Abstract
Background:
The objective of this study was to evaluate Octamer-binding transcription factor 4 (Oct-4), neutrophil to lymphocyte ratio (NLR) and body mass index (BMI) as independent prognostic biomarkers for prediction of urinary bladder cancer (UBC) outcomes. With the advancement in prognostic biomarker discovery, tumor recurrence is difficult to accurately predict in UBC. UBC is costly to treat due to the requirement of frequent invasive follow-up sessions. Therefore, it is of utmost importance to evaluate good prognostic biomarkers for UBC surveillance.
Methods:
We studied 39 UBC tissue samples. Oct-4 protein expression was evaluated semiquantitatively by immunohistochemistry (IHC). Complete blood count data and body weight as well as the height of the patients were retrieved and recorded before the date of the first transurethral resection of bladder tumor (TURBT). The follow-up period was 48 months for recurrence-free survival (RFS), progression-free survival (PFS), and overall survival (OS).
Results:
Oct-4 expression profile was found to be significantly associated with gender (p = 0.028), tumor grade (p = 0.038), tumor stage (p = 0.003), lymph node status (p = 0.029), recurrence (p = 0.004), progression (p = 0.011), and treatment modality (p = 0.016). Tumor grade and progression were found significant with NLR values (tumor grade, p = 0.006; progression, p = 0.038) and BMI (tumor grade, p = 0.036; progression, p = 0.014). Moreover, BMI was also significantly associated with UBC recurrence (p = 0.014). Kaplan–Meier survival analysis showed poor prognosis with both high Oct-4 expression (RFS, p = 0.001; PFS, p = 0.004; OS, p = 0.014) and high NLR values (RFS, p = 0.049; PFS, p = 0.004; OS, p = 0.005). Patients with high BMI too had poor RFS (p = 0.025) and poor PFS (p = 0.032). Furthermore, multivariate Cox regression analysis, indicated Oct-4 as an independent prognostic biomarker for RFS (HR = 0.240, 95% CI, 0.072–0.804, p = 0.021).
Conclusions:
We conclude that the expression profile of Oct-4 will be beneficial in prediction of UBC recurrence, and could have profound implications on the development of new therapeutic targets for UBC treatment.
Keywords
Introduction
The presence of cancer stem cells (CSC) was first acknowledged by Bonnet and colleagues in leukemic cells. 1 Since then, CSCs have been identified in a number of solid tumors, such as breast, colon, brain, and prostate cancer. The maintenance of ‘stemness’ in cancer cells is controlled by various signaling pathways, such as Hedgehog, WNT/β- catenin, Notch, PI3K/Akt, NFkB, JAK-STAT, and PTEN (Supplementary Figure 1). 2 CSCs are cells within a tumor with the capacity for self-renewal, tumor-initiation, and cancer therapy resistance, thus promoting tumor aggressiveness and metastasis. 3 Epithelial to mesenchymal transition (EMT) is achieved by the loss of E-cadherins, β-catenins, and basal-apical polarity, leading to the formation of CSCs, which may also result in invasiveness, angiogenesis, and metastasis. 3 Recently, the relationship between inflammation and CSCs has attracted more attention. The systemic inflammatory response alters immune cells such as basophils, neutrophils, and lymphocytes, and is found to be involved in tumor initiation, angiogenesis, and progression. 4 Chronic/systemic inflammation may also lead to the production of reactive oxygen species (ROS), promoting EMT and progenitor stem cell activation. This inflammatory response is involved in the propagation of CSCs and causes the production of pro-inflammatory cytokines by adipocytes.5,6 Thus, a vicious cycle develops, which worsens the oncological outcome in patients. Chronic/systemic inflammation in cancer can be assessed by evaluation of neutrophil to lymphocyte ratio (NLR), absolute basophil count, absolute monocyte count, and absolute eosinophil count.7,8 High NLR is a predictor of recurrence-free survival (RFS), progression-free survival (PFS), and overall survival (OS) in various malignancies, including urinary bladder cancer (UBC), colorectal cancer, and prostate cancer.9–11 Body mass index (BMI) is the calculative marker for obesity and its relation to UBC prognosis has been recently studied by Ferro and colleagues.12,13
Octamer-binding transcription factor 4 (Oct-4), also known as POU domain, class 5, transcription factor 1 (POU5F1), is a transcription factor and a well established marker of CSC. 14 High/positive Oct-4 expression is found to be associated with tumor aggressiveness in non-small cell lung cancer, breast cancer, prostate cancer, bladder cancer, and rectal cancer.15–19 Atlasi and colleagues were the first to investigate correlation of positive Oct-4 expression (r = 0.6) in UBC. 20 Oct-4 has been associated with tumor recurrence and progression.18,21 Oct-4 expression analysis holds profound implications in UBC, as it is marked by frequent recurrence in non-muscle invasive bladder cancers (NMIBC) and early progression of muscle invasive bladder cancers (MIBC). Therefore, we aimed to study protein expression of Oct-4, along with hematological and metabolic involvement, in UBC prognosis.
Materials and methods
Study design
This study was approved by the Institutional Ethics Committee, Era’s Lucknow Medical College and Hospital (ELMC&H), Lucknow, India (ELMC/R_Cell/EC/2014/02, August 11, 2014) and written consent was obtained from all enrolled patients. Patients aged ⩾30 years were included, while those who were operated earlier, presented with secondary cancer, or who had received prior chemotherapy or radiotherapy, or had immunodeficiency disorders, were excluded from the study. A total of 39 patients with histopathologically proven UBC presented in Department of Surgery, ELMC&H, Lucknow, India and in Department of Urology, King George’s Medical University, Lucknow, India, between September 2014 and December 2014 were enrolled in the study, and the last follow up was conducted in December 2018. Tissue samples obtained from either transurethral resection of bladder tumor (TURBT) or radical cystectomy (RC) were fixed in 10% formalin for histopathological examination. Adjuvant therapy was provided to 56.4% (22/39) of UBC patients, of which 86.3% had NMIBC; the latter include 73.6% of low grade UBC cases, while 43.5% (17/39) received neoadjuvant therapy and all comprised high grade MIBC cases.
The complete blood count data before the date of first TURBT was retrieved and recorded. Pretreatment NLR data was collected and based on the cut-off value, that is, <3, ⩾3 categorization identified low and high level groups, respectively. 22
Weight (kilograms) divided by the square of height (meters) was defined as BMI. The pre-TURBT data of weight and height were collected, and patients were classified as underweight (BMI <18.5), normal weight (BMI 18.5–24.99), overweight (BMI 25–29.99), or obese (BMI ⩾30). 12 Tumor grading and staging was performed according to the World Health Organization/International Society of Urological Pathology 2004 grading system and the American Joint Committee on Cancer/TNM 2010 stage, respectively.
Oct-4 expression analysis by immunohistochemistry
All formalin-fixed paraffin-embedded (FFPE) cut sections were 3–5 µm thick and dried at 60°C for 1 h. The sections were deparaffinized and dehydrated in three steps of a xylene and gradient alcohol series (100%, 70%, and 50%), respectively. A microwave antigen retrieval procedure using antigen retrieval buffer (pH 9, EnVisionTM FLEX, DAKO) was performed. Washing was done with Tris buffer (pH 7.4) and blocking by hydrogen peroxide, followed by incubation with anti-Oct-4 (DAKO, Glostrup, Denmark) at room temperature for 1 h. Next, the sections were incubated with secondary antibody (EnVisionTM, FLEX/ HRP, DAKO, Carpinteria, CA, USA) for 30 min, followed by washing with Tris buffer three times and incubation for 5 min with chromagen [3,3′-diaminobenzidine (DAB)]. Counter staining was performed with hematoxylin for 3–5 min.
Scoring and evaluation of Oct-4
Oct-4 staining was both nuclear and cytoplasmic. Staining intensity was evaluated as follow: 0, no staining; 1, weak staining; 2, moderate staining; 3, strong staining, while the proportion of stained tumor cells was graded as: 0, no positive tumor cells; 1, 10% positive tumor cells; 2, 10–50% positive tumor cells; 3, 50% positive tumor cells. The final score was obtained by multiplying the percentage of positive cells by the intensity of staining. Based on this, staining scores of ⩽4 and ⩾6 were defined as low and high Oct-4 expression, respectively. 23 Positive controls included testicular seminoma tissue samples for Oct-4. 24 The slides were examined by two pathologists under ×400 magnification.
Follow-up
NMIBC patients underwent cystoscopy and urine cytology every 3 months for 2 years, then every 6 months for the last year. MIBC patients who underwent TURBT earlier also followed the same process, as well as random biopsies at every 3–6 months for 2 years. Those who underwent RC had urine cytology, along with renal ultrasound (RU) and thoracoabdominal computed tomography/magnetic resonance imaging (CT/MRI) every 3–6 months for 2 years. 25
Statistical analysis
All data were analyzed using SPSS software (version 20; IBM, Chicago, IL, USA). Mann–Whitney U test was performed to compare difference between groups. Association between Oct-4, NLR, and BMI with clinicopathological characteristics was determined by chi-square test. For survival analysis, the Kaplan–Meier method was performed using log-rank test. Survival data was also evaluated using Cox’s multivariate regression analysis along with hazard ratio (HR) and corresponding 95% confidence interval (CI). We defined OS from the day of treatment until the completion of follow-up period, that is, 48 months or death, while RFS was evaluated from the day of treatment until the first episode of recurrence or death. The PFS was calculated from the day treatment started until the progression of tumor or death. Tumor progression was determined by histopathological findings, or renal ultrasound/thoracoabdominal CT/MRI. All statistical analysis if had p < 0.05 were considered statistically significant and were two-sided.
Results
A total of 39 UBC patients were studied, of which 87.2% were male. The average age of male patients was found to be 58.54 ± 10.741 (mean ± SD). Mean tumor size was 3.91 ± 1.747. Table 1 depicts clinicopathological features association with Oct-4 expression, NLR, and BMI in enrolled UBC cases. The mean expression values of Oct-4 were found be significantly associated with tumor grade (p = 0.039), tumor stage (p = 0.021), lymph node status (p = 0.048), recurrence (p = 0.002), and progression (p = 0.020), as tabulated in Table 2. Figure 1 shows tissue expression of Oct-4.
Clinicopathological features.

BMI, body mass index; NLR, neutrophil to lymphocyte ratio; Oct-4, Octamer-binding transcription factor 4; RC, radical cystectomy; TURBT, transurethral resection of bladder tumor.
Association of Oct-4, hematological parameters and BMI with tumor grade, tumor stage, lymph node status, recurrence, and progression.

Significance between groups determined by Mann-Whitney U test (p < 0.05).
BMI, body mass index; NLR, neutrophil to lymphocyte ratio; Oct-4, Octamer-binding transcription factor 4.
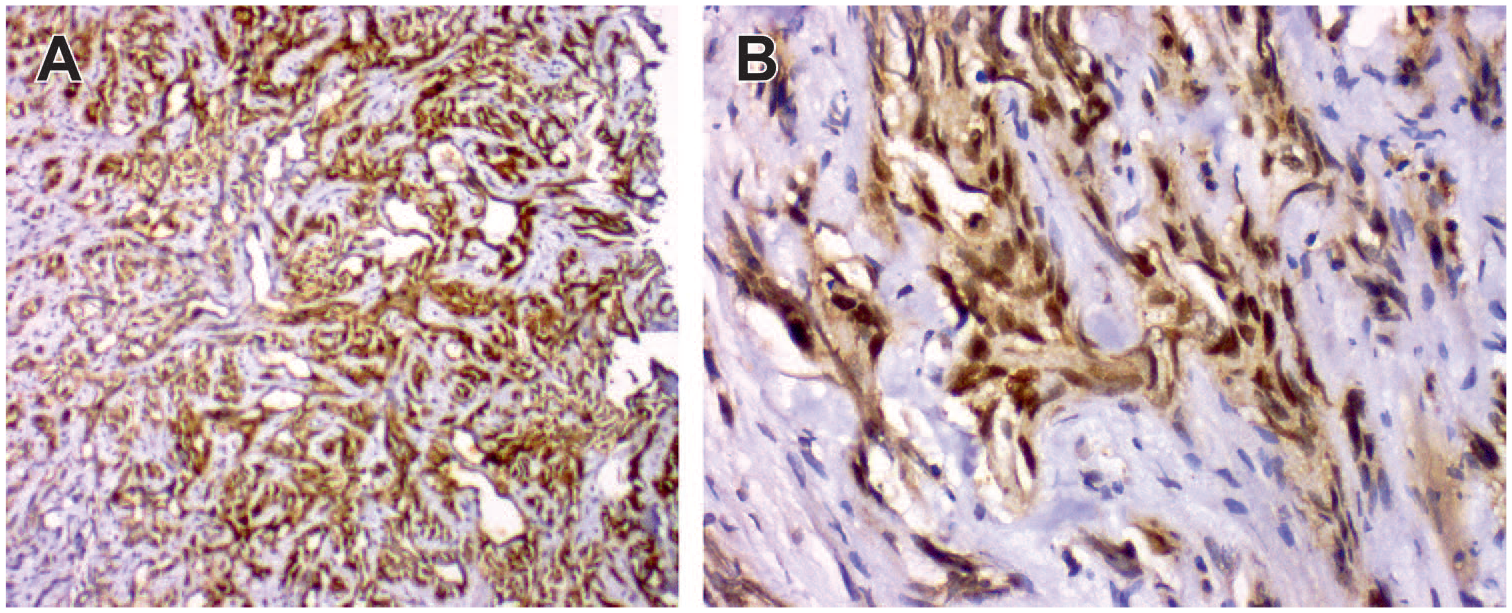
Tissue expression of Octamer-binding transcription factor 4.
High mean NLR values were significantly associated with tumor grade (p = 0.012), tumor stage (p = 0.041), and progression (p = 0.027). Similarly, high mean BMI was associated with tumor grade (p = 0.012), tumor stage (p = 0.019), recurrence (p = 0.002), as well as progression (p = 0.005).
Correlation of NLR and BMI with Oct-4 (low versus high) expression
The correlation between NLR and Oct-4 was found to be significant and spearman correlation coefficient(r) value was found to be 0.371 (p = 0.020). BMI too had significant correlation with Oct-4 expression (r = 0.502; p = 0.001). Furthermore distribution of NLR and BMI was significantly associated with Oct-4 low and high expression groups (p = 0.007; p = 0.002 respectively), as shown in Figure 2.
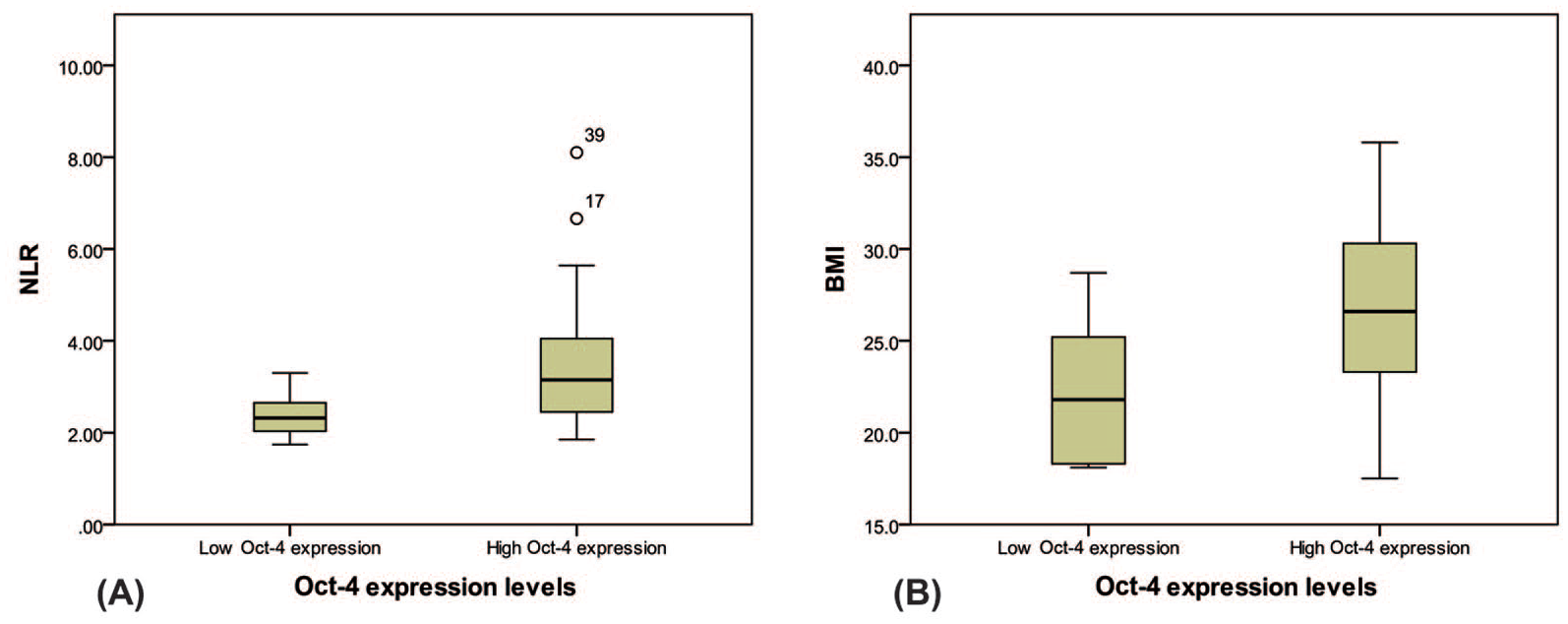
Distribution of NLR and BMI is significantly associated with Oct-4 low and high expression groups.
Survival analysis
The RFS at 1 year, 2 years, and 3 years was 53%, 44%, and 38%. Low Oct-4 expression was found to be associated with higher mean RFS (Figure 3a) of 41.7 ± 2.987 (months ± SEM) with 95% CI between 35.860 and 47.569 in compare to high Oct-4 expression with mean RFS of 22.2 ± 3.057 (95% CI, 16.226–28.208). NLR with ⩾3 value was significantly associated with shorter mean RFS (Figure 3b) of 23.8 ± 3.766 (95% CI, 16.420–31.184) while NLR with <3 value had mean RFS of 34.1 ± 3.541 (95% CI, 27.202–41.084). The PFS was 74%, 65%, and 47% at 1 year, 2 years, and 3 years respectively. The mean PFS was 31.3 ± 3.060 (95% CI, 25.376–37.372) and 29 ± 3.586 (95% CI, 22.039–36.095) for high Oct-4 expression (Figure 3d) and for NLR with ⩾3 (Figure 3e), respectively. The PFS months were high for patients with NLR <3 and low Oct-4 expression, that is, 42.1 ± 2.385 (95% CI, 37.481–46.830) and 45 ± 1.843 (95% CI, 41.433–48.658) respectively. The overall survival rates were 82%, 64%, and 53% at 1 year, 2 years, and 3 years respectively. High Oct-4 expression had a mean survival (Figure 3g) of 35 ± 2.498 (95% CI, 30.111–39.903) whereas low Oct-4 expression had a higher mean OS of 45.2 ± 1.817 (95% CI, 41.725–48.846). The UBC patients with high NLR (⩾3) had mean OS (Figure 2H) of 32.8 ± 2.915 (95% CI, 27.090–38.516) and those with low NLR (<3) had mean OS of 43.6 ± 1.918 (95% CI, 39.907–47.426). But we found no significant association of BMI with overall survival. Our result suggest that patient’s with high BMI have shorter RFS (p = 0.025) as well as shorter PFS (p = 0.032), as illustrated in Figure 3(c) and (f), respectively.

Association of Oct-4 expression, NLR, and BMI with PFS, RFS, and OS.
None of the UBC patient’s characteristics were significant in Multivariate Cox regression analysis (Table 3) for RFS, PFS, and OS except for Oct-4 expression, which proves to be an independent prognostic biomarker for RFS (p = 0.021).
Associations of study parameters with overall survival, recurrence-free survival, and progression-free survival.

BMI, body mass index; NLR, neutrophil to lymphocyte ratio; Oct-4, Octamer-binding transcription factor 4.
Discussion
In this study, we have shown the usefulness of CSC, chronic inflammatory markers, and BMI as promising prognostic biomarkers for prediction of UBC outcomes. On comparing with previously analyzed studies, measurement of these three components makes them appealing prognostic targets. First, this concept is derived from the fact that the urothelium comprises a basal layer, intermediate layer, and superficial layer that exhibit a hierarchy of differentiation. The basal cells of the basal layer are thought to comprise urothelial stem cells, which differentiate into transit-amplifying cells of the intermediate cell layer, which, in turn, differentiate into the umbrella cells of the superficial layer. 26 Thus, the CSCs of the bladder arise from the basal layer and differentiate with successive layers. Second, the tumor microenvironment mediates CSC survival by activation of the inflammatory response; therefore, the prevalence of chronic inflammation is the key to the activation of immune cells, generation of ROS and secretion of pro-inflammatory cytokines by adipocytes during obesity. Thirdly, the nearby adipocytes communicate with the cancer cells and promote tumor initiation, growth, and recurrence along with chemoresistance. 6
The first aspect of our study includes the prognostic evaluation of Oct-4, which is a well known regulator of CSC of UBC, and promotes stem-cell like properties by regulating self renewal of undifferentiated embryonic stem cells, adult human stem cells, and germ cells. Oct-4 regulates a variety of functions such as tumor initiation, proliferation, pluripotency, migration, resistance to cancer therapy and self-renewal.26,27 Oct-4 expression was found to be associated significantly with tumor grade, stage, and size but Atlasi and colleagues found no association with these clinicopathological parameters, although a highly significant Oct-4 expression correlation was found between tumor and nontumor tissue samples. 20 In an another study, 81.6% of UBC tissue samples showed approximately 0.6% of Oct-4 positive cancer cells. 28 High Oct-4 expression was present in 64.1% of UBC cases, while Hafeti and colleagues observed 44% of high Oct-4 expression and a significant association with tumor grade and stage. 29 A 64.2% Oct-4 positivity was found in UBC cases by Asar and colleagues. 30 Several cell lines such as Caco-2, HT-29, HepG2, LNCap, and HT-1376 of colon cancer, hepatocarcinoma, prostate cancer, and bladder cancer, respectively, were evaluated in a single study with ubiquitous Oct-4 expression. 31 A study examined bronchioloalveolar (BA) carcinoma and adenocarcinoma (AD) tissue samples for Oct-4 positivity by IHC, and found its positive expression in 38.4% (5/13) samples. Further evaluation of intensity and number of cells with Oct-4 expression was carried out by flow cytometry in BA carcinoma and AD-derived cultures. AD showed significant difference in derived cultures and tissue samples with respect to intensity and number of positive Oct-4 cells, while BA carcinoma showed no such difference. 15 In a mouse breast cancer cell line (4T1), a large and elevated number of tumor spheres in high Oct-4 expressing cells was seen. But, when Oct-4high 4T1 cells were injected in immune-competent mice, a 2-fold increase was observed compared with Oct-4low4T1 cells. 16 In another study, UBC recurrence had tumor size as the risk factor, and its progression was significantly associated with intensity of Oct-4 expression. 18 Median RFS is found to be shorter with high Oct-4-expressing UBC cases, that is, 13 months compared with 34.5 months for low Oct-4 expression, which is similar to our findings. Recurrent bladder tumors have significant higher Oct-4 expression than primary bladder tumors. 21 A multivariate analysis revealed high Oct-4 expression to be an independent recurrence prognostic marker of prostate-specific antigen (HR = 5.436, 95% CI, 2.492–11.859). 17 Oct-4 proves to be an independent prognostic factor for both OS (HR = 8.988, 95% CI, 4.084–19.779) and RFS (HR = 2.722, 95% CI, 1.502–4.932) intrahepatic cholangiocarcinoma. 32 However, we found that UBC patients with high Oct-4 expression have shortened RFS (HR = 0.240, 95% CI, 0.072–0.804, p = 0.021). In cervical cancer, Oct-4 is an independent risk factor of OS (HR = 11.23, 95% CI, 1.31–95.6; p = 0.027). 33 Poorer OS was observed with Oct-4 positivity in HER2+ breast cancer tissue. 34 We also observed a shortened OS with high Oct-4 expression (p = 0.014).
Second is the assessment of chronic inflammatory markers, that is, absolute monocyte count, absolute eosinophil count, absolute basophil count, and NLR. No significant association was found between mean absolute monocyte count, absolute eosinophil count, absolute basophil count values with tumor grade, tumor stage, lymph node status, recurrence, and progression. Similarly, univariate analysis showed no significant association with RFS, PFS, and OS. A multivariate analysis of high grade T1 UBC cases revealed a 30% increased recurrence hazard per unit rise of logarithmic basophil count. 35 In gastric cancer, treatment modality has an impact on total white blood cells count, and neutrophil, lymphocyte, monocyte, eosinophil and basophil counts. 8 NLR was found to be significantly associated with tumor grade and progression, but its mean values in different groups proved to be significant with tumor grade, tumor stage, and progression. A random model (I2 = 57.8%) showed that pretreatment high NLR has poor RFS (pooled HR = 2.31; 95% CI = 1.27–2.22; p = 0.006) and poor PFS (pooled HR = 2.14, 95% CI = 1.59–2.87; p = 0.001). 36 Likewise, pretreatment high NLR is associated with pathological stage, lymph node positivity (odd ratio = 1.09; p = 0.02), and extravesical tumor extension (odd ratio = 1.07; p = 0.03). Multivariate analysis portrays high NLR as an independent risk factor for UBC recurrence (HR = 1.04; p = 0.02) and death from UBC (HR = 1.04; p = 0.01). 7 For high grade T1 NMIBC, a systemic inflammatory marker (SIM) score was calculated based on the cut off values of NLR, lymphocyte/monocyte ratio, and platelet/lymphocyte ratio. The RFS (median follow up = 48 months) for SIM score 0,1,2,3 was 80.8%, 47.35%, 20.67%, and 17.06%, respectively, and for PFS (median follow up = 48 months) 92.0%, 75.67%, 72.85%, and 63.1%, respectively. 9 The high NLR values in colorectal cancer patients receiving neoadjuvant chemoradiotherapy is an independent predictor for OS (HR 2.336, p = 0.017) and PFS (HR 2.243, p = 0.034). 10 A meta-analysis of 14 studies showed that high NLR is a predictor of poor PFS/RFS (HR = 1.24, 95% CI = 1.05–1.46) and OS (HR = 1.38, 95% CI = 1.22–1.56) in prostrate cancer. 11
The third component is the significance of BMI in UBC. We depicted its significance with tumor grade, recurrence, and progression. Obese people have approximately five times more risk of UBC recurrence. 13 UBC cases with higher BMI had poor RFS(p = 0.025) as well as poor PFS (p = 0.032) in our study.
Few studies have established a correlation between CSCs and chronic inflammatory markers in cancer. ALDH1 expression in head and neck squamous cell carcinoma had a highly significant association with NLR (p < 0.001). 37 Our data shows significant distribution of pretreatment NLR with Oct-4 (low versus high) expression levels (p = 0.007). BMI too has significant distribution with Oct-4 (low versus high) expression levels (p = 0.001).
Conclusion
The cause and effect relationship of chronic inflammation, CSC, and obesity in cancer needs to be established and studied in today’s era of personalized therapy. Hence, we evaluated the prognostic significance of Oct-4, NLR, and BMI in UBC, but only Oct-4, a marker of CSC, proved to be a promising independent prognostic marker for determination of RFS.
Supplemental Material
Suplementary_Figure_1_jpg – Supplemental material for Oct-4: a prognostic biomarker of urinary bladder cancer in North India
Supplemental material, Suplementary_Figure_1_jpg for Oct-4: a prognostic biomarker of urinary bladder cancer in North India by Zainab Siddiqui, Anand N. Srivastava, Satya N. Sankhwar, Noorin Zaidi, Naseem Fatima, Shivangi Singh and Mohd. Yusuf in Therapeutic Advances in Urology
Footnotes
Acknowledgements
The authors are grateful to the Department of Statistics, Lucknow University, Lucknow, India for granting permission to use their SPSS software.
Authors’ Note
Zainab Siddiqui is also affiliated with Department of Biotechnology, Dr. A. P. J. Abdul Kalam Technical University, Lucknow, India.
Contributorship
The present study was conceptualized by Anand N. Srivastava and Satya N. Sankhwar, Zainab Siddiqui and Naseem Fatima. Data collection and all the experimental work was done by Zainab Siddiqui. The data interpretation was done by Anand N. Srivastava, Noorin Zaidi, Zainab Siddiqui, Naseem Fatima, and Mohd. Yusuf. The manuscript drafting was performed by Zainab Siddiqui and Shivangi Singh. All the authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and publication of this article: The study was supported by research grants in part from Era’s Lucknow Medical College and Hospital, Era University, Lucknow. Zainab Siddiqui. received Maulana Azad National Fellowship-Junior Research Fellow (MANF-JRF) fellowship (2013-14/MUS-DEL-22727/SA-III) from University Grants Commission, Government of India, New Delhi.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Informed consent
Written consent was obtained from all individual participants included in the study
Ethical approval
The study was approved by the Institutional Ethics Committee, Era’s Lucknow Medical College and Hospital, Lucknow, India (ELMC/R_Cell/EC/2014/02, August 11, 2014).
Guarantor
A.N. Srivastava
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
