Abstract
There has been an increase in the number of urologic procedures performed robotically assisted; this is the case for radical prostatectomy. Currently, in the USA, 67% of prostatectomies are performed robotically assisted. With this increase in robotic urologic surgery it is clear that there are more surgeons in their learning curve, where most of the complications occur. Among the complications that can occur are vascular injuries. These can occur in the initial stages of surgery, such as in accessing the abdominal cavity, as well as in the intraoperative or postoperative setting. We present the most common vascular injuries in robot-assisted radical prostatectomy, as well as their management and prevention. We believe that it is of vital importance to be able to recognize these injuries so that they can be prevented.
Introduction
Given the enhanced dexterity and 3D vision afforded by the robotic platform and the relatively short learning curve, the number of radical prostatectomies being performed robotically is rapidly increasing. Since 2010, 67% of prostatectomies have been robotically assisted in the USA [Lowrance et al. 2012]. With this increase in robotic urologic surgery, it is clear that more surgeons are in the process of gaining experience with the technique. This change may result in an increase in the complication rate, particularly during a surgeon’s learning curve.
Vascular injuries are a potentially devastating complication of robotic-assisted radical prostatectomy (RARP). These injuries can occur in the initial stages of surgery, such as while gaining access to the abdominal cavity, as well as in the intraoperative or postoperative setting. Herein, we present a review of the most common vascular injuries during RARP, and their management and prevention. We believe that it is of vital importance to be able to recognize these injuries so that they can be prevented. The overall complication rate of RARP has been shown to be 4.3–12% [Patel et al. 2008; Jeong et al. 2010; Lebeau et al. 2011].
The cumulative analysis of comparative studies described by Tewari and colleagues revealed a mortality rate of 0.1% for the conventional open radical prostatectomy (ORP) approach, compared with 0.04% for laparoscopic radical prostatectomy (LRP) and robotic-assisted approaches. The researchers also found significant advantages (p < 0.0001) for RARP compared with other approaches in terms of the incidence of global perioperative complication rates (17.9%, 11.1%, and 7.8% for ORP, LRP, and RARP, respectively), and they described a lower incidence of vascular injury, obturator nerve damage, ureteral injury, rectal injury, deep vein thrombosis, anastomotic urine leak, hematoma, lymphocele, and wound infection with the robotic approach [Tewari et al. 2012].
The complication rate covers the time from gaining access to the abdominal cavity through the surgery and the postoperative period. The modified Clavien classification of surgical complications is used to grade complications as shown below [Lasser et al. 2010; Clavien et al. 1992; Dindo et al. 2004].

Access complications
Veress needle injuries
The prevalence of injuries caused by the Veress needle, or the ‘blind access’ described in the reviewed literature, is 0.23% [Azevedo et al. 2009]. A meta-analysis revealed a mean rate of 0.044% for vascular injury during laparoscopic access [Larobina and Nottle, 2005].
For umbilical access by the Veress technique, the needle should be placed at a 45° angle in the horizontal plane of the patient. In obese patients, the angle of the Veress needle should be changed from 45° to 90° in order to avoid vascular injury (Figures 1 and 2).
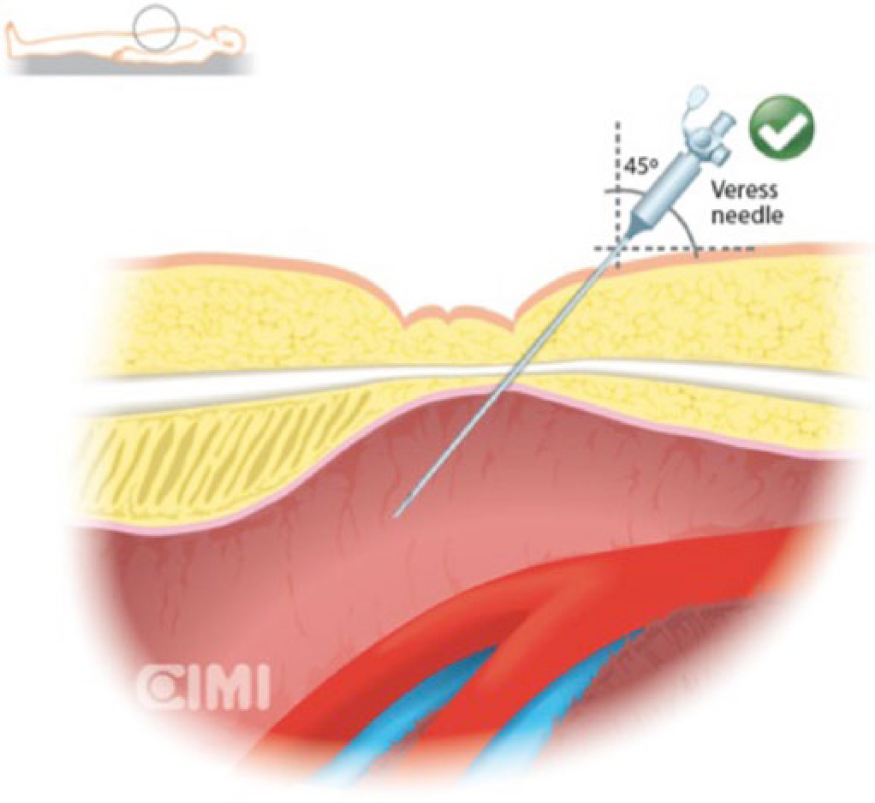
Umbilical access by the Veress technique at 45°.

Umbilical access by the Veress technique at 90°.
The umbilicus usually coincides with the sacral promontory, which in turn coincides with the bifurcation of the great vessels, so it is important to take great care to avoid injuring these vessels. In obese patients, the umbilicus will be located lower anatomically, which should be kept in mind while accessing the abdomen.
Most of the complications due to the Veress needle can be recognized by direct visualization, such as observing free blood in the abdominal cavity or, more commonly, a retroperitoneal hematoma. In the latter case, there will be a visible bruise that can be monitored for expansion.
Often, a hematoma may bleed if it is opened, so the surgeon must be prepared for this possibility. The assistance of a vascular surgeon may be required, and an open laparotomy may also be required.
If vascular injury is present, its management should be tailored to the specific situation. If there is a small, nonexpanding hematoma, it can be outlined with clips and monitored while continuing the surgery. At the end of the surgery, re-inspect the size of the hematoma. If the hematoma has expanded, open the hematoma and amply expose the bleeding site. Laparoscopic or robotic techniques can be used for the repair. Additional trocars may be inserted and a small gauze can be introduced to tamponade the bleeding.
If the injury cannot be easily repaired laparoscopically or robotically, apply compression and perform prompt laparotomy. Doing this is preferable to losing time trying to repair laparoscopically a vascular injury, which could potentially lead to undesired consequences.
A useful method to gain access with the Veress needle is to insert it while it is connected to the pneumoperitoneum machine. In this way, it is possible to observe how the pressure rises as the needle is slowly advanced through the abdominal wall, with constant pressure. If the pressure constantly changes, with wide fluctuations, try to go slightly deeper. If there is doubt about the position of the Veress needle, the needle can be aspirated. If there is blood, leave the needle in place, gain open access and then inspect the zone for damage. If the Veress needle is in the correct position, the pressure will fall below 5 cm H2O, coinciding with a click of the needle. At this time, you should not go deeper, and the pressure should begin to rise slowly. As the peritoneum is more firmly adherent underneath the umbilicus, obtaining access at this level could avoid insufflating the preperitoneal space [Ahmad et al. 2007].
Hasson open technique
Another way to access the abdominal cavity is by using the Hasson open technique, which was described in 1971 [Hasson, 1971]. This technique can be used in patients with prior surgeries in whom adhesions are expected. To avoid damage, perform the open access away from the scar, where adhesions are most likely to be found. A meta-analysis published in 2003 reported a 0.03% incidence of vascular lesions when using this technique [Merlin et al. 2003]. Caution should be taken in patients with prior surgeries or atypical anatomy. Preoperative imaging is helpful in identifying any variations.
Trocar injury
The average incidence of major vascular injuries from trocars and Veress needles is approximately 0.1%. A study from the US Food and Drug Administration carried out from 1993 to 1996 reported a total of 32 deaths out of 629 trocar injuries, with 81% of the deaths due to major vascular injuries, and 19 resulting from bowel injuries. Most of the cases of trocar injuries were nonfatal vascular injuries, followed by nonfatal visceral injuries such as bowel and abdominal wall hematomas [Bhoyrul et al. 2001]. Among the vascular injuries, the most common vessels injured were the aorta, the inferior vena cava, the iliac vessels [Castillo et al. 2008], and the epigastric vessels (lateral trocars) [Pereira Arias et al. 2010].
To avoid damage to intra-abdominal structures, ensure that the pneumoperitoneum is between 16 mmHg and 20 mmHg before inserting the first trocar. Make a skin incision long enough to allow the trocar to pass and then apply mild force. If the safe button on the trocar activates after inserting it, remember to reset it. For surgeons new to the technique, inserting the trocar while controlling it with both hands is recommended. One hand should apply pressure to progress the trocar, with the index finger over the body of the trocar, and the other hand should act as a top. This maneuver allows better control of depth, avoiding sudden progression of the tip of the trocar.
Injury to the aorta, vena cava, or iliac vessels is a life-threatening situation in which visualization of the damage and quick decision making regarding whether to convert to open surgery are needed. This decision should take into consideration the amount of bleeding and the surgeon’s laparoscopic skills. Surgeons experienced in laparoscopy may try to repair the injury using this method. In such cases, elevate the pneumoperitoneum to 20–25 mmHg, insert additional trocars if needed, apply pressure with a sponge or gauze, widely expose the site of injury, clamp above or below the lesion, and repair. Injury to the inferior epigastric vessels is the most common vascular complication, often recognized intraoperatively, and is usually caused during insertion of the pararectal trocars [Stolzenburg and Truss, 2003]. Bipolar coagulation and clipping are often effective in controlling any bleeding. If the bleeding is persistent, suturing through the abdominal wall with the aid of a straight needle, encaging the bleeding vessel, is very useful. The suture should be released 2 days after the initial operative procedure [Stolzenburg et al. 2006]. Remember to inspect all trocar sites after removal because bleeding may not be apparent until trocar removal and lowering the pneumoperitoneal pressure [Stolzenburg et al. 2006].
In laparoscopic surgery, it should be kept in mind that accidents can occur at any time, suddenly and without warning. A large-needle guided multifilament suture is easier to tie than a monofilament suture, making it easier to quickly control bleeding. This topic is controversial because many vascular surgeons state that vessels should not be repaired with multifilament sutures. However, during an emergency, it is easier to tie sutures without memory, and a large needle is much easier to visualize in the midst of bleeding. For these repairs, the ‘rescue stitch’ is a useful maneuver, requiring a CT-1 needle (15 cm long, 0-polyglactin) with a Hem-o-lok clip tied to the end, as reported by Aron and colleagues [Abreu et al. 2013] (Figure 3).

Rescue stitch.
If the bleeding cannot be controlled and open surgery is needed, the laparoscope can be directed toward the body wall to allow for a quick incision over the laparoscope [Gill et al. 1995]. Whenever possible, the blood flow should be controlled with upstream clamps. A separate sterile tray containing specific vascular repair tools and materials in case of injury should be immediately available.
Hemorrhage tray
Rescue stitch
Lapra-Ty or Hem-o-lok clip applier
Two needle holders
Klein bulldogs and Klein applicator
Satinsky clamps
Surgicel
Bolsters
Intraoperative injuries
The overall rate of intraoperative complications was found to be 0.4% [Trinh et al. 2012]. Vascular injuries can occur at different times during RARP. Most frequently, these situations occur during trocar insertion and lymphadenectomy [Zorn et al. 2009], but they can also occur while handling the dorsal vein complex (DVC) or the lateral pedicles, and during neurovascular bundle (NVB) dissection.
During lymphadenectomy, robotic surgeons still gaining experience should try to use delicate and controlled maneuvers and ensure that the instruments are kept within the field of vision. Experienced robotic surgeons can move their instruments without having to reposition the camera, but this skill is only acquired over time. Direct contact of energy-based instruments with vessels should be avoided. There have been reports of failure of the insulation of laparoscopic instruments, with the electrocautery electricity passing directly through the vessel and burning it [Lorenzo et al. 2011]. A direct cut to the iliac vessels during lymphadenectomy is another scenario that may occur. Compression is the first line of treatment. After that, increase the pneumoperitoneum to 20 mmHg and quickly change to needle holders. In cases of venous injury, this action will stop the bleeding, allowing for repair [Siqueira et al. 2002]. For arterial bleeding, utilize rolled gauze sponges to stop the bleeding by tamponade. A grasper or other blunt laparoscopic-robotic instrument can be placed at the site of the injury as a tamponade or to grasp the bleeding artery [Gill et al. 1995; Siqueira et al. 2002]. Be cautious with suction, as it decreases the effect of pneumoperitoneum compression. Add an additional port if needed. The rescue stitch is useful in these cases.
There are primarily two techniques used to control the DVC: one is suture ligature, and the other is stapling using an endoscopic gastrointestinal anastomosis stapler. Variability in the pelvic space and the anatomy of the pubic bones can make placing the stapler more difficult in certain cases [Nguyen et al. 2008]. A malfunction of the stapler can also occur: there may be incomplete stapling, or the stapler may stay locked. These are situations in which bleeding may occur. A third way to handle the DVC is by cutting it, increasing the pneumoperitoneum temporarily and then suturing the DVC. The most important aspect is to verify that there is no bleeding after decreasing the pneumoperitoneum. If hemorrhage is present while handling the DVC, increase the pressure of the gas to 20 mmHg. Meticulous bipolar coagulation should then be performed, and an additional suture is recommended. In case of persistent bleeding, complete the dissection of the ventral urethral wall and retract the Foley catheter with the balloon inflated to stop the bleeding by tamponade [Stolzenburg et al. 2007]. Finally, but no less important, lateral pedicle control using electrocautery, ultrasonic thermal energy, or clips has been described, and bioadhesives have been used to secure hemostasis after controlling the lateral pedicle. Gill and colleagues described a laparoscopic technique in which a bulldog clamp was used for pedicle control and cold cutting was performed followed by suturing with a 4-0 hemostatic suture [Gill et al. 2005]. Additionally, Beck and colleagues introduced a technique known as athermal tension adjustable suture ligation of the vascular pedicle, in which the vascular pedicle suture, placed for ligation, can be ‘recinched’ during and after transecting the vascular pedicle, as needed [Beck et al. 2012]. The most important step is to verify that there is no bleeding before performing the anastomosis. A possible source of postoperative hematoma is the small vessels adjacent to the NVBs, and intraoperative coagulation should be avoided. Management of bleeding can be performed either by selective suturing or by using matrix hemostatic sealants along the entire length of the NVBs [Stolzenburg et al. 2007].
If there are accessory pudendal arteries during dissection, the arterial branches may be transected, causing them to collapse and making it difficult to find the source of bleeding. Thus, when an accessory pudendal artery is present, follow the artery and inspect for bleeding sites.
Prevention
Careful inspection of the abdominal wall via the laparoscope before trocar insertion is useful. It is also useful to prepuncture and visualize the site of planned trocar insertion [Stolzenburg et al. 2006]. The first robotic instrument has to be inserted under direct vision because it has no memory and can go further than desired. Inserting the instrument under direct vision is also required because touching the clutch of the robotic arm causes it to lose its memory. It is important to avoid injury when inserting the instruments. Prevention of bleeding through careful dissection is the top priority in laparoscopic-robotic surgery. Avoid ever-narrowing, tunnel-like operating fields that limit good visualization and access to surrounding structures. In the case of vascular injury, be prepared to quickly change the robotic instruments to those that will enable vascular repair to be performed. To ensure that all the bleeding sites are well controlled, at the end of the procedure, reduce the gas pressure gradually (from 12 mmHg to 10 mmHg to 8 mmHg to 6 mmHg), and wait 1–2 min between each step to detect bleeding previously stopped by tamponade by the gas pressure [Stolzenburg et al. 2007]. Large abdominal or scrotal hematomas have been described due to small amounts of bleeding that were not seen during surgery (Figure 4).
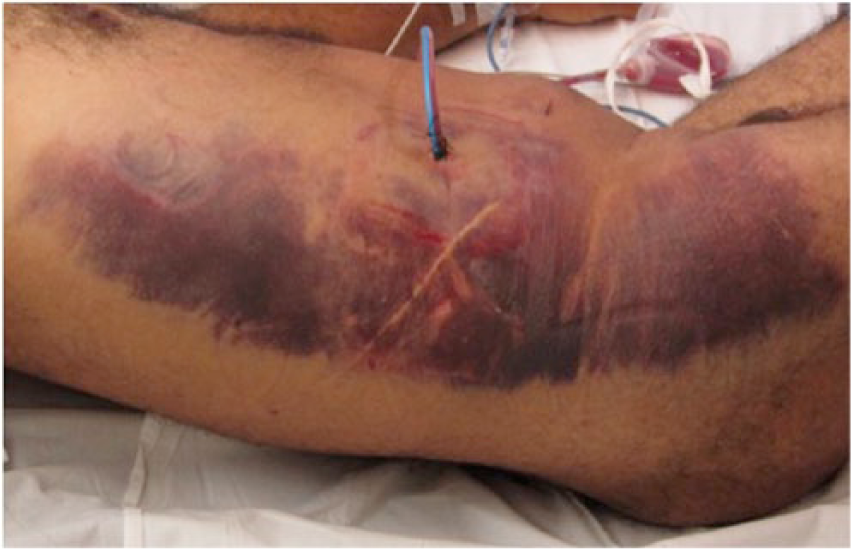
External epigastric vessel injury.
Care must be taken with the position and movement of the robotic arms during surgery, especially when one of the arms is placed outside the field of view, because the pressure of the instrument on vascular structures could cause delayed injuries due to intramural hematomas or thrombosis due to blood stasis.
Postoperative complications
The incidence of postoperative complications has been reported to be 1.9–6.8% [Carlsson et al. 2010; Hu et al. 2006], and these can occur without noticing. Thus, it is crucial to evaluate the patient in the first 2–3 h postoperatively. In the recovery room, assess how the patient is waking up and their heart rate, blood pressure, and level of consciousness. Also inspect for bruises around the trocar sites, scrotum, or perianal region. Assess the postoperative hemoglobin level, drainage speed, and tenderness of the abdomen. In the postoperative period, immediate or delayed complications may occur.
During the postoperative period, the incidence of significant bleeding in the dissection bed ranges from 0.3% to 3.2% [Dall’Oglio et al. 2003]. This bleeding may form pelvic hematomas. The formation of small hematomas requires only conservative management [Shekarriz et al. 2001]. In contrast, bulky hematomas can compress and displace the bladder, causing disruption of a vesicourethral anastomosis if they are not drained [Hedican and Walsh, 1994]. To avoid infection and severe perineal pain due to a large hematoma, drainage of the hematoma is an option [Dall’Oglio et al. 2003]. Surgical exploration for hematoma drainage with revision of the anastomosis has a better outcome than conservative management, in which sclerosis of the vesical neck and prolonged urinary incontinence are more likely to occur [Hedican and Walsh, 1994]. One of the advantages of extraperitoneal access is that minor postoperative bleeding can be stopped by a natural tamponade effect. If the hematoma expands, the same ports should be used with a 10 mm suction cannula to be able to aspirate all of the clots [Stolzenburg et al. 2007]. If there is minor bleeding from the urethral catheter, the balloon should be inflated with 20 ml, and minor traction should be applied to the catheter [Stolzenburg et al. 2007]. Urinary retention caused by clots can jeopardize the anastomosis; thus, forced diuresis is suggested if hematuria is present. Continuous irrigation of the bladder with a 3-way Foley catheter is also an option to prevent clot formation [Stolzenburg et al. 2007]. In rare cases in which gross hematuria continues and cannot be stopped with conservative measures, cystoscopy and fulguration are advised when bladder-neck vessel bleeding is the cause of the hematuria [Stolzenburg et al. 2007]. In patients with multiple prior surgeries, adhesions might be present. These involve important vessels, and after adhesiolysis, bleeding can occur in the postoperative period. Thus, it is important to inspect the area before exiting the abdominal cavity. Iatrogenic arteriovenous fistulae and pseudoaneurysms have been reported as rare complications of laparoscopic surgery by Ovroutski and colleagues [Ovroutski et al. 2001], serving as sequelae of penetrating injuries to the vessels. If the vascular damage is not repaired, late intermittent claudication may be present due to ischemia and venous thrombosis [Barbosa Barros et al. 2005].
Blood transfusion
In a recent meta-analysis, Novara and colleagues demonstrated statistically significantly lower transfusion rates for robotic surgery compared with laparoscopic and open approaches, with 16.5%, 4.8%, and 1.8% for ORP, LRP, and RARP, respectively [Novara et al. 2012].
The reported incidence of blood transfusions is 1.5%. The indication for transfusion is based on the clinical presentation [Ahme et al. 2012; Coelho et al. 2010]. Tachycardia, hypotension, heart disease in patients who need a hematocrit over 30%, and a hemoglobin level of 70 g/L or less are criteria for transfusion. In patients with hemoglobin between 70 g/L and 100 g/L, or with risk factors for complications due to poor oxygenation, consider transfusion to treat potential or active bleeding.
Thromboembolic complications
These events include deep venous thrombosis (DVT) and resultant pulmonary embolism. Sporadic cases have been reported, with a low incidence (below 1%). However, the development of DVT is usually due to predisposing factors, such as vascular injury, hypercoagulability, and venous stasis. Prophylaxis is advised, such as intermittent compressive devices or low-molecular-weight heparin [Keegan and Cookson, 2011].
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
