Abstract
Background:
The aim of this study was to compare oncologic outcomes of radical prostatectomy (RP) with brachytherapy (BT).
Methods:
A literature review was conducted according to the ‘Preferred reporting items for systematic reviews and meta-analyses’ (PRISMA) statement. We included studies reporting comparative oncologic outcomes of RP versus BT for localized prostate cancer (PCa). From each comparative study, we extracted the study design, the number and features of the included patients, and the oncologic outcomes expressed as all-cause mortality (ACM), PCa-specific mortality (PCSM) or, when the former were unavailable, as biochemical recurrence (BCR). All of the data retrieved from the selected studies were recorded in an electronic database. Cumulative analysis was conducted using the Review Manager version 5.3 software, designed for composing Cochrane Reviews (Cochrane Collaboration, Oxford, UK). Statistical heterogeneity was tested using the Chi-square test.
Results:
Our cumulative analysis did not show any significant difference in terms of BCR, ACM or PCSM rates between the RP and BT cohorts. Only three studies reported risk-stratified outcomes of intermediate- and high-risk patients, which are the most prone to treatment failure.
Conclusions:
our analysis suggested that RP and BT may have similar oncologic outcomes. However, the analysis included a limited number of studies, and most of them were retrospective, making it impossible to derive any definitive conclusion, especially for intermediate- and high-risk patients. In this scenario, appropriate urologic counseling remains of utmost importance.
Keywords
Introduction
Prostate cancer (PCa) is the most common malignancy in elderly males in Europe. It represents a major health concern, especially in developed countries with their greater proportion of elderly men in the general population. 1 However, during the last decade, the 5-year relative survival percentages for PCa steadily increased from 73.4% in 1999–2001 to 83.4% in 2005–2007. 2
As a result of prostate-specific antigen (PSA) screening, the incidence of PCa (especially localized PCa) keeps growing while the death rate from PCa decreases, posing a question on which is the appropriate management option for each PCa patient.
Radical prostatectomy (RP), which involves the removal of prostate and seminal vesicles with or without pelvic lymphadenectomy, represents the surgical treatment option for PCa.3,4 Other treatment options include external-beam radiation therapy (EBRT) combined with androgen deprivation therapy (ADT) 5 and brachytherapy (BT).
At present, RP represents the standard for long-term cure of localized PCa, with cancer-specific survival approaching 95% at 15 years after radical surgery. 6
Currently, RP is the only treatment for localized PCa to show a benefit for overall- and cancer-specific survival, compared with conservative management, as shown in one prospective randomized trial. 3 With regard to functional outcomes, a recent systematic review found that the mean continence rates at 12 months were 89–100% for patients treated with robotic-assisted RP (RARP) and 80–97% for patients treated with retropubic RP (RRP). 7 A similar study reported mean potency recovery rates at 12 months of 55–81% for patients treated with RARP and 26–63% for patients treated with RRP. 8
Transperineal low-dose rate (LDR) BT as a monotherapy for localized PCa, using iodine-125 or palladium-103, has potential advantages in that it is a single, relatively cost-effective and safe outpatient treatment. 9 High-dose rate (HDR) BT is recommended as a dose escalation technique combined with EBRT for patients with intermediate or high risk of failure with life expectancy longer than 5 years.
BT, combined with EBRT or alone, remains one of best tools for absolute dose escalation inside prostate. Due to low alpha/beta ratio for PCa these protocols are favorable, in which hypofractionation is used. 10
Dose prescription to 100% isodose should be 145 Gy for 125I and 125 Gy for 103Pd seeds. Previously used 198Au is no longer recommended for routine practice. 11
Recurrence-free survival after 5 and 10 years has been reported to range from 71% to 93% and from 65% to 85%, while there is no benefit in adding neoadjuvant or adjuvant ADT. 1 BT is particularly efficacious in low-risk patients: the 10-year disease-specific survival for patients with Gleason 6 disease treated with BT is 98%. 12 In high-risk patients treated with BT directed at the prostate and seminal vesicles, 11 the overall biochemical control at 10–15 years is 60–70%. Among patients with PSA failure, about one third have biopsy-proven local persistence while one quarter develops clinical evidence of distant metastasis at 10 years. 13
Some patients experience significant urinary complications following implantation, such as urinary retention (1.5–22%), post-implantation transurethral resection of prostate (TURP), which is required in up to 8.7% of cases, and incontinence (0–19%). 14
According to the European Association of Urology (EAU) Guidelines, management decisions should be made after all treatments have been discussed by a multidisciplinary team (including urologists, radiation oncologists, medical oncologists and radiologists), and after the balance of benefits and side effects of each therapy modality has been considered by the patients with regard to their own individual circumstances. 1 This could avoid biased advice to patients. 15
To our knowledge there have been no modern randomized trials demonstrating superiority of any definitive local treatment for clinically localized PCa. A randomized trial published in 1982 between radiotherapy (RT) and RP showed an advantage to RP, but was never widely accepted, because of randomization artifacts and worse than previously reported RT results. 16 The phase III SPIRIT (Surgery versus Internal Radiation in Treating Patients With Stage II PCa; ClinicalTrials.gov identifier: NCT00023686) randomized trial comparing seed implant to surgery closed due to lack of accrual. 11 Only one recent randomized controlled trial, the ProtecT study, examined the comparative effectiveness of different treatment modalities: the results of this trial are still not available. 17
A comparison of different therapies for localized PCa should include issues of cancer control, morbidity, quality of life, salvage of primary treatment failure, late effects, and cost. Of these, cancer control is the most important, because most patients may be willing to endure some morbidity or sacrifice some quality of life for a more efficacious therapy. 18
Purpose of the present study was to compare oncologic outcomes of patients managed with RP or BT.
Material and methods
A literature review was conducted according to the ‘Preferred reporting items for systematic reviews and meta-analyses’ (PRISMA) statement. 19 The literature search was performed in January 2015 using the Pubmed/Medline database. We searched Medline using the terms “prostatectomy versus brachytherapy” across the ‘Title’ and ‘Abstract’ fields of the records with the following limits: humans, sex (male), and language (English).
In the present meta-analysis, we included studies reporting comparative oncologic outcomes of RP versus BT. Studies published only as abstracts and reports from meetings were not included in the review. From each comparative study, we extracted the number of analyzed patients, the study design, the features of the included patients, and the oncologic outcomes expressed as all-cause mortality (ACM), PCa-specific mortality (PCSM) or, when the former were unavailable, as biochemical recurrence (BCR). All of the data retrieved from the selected studies were recorded in an electronic database.
Cumulative analysis was conducted using the Review Manager version 5.3 software, designed for composing Cochrane Reviews (Cochrane Collaboration, Oxford, UK). Statistical heterogeneity was tested using the Chi-square test. A p-value < 0.10 was used to indicate heterogeneity. Random effects models were used in case of heterogeneity.
Results
A total of 84 records were retrieved from the Pubmed/Medline database. The authors screened the papers dealing with oncologic outcomes of RP and BT, and 14 full-text papers were assessed for eligibility. Overall, three records consisting of reviews, meta-analysis or reports of overlapping cohorts were excluded. In addition, two records not providing data suitable for quantitative analysis were excluded. A total of nine studies reporting comparative oncologic outcomes of RP versus BT were included (Figure 1).20–28
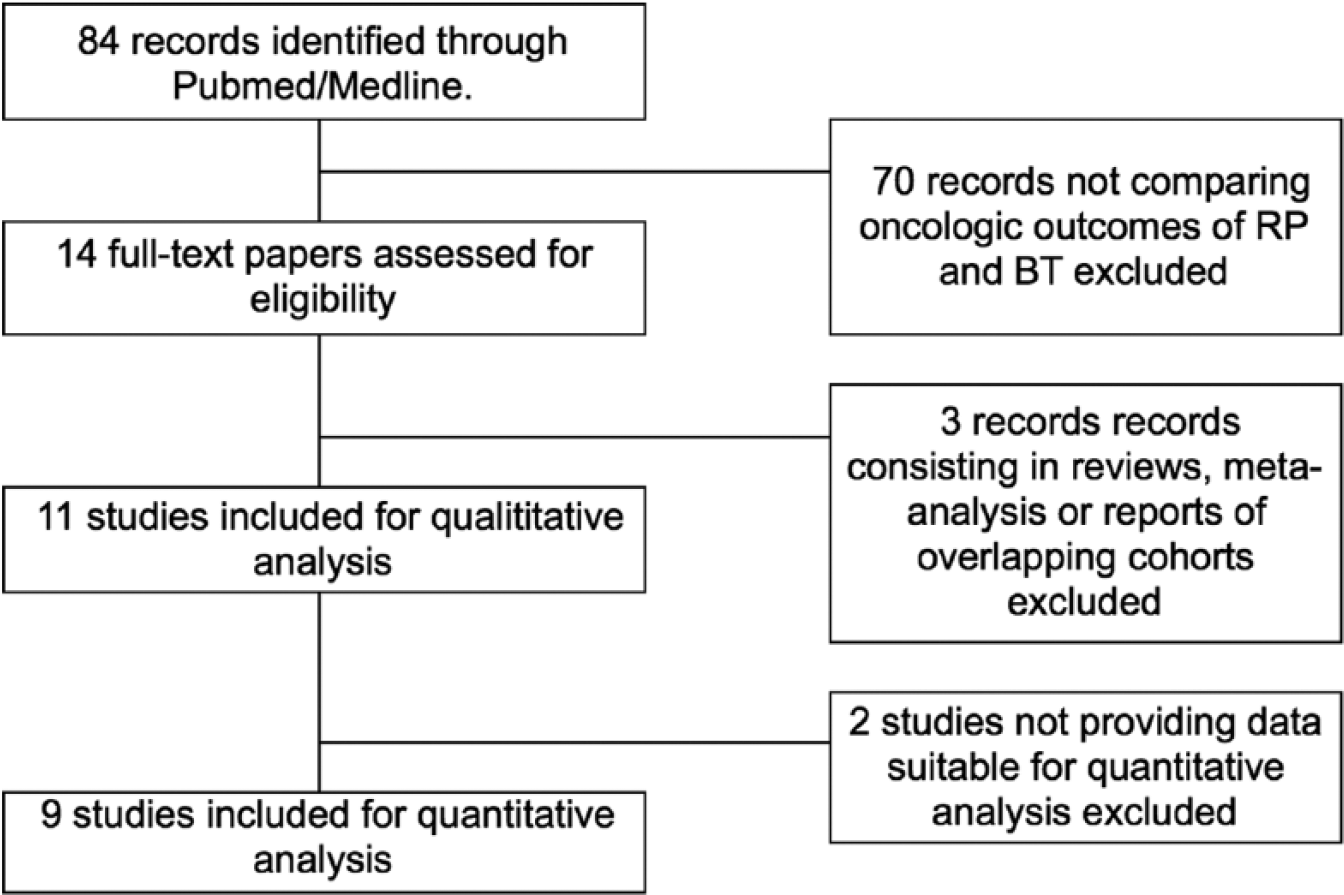
PRISMA flow diagram.
The design of all these studies was retrospective (level of evidence (LoE) 3 according to the Oxford Centre for Evidence-Based Medicine (OCEBM)), 29 except for the study carried out by Giberti and colleagues 27 who performed a randomized trial (LoE 2 according to OCEBM).
The features of the populations described by the included studies are reported in Table 1, while oncologic outcomes, expressed in terms of BCR, ACM and PCSM rates are reported in Tables 2 and 3.
Features of the populations described by the included studies.

BT, brachytherapy ; NA, not available; PSA, prostatic-specific antigen; RP, radical prostatectomy.
Biochemical recurrence (BCR) of PCa after RP and BT.

BCR, biochemical recurrence; BT, brachytherapy; NA, not available; PCa, prostate cancer; RP, radical prostatectomy.
All-cause mortality (ACM) and cancer-specific mortality (CSM) after RP and BT.
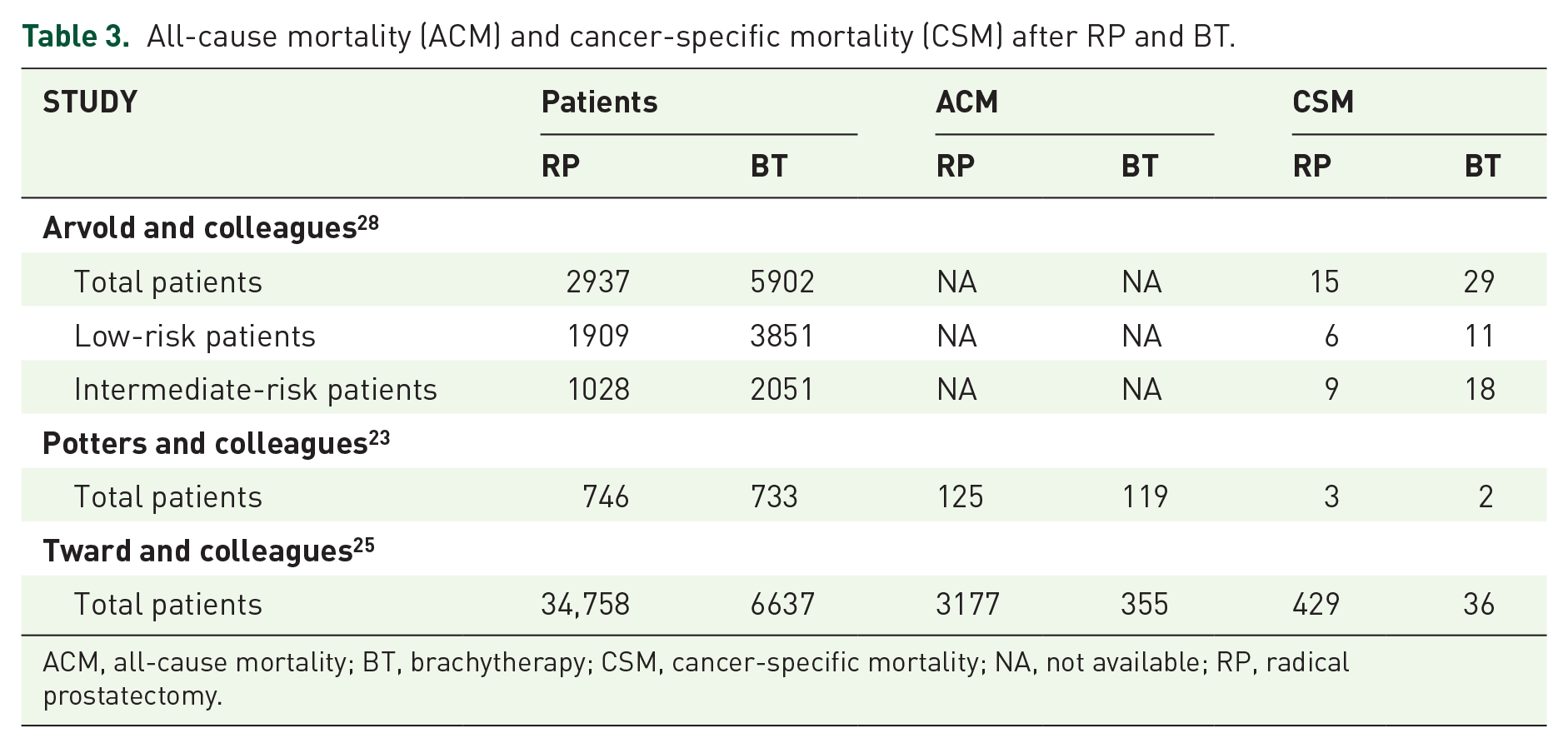
ACM, all-cause mortality; BT, brachytherapy; CSM, cancer-specific mortality; NA, not available; RP, radical prostatectomy.
A total of seven studies reported BCR rates in 4905 total patients, 2115 treated with RP and 2790 with BT. The overall cumulative analysis showed no significant differences between RP and BT (Figure 2).

BCR in RP and BT cohorts, not risk-stratified: no statistically significant differences were found. (RR: 0.77; 95% CI of RR: 0.47–1.26; p = 0.30).
Only two studies reported BCR rates in low-risk patients (1383 total patients), while three studies reported BCR rates in intermediate- and high-risk patients (913 and 307 total patients respectively). In all cases, no significant differences were found (Figures 3–5).
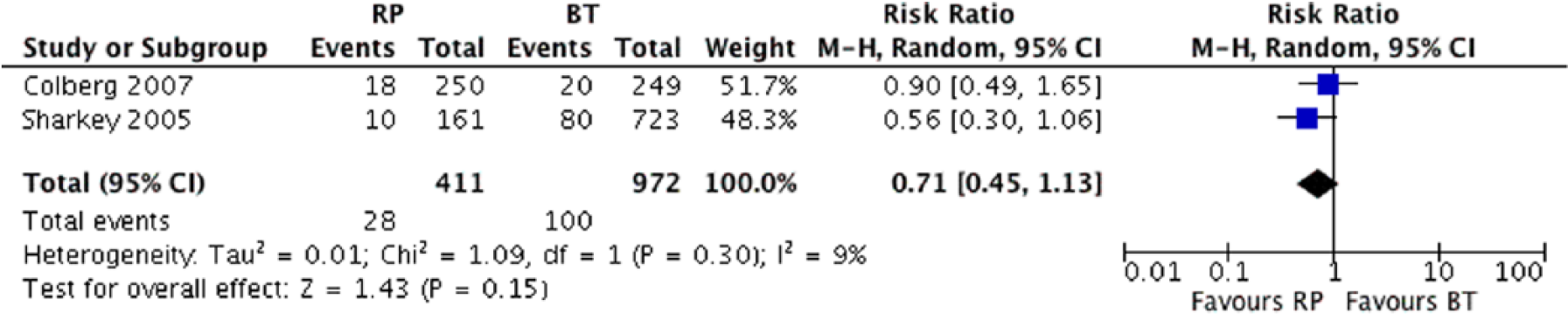
BCR in RP and BT cohorts reporting outcomes for low-risk patients: no statistically significant differences were found. (RR: 0.71; 95% CI of RR: 0.45–1.13; p = 0.15).

BCR in RP and BT cohorts reporting outcomes for intermediate-risk patients: no statistically significant differences were found. (RR: 1.02; 95% CI of RR: 0.25–4.11; p = 0.98).
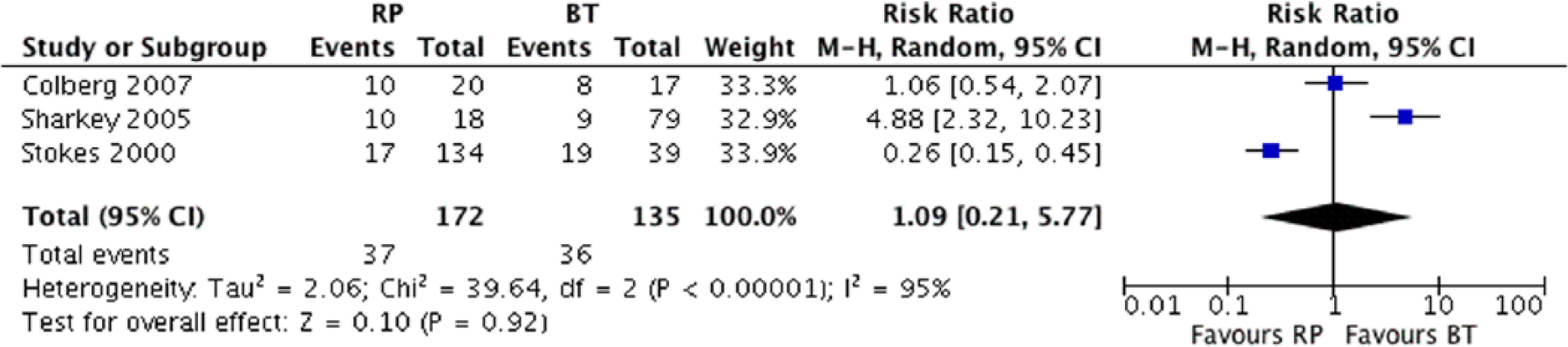
BCR in RP and BT cohorts reporting outcomes for high-risk patients: no statistically significant differences were found. (RR: 1.09; 95% CI of RR: 0.21–5.77; p = 0.92).
Only two studies reported comparative mortality outcomes of RP versus BT in terms of ACM and PCSM: the cumulative analysis did not show significant differences (Figures 6 and 7).

ACM in RP and BT cohorts, not risk-stratified: no statistically significant differences were found. (RR: 1.34; 95% CI of RR: 0.82–2.21; p = 0.25).
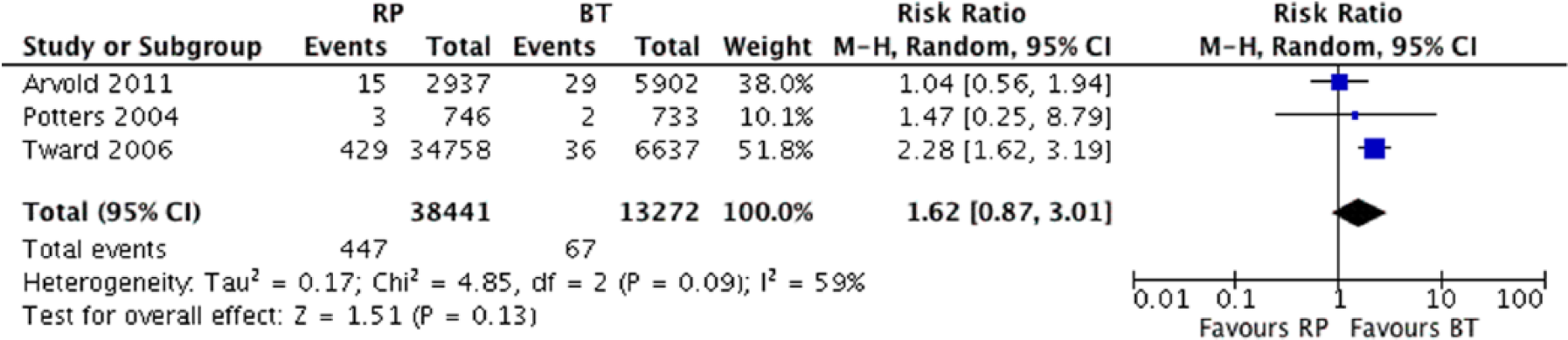
PCSM in RP and BT cohorts, not risk-stratified: no statistically significant differences were found. (RR: 1.34; 95% CI of RR: 0.82–2.21; p = 0.25).
Discussion
When the need for an active treatment for PCa is established by the clinician or chosen by the patient, uncertainty regarding the optimal treatment option for localized PCa produces wide and excessive local and regional variation in the use of various interventions. 30 This variation is not evidence-driven; indeed, given the dearth of high-quality comparative data, the Institute of Medicine recently included treatment for localized PCa among the 25 most important topics for comparative-effectiveness research. 31
Randomized trials in localized PCa face challenges related to high costs associated with long follow up and patient or clinician biases a priori in favor of any given approach or another. 32 Thus, meta-analyses may represent an acceptable way to provide evidence for decision-making.
Our cumulative analysis did not show any significant difference in terms of BCR, ACM or PCSM rates between the RP and BT cohorts. The study that provided the best evidence quality was the one conducted by Giberti and colleagues 27 In fact, these authors conducted a randomized trial comparing BCR rates of 100 patients undergoing RRP versus 100 patients undergoing BT. Mean age and baseline PSA were comparable in the two cohorts (65.2 versus 65.6 years and 7.8 versus 7.5 ng/ml). Mean follow up was 5 years and BCR rates resulted to be the same in both cohorts (8% versus 7%).
Interstingly, some studies, like the ones conducted by Arvold and colleagues, 28 Stokes 22 and Colberg and colleagues, 26 included intermediate- and high-risk patients, which is in clear contrast with the EAU Guidelines. Arvold and colleagues 28 and Colberg and colleagues 26 found no significant differences in terms of PCSM and BCR rates respectively, while Stokes reported improved BCR rates for RP patients. 22
Unfortunately, only these three studies reported risk-stratified outcomes of intermediate- and high-risk patients, which are the most prone to treatment failure.
Comparing oncologic outcomes of RP with BT is difficult for several reasons.
First, among the included studies, mean follow up was 50–84 months, which is a relatively short time when dealing with ACM and PCSM considering the long natural history of PCa.
Second, men with similar pretreatment characteristics treated with BT do not have the opportunity to assess for upgrading/up-staging and, thus, typically are not offered adjuvant therapy. On the other hand, adjuvant therapies administered after surgery (like ADT or EBRT) may decrease the risk of PCSM favoring RP. 28
Additionally, the timing of initiation of salvage therapy varies between men initially treated with RP versus BT. Patients undergoing RP experience an immediate PSA nadir and, thus, those with a detectable or increasing PSA postoperatively are able to begin salvaging EBRT or ADT early when PSA is only 0.2–0.4 ng/ml. This scenario is in contrast with men receiving BT, who experience a more protracted time to PSA nadir, followed by a PSA increase in those with treatment failure. Thus, for men treated with BT there is a longer time until PSA failure and subsequent salvage therapy. As a result, the timing of death from PCa may sometimes appear earlier in the BT group. 33 Anyway, this scenario did not present in our analysis.
Interpretation of biopsy findings after BT is also difficult. Although the appropriate time has not been established, biopsy is not recommended before 18–24 months after seed implantation. 21
Furthermore, BT implant quality has been shown to be associated with outcome. 28 Earlier reports of brachytherapy as treatment for clinically localized PCa were distinguished by high rates of local failure and treatment-related complications. Since then, several technical advances have been introduced attempting to overcome many of these initial limitations. These advances include the development of computerized algorithms to calculate three-dimensional dosimetry based on transrectal ultrasound or computed tomography-derived prostate volumes, and techniques for template-based, ultrasound-guided transperineal implantation with prostate immobilization. 20 Thus, more recent BT cohorts may be expected to achieve better oncologic outcomes.
The main limitation of our analysis is the design of the included study: all the included studies, except the one conducted by Giberti and colleagues, 27 were retrospective.
Second, we could not compare mean age and PSA in the RP and BT cohorts, since the included studies did not report these data in a suitable format (e.g. mean ± standard deviation).
Furthermore, no information was available about race, comorbidities and socioeconomic status of the described population. This is important since afroamerican men often present with more aggressive disease, while comorbidities and socioeconomic status are known to be associated with life expectancy. 34
Conclusion
Choosing the best management option for localized PCa remains a challenge for the urologist. Our cumulative analysis showed no differences in terms of BCR, ACM and PCSM rates between RP and BT.
There is still no evidence enough to prefer one kind of curative therapy on another, mainly for the lack of randomized trials, which are very difficult to perform in the field of PCa. In this scenario, appropriate counseling remains of utmost importance in order to put the patient in the right condition to decide among the different treatment options taking into account the specific drawbacks.
Footnotes
Acknowledgements
Gabriele Cozzi thanks Elena Collarin and Serena Detti for their help every day.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
