Abstract
Mirabegron is a novel, once-daily, orally active, first-in-class, potent β3-adrenoceptor agonist recently approved by Food and Drug Administration for overactive bladder therapy. Phase II studies and four large-scale phase III multinational randomized, controlled trials have supported the efficacy and tolerability of mirabegron in the clinical trial setting of patients with overactive bladder for up to 12 weeks of therapy and in the long term (12 months). The reported incidence and severity of treatment-emergent and serious adverse effects were similar to antimuscarinics, but with a more than threefold lower incidence of dry mouth compared with tolterodine. However, the effects on the cardiovascular system, pharmacokinetic interactions with other drugs, and increased incidence of new malignant events will require careful evaluation in the near future.
Introduction
Overactive bladder (OAB), defined by the International Continence Society as urgency with or without urinary incontinence, usually associated with frequency and nocturia [Abrams et al. 2002], is a multifactorial and common health disorder associated with detrimental effects on quality of life and huge economic burden [Irwin et al. 2006; Sacco et al. 2010]. Irrespective of the pathophysiology, the aim of treatment of OAB is to obtain symptom relief without affecting the empty phase of the micturition cycle. Current first-line treatment relies mainly on conservative therapeutic strategies, such as behavioural therapies, functional electrical stimulation, clean intermittent catheterization and pharmacological treatment. Second-line treatment options in refractory patients are neuromodulation, botulinum toxin and surgery. Antimuscarinic drugs are US Food and Drug Adminstration (FDA)-approved medications for OAB and represent the mainstay of pharmacological treatment [Chapple et al. 2008a]. Unfortunately, antimuscarinics are not always effective in controlling OAB symptoms and rarely ‘cure’ OAB. Furthermore, bothersome adverse effects (AEs), including dry mouth, nausea, constipation and central nervous system AEs, due to their lack of bladder selectivity, cause poor long-term adherence to treatment [Milsom et al. 2001; D’Souza et al. 2008].
Recent advances in the understanding of the physiopathology of OAB have driven a huge amount of basic and clinical research into novel pharmacological compounds. Among potential novel peripherally acting drugs, β3-adrenoceptor (AR) agonists appear very promising [Sacco et al. 2008].
Mirabegron (also known as YM178) is a novel, once-daily orally active, first-in-class, potent β3-AR agonist [Takasu et al. 2007]. Regulatory applications for mirabegron are under review in several countries. In July 2011, mirabegron was granted marketing approval in Japan and was launched in September 2011. In June 2012 FDA also approved mirabegron (Myrbetriq, Astellas Pharma US, Inc.). Thereafter we reviewed clinical data on the safety and efficacy of mirabegron by searching English language full papers and abstracts in MEDLINE, ClinicalTrials.gov, controlled-trials.com, clinicaltrialsfeeds.org and proceedings of international scientific meetings. The reports of the Division of Reproductive and Urologic Products Office of the FDA [FDA, 2012], which recently reviewed efficacy and safety of mirabegron for marketing approval, has also been used as a source of data.
Mechanism of action
In mammalian species detrusor relaxation is mainly mediated by the cyclic adenosine monophosphate pathway, which is activated via the fixation of noradrenalin to β -ARs [Andersson et al. 2009]. β1,2,3-ARs have all been demonstrated in both animal and human bladder [Andersson et al. 2009]. However, β3-ARs account for more than 95% of all β-AR mRNA in the human bladder and are thought to be the main β-ARs mediating human detrusor relaxation [Takeda et al. 1999, 2000; Igawa et al. 2010]. β3-ARs have been found to be highly and preferentially expressed on urinary bladder tissues, including the urothelium, interstitial cells, and detrusor smooth muscle [Andersson et al. 2009; Andersson and Arner, 2004; Otsuka et al. 2008; Limberg et al. 2010; Fujimura et al. 1999].
In animal studies β3-AR agonists directly cause dose-dependent detrusor relaxation during the storage phase of the micturition cycle and inhibited neurogenic detrusor overactivity and experimentally induced or bladder outlet obstruction (BOO)-associated OAB [Woods et al. 2001; Hicks et al. 2007; Takasu et al. 2007]. It has been shown that, compared with other agents (including antimuscarinics), β3-AR agonists increase bladder capacity with no change in micturition pressure and residual volume, supporting the principle of β3-AR agonism as a new therapeutic approach to OAB.
Receptors on smooth muscle are thought to be the main site of action for these agents in treating detrusor overactivity, although they also promote relaxation by directly influencing urothelial functions [Yamaguchi and Chapple, 2007; Birder et al. 1998]. In addition, it has been demonstrated that β3-AR agonists can directly inhibit afferent nerve firing in spinal cord transected rats [Kanai et al. 2011]. These drugs have many fewer, if any, cardiovascular side effects compared with β1,2-AR agonists [Kaidoh et al. 2002]. Pilot studies already reported beneficial effects with terbutaline [Lindholm and Lose, 1986] and clenbuterol [Gruneberger, 1984] in patients with OAB. Consequently, several pharmaceutical companies are developing a number of β3-AR selective agonists aimed at treating OAB. In particular, an extensive clinical development and clinical pharmacology programs which consist of 41 studies were involved in the development of mirabegron by Astellas Pharma (Ibaraki, Japan).
Pharmacokinetics
The manufacturer conducted 23 human pharmacocynetic studies, including extensive pharmacocynetic/pharmacodynamic modelling and simulation [FDA, 2012]. The plasma protein binding is 71%. The drug is widely distributed in the body with a large volume of distribution of approximately 1670 liters. The terminal elimination half life of mirabegron is approximately 50 h. The time to reach maximum concentration (T max) is 3–4 h. The absolute bioavailability is approximately 29% at a 25 mg dose and 35% at a 50 mg dose. The bioavailability appears to be higher in women (40–50% higher exposure) than in men; however, when normalized for body weight the difference is reduced to around 20–30%. The bioavailability is reduced when given with food, depending on fat content, with low-fat food having greater effect on the absorption than high-fat food [FDA, 2012].
Absorption and elimination kinetics and metabolite profiles of mirabegron were investigated in four healthy men by Takusagawa and colleagues [Takusagawa et al. 2012]. Mirabegron, administered as an oral solution (160 mg of radiolabelled [14C]mirabegron, 1.85 MBq) was rapidly absorbed. A double-peak phenomenon in the plasma mirabegron concentration–time profile was seen with distinct peaks at approximately 0.5–1 h and 2–4 h after administration, likely caused by two separated ‘absorption windows’ along the small intestine. Unchanged mirabegron was the most abundant fraction of radioactivity, accounting for approximately 22% of plasma radioactivity. The mean cumulative excretion of radioactivity by 408 h after dosing was 55.0% in urine, 34.2% in faeces and 89.2% in total, showing that at least a 55.0% dose of mirabegron was absorbed from the gastrointestinal tract. Mirabegron was recovered in urine and faeces mainly in the unchanged form. Thus, unchanged mirabegron excreted in urine accounted for 45% of the excreted radioactivity, while 10 metabolites accounted for most of the remainder of the urinary radioactivity. The unchanged form accounted for almost all radioactivity in the faeces. Eight urinary metabolites were also detected in the plasma. There appear to be several metabolic pathways involved in the metabolism of mirabegron, including dealkylation, oxidation, glucuronidation, and amide hydrolysis. Also, these pathways involve multiple enzymes and isoenzymes such as butyrylcholinesterase, uridine diphospho-glucuronosyltransferase, cytochrome P450 (CYP3A4) (the primary responsible isoenzyme), CYP2D6, and alcohol dehydrogenase. As a result, the possibility that coadministered drugs with the potential to inhibit/induce one specific enzyme or transporter may affect the pharmacokinetics of mirabegron is low. Polymorphism of CYP2D6 has been shown to cause pharmacokinetic differences between poor and extensive metabolizers, although they are not considered clinically relevant [van Gelderen et al. 2009]. Recently, methods for the determination of mirabegron and its metabolites in human plasma have been developed and validated [Teijlingen et al. 2012]. Systemic exposure was higher in patients with severe renal impairment or moderate hepatic impairment than in healthy controls; the dose proposed by the manufacturer is 25 mg in these patients.
Mirabegron is formulated as oral controlled absorption system (OCAS) tablets. OCAS is a modified release system (also referred to as extended release or prolonged release) that allows the release of drug from the tablets for an extended period. The drug product is available in two strengths of 50 mg (recommended, to be marketed orally once daily dose, with or without food) and 25 mg (for patients with severe renal or moderate hepatic impairment).
Clinical efficacy
Mirabegron has been studied extensively in more than 10,000 individuals over the last 10 years. The safety and efficacy in patients with OAB were evaluated in six global, 12-week, phase IIb and phase III studies; however, most of these studies are still unpublished as full-paper articles in peer-reviewed journals.
Chapple and colleagues reported the results of a proof-of-concept, randomized, double-blind, parallel group, phase IIa dose-ranging trial (BLOSSOM trial) with mirabegron in patients with OAB [Chapple et al. 2008b]. Tolterodine and placebo were used as controls. Thirty-one sites of six European countries enrolled 260 patients who, after a 2-week placebo run-in period, were randomly assigned to four study groups: placebo (n = 66), mirabegron 100 mg twice daily (n = 65), mirabegron 150 mg twice daily (n = 65), and tolterodine 4 mg four times daily (n = 64) for a 4-week period. Analysis of the primary efficacy measure showed a statistically significant reduction from baseline in mean micturition frequency with 100 and 150 mg of mirabegron (−17% and −18% respectively) compared with placebo (−9%) and tolterodine (−11%). Mirabegron was also significantly superior than placebo with regard to mean volume voided per micturition, mean number of incontinence episodes, nocturia episodes, urgency incontinence episodes, and urgency episodes per 24 h.
Based on these results, Chapple and colleagues conducted a European dose-ranging phase IIb trial (DRAGON trial) enrolling 919 patients randomized in five study groups: placebo, once-daily mirabegron 25 mg, 50 mg, 100 mg, and 200 mg for a 12-week period [Chapple et al. 2010]. Analysis of the primary efficacy measure showed statistically significant dose-dependent reduction in mean micturition frequency with 50, 100, and 200 mg of mirabegron compared with placebo (−2.1, −2.1, and −2.2 respectively; p < 0.05). Furthermore, compared with placebo, mirabegron significantly increased mean volume voided per micturition (dose-dependent effect), decreased the mean number of incontinence episodes, urgency incontinence episodes, and urgency episodes per 24 h.
Three pivotal large-scale phase III studies conducted by Astellas have been considered for the efficacy analysis in the report of the Division of Reproductive and Urologic Products of FDA [FDA, 2012; Khullar et al. 2011; Nitti et al. 2011]. Efficacy analysis of these studies and of pooled results were based on two coprimary efficacy endpoints: the change from baseline to endpoint in the mean number of incontinence episodes per 24 h; and the mean change from baseline to endpoint in the mean number of micturitions per 24 h.
Khullar et al. carried out a European–Australian multicentre, randomized, double-blind, parallel-group, placebo and active controlled phase III trial (SCORPIO trial) [Khullar et al. 2011]. The study enrolled 1978 patients with OAB who, after a 2-week placebo run-in period, were randomized to receive one of the following once daily for 12 weeks: placebo (n = 494), mirabegron 50 mg (n = 493), mirabegron 100 mg (n = 496), or tolterodine slow release (SR) 4 mg (n = 495). Analysis of the coprimary efficacy measures showed a statistically significant decrease from baseline in mean number of micturitions with 50 mg and 100 mg of mirabegron (−1.93, −1.77, respectively) and mean number of incontinence episodes (−1.57, −1.46, respectively ) per 24 h compared with placebo (Figure 1). Although improvements in both coprimary endpoints were also observed with tolterodine SR, these did not reach statistical significance.
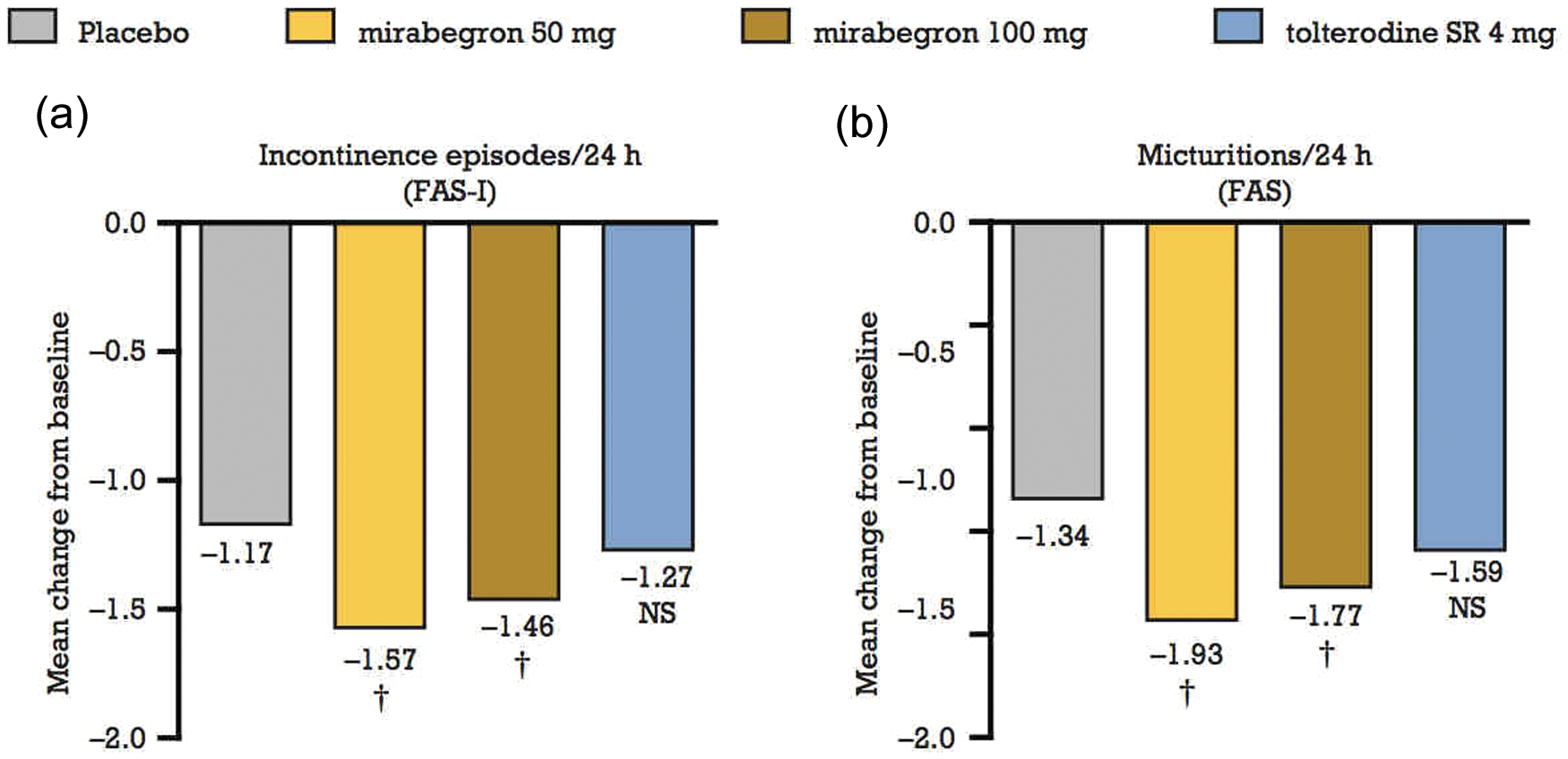
Coprimary efficacy variables in the SCORPIO trial. Mean change from baseline to final visit in the number of (a) incontinence episodes per 24 h and (b) micturitions per 24 h. Data are least squares mean adjusted for baseline, sex and geographical region. †Statistically significantly superior versus placebo at the 0.05 level with multiplicity adjustment. FAS, full analysis set; FAS-I, all full analysis set patients who had at least one incontinence episode at baseline; NS, not statistically significant versus placebo. Reprinted from [Khullar et al. 2012], with permission from Elsevier.
Nitti et al. confirmed the aforementioned results in a multicentre, randomized, double-blind, parallel-group, placebo-controlled phase III trial (the ARIES trial) conducted in North America [Nitti et al. 2011]. This study enrolled 1328 patients with OAB and randomly assigned to them three treatment arms: placebo, mirabegron 50 mg and mirabegron 100 mg for 12 weeks. Statistically significant reductions from baseline were observed with both doses of mirabegron in the number of incontinence episodes (−1.13, −1.47, and −1.63 for placebo, mirabegron 50 mg, and 100 mg, respectively; p < 0.05) and in the number of micturitions per 24 h (−1.05, − 1.66, and −1.75 for placebo, mirabegron 50 mg, and 100 mg, respectively; p < 0.05) .
A post hoc analysis of the SORPIO trial [Khullar et al. 2011] showed that both mirabegron doses (50 mg and 100 mg once daily) were effective in improving coprimary efficacy endpoints versus placebo in antimuscarinic treatment-naïve patients and in patients whose condition failed to respond to previous antimuscarinic therapy, regardless of the reason for discontinuation [Khullar et al. 2012].
The third pivotal phase III study is a European–North American, randomized, double-blind, placebo-controlled trial (the CAPRICORN trial) comparing mirabegron 25 mg and 50 mg with placebo. The results of this trial, submitted to FDA by Astellas, are still unpublished [FDA, 2012].
Tables 1 and 2 display the results of a primary pooled efficacy analysis performed by FDA and including data from the three aforementioned pivotal phase III studies [FDA, 2012]. The data relate to the 50 mg mirabegron OCAS once daily regimen, the proposed dosing regimen under evaluation for FDA marketing approval.
Pooled phase III pivotal studies: mean number of micturitions per 24 h.

p Values are nominal from paired comparisons versus placebo within stratified rank analysis of covariance model. Reprinted from [FDA, 2012].
Statistically significantly superior compared with placebo at the 0.05 level with multiplicity adjustment.
CI, confidence interval; LSM, least square mean; SE, standard error.
Pooled phase III pivotal studies: mean number of incontinence episodes per 24 h.
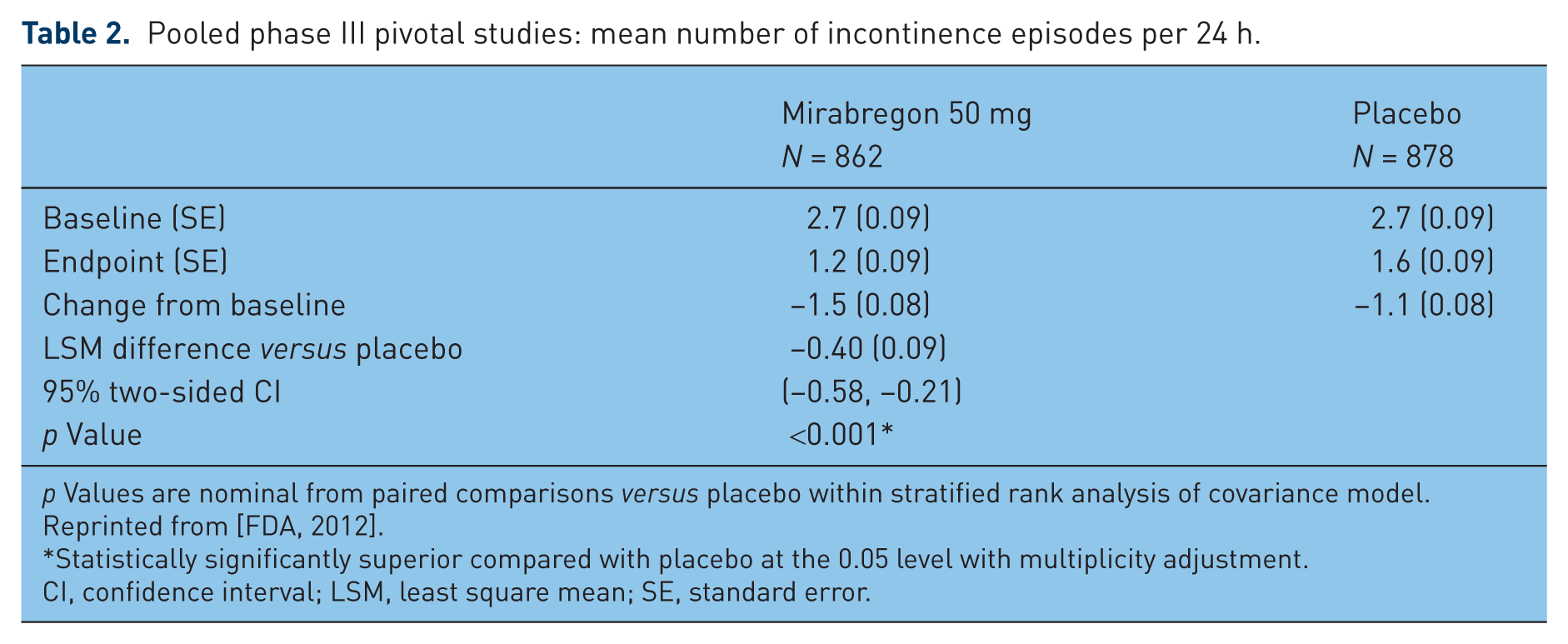
p Values are nominal from paired comparisons versus placebo within stratified rank analysis of covariance model. Reprinted from [FDA, 2012].
Statistically significantly superior compared with placebo at the 0.05 level with multiplicity adjustment.
CI, confidence interval; LSM, least square mean; SE, standard error.
Further data coming from subpopulation analyses showed that mirabegron 25 mg and 50 mg was effective in men and women, although a larger reduction versus placebo in mean number of incontinence episodes was observed in women compared with men. In addition, mirabegron 25 mg and 50 mg resulted in reduction in mean number of incontinence episodes per 24 h that was significantly lower in the age group under 65 years compared with those aged 65 years and over. Mirabegron 50 mg and 100 mg did not appear effective in decreasing the mean number of incontinence episodes in men with benign prostatic hyperplasia [FDA, 2012].
Another multinational randomized, double-blind, parallel group, active-controlled, phase III trial has been conducted in North America, Europe and other countries (the TAURUS trial) and assessed the long-term (12 months) safety and efficacy of mirabegron 50 mg and 100 mg and tolterodine SR 4 mg [Chapple et al. 2012]. The study was not designed to demonstrate a statistically significant difference in efficacy between treatment groups. After a single-blind placebo run-in period of 2 weeks, 2444 patients with OAB were randomized to the four study groups for 12 months. The authors reported that for both doses improvements in OAB symptoms were observed by month 1, with continued improvement until at least month 3 and maintenance of the effect through month 12, as measured by the change from baseline for mean number of micturitions per 24 h, mean number of incontinence episodes per 24 h, and improvements in mean volume voided/micturition (Figure 2) [Chapple et al. 2012].

Coprimary efficacy variables in the long-term (12 months) TAURUS trial. Mean number of (a) incontinence episodes per 24 h and (b) micturitions per 24 h. Adjusted mean changes from baseline at each visit, based on the repeated measures model. Reprinted from [Chapple et al. 2012], with permission from Elsevier.
Results from further studies are awaited, such as those from a phase II trial evaluating mirabegron and solifenacin alone and in combination for treating OAB (ClinicalTrials.gov identifier: NCT01340027).
Safety and tolerability
Chapple and colleagues reported in the BLOSSOM trial an incidence of treatment-emergent adverse effects (TEAEs) of 39.2% compared with 36.4% for the placebo group and 48.4% for the tolterodine group [Chapple et al. 2008b]. AEs were mild or moderate in intensity, and headache and gastrointestinal disorders were the most common (6.9%, 13.8%, respectively) in the mirabegron group, but their incidence was lower compared with the tolterodine group (9.4%, 23.4% respectively). Treatment-related dizziness and palpitations were more common with mirabegron compared with placebo and tolterodine. Of note, no episodes of acute urinary retention were reported. Discontinuation rates caused by adverse effects were 4.6% and 7.7% with mirabegron 100 mg and 150 mg, respectively, 1.5% with placebo and 3.1% with tolterodine.
In the DRAGON trial [Chapple et al. 2010] the incidence of one or more TEAEs in the mirabegron groups (25, 50,100, and 200 mg) was between 43.8% and 47.9% compared with 43.2% in the placebo group. The most commonly reported class of TEAEs were gastrointestinal disorders, including constipation, dry mouth, dyspepsia and nausea (7.2–8.3% in the mirabegron groups versus 5.3% in the placebo group). Of note, the incidence of dry mouth was lower than has been seen with antimuscarinics [Chapple et al. 2008a] and, again, no episodes of acute urinary retention were reported. No differences between treatment groups were observed in electrocardiogram (ECG) parameters but a statistically significant increase from baseline in mean pulse rate versus placebo was observed with 100 mg and 200 mg mirabegron [1.6 and 4.1 beats per minute (bpm) respectively in the morning; 2.7 and 4.7 bpm in the afternoon]; this change in pulse rate was not associated with an increase in cardiovascular AEs. Discontinuation rates were between 2.4% and 5.3% in the mirabegron groups and 3.0% in the placebo group.
Khullar and colleagues reported in their large-scale European–Australian trial an incidence of TEAEs similar across the placebo, mirabegron 50 mg, mirabegron 100 mg, and tolterodine SR groups (43.3, 42.8, 40.1, and 46.7%, respectively) [Khullar et al. 2011]. The most common TEAEs were hypertension (7.7, 5.9, 5.4, and 8.1%), dry mouth (2.6, 2.8, 2.8, and 10.1%), headache (2.8, 3.7, 1.8, and 3.6%), and nasopharyngitis (1.6, 2.8, 2.8, and 2.8%). Although the incidence of dry mouth was similar in the placebo and mirabegron groups, and markedly lower than observed in patients receiving tolterodine SR, the incidence of constipation was similar across all treatment groups.
In the North American phase III trial a similar incidence of TEAEs was observed for placebo, mirabegron 50 mg and 100 mg groups (50.1%, 51.6% and 46.9% respectively) [Nitti et al. 2011]. The incidence of hypertension was 6.6%, 6.1%, and 4.9%, and headache 2.0%, 3.2%, and 3.0% in the placebo, mirabegron 50 mg, and 100 mg groups respectively. Discontinuation rates due to adverse events were 3.8%, 4.1%, and 4.4% in the placebo, mirabegron 50 mg, and 100 mg groups. Changes in laboratory assessments, vital signs, physical examination, electrocardiograms, and post-void residual volume were small and consistent across treatment groups.
Long-term (12 months) safety and tolerability were the primary outcome of the TAURUS trial [Chapple et al. 2012], showing that the incidence and severity of treatment-emergent and serious AEs were similar across the mirabegron 50 mg (59.7%), mirabegron 100 mg (61.3%), and tolterodine SR 4 mg (62.6%) groups (Table 3). The most frequent TEAEs were hypertension, dry mouth, constipation, and headache, which occurred at a similar incidence across all treatment groups, while the incidence of dry mouth was more than threefold lower compared with the tolterodine SR 4 mg group [Chapple et al. 2012]. Most TEAEs were mild or moderate in severity across all treatment groups. Five deaths were reported; three in the mirabegron 50 mg group and two in the tolterodine SR 4 mg group, but all were considered unrelated to the study drug. No consistent trends in ECG changes were identified and there was no prolongation of the QTc interval in mirabegron-treated patients compared with tolterodine-treated patients. Incidentally, a thorough QT study showed no QT prolongation at the 50 mg dose [FDA, 2012].
Most frequent (≥2% in any treatment group) treatment-emergent adverse events (the TAURUS trial).

Reprinted from [Chapple et al. 2012], with permission from Elsevier.
AE, adverse event; SR, sustained release.
Although mirabegron appeared to be well tolerated in patients with OAB and exhibited a low incidence of TEAEs across the reported trials, some safety issues still need to be clarified. A mirabegron-related, dose-responsive elevation in blood pressure of approximately 3-4 mmHg was observed at T max in phase I studies and raised concerns, although the mirabegron-related elevation in systolic and diastolic blood pressures was only approximately 1 mmHg in phase III studies, generally comparable across all treatment groups. A similar discrepancy exists between findings in phase I studies and phase III studies relating to effects on heart rate. The reasons for these discrepancies and the extent and clinical relevance of these cardiovascular effects are not completely clear, and according to FDA [FDA, 2012], represent a potential cardiovascular safety signal. In relation to its mechanism of action, mirabegron also increases the incidence of mild-severity urinary tract infections and there have been postmarketing reports of urinary retention. Furthermore, in the phase III trials, an increased incidence of a variety of neoplasms occurred in the mirabegron 100 mg group compared with the placebo group, but not in the to-be-marketed mirabegron 50 mg group; whether these data represent a true mirabegron-related effect is still unknown [FDA, 2012]. Finally, evidence of CYP2D6 moderate inhibition in clinical trials highlighted a concern for pharmacokinetic interaction with other drugs that are CYP2D6 substrates; therefore mirabegron increases the systemic exposure of desipramine [Krauwinkel et al. 2010].
Conclusion
Overall, the results of phase III trials are promising for mirabegron becoming a novel effective and safe drug for patients with OAB. Treatment with the to-be-marketed dose of 50 mg achieved the primary efficacy objectives, although it resulted only in a reduction of 0.55 micturitions per 24 h and 0.40 incontinence episodes per 24 h compared with placebo. The drug is well tolerated overall, with a lower incidence of the well known side effects of antimuscarinics, such as dry mouth, constipation, and blurred vision.
However, some critical safety issues are still to be clarified. The increase in blood pressure and heart rate, mainly observed in phase I studies, pharmacokinetic interaction with other drugs, and the increased incidence of new malignant events observed with the 100 mg dose have raised concerns. Further clinical and long-term studies are especially needed for a thorough representation of the safety profile.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
