staghorn stones and calcium phosphate stones
Staghorn stones are branched stones which occupy a large portion or even all of the branches in the renal collecting system. The EAU guideline on urolithiasis defines staghorn stone as a stone with a central body at the renal pelvis with at least one caliceal branch. 52 Staghorn stones are usually composed of a mixture of magnesium ammonium phosphate (struvite) and/or calcium carbonate apatite. 52 The underlying mechanism in the formation of staghorn stones is associated with urinary infection. 52 Chronic urinary infection with urease-producing bacteria (Proteus, Providencia, Morganella spp, Ureaplasma urealyticum, Corynebacterium urealyticum, Klebsiella, Pseudomonas, Mycoplasma, Anaerobic clostridia, Staphylococcus, and Serratia) leaves residues in the urine and hydrolyzes urine urea constantly. The hydrolysis of urea yields ammonium, hydroxyl ions, and persistently alkaline urine. The resulting alkaline urinary environment and high ammonia concentration, along with abundant phosphate and magnesium in urine, promotes the crystallization of magnesium ammonium phosphate and lead to the formation of large branched stones.
As previously mentioned, calcium-containing stones, which include calcium oxalate and calcium phosphate stones, account for 70–80% of urinary calculi. 53 Most calcium stones are composed of calcium oxalate, either by itself or more commonly in combination with calcium phosphate or calcium urate. 53 Calcium oxalate is usually the main constituent and calcium phosphate is present in amounts ranging from 1% to 10%. 54 When calcium phosphate becomes the main constituent (>50%) of the stones, the stones are called calcium phosphate stones, and patients who form calcium phosphate stones are referred to as calcium phosphate stone formers. 55 Calcium phosphate stones are a less common type of urolithiasis, only accounting for 5–10% of total stones. The most common crystalline phases found in calcium phosphate stones are carbapatite (carbonated apatite, often improperly termed apatite) and brushite (dicalcium phosphate dihydrate), and they, respectively, present as the main components in 67% and 14% of phosphate stones; struvite (magnesium ammonium phosphate hexahydrate), amorphous carbonated calcium phosphate, octacalcium phosphate pentahydrate, and whitlockite (mixed calcium and magnesium phosphate) are less frequent. 56 Phosphate stones are pH and calcium dependent, 56 and because calcium phosphate stones are often associated with acidification disorders such as RTA, when calcium phosphate stones are found and urinary pH is above 6.8, RTA should be suspected. 57
Pharmacotherapeutic strategies for urolithiasis
The optimal pharmacotherapeutic strategy can only be found after a careful medical evaluation of the urolithiasis is conducted. When the stone size is <5 mm and located in the distal ureter, medical expulsive therapy is recommended to promote passage of the stones. 58 However, if the stone size is larger than 7 mm, surgical intervention or ESWL is the optimal choice in most cases as they can remove stones effectively. 59 The purpose of pharmacotherapy is to prevent stone recurrence or dissolve specific stones. Based on the stone composition and potential metabolic abnormalities, different pharmacotherapeutic strategies should be made to correct the underlying cause (Table 3). Combining surgical intervention, ESWL, and pharmacotherapy provides the best solution for the management of urolithiasis in current clinical practices.
Pharmacological interventions for urolithiasis.
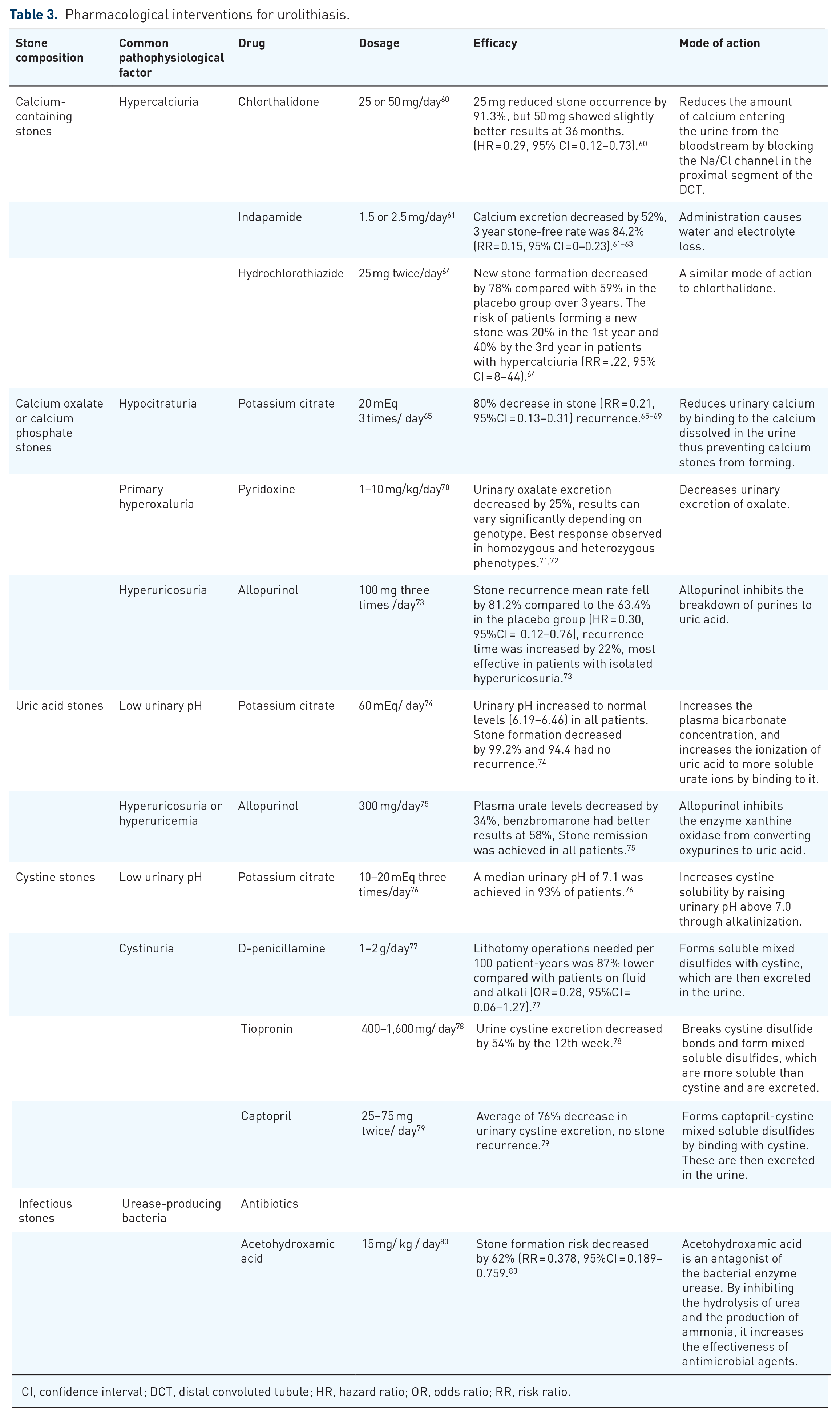
CI, confidence interval; DCT, distal convoluted tubule; HR, hazard ratio; OR, odds ratio; RR, risk ratio.
General recommendations for the management of urolithiasis
Before pharmacotherapy is recommended, there are some general recommendations for all stone formers. Stone formers should drink enough fluid to maintain a urine output of at least 2.5 L per day, as low urine volume is the main cause of supersaturation. 81 A low-salt, normal calcium diet and moderate animal protein diet is also advised. 53 Systemic disorders such as hyperparathyroidism, distal RTA, and gout must be excluded.
Medical expulsive therapy for stone size less than 5 mm
The spontaneous passage rate in 20 weeks was 312 out of 392 stones, 98% in 0–2 mm, 98% in 3 mm, 81% in 4 mm, 65% in 5 mm, 33% in 6 mm, and 9% in ⩾6.5 mm wide stones. 82 The stone size and location can be used to predict spontaneous ureteric stone passage. 82 However, medical expulsive therapy may increase this possibility. Calcium-channel blockers and α-blockers have emerged as the most promising agents for medical expulsive therapy. 83 A multicenter clinical study has suggested that tamsulosin is more efficient than nifedipine in medical expulsive therapy for distal ureteric stones. 84 Due to its low cost and high safety profile, the Combined AUA/EAU Ureteral Stones Clinical Guidelines Panel recommends that patients with newly diagnosed ureteral stones <5 mm and well-controlled symptoms should be treated by medical expulsive therapy.
Pharmacotherapy for uric acid or cystine stones
Both uric acid stones and cystine stones can be dissolved completely using pharmacotherapy, therefore it is important to differentiate them from other types of stones in vivo. Dual-energy CT, conventional KUB, urine pH, and metabolic evaluation are useful for differentiation.
Uric acid stones are associated with three main factors: Low urinary pH (<5.5), low urinary volume, hyperuricosuria, or all of the above. Therefore, the alkalization of urine using potassium citrate to raise urinary pH (>6) and hydration are reasonable treatment recommendations. If hyperuricosuria or hyperuricemia are present, allopurinol can be added.3,85,86 Recent studies have suggested that uric acid stones are not only related to gout but are also related to other metabolic disorders. High body-mass index, hypertriglyceridemia, obesity, glucose intolerance, insulin resistance, and overt type 2 diabetes are common in uric acid stone patients.32,87–90 Therefore, controlling these disorders may be helpful in the management of uric acid stones.
Similar to uric acid stones, the formation of cystine stones is also related to low urinary pH and low urinary volume. Hydration and potassium citrate are the mainstays of therapy. For moderate cystinuria (<500 mg/dL), raising urine pH to 7.0 and increasing fluid intake (>4 L/dL) is sufficient; however, in more severe cystinuria (>500 mg/dL), administration of chelating agents such as D-penicillamine, tiopronin, or captopril is recommended.39,91,92
Although uric acid stones and cystine stones may be dissolved completely by pharmacotherapy, it is a slow process. Pharmacotherapy is a reasonable first choice for the treatment of uric acid stones or cystine stones, which are small-sized and do not significantly obstruct the urinary tract. However, if the stone size is large or the urinary tract is obstructed, ESWL or endoscopic intervention is necessary for removal of the stone. As cystine stones are quite resistant to ESWL, minimally invasive endoscopic procedures should be the first-line treatment if a cystine stone is suspected. After the stones are removed, pharmacotherapy should be considered as it is efficient in the prevention of stone recurrence.
Pharmacotherapy focus on the prevention of calcium oxalate stones and calcium phosphate stones recurrence
ESWL or minimally invasive endoscopic surgeries are the mainstays in the management of calcium-containing stones. The purpose of pharmacotherapy is for the prevention of stone recurrence. A urinary metabolic evaluation can provide key evidence for pharmacotherapy. 93
Together with a lower urinary volume, four factors including hypercalciuria, hypocitraturia, hyperoxaluria, and hyperuricosuria may contribute to the formation of calcium oxalate; 94 these factors can be revealed by conducting a metabolic evaluation of the urine. Hypercalciuria is typically the most common factor found and can be treated with thiazide diuretics. However, it must be recognized that hyperparathyroidism can cause hypercalciuria and urolithiasis. Therefore, hyperparathyroidism should be excluded before administering thiazide diuretic treatment. Hypocitraturia can be treated using potassium citrate; however, caution should be exercised when using potassium citrate as high urinary pH is positively associated with the possibility of calcium phosphate stone formation. As per the EAU guidelines, patients suffering from hyperoxaluria should reduce their intake of animal protein, purine-rich foods, oxalate, xalate, and dietary fat. For enteric hyperoxaluria, calcium, magnesium, and alkaline citrates supplements are recommended. Patients with primary hyperoxaluria should be referred to a specialist, as successful treatment would require an experienced interdisciplinary team. The treatment of choice is pyridoxine 5–20 mg/kg/day measured according to the patient’s urinary oxalate excretion and the patient’s tolerance levels, 9–12 g/day of alkaline citrate for adults and 0.1–0.15 mg/kg/day for children, and 200–400 mg/day of magnesium. Dietary modifications (restriction of oxalate intake and normal calcium intake) and increasing fluid intake to 3–4 L/day is helpful for the alleviation of hyperoxaluria. 95 The outcomes associated with the probiotic treatment approach for Oxalobacter formogenes or a direct administration of a recombinant oxalate decarboxylase still requires further studies. 96 A persistently elevated urinary oxalate of >0.7 mmol/1.73 m2/day should prompt consideration of primary hyperoxaluria. Although it is a rare inherited disorder, type 1 patients can be treated with pyridoxine. 97 If hyperuricosuria is present, allopurinol is used to decrease stone recurrence. 98
The mechanism of calcium phosphate stone formation is different from that of calcium oxalate formation. Hypercalciuria, hypocitraturia, and high urinary pH are the main factors used to determine the formation of calcium phosphate stones. After excluding systemic disorders such as hyperparathyroidism, sarcoidosis, and RTA, pharmacotherapy can be applied to prevent the formation of calcium phosphate stones. The treatment of calcium phosphate stones is similar to calcium oxalate stones. Thiazide diuretics are used for the management of hypercalciuria and potassium citrate is used to restore urinary citrate concentration. Urinary pH, citrate, and the degree of supersaturation should be assessed after pharmacotherapy as increasing urinary pH can promote calcium phosphate stone formation.99,100
Pharmacotherapy for infectious stones
Managing infectious stones is challenging as these stones grow quickly and extensively. 101 The stone burden is high and the kidney usually sustains severe damage from a combination of factors such as obstruction, pH disturbance, and abnormal excretion of minerals. 102 Urinary culture and staghorn formation imply that the infectious stones have a struvite/carbonate apatite composition.103,104 Surgical interventions such as PCNL or the combination of PCNL and ESWL are the primary modality for the treatment of infectious stones. 105 Complete stone removal should be the therapeutic goal. Additionally, three aspects should be considered in pharmacotherapy: (1) Antibiotic treatment to control urinary infection; (2) if metabolic abnormalities exist, the correction of potential defects is necessary; (3) urease inhibitors such as acetohydroxamic acid may be reserved for patients who are intolerant to surgical intervention. 106
Conclusion
Urolithiasis is a common disease that has a significant social and financial burden. Management of urolithiasis should be unique to each individual. Multiple factors such as stone location, stone burden, stone fragility, stone composition, potential metabolic abnormalities, and patient condition should be considered to develop the best treatment plan for this disease. When the stone diameter is less than 3–5 mm, most stones can pass spontaneously with hydration and pain control. Although ESWL and surgical interventions are both effective methods to remove stones in most cases after the initial treatment, patients should be evaluated for the risk of recurrence. For patients without urinary tract infection, hypertension, diabetes, obesity, family history of urinary calculi, and other high-risk factors, after their stones are removed, general recommendations such as increasing fluid intake and adjustment of diet should be given to the patient and no further pharmacotherapy is required. However, in high-risk patients, after stones are removed, specific pharmacotherapy which was established after a careful medical evaluation should be discussed with patients. A combination of surgical intervention, ESWL, and pharmacotherapy is currently the best treatment plan for the management of urolithiasis with a high risk of recurrence and should be recommended in urological daily practice.
Footnotes
Acknowledgements
The comments from Dr. Xiaolong Jia (Department of Urology, Ningbo First Hospital, Ningbo, China) in modifying the manuscript were greatly appreciated.
Authors’ contributions
MND, HM, MA searched the literature, extracted the data, and wrote the manuscript. KYW, JFP searched the literature. YC, QM revised and approved the final manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Ningbo Clinical Research Center for Urological Disease (2019A21001).
