Abstract
Storage lower urinary tract symptoms (LUTS) are characterized by an altered bladder sensation, increased daytime frequency, nocturia, urgency and urgency incontinence. Some evidence underlines the role of metabolic factors, pelvic ischemia, prostatic chronic inflammation and associated comorbidities in the pathophysiology of storage LUTS. A detailed evaluation of the severity of storage LUTS, and the concomitance of these symptoms with voiding and postmicturition symptoms, is mandatory for improving the diagnosis and personalizing treatment.
A detailed medical history with comorbidities and associated risk factors, a physical examination, a comprehensive analysis of all the features of LUTS, including their impact on quality of life, and a frequency–volume chart (FVC) or bladder diary, are recommended for men with storage LUTS.
Several drugs are available for the treatment of LUTS secondary to benign prostatic obstruction (BPO). Alpha-blockers (α-blockers), 5-α-reductase inhibitors and phosphodiesterase type 5 inhibitors are commonly used to manage storage LUTS occurring with voiding symptoms associated with BPO.
Muscarinic receptor antagonists and Beta 3-agonists (β3-agonists) alone, or in combination with α-blockers, represent the gold standard of treatment in men with predominant storage LUTS. There is no specific recommendation regarding the best treatment options for storage LUTS after prostatic surgery.
Keywords
Introduction
Lower urinary tract symptoms (LUTS) are highly prevalent in men, and can be associated with a remarkable reduction in quality of life (QoL) and interference with daily activities, 1 moreover, left untreated, LUTS can progress to urinary retention and related surgery, urinary tract infections or renal insufficiency. 2
Based on anatomical, physiological and urodynamic concerns, LUTS are conventionally divided into storage, voiding and postmicturition symptoms. 3
In a recent cross-sectional analysis of more than 100,000 middle-age and older men, 18.3% in the cohort reported moderate LUTS, and 3.6%, severe LUTSs.4 LUTS have been defined according to the modified International Prostate Symptom Score (m-IPSS) with identical questions regarding symptoms to the IPSS, but with a 4-point response scale: in particular, the cut-off values were 6–11 for moderate symptoms and 12–21 for severe symptoms.
The percentage of men with moderate–severe LUTS increased with age, from 10.6% among men aged 45–49 to 35.4% for men aged more than 80, while the prevalence of severe storage and voiding symptoms increased with age, with a remarkable occurrence of high storage symptoms from the age of 70. 4 Moreover, among those with severe LUTS, 90% had high voiding symptom scores, 76% had high storage symptom scores, while 66% had both high voiding and high storage symptom scores. 4 In the Tampere Ageing Male Urological cohort Study, the overall prevalence of moderate-to-severe storage LUTS in men aged 30–80 years were: urgency (10.9%), increased daytime frequency (5.4%), nocturia (⩾twice per night; 4.3%), and incontinence (1.7%). 5
Storage symptoms are characterized by bladder dysfunction, most commonly, detrusor overactivity. They are defined as the presence of urinary urgency, usually accompanied by frequency and nocturia, with or without urinary incontinence, in absence of urinary tract infection or other urethro-vesical dysfunctions, 3 and are now recognized as part of the overactive bladder (OAB) symptom complex, being as prevalent in men as in women.6,7
As reported in the EpiLUTS study, 45.7 % of the 14,139 men evaluated had storage LUTS. However, in this large population-based study of three countries, only 9.1 % had storage symptoms alone; the others also reported concomitant voiding and or postmicturition LUTS (Figure 1). 8
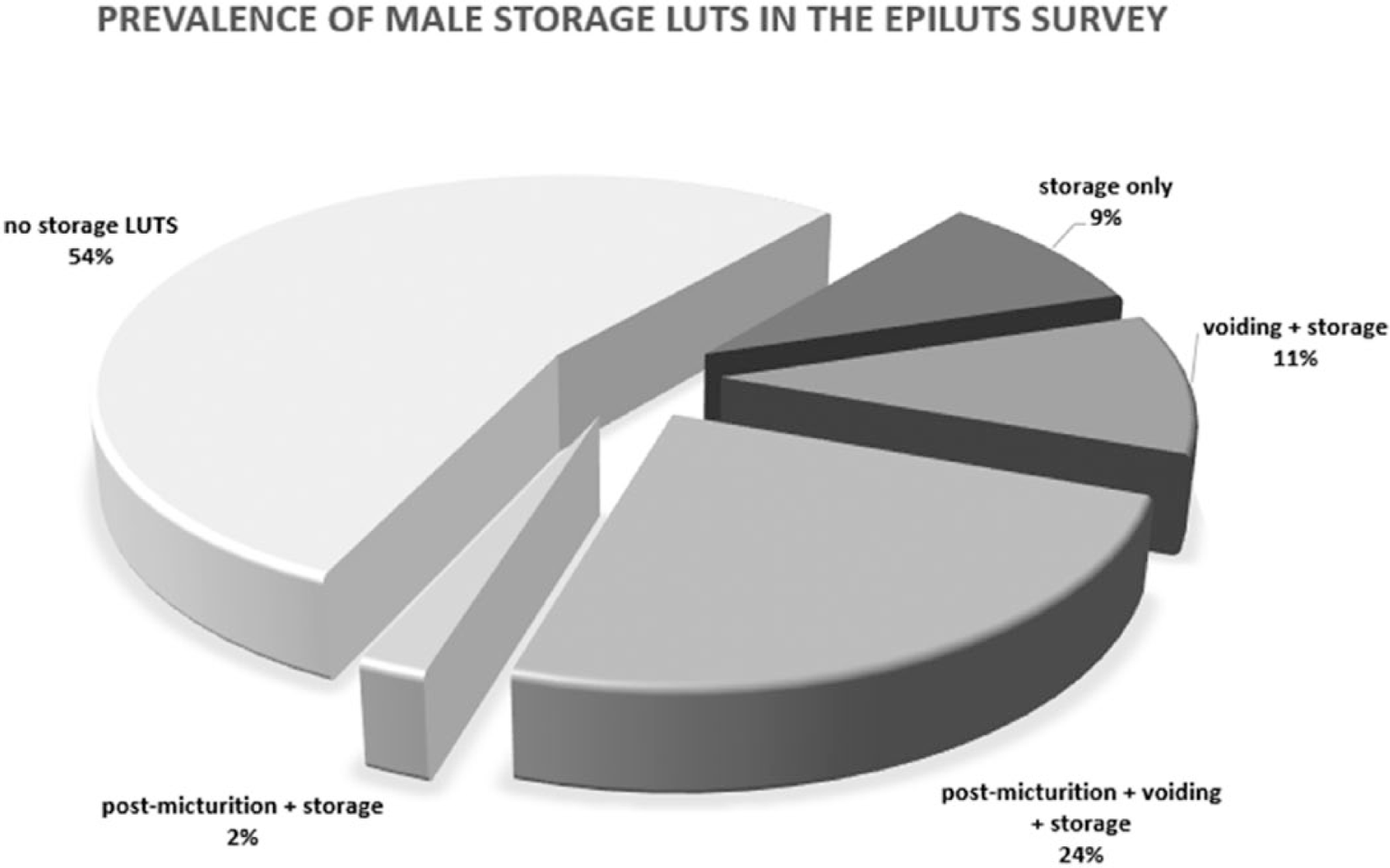
Prevalence of male storage lower urinary tract symptoms, data from epiLUTS survey.
Several data indicate that lifestyle behaviors may be significant for LUTS etiology and progression. 9 In particular, high fat diet, low physical activity and smoking, which affect metabolism and promote inflammatory processes, resulted in increased incidence and severity of LUTS in several studies of men. 10 At the same time, diet, physical activity and smoke reduction, seem to be potential targets for prevention of disease progression. 4
The aim of the present review is to frame the storage aspect of male LUTS, define the pathophysiologic mechanisms underlying this condition, propose a tailored diagnostic approach, critically analyze the impact of medications currently used and the gold standard of treatments, and the management of storage LUTS, including after prostatic surgery.
Storage lower urinary tract symptoms: definition and framing
LUTS in men can have a variety of underlying causes and contributing factors. Many years ago, male LUTS were considered to be a consequence of prostate obstruction. This prostate-centred approach did not allow explanation of the persistence of LUTS in a considerable proportion of men after transurethral resection of the prostate (TURP) or medical treatment. In recent years, urologists have realised that LUTS are not only caused by prostatic obstruction, but also by other factors such as nocturnal polyuria, strictures, neurogenic bladder, detrusor overactivity or detrusor underactivity during voiding. Comorbidities such as diabetes mellitus, renal disease, cardiac failure, obesity and metabolic syndrome can also play a role in the pathophysiology.
The European Association of Urology has reviewed the definitions related to male LUTS. 11
Bladder outlet obstruction (BOO) is a term for obstruction during voiding. An increased detrusor pressure and reduced urinary flow rate are the hallmarks of this phenomenon.
Benign prostatic obstruction (BPO) is a form of BOO and is caused by benign prostatic enlargement (BPE). Benign prostatic hyperplasia (BPH) should only be used to refer to a histological pattern.
The bladder also contributes to male LUTS. OAB syndrome is a symptom complex consisting of urgency with or without urgency incontinence, usually with increased frequency and nocturia, in the absence of proven infection or other pathology. Detrusor overactivity is a urodynamic observation, characterized by involuntary detrusor contractions during the filling phase.
Underactive bladder is a symptom complex that is characterized by prolonged urination time with or without sensation of incomplete bladder emptying, usually with hesitancy, reduced sensation of filling and slow stream. 12
When describing LUTS in men, it is important to distinguish storage symptoms from voiding and postmicturition symptoms.13,14 Storage symptoms are characterized by an altered bladder sensation, increased daytime frequency, nocturia, urgency or urgency incontinence. Voiding symptoms consist of hesitancy, intermittency, slow stream, splitting or spraying of the stream, straining and terminal dribble. Postmicturition symptoms include the feeling of incomplete bladder emptying and postmicturition dribble. Distinguishing these three symptom groups is important because they may lead to different medical or surgical treatment.
Refining the symptom presentation of the patient is crucial for the further assessment of the condition and the subsequent treatment. When symptoms are measured by validated questionnaires, such as the IPSS or the International Consultation on Incontinence Questionnaire on male lower urinary tract symptoms (ICIQ-MLUTS), it is important not only to look at the total symptom score but also at the breakdown of the storage and voiding subscores. In patients with mixed symptoms, storage, voiding and postmicturition symptoms may be present at the same time (Figure 1), and the dominance of certain symptoms can be used to guide the initial therapy.
Pathophysiology of storage lower urinary tract symptoms
BOO related to age-dependent progressive prostate overgrowth has been historically considered the main pathogenetic factor causing storage LUTS. However, convincing evidence published in the last few years has clearly documented that age-related conditions and in particular, metabolic derangements, play an independent and crucial role in the development of BPH, which may progress to BPO, and storage LUTS pathogenesis.15–17 Several reports have documented that obesity, and in particular, visceral obesity, is frequently comorbid with BPO/storage LUTS.15–17 In addition, a possible link between obesity-associated hyperinsulinemia/glucose intolerance and BPO has been documented since 1966.15–17 More recently, Nandeesha and colleagues 18 reported that insulin levels were an independent predictor of prostate volume. Similar results were documented in a large sample of patients seeking medical care for couple infertility. 19 The specific mechanisms linking obesity and hyperglycemia to BPE have been not completely clarified but hyperinsulinemia might play a crucial role, most probably stimulating prostate growth acting on insulin-like growth factor receptors.15–17 Dyslipidemia represents another independent risk factor associated with LUTS. By analyzing the data of the cohort in the Rancho Bernardo Study, a survey performed in middle-to-upper-middle class, community-dwelling adults in southern California, US, Parsons and colleagues 20 reported a fourfold increased risk of LUTS among diabetic men with elevated low-density lipoprotein (LDL) cholesterol. Similarly, Nandeesha and colleagues 18 reported that circulating total and high-density lipoprotein (HDL) cholesterol were associated, in a positive and negative manner, respectively, with prostate enlargement in a series of 50 symptomatic BPO cases, diagnosed by digital rectal examination and transabdominal ultrasound and admitted for prostatectomy, and 38 controls. Interestingly, it has been reported that lipids (oxidized LDL) increase in vitro the secretion of growth (e.g. beta basic fibroblast growth factor) and pro-inflammatory factors [interleukin-6 (IL-6), IL-8, and IL-7] by isolated stromal BPH cells.15–17 Hence, it can be speculated that dyslipidemia, could induce the development of an inflammatory response within the prostate, leading to BPO progression and LUTS.15–17 Considering the above evidence, it is not surprising that a recent meta-analysis of the available evidence documented that metabolic syndrome (MetS), is tightly associated with a higher total and transitional zone prostate volume. 21 Interestingly, the latter analysis also showed that central obesity and reduced HDL cholesterol were the most important predictors of MetS-associated enlarged prostate volume. 21 Finally, it is important to emphasize that obesity and MetS are frequently associated with reduced testosterone volume. 22 MetS-related hypogonadism has been suggested as a contributor of the chronic prostatic inflammatory response associated with metabolic derangements 23 (Figure 2).
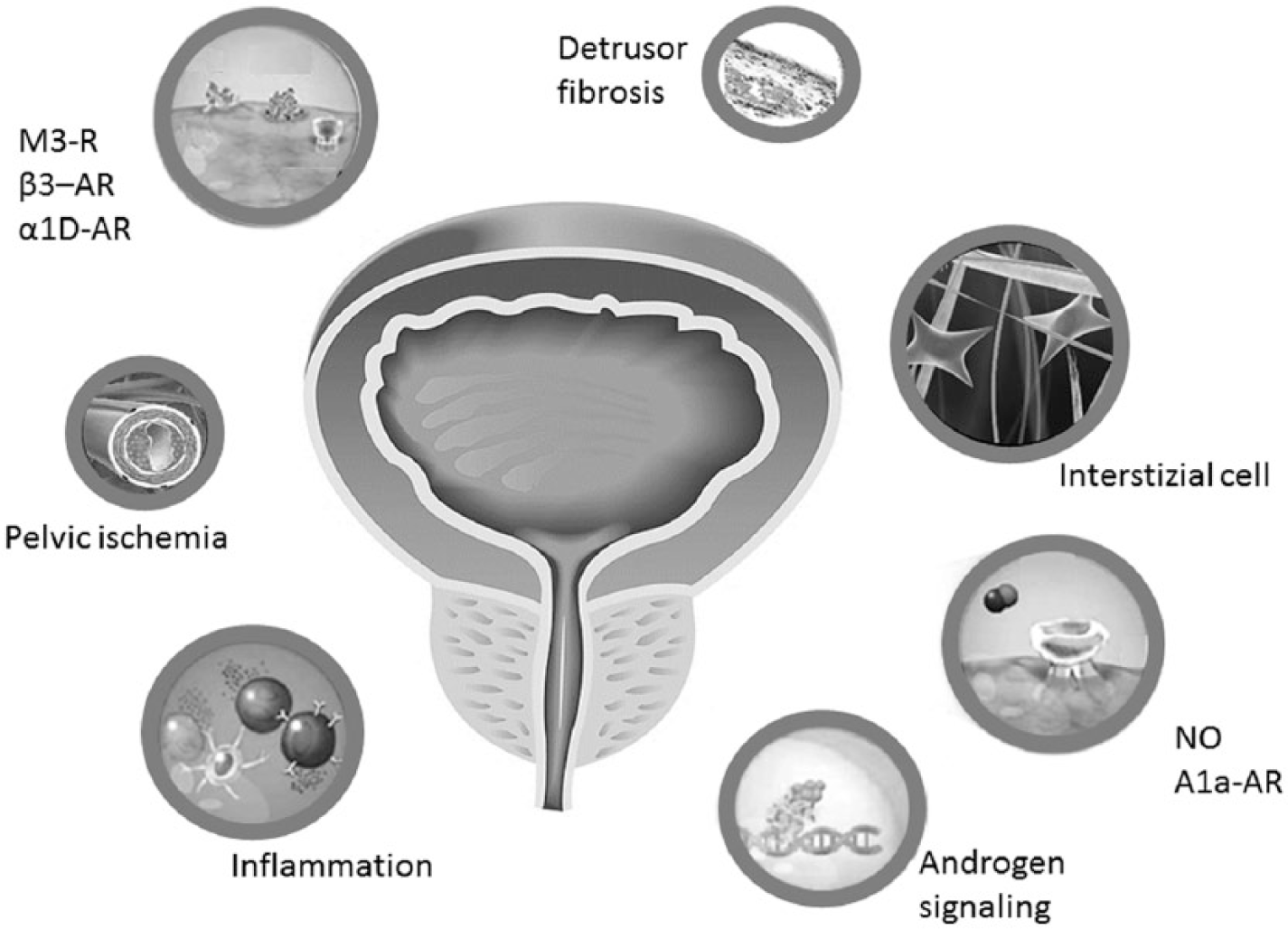
Pathophysiological pathways underlying storage lower urinary tract symptoms.
Assessment of male lower urinary tract symptoms: focus on storage lower urinary tract symptoms
In the past, LUTS in men were simplistically attributed to the prostate gland, and terms such as BPH, BPE, BPO, and BOO have been used to describe the underlying etiology. 24 However, it is only in recent years that urologists have begun to acknowledge that LUTS can result from several urological and nonurological conditions. It seems that contribution from both detrusor and outlet plays a dominant role in the etiology of male lower urinary tract storage and voiding symptoms.
This understanding has a significant impact on the assessment of LUTS since tests are used to make the differential diagnosis of the potential causes of symptoms. In addition, the diagnostic evaluation should be able to define the clinical profile of men with LUTS. 13 In this context, the identification and recognition of the predominant type of symptoms (storage or voiding) is essential in order to provide the appropriate care.
Initial investigational tests of patients with LUTS include medical history, symptom score questionnaires and bladder diary, physical examination, digital rectal examination, urinalysis, serum prostate-specific antigen (PSA) and postvoid residual urine (PVR). 13 A structured medical history aims to identify conditions relevant to the etiology and capture current medication, lifestyle habits, emotional and psychological factors. Symptom scores are helpful in quantifying LUTS and in identifying which type of symptoms are predominant. PVR is not necessarily associated with BOO, since high PVR volumes can be a consequence of obstruction or poor detrusor function. 25 A high baseline PVR has been associated with an increased risk of symptom progression 26 and it is of particular importance when antimuscarinics are used for the treatment of storage symptoms. The bladder diary is essential in determining the cause of nocturia and in documenting frequency, leakage episodes and voided volumes.
Bothersome symptoms requiring treatment will result in an escalation of diagnostic tests to allocate patients for medical or surgical treatment. Such tests are frequency–volume charts (FVCs), renal function assessment, imaging of prostate and upper urinary tract, uroflowmetry, and pressure–flow studies. Not all of these tests are mandatory in the assessment of each individual patient, but they are tailored according to each situation. With regard to storage symptoms, FVCs or bladder diaries are recommended. 13 Parameters that can be derived from the FVC and bladder diary include: daytime and night-time voiding frequency, total voided volume, the fraction of urine production during the night (nocturnal polyuria index), and volume of individual voids. The recommended duration of the FVC is 3 or more days. 27
Impact of medications for benign prostatic obstruction on storage lower urinary tract symptoms
Currently, several drugs are available for the treatment of LUTS secondary to BPO. Alpha-blockers (α-blockers) are considered the first-line therapy for the treatment of LUTS, since they can offer an improvement of LUTS within few days with a good safety profile. Controlled studies show that α1-blockers typically reduce IPSS by approximately 30–40% and increase maximum urinary flow (Qmax) by approximately 20–25%. However, they have no impact on BPO progression, and their efficacy usually decreases after 2 years.
Recently, Aikawa and colleagues analyzed more than 4800 patients treated with an α-blocker for LUTS. Interestingly, they confirm the reduction of 30% of total IPSS after treatment, also in the subgroup of patients with prevalent storage LUTS. 28
Indeed, α-blockers reduce the dynamic component of BOO to decreasing the smooth muscle tone at the bladder neck and inside the prostate. Moreover, they increase arterial perfusion in the LUT and cystometric capacity (C max) and may counteract, as suggested by Pinggera and colleagues, arterial occlusive disease and concomitant chronic bladder ischemia that may produce bladder dysfunction and storage LUTS. 29
Five-α-reductase inhibitors (5-ARIs) are a recommended treatment option in men with moderate-to-severe LUTS and large prostate. They reduce prostate volume and the fixed component of BPO after 3–6 months of treatment. Long-term treatment with 5-ARIs is well tolerated and reduces the risk of acute urinary retention (AUR) and the need for BPO-related surgery. Only a few studies have evaluated the impact of 5-ARIs on storage LUTS. Matsukawa and colleagues showed a significant reduction of 2.5–3.0 points in storage IPSS subscore and the disappearance of involuntary detrusor contraction in 52.0% of patients evaluated with pressure flow study after 12 months of treatment with dutasteride. 30 Moreover, the IPSS storage subscore reduction with α1-blocker and 5-ARI combination therapy was significantly better than dutasteride alone from 3 months onwards, and significantly better than tamsulosin alone from 12 months onwards. 31 A proposed therapeutic effect of 5-ARIs on storage LUTS is the reduction of the detrusor’s mass and therefore of the remodeling of bladder structure and function. 32
Phosphodiesterase type-5 inhibitors (PDE5Is) are an effective treatment option for men affected by bothersome LUTS with concomitant erectile dysfunction (ED). They have a relaxant effect on smooth muscle fibers of the prostate, urethra, and bladder neck, and a concomitant vasodilatory effect that may increase oxygenation of the LUT. 33 Furthermore, they decrease the afferent signaling in the micturition reflex, downregulate inflammatory processes in LUT and exert antiproliferative effects.
The only PDE5I licensed for the treatment of male LUTS with or without ED is tadalafil, at a dose of 5 mg daily. 11 Long-term experience with tadalafil is limited, so we have no data about its efficacy on disease progression. Brock and colleagues reported that tadalafil significantly reduced BPO-associated LUTS compared with placebo in men without ED (IPSS −5.4 versus −3.3, p < 0.01; IPSS storage subscore −1.9 versus −1.3, p < 0.05). 34
In a recent review, Gacci and colleagues reported a similar significant improvement of IPSS storage subscore compared with placebo. 33 The combination of PDE5Is with α-blockers induce a small, but statistically significant improvement of maximum flow rate as compared with α-blockers alone, in addition to the positive effect on IPSS. PDE5Is alone or in combination with α-blockers are well tolerated and in contrast to other treatment modalities for LUTS, PDE5Is induce beneficial effects in erectile function parameters.
Recommended treatment for predominant storage lower urinary tract symptoms
In men with nonbothersome storage LUTS, watchful waiting (WW) is a viable option as most of them will remain stable for years. However, first-line treatment for patients with storage LUTS should be lifestyle advice and behavioral modifications. 35 The most commonly prescribed strategies are weight loss, caffeine and alcohol restriction, bladder training, bladder control strategies, pelvic floor muscle training, and fluid intake restriction at specific times. If conservative measures fail, different drugs can be used.
Anticholinergic drugs
Currently, the pharmacological treatment pivot for storage LUTS is represented by anticholinergic drugs such as oxybutynin, tolterodine, fesoterodine, darifenacin, solifenacin, or trospium. 35
Anticholinergic agents or antimuscarinics act as m-cholinoceptor antagonists, by blocking M2 and M3 receptors on smooth muscle cells of the bladder, mainly during the urinary storage phase, reducing involuntary detrusor contractions or altering contraction thresholds, 36 resulting in decreased urgency and increased bladder capacity. However, muscarinic receptors are also found in other parts of the body, including brain, heart, gut, salivary glands, and tear ducts, leading to several treatment adverse events (TAEs) such as dry mouth, constipation, tachycardia, accommodation disorder, and cognitive dysfunction. As patient tolerability represents a fundamental parameter for the administration of antimuscarinics, after the introduction of oxybutynin, more selective agents have been marketed and extended release (ER) formulations were developed.
Since its introduction in clinical practice, tolterodine has been the active comparator in several studies for its good efficacy profile with a lower occurrence of TAEs compared with oxybutynin, above all, in relation to dry mouth (22.3% versus 29.7%, p = 0.02). 37
The STAR Study compared tolterodine 4 mg with flexible dosing of solifenacin, which proved to be superior in reducing the numbers of urgency episodes per 24h (–2.85 versus –2.42), urgency incontinence episodes per 24h (−1.42 versus −0-83), incontinence episodes per 24h (–1.60 versus 1–11), and pad use (−1.72 versus −1.19). However, dry mouth and constipation were significantly more common in the solifenacin arm (18.2 versus 14.5% and 3.0 versus 1.2%, respectively), although they were mainly of mild-to-moderate severity. 38
An active control arm with tolterodine was evaluated by Chapple and colleagues to compare fesoterodine 4 mg or fesoterodine 8 mg once daily with placebo in a randomized controlled trial. 39 Fesoterodine 8 mg was superior to tolterodine 4 mg for median change in number of urgency urinary incontinence episodes (p < 0.05) and volume voided per micturition (p < 0.05) from baseline, while similar efficacy was shown for fesoterodine 4 mg and tolterodine 4 mg. Fesoterodine 4 mg and tolterodine ER 4 mg had a similar safety profile, while fesoterodine 8 mg was associated with significantly higher rates of dry mouth (p < 0.0001) and dry eyes (p = 0.02) compared with tolterodine 4 mg. 40 In 2013, Ginsberg and colleagues 41 evaluated the efficacy of fesoterodine 8 mg versus tolterodine ER 4 mg for OAB symptoms in terms of patient-reported outcomes, supporting the superiority of fesoterodine 8 mg over tolterodine ER 4 mg in improving severe urgency and symptom bother in men.
Several studies have evaluated the efficacy of α-blocker/antimuscarinic combination therapy. The TIMES Study 42 showed that patients treated with tamsulosin/tolterodine combination therapy, but not with tamsulosin, tolterodine or placebo alone, experienced a significant improvement, as defined by patient perception questionnaire (80% versus 71%, 65%, and 62%, respectively). Moreover, only combination therapy significantly improved total IPSS and QoL as well as the IPSS storage subscore, and in patients with large prostates (prostate volume ⩾30 ml) significantly reduced 24 h voiding frequency (2.8 versus 1.7 with tamsulosin alone, 1.4 with tolterodine alone, or 1.6 with placebo). Antimuscarinics TAEs (e.g. dry mouth or constipation) occurred more often in the combination therapy group than in patients receiving monotherapy. However, no clinically relevant increase in PVR or AUR was reported. The VICTOR 43 trial evaluated the safety and tolerability of adding solifenacin in men with residual OAB symptoms after tamsulosin monotherapy. Solifenacin add-on led to significantly less urgency episodes than placebo plus tamsulosin, with the rare occurrence of TAEs, including low incidence of AUR. Accordingly, the combination of tamsulosin 0.4 mg plus solifenacin 6 mg resulted in significant improvement of storage symptoms and overall QoL in men with both storage and voiding LUTS compared with tamsulosin monotherapy, with no further risk of AUR, despite a documented but nonclinically significant rise of PVR. 44 Nevertheless, according to the European Association of Urology guidelines, combination treatment with an α1-blocker and a muscarinic receptor antagonist has to be prescribed with caution in men with a PVR > 150 ml (Table 1).
Lower urinary tract symptoms treatment according to guidelines and current strongest supporting evidences.

AUA, American Urological Association; BOO, bladder outlet obstruction; BPE, benign prostatic enlargement; BPH, benign prostatic hyperplasia; DRE, Digital Rectal Examination; EAU, European Association of Urology; 5-ARI, 5-α-reductase inhibitor; GR, grade of recommendation; HRQoL, health-related quality of life; IIEF, International Index of Erectile Function; IPSS, International Prostate Symptom Score; LE, level of evidence; LUTS, lower urinary tract symptoms; OAB, overactive bladder; PDE5I, phosphodiesterase type-5 inhibitors; QoL, quality of life; PSA, prostate-specific antigen; PVR, postvoid residual urine; Qmax, maximum urinary flow; RCT, randomized controlled trial; SI, symptom index; UUI, urgency urinary incontinence.
β3-adrenoreceptor agonists
More than 60% of patients discontinue antimuscarinic therapy over a 12-month period due to inadequate symptom control or intolerable AEs (e.g. dry mouth, constipation). 49 To overcome this limitation, the role of β-adrenoceptor (AR) agonists (β1, β2, β3) in relaxation of the detrusor has been recently evaluated: in particular, β3-AR, which represents the main actor in mediating human detrusor relaxation. Indeed, the urinary bladder is innervated by both sympathetic and parasympathetic nervous systems. Activation of sympathetic nerves contributes to urine storage by relaxing the detrusor muscle through activation of β-ARs.
Mirabegron, a selective β3-AR agonist, was approved for the treatment of OAB symptoms by the US Food and Drug Administration (FDA) in 2012, and is the first of this class of compounds with a different mechanism of action from antimuscarinics. Indeed, mirabegron improves the storage capacity of the bladder without impairing bladder contraction during the voiding phase. 50
Nitti and colleagues demonstrated that treatment with mirabegron 50 and 100 mg was non-inferior to placebo for Qmax and detrusor pressure at Qmax in men with LUTS and BOO. The adjusted mean difference of mirabegron 50 mg and 100 mg versus placebo was 0.40 and 0.62 ml per second for Qmax, and −5.94 and −1.39 cm H2O for detrusor pressure at Qmax, respectively. Mirabegron at both doses of 50 and 100 mg was associated with significant improvements and reduced the mean number of micturitions per 24 h versus placebo, and the mean number of urgency episodes (severity 3 or more) per 24 h, from baseline to treatment end. The incidence of AEs was similar for mirabegron and placebo. However, a dose-dependent increase in PVR volume was observed at the final visit. 47
Recently, 50 male LUTS patients were enrolled to evaluate mirabegron 50 mg as additional therapy to an α1-blocker. The combination therapy led to the improvement of total Overactive Bladder Symptom Score (from 6.0 to 4.4), total IPSS (from 15.1 to 11.8), and IPSS-QoL score (from 4.2 to 3.4), as well as voided volume (from 137.3 ml to 154.1 ml) and Qmax (from 9.1 ml/s to 11.0 ml/s), without any significant change in PVR. 51
Storage lower urinary tract symptoms after prostatic surgery
Simple and radical prostatectomy (RP) are the most common surgical procedures in urologic units all over the world for BPO and prostate cancer (PCa), respectively. After both interventions, patients can present with bothersome storage LUTS postsurgery that are persistent and can worsen the QoL outcomes.
After RP, data on bladder-filling sensation, cystometric capacity, impaired detrusor contractility, detrusor overactivity, and BOO are limited and often contradictory. Impaired bladder compliance was present in 8–39% of patients and was de novo in about 50%. Impaired detrusor contractility was found in 29–61% of patients, was de novo in 47%, and recovered in about 50% of patients. Detrusor overactivity was a de novo dysfunction in 2–77% of patients. 52
Likewise, Hosier and colleagues 53 found that 29% of patients developed de novo storage LUTS after RP, including urgency, urgency incontinence, frequency, and nocturia. After RP, several items, including preoperative patient characteristics, surgeon experience and surgical technique, can influence the prevalence of postprostatectomy LUTS. 52 After catheter removal, most patients reported some level of transitory bothersome LUTS. In particular, in the MIRROR Study, a multicenter Italian survey on men treated with RP for PCa, only 32.7 % of men were continent 1 month after RP. After RP, regardless of surgical procedure, urinary continence outcomes were significantly lower in obese men at either 12- or 24-month follow up compared with nonobese ones. Furthermore, it was demonstrated that higher BMI was associated with a worsening of LUTS at 3 months postoperatively [odds ratio (OR) 1.148, p = 0.048]. At 1 year after RP, a higher BMI was negatively associated with improvement in storage symptoms. 52 Current evidence suggests that the rate of medical treatment for storage LUTS after RP is low. The absence of a gold standard treatment and the risk of potentially bothersome side effects, such as constipation, dry mouth, and urinary retention, justify the delay of medication. 53 However, treatment based on urodynamic findings could be recommended if incontinence persists more than 12 months after RP. 52
After simple prostatectomy for severe LUTS due to BPO, the full recovery of urinary symptoms, above all storage symptoms, can be more easily achieved in men with a waist circumference (WC) of <102 cm compared with those having a WC of ⩾102 cm. 54 Moreover, patients with MetS reported a lower recovery in total and storage IPSS compared with those without MetS after prostatic surgery. Persistence of storage LUTS in obese men after prostate surgery was independent from the full recovery of the voiding function postsurgery.
In addition, an increasing number of MetS components resulted in increased postoperative storage IPSS scores. Several studies have been focused on the impact of MetS parameters on storage LUTS, risk of LUTS progression and risk of prostatic surgery. Thus, MetS can be considered as one of the main preoperative risk factors of postprostatectomy persistence of storage LUTS. 55 Moreover, Han and colleagues showed that age > 70 years (OR 2.474; p = 0.001), history of diabetes (OR 1.949; p = 0.040), history of cerebrovascular accident (OR 5.932; p = 0.001), any previous LUTS/BPO medication use (OR 5.384; p = <0.001), and previous antimuscarinic drug use (OR 2.962; p = 0.016) were significantly associated with symptom persistence and continuing medical therapy after surgery. 56
Currently, there are no specific medical treatment recommendations for persistent storage LUTS post simple prostatectomy or endoscopic surgery. However, α-blocker monotherapy has been considered for its direct effect on smooth muscle but also at the level of the spinal cord, ganglia, and nerve terminals that influence various neuronal outflows to the LUT. 48 Although antimuscarinics are the mainstay of treatment for storage LUTS, in clinical practice they are rarely prescribed, for the theoretical concern of AUR increased risk. 56
Conclusion
Storage LUTS (urinary urgency, usually accompanied by frequency and nocturia, with or without urgency incontinence, in absence of urinary tract infection or other urethro-vesical dysfunctions) alone, or in combination with voiding and postmicturition LUTS, is a common condition in elderly men.
New evidence regarding the role of metabolic factors, pelvic ischemia, prostatic chronic inflammation and associated comorbidities, in the pathophysiology of storage LUTS, has allowed better understanding of the complexity of the clinical approach needed to manage these patients. The identification and recognition of the predominant storage symptoms, based on a tailored clinical and instrumental workup, is mandatory in order to provide the appropriate care.
First-line treatment for patients with storage LUTS should be lifestyle advice and behavioral modifications. α-blockers, 5-ARIs and PDE5Is allow some improvement in storage LUTS in men with concomitant voiding LUTS due to benign prostatic obstruction.
Muscarinic receptor antagonists and β-3 agonists alone or in combination with α-blockers represent the gold standard of treatment in men with predominant storage LUTS due to detrusor overactivity.
There is no specific recommendation regarding the best treatment options for storage LUTS after prostatic surgery, due to the lack of high level of evidence.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship or publication of this article: M Gacci: Consulting, honoraria, research support from Lilly, Bayer, Pfizer, Ibsa, GSK, Astellas, Pierre Fabre, Konpharma. D De Ridder: Consulting, honoraria, research support from Astellas, Medtronic, Axonics, Allergan. S Gravas: Consulting, honoraria, research support from Pierre Fabre Medicament, GSK, Astellas. P Abrams: Consulting, honoraria, research support from A Sebastianelli, P Spatafora and S Serni declare no conflict of interest.
