Abstract
Iatrogenic injury to the ureter is a potentially devastating complication of modern surgery. The ureters are most often injured in gynecologic, colorectal, and vascular pelvic surgery. There is also potential for considerable ureteral injury during endoscopic procedures for ureteric pathology such as tumor or lithiasis. While maneuvers such as perioperative stenting have been touted as a means to avoid ureteral injury, these techniques have not been adopted universally, and the available literature does not make a case for their routine use. Distal ureteral injuries are best managed with ureteroneocystostomy with or without a vesico-psoas hitch. Mid-ureteral and proximal ureteral injuries can potentially be managed with ureteroureterostomy. If the distal segment is unsuitable for anastomosis then a number of techniques are available for repair including a Boari tubularized bladder flap, transureteroureterostomy, or renal autotransplantation. In rare cases renal autotransplantation or ureteral substitution with gastrointestinal segments may be warranted to re-establish urinary tract continuity. Laparoscopic and minimally invasive techniques have been employed to remedy iatrogenic ureteral injuries.
Introduction
Iatrogenic injuries bring to mind the Latin phrase
Incidence of ureteral injuries
Gynecologic surgery
While ureteral injuries are an uncommon surgical complication, it is estimated that 52–82% of iatrogenic injuries occur during gynecologic surgery [Lee et al. 1988; Dowling et al. 1986; St Lezin and Stoller, 1991]. The rate of ureteral injury for vaginal hysterectomy is 0.2 injuries per 1000 cases, and 1.3 injuries per 1000 cases for total abdominal hysterectomy [Gilmour et al. 2006]. It has been suggested that this difference in rates between procedures is a result of the selection bias towards transabdominal hysterectomy for cases complicated by infection, previous surgery, and malignancy [Elliott and McAninch, 2006]. Other clinical risk factors for ureteral injury during hysterectomy include a large uterus, endometriosis, pelvic organ prolapse, and prior pelvic surgery [Vakili et al. 2005; Dandolu et al. 2003]. The most common sight of ureteral injury is near the ureterosacral ligaments [Grainger et al. 1990].
General and vascular surgery
Other surgical procedures that account for iatrogenic ureteral injuries include pelvic surgeries for colon and rectal pathology, and vascular surgery including aortoiliac and aortofemoral bypass. Colon and rectal procedures, such as low anterior resection (LAR) and abdominal perineal resection (APR), are responsible for 9% of all ureteral injuries [St Lezin and Stoller, 1991]. APR or LAR are complicated by an iatrogenic ureteral injury in 0.3–5% of cases [Coburn, 1996]. A recent retrospective review of accidental punctures or lacerations during colorectal surgery revealed nine ureteral injuries in a series of 2897 colorectal operations. Interestingly, history of prior surgery was not associated with an increased incidence of extraintestinal organ injury [Kin et al. 2013]. Vascular procedures may cause an intense inflammatory response around the ureters and result in either stricture or fistula as shown in Figure 1.

Retrograde pyelogram illustrating a ureteral arterial fistula.
Urologic procedures
With the introduction of endoscopic ureteric techniques, iatrogenic ureteric injury during routine urologic procedures for mucosal lesions and urolithiasis are relatively uncommon. Ureteral avulsion is probably the most catastrophic complication of ureteroscopy, and most often occurs during ureteroscopic stone retrieval. In three large retrospective studies it was found to occur less than 1% of the time [Weinberg et al. 1987; Stoller and Wolf, 1996; Grasso, 2001]. Mucosal abrasions and ureteral perforation are much more common during ureteroscopy and are generally managed with prolonged stenting, with surprisingly little consequence [de la Rosette et al. 2006]. Fortunately, with the advent of flexible ureteroscopy and durable access sheaths, ureteral injury is a rare event. A review of 150 applications of a 12/14 French access sheath revealed a single resulting ureteral stricture [Stern et al. 2007].
Prevention of ureteral injuries
Ureteral injury can cause catastrophic complications, so its prevention has garnered keen interest. Preoperative ureteral catheterization has long been proposed as a technique to identify the ureters and avoid iatrogenic injury. In a recent randomized trial comparing preoperative ureteral catheterization
Diagnosis
Ideally an iatrogenic ureteral injury is discovered in the intraoperative setting and dealt with immediately. Unfortunately, 50–70% of ureteral injuries are not diagnosed in the acute setting [Dowling et al. 1986; Ostrzenski et al. 2003]. Intraoperatively, if a ureteral injury is suspected, then a low threshold should be maintained for visual inspection of the ureters through meticulous dissection in an open operation or laparoscopically. The ureters may also be interrogated by cystoscopy and passage of a ureteral catheter in a retrograde fashion. When the patient is in a supine position and cystoscopy is difficult, a cystotomy may be performed, and ureteral catheters passed in a retrograde manner. The most sensitive diagnostic study for diagnosis of a ureteral injury is a retrograde pyelogram. This procedure also allows for possible treatment of a ureteral injury with retrograde indwelling stent placement. Often in an intraoperative setting, patient positioning or the operative suite does not allow for fluoroscopic examination or cystoscopy.
When iatrogenic ureteral injuries present later in the postoperative course the most common presenting signs and symptoms are abdominal pain with peritonitis, leukocytosis, and fever. Flank pain may or may not be present depending on the nature of the injury and whether the ureter is occluded, or has fistulized to the peritoneal cavity or retroperitoneal space [Grainger et al. 1990]. Occasionally a urinoma may form in the confined space of the retroperitoneum and present as a flank mass. Immediate cystoscopy and retrograde pyelogram should be performed for diagnosis of a suspected ureteral injury and possible stenting. If cystoscopy and retrograde pyelogram cannot be performed then the preferred imaging technique is computed tomography with intravenous pyelogram (CT-IVP). A CT-IVP allows for visualization of the relevant anatomy and continuity of the upper urinary tract as well as the location of extravasated urine that often develops near a ureteral injury. Delayed scans using CT-IVP often reveal an enhancement of fluid collections as contrast leaks from a ureteric injury [Gayer et al. 2002].
Surgical management
Distal ureteral injuries
Ureteroneocystostomy
The majority of operative injuries occur in the distal ureter. The distal ureter is most often injured during attempts to ligate branches of the internal iliac vessels, and the injured ureter may have an even more tenuous blood supply than usual after these maneuvers. As the blood supply of the distal ureteral segment may be disrupted, ureteroneocystostomy is an ideal option for distal ureteral repair. When preparing the ureter for implantation, it should be judiciously debrided back to viable tissue and spatulated. Reimplantation can be performed in a refluxing or nonrefluxing manner and the consensus is that a nonrefluxing reimplantation is preferred [Png and Chapple, 2000]. Despite the consensus towards a nonrefluxing reimplantation, neither technique has a detrimental effect on renal function or has an increased risk of stenosis [Stefanovic et al. 1991]. If a nonrefluxing reimplantation is planned, the ureter should be tunneled in the bladder wall as in the Politano–Leadbetter technique with the length of the tunnel three times the diameter of the ureter as shown in Figure 2 [Shokeir and El-Hammady, 1996]. The reimplantation site should be on the posterior or anterior dome of the bladder and not on the lateral aspects. Reimplantation of the ureter on the lateral aspects of the bladder is prone to kinking with bladder filling [Hensle et al. 1982]. The reimplanted ureter should be stented and a Foley catheter maintained in the bladder perioperatively together with a Jackson–Pratt (JP) drain near the anastomosis. The Foley catheter may be removed in approximately 1 week and the JP drain removed once it is determined that the repair is not leaking. The stent is removed in 6 weeks.
A tunneled ureteroneocystostomy for distal ureteral injury.
Vesico-psoas hitch
When a lengthy defect in the distal ureter is discovered, such that performing a simple ureteroneocystomy would result in tension on the anastomosis, often this distance can be bridged with a vesico-psoas hitch, first described by Zimmerman and colleagues [Zimmerman et al. 1960]. Mobilization of the bladder in the space of Retzius and ligation of the contralateral bladder pedicle aids in positioning the bladder near the psoas muscle. The bladder detrusor is then ‘hitched’ to the psoas muscle with longitudinal, nonabsorbable monofilament sutures to make up the length between the bladder and proximal ureteral stump as shown in Figure 3. Care is taken to avoid the genitofemoral nerve that lies on the anterior surface of the psoas muscle. The ureter is then reimplanted into the repositioned bladder in a similar manner to the previously mentioned ureteroneocystostomy. Some authors have advocated the routine use of a vesico-psoas hitch for ureteral reimplantation because it provides a long, fixed, straight submucosal tunnel that is unlikely to be affected by the degree of bladder filling [Middleton, 1980]. A large series of 181 patients for which a vesico-psoas hitch was employed for ureteroneocystostomy reported a 97% success rate at a mean follow up of 4.5 years. In this series the authors stated that the keys to success are a tension-free anastomosis, an antirefluxing, long submucosal tunnel, and a straight course of ureteral reimplantation to prevent kinking at the point of entrance to the bladder [Riedmiller et al. 1984]. A more contemporary series of 24 patients reported no difference in outcomes when the repair of an iatrogenic injury was treated with prompt repair (less than 6 weeks) using a psoas hitch or a delayed repair (more than 6 weeks) [Ahn and Loughlin, 2001].

Vesico-psoas hitch for distal ureteral reimplantation.
Upper and mid-ureteral injuries
Ureteroureterostomy
Often with small (2–3 cm) defects of the mid-ureter and upper ureter a primary ureteroureterostomy can be performed [Coburn, 1996]. The distal and proximal ureteral ends are debrided back to viable tissue and a standard running or interrupted anastomosis is performed as shown in Figure 4. The anastomosis should be stented and, if possible, covered with peritoneum or other tissue. One study of nine patients treated for ureteral injuries suffered during gynecologic and general surgical procedures reviewed outcomes when treated with ureteroureterostomy. They reported an average length of 3 cm of ureteral loss and reported no adverse events with a mean follow-up period of 33 months [Paick et al. 2006].
Ureteroureterostomy: the ends of the ureters are trimmed and a running anastomosis is performed.
Boari tubularized bladder flap
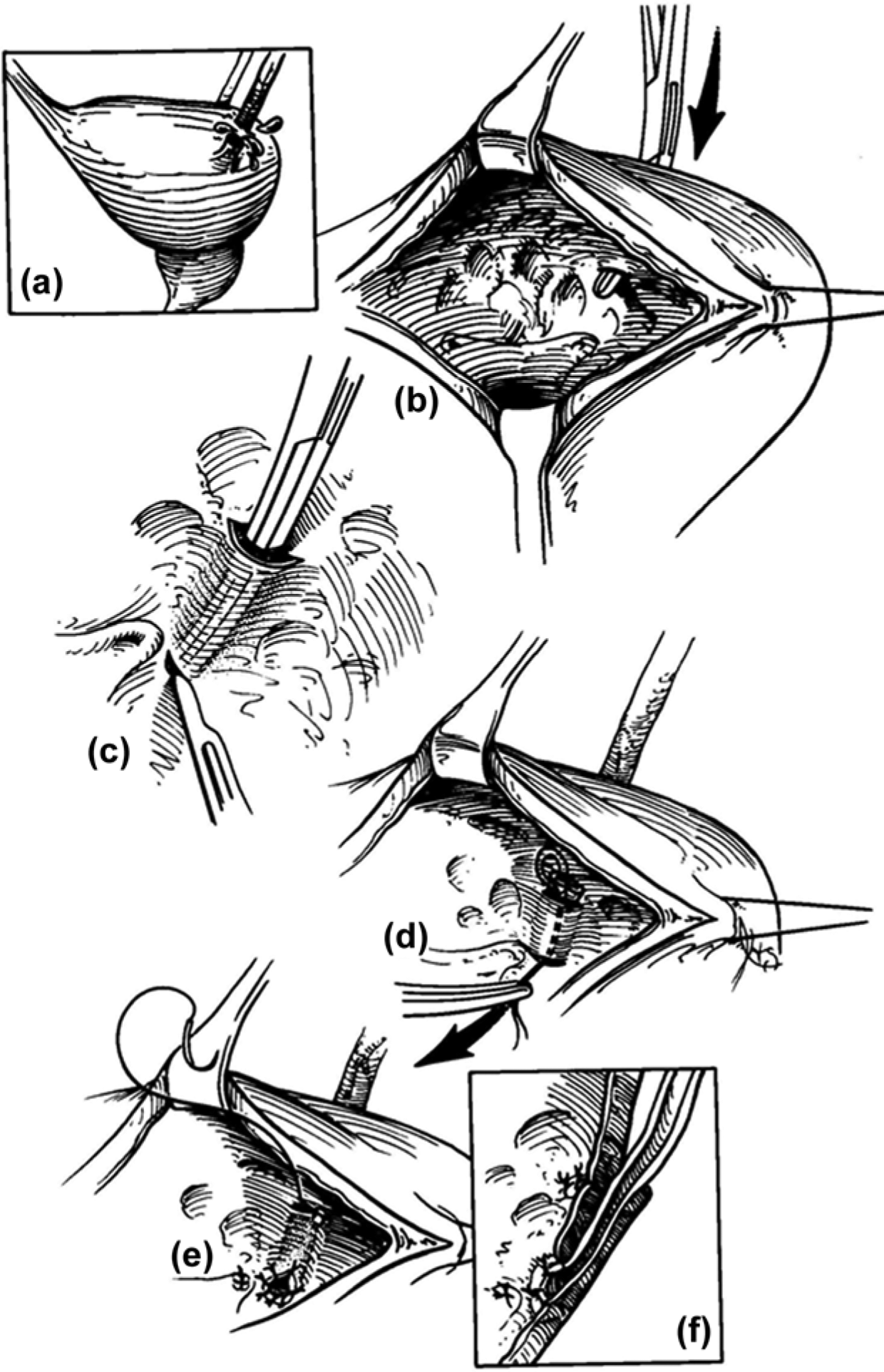
When a mid-ureteral or proximal ureteral injury occurs and the distal ureteral segment is not suitable for anastomosis, a Boari tubularized bladder flap is often a viable alternative. The bladder is opened on its anterior surface, and a full thickness bladder flap is swung cranially and tubularized for anastomosis to the proximal ureteral segment as shown in Figure 5. This is a technically challenging procedure and should be referred to a center with urologic reconstruction experience if necessary.
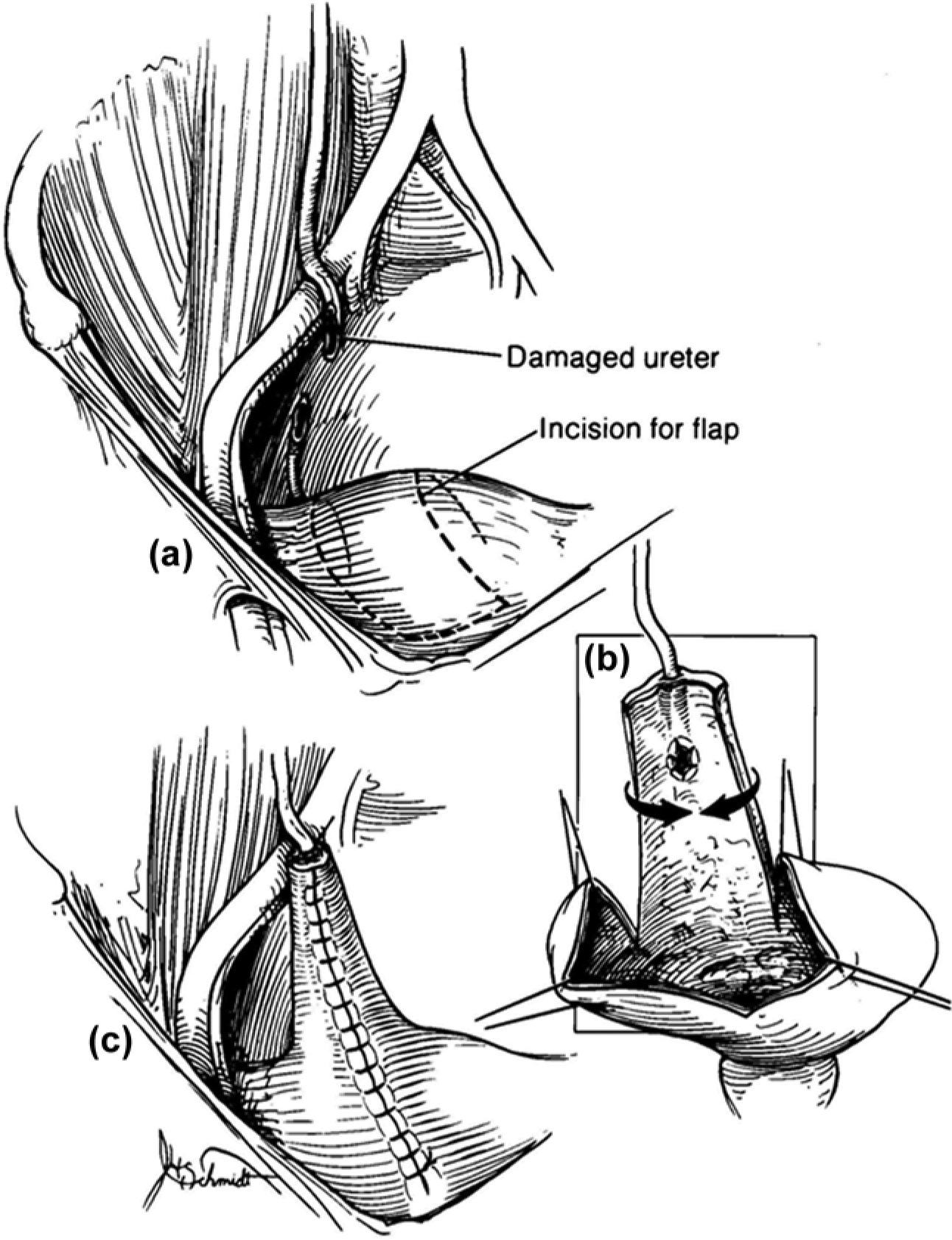
A Boari flap: a flap of bladder is fashioned into a tube and an anastomosis is created between it and the proximal ureter.
Transureteroureterostomy
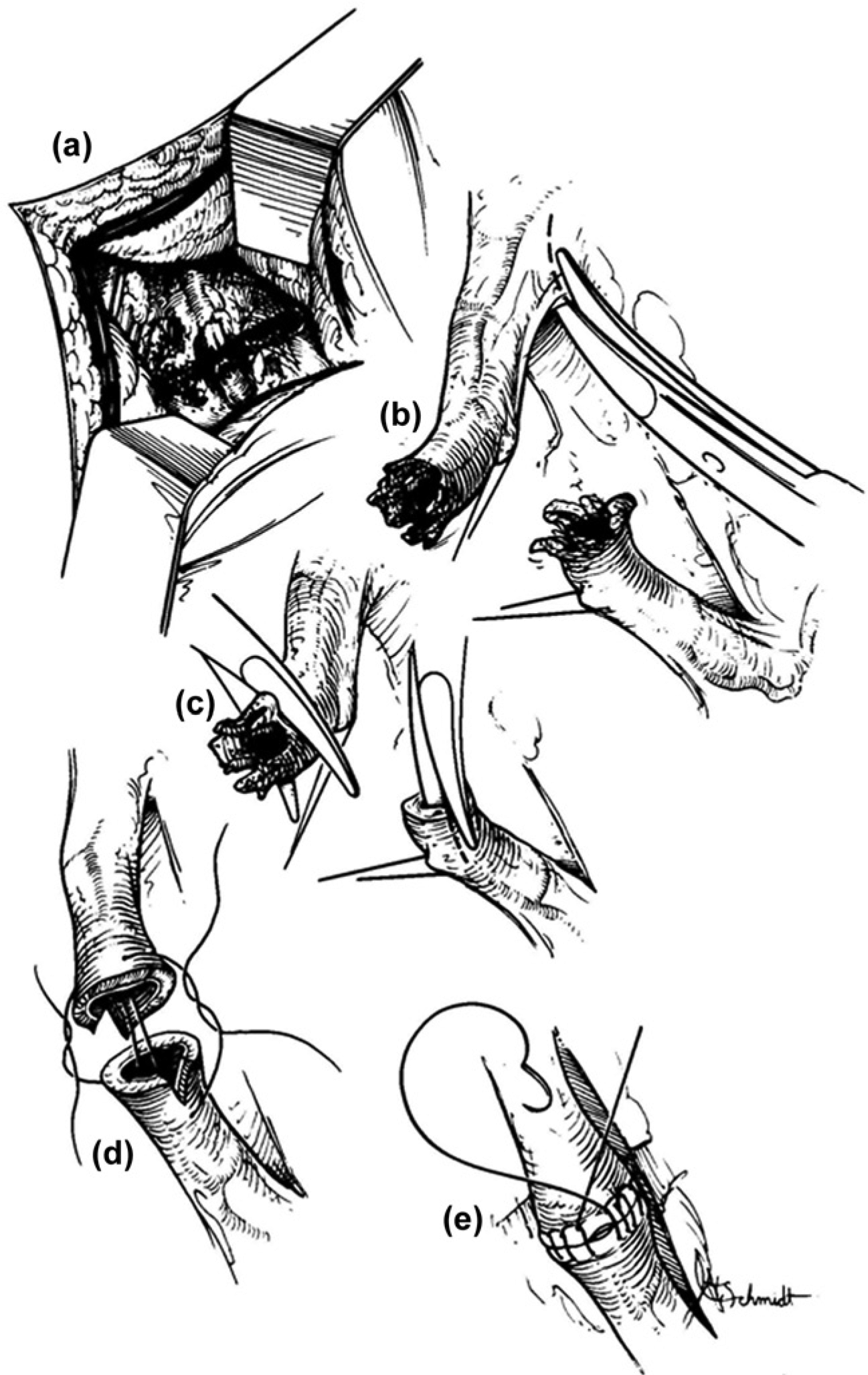
Most often the length of ureteral loss as a result of iatrogenic ureteral injury is quite short making transureteroureterostomy (TUU) unnecessary. In cases when primary reanastomosis to a distal segment is not feasible, or if a ureteroneocystostomy is precluded (i.e. rectal injury, major vascular injury, or extensive bladder injury), then TUU is an acceptable option. Contraindications to TUU include inadequate donor ureter length, which would create tension on the anastomosis, or disease of the recipient ureter such as urothelial carcinoma, urolithiasis, retroperitoneal fibrosis, or pelvic tumors with ureteral involvement [Iwaszko et al. 2010]. The donor ureter should be extensively mobilized, but care should be taken not to disrupt the longitudinal blood supply in the adventitial layer. Very little of the recipient ureter should be mobilized so that the area of anastomosis has a sufficient blood supply. The donor ureter should be passed under the sigmoid colon through the mesentery to the location of the recipient ureter. The donor ureter is then spatulated approximately 2 cm and the recipient ureter is opened to match the donor ureter opening [Barry, 2005]. An end-to-side anastomosis is performed as shown in Figure 6. The donor ureter should be stented with the stent passing through the distal portion of the recipient ureter down to the bladder. An attempt should be made to try to reperitonealize the ureteral anastomosis if possible.

Transureteroureterostomy. (a) The donor ureter is tunneled through the mesentery. (b) End-to-side anastomosis of the donor ureter and recipient ureter. (c) The final transureteroureterostomy configuration.
A recent retrospective review of 63 patients for which TUU was performed revealed that 24% of patients experienced a postoperative complication. The most common perioperative complication was urine leak from the anastomosis (9.5%). Postoperative imaging was available for 56 patients and revealed a patency rate of 96% and 4% of patients with a radiographic obstruction at the anastomosis. Eventually 10% of patients required subsequent intervention or revision for obstruction over a mean follow up of 6 years [Iwaszko et al. 2010]. The authors emphasized that because of the high risk of complication/revision, and concern for injuring the contralateral ureter, they seldom elect to perform TUU.
Renal autotransplantation
Renal autotransplantation is rarely if ever indicated at the time of intraoperative consultation for ureteral injury. If an iatrogenic ureteral injury precludes ureteral reconstruction and renal autotransplantation is being considered, then the ureter should be ligated and a percutaneous nephrostomy tube placed for renal drainage. Renal autotransplantation requires an extensive discussion with the patient about the potential complications and the options for elective repair. Meng and colleagues reported on series of seven patients who underwent laparoscopic nephrectomy and autotransplantation for severe ureteral injury. Of the renal units harvested, six were adequate for transplantation. At a follow up of 17 months imaging demonstrated that all renal autographs had normal function. The authors highlighted the need for coordination with transplantation personnel for graft preservation and preparation [Meng et al. 2003].
Ureteral substitution
The use of gastrointestinal segments, such as ileum for ureteral substitution, was first popularized in the 1950s by Goodwin and colleagues [Goodwin et al. 1959]. Other reported tissue sources for ureteral substitution include appendix, tubularized stomach, and colon [Moreira et al. 2004]. By far the most common source of tissue for ureteral replacement is the ileum as shown in Figure 7. Armatys and colleagues retrospectively reviewed the outcomes of ileal ureteral substitution in a large series of 91 patients. The majority of patients in the series received an ileal ureter as a treatment for ureteral stricture as a result of genitourinary surgery (32%) and radiation treatment (19%). Nonurologic surgical iatrogenic injury was the indication for an ileal ureter in 18% of patients. The authors reported long-term complications of anastomotic stricture in 3% of patients and fistula in 7% with a mean follow up of 36 months. It was also reported that 75% of patients had a serum creatinine level that either decreased or remained stable, and no patients complained of excessive urinary mucous [Armatys et al. 2009].

(a) The ileal ueter kidney anastomosis. (b) The ileal ureter bladder anastomosis.
Laparoscopic and minimally invasive treatment of ureteral injuries
Laparoscopy and minimally invasive surgical techniques have transformed modern surgical care. Not surprisingly the management of iatrogenic ureteral injuries also encompasses these techniques. Possibly the most minimally invasive treatment of an iatrogenic ureteral injury is cystoscopy and retrograde ureteral stent placement or antegrade placement of a ureteral stent. A recent series describes the management of iatrogenic ureteral injuries with minimally invasive techniques such as percutaneous nephrostomy tube placement, wire recanalization of the ureteral lumen, antegrade ureteral dilation, and ureteral stent placement. A total of 25 ureters were treated in this manner for iatrogenic injuries such as ureteral laceration and ureteral obstruction. Successful recanalization or dilation of the affected ureter was achieved in 18 out of 25 patients (72%). While two patients died from unrelated causes, the remaining patients suffered no major complications. At a mean follow up of 13 months six patients had a patent ureter, while the remaining patients required repeat ureteral dilation [Koukouras et al. 2010].
Many reconstructive techniques have been performed laparoscopically and reconstruction of the ureter after iatrogenic injury is no exception. Laparoscopic techniques for the management of ureteral injuries include ureteroureterostomy, ureteroneocystostomy, and Boari-flap procedures. In a comparative retrospective review of patients undergoing laparoscopic and open techniques for ureteral reimplantation, patients under going distal ureteral reimplantation laparoscopically required less pain medication, had a shorter length of stay, and less blood loss [Rassweiler et al. 2007]. As ureteral reconstruction often requires intracorporeal suturing, the use of robotics in ureteral reconstruction is an area of keen interest. Schimpf and Wagner described a series of 11 patients who underwent laparoscopic, robotic-assisted, distal ureteral reimplantation. The authors reported no perioperative complications and follow-up imaging revealed patency of all ureters at a follow up of 24 months [Schimpf and Wagner, 2009]. Do and colleagues recently published a series of patients who underwent a robotic-assisted technique for Boari-flap ureteral reimplantation. In their series eight patients underwent the robotic-assisted Boari flap for ureteral pathology with four of these patients having ureteral pathology as an iatrogenic etiology. No procedures were converted to an open procedure and there were no intraoperative complications. One patient suffered from a prolonged anastomotic leak [Do et al. 2014].
Summary
Iatrogenic ureteral injury is uncommon, but often requires the expertise of a urologist to repair. Gynecologic, general surgical, and urologic procedures, especially in the pelvis, put the ureter at risk for injury. Clinicians should have a high suspicion for ureteral injury in the presence of postoperative fever, flank pain, leukocytosis, and peritonitis. Surgical management of the ureter is dictated by the location of the ureteral injury. In the case of distal ureteral injuries ureteroneocystostomy, with or without a vesico-psoas hitch, is the most commonly employed technique for management. If the injury is in the mid or proximal ureter then a ureteroureterostomy, transureteroureterostomy, or a Boari flap are commonly utilized. Rarely renal autotransplantation and ureteral substitution techniques are used to remedy an iatrogenic injury. Ureteral iatrogenic injuries continue to be managed with minimally invasive techniques and laparoscopic forms of traditional ureteral reconstruction techniques. As long as surgeons operate near the ureters the potential for iatrogenic ureteral injury will exist and requires a dynamic approach for management.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
