Abstract
Treatment of men with high-risk prostate cancer (PCa) remains challenging for urologists. The complex natural history of high-risk PCa and the lack of specific and accurate definitions for high-risk disease impede treatment decision making. Historically, surgery in this patient group has been avoided based on the perception of ostensibly higher complication rates associated with inferior functional and oncological outcomes. To date, no randomized data comparing different therapy approaches have been made available. Several investigators have reported that continence rates in patients after radical prostatectomy (RP) for high-risk disease seem to be unaffected. Similarly, in a large proportion of these men, a nerve-sparing procedure can be performed without a significant negative impact on surgical margin rates and with comparable potency results. Moreover, extended pelvic lymph node dissection (EPLND) contributes to accurate pathological staging with a marginal effect on perioperative morbidity. With regards to the benefits of RP on local recurrence and cure rates, realistic expectations regarding the success of RP alone or in the context of a multimodal approach should be provided during patient counseling.
Introduction
Prostate cancer (PCa) continues to be the most common cancer in men, and the second most frequent cause of cancer death in the United States [Jemal et al. 2010]. Oncological outcomes of PCa are strongly related to its stage, grade and preoperative prostate specific antigen (PSA) levels [Pound et al. 1997]. Patients with advanced localized disease, high-grade PCa on biopsy, and high pretreatment levels of prostate-specific antigen (PSA) have inferior outcomes compared with their counterparts with more favorable clinical characteristics [Chun et al. 2006; Bianco et al. 2005; Hull et al. 2002; Pound et al. 1997]. Depending on the definition used, between 15% and 38% of patients with PCa initially present with high-risk disease [Cooperberg et al. 2008; Yossepowitch et al. 2007; Greene et al. 2005]. Specifically, PSA screening has led to a decrease in clinically advanced PCa and has changed the characteristics of high-risk tumors. Patients are nowadays more likely to be categorized as high-risk according to Gleason grade alone rather than elevated PSA or abnormal digital rectal examination [Kane et al. 2007].
Despite the lack of randomized, prospective trials and large observational series comparing alternative treatment methods for high-risk PCa patients, many urologic surgeons favor a nonsurgical approach, such as radiation therapy and androgen-deprivation therapy, when confronted with patients showing unfavorable clinical characteristics [Boccon-Gibod et al. 2003; Bolla et al. 1997, 2002; Lawton et al. 2001; Hodgson et al. 1998]. According to population-based data from the United States, the proportion of patients undergoing radical prostatectomy (RP) for clinical T3 PCa decreased significantly from 18% to 9% between 1995 and 2001 while the proportion of patients undergoing radiotherapy within this group increased by 20% during the same period [Denberg et al. 2006].
In this review, we explore the definitions of high-risk PCa, the technical aspects of RP in these patients, as well as the morbidity, functional and oncological outcomes related to this procedure.
Risk factors of prostate cancer and definition of high-risk disease
The primary risk factors for PCa are clinical stage, initial PSA level, and biopsy Gleason score at presentation. Clinical stage is usually defined in accordance with the American Joint Committee on Cancer (AJCC) staging system [American Joint Committee on Cancer, 2002]. Patients described as T3 harbor extensive palpable disease compared with patients with incidentally discovered (T1) or small-volume palpable (T2) disease. The major drawback of clinical staging is its subjectivity and lack of precision. For example, several studies showed that many patients with clinical T3 PCa have favorable pathological features (organ-confined or specimen-confined disease) and some will even experience long-term biochemical recurrence (BCR)-free survival after RP [Yossepowitch et al. 2007; Carver et al. 2006; Ward et al. 2005; Van Poppel et al. 2000; Van Den Ouden et al. 1998]. A better correlation with oncological outcomes has been shown for histological grade [Gleason and Mellinger, 1974]. To date, it is widely acknowledged that patients with Gleason score 8–10 tumors are at higher risk of adverse outcomes [Bastian et al. 2006]. Finally, PSA remains an important variable in predicting risk of biochemical failure after RP [Mitchell et al. 2006]. Preoperative PSA level of 20 ng/ml or more is known to be a clinical indicator of high risk of postoperative PSA failure [D’Amico et al. 1995; Oesterling et al. 1988]. For example, these patients harbor organ-confined disease in 27%, and have extracapsular extension and seminal vesicle invasion in 73% and 36%, respectively [D’Amico et al. 1995].
BCR-free survival at 5 and 10 years in series reporting on ‘high-risk’ prostate cancer patients.

BCR, biochemical recurrence; PSA, prostate-specific antigen; NA, not available
Similarly, both the Radiation Therapy Oncology Group (RTOG) and the National Comprehensive Cancer Network (NCCN) use risk stratification schemes comparable to those of D’Amico and colleagues to identify patients with high-risk PCa. Commonly used criteria include patients with combinations of Gleason score 8–10, PSA levels >20 ng/ml, and clinical stage T2c–T4 disease.
Taken together, clinically localized high-risk PCa does not have a uniformly poor prognosis after RP. Many patients have organ-confined disease and remain BCR-free 5 years after surgery. The rates of favorable pathological stage and of BCR-free survival vary depending on the definition used.
Nerve-sparing radical prostatectomy in high-risk patients
From an oncological standpoint, a nerve-sparing technique may interfere with the paramount aim to avoid positive surgical margins, as large tumors may not be confined to the prostatic capsule. Nonetheless, the neurovascular bundle can be partially preserved in select patients with high-risk cancers, as the dissection of the erectile nerves is not an ‘all-or-nothing’ procedure. Preoperative imaging, precise knowledge about location and tumor grade of positive biopsy cores, different nerve-sparing techniques (intrafascial versus interfascial versus extrafascial approaches), and intraoperative frozen sections allow the preservation of the neurovascular bundle accurately in an individualized fashion.
Loeb and colleagues examined 288 men undergoing RP for high-risk or locally advanced cancers [Loeb et al. 2007]. In this series, a bilateral nerve-sparing procedure was performed in 75% of patients, which resulted in a potency rate of 64%. Men who underwent a nerve-sparing procedure had a significantly lower rate of positive surgical margins than those where nerve sparing was omitted (37% versus 54%, p = 0.002), most likely due to a selection bias. Lavery and colleagues reported on 1503 high-risk PCa patients undergoing RP [Lavery et al. 2010]. A nerve-sparing procedure was performed in 73% of cases. The overall positive surgical margin rate was 31% and was unrelated to nerve sparing. Moreover, no difference in BCR rate between the nerve-sparing and non-nerve-sparing groups was reported. Overall, 56% of patients were potent 1 year after surgery. Taken together, a nerve-sparing procedure in high-risk disease is feasible and safe in select patients, and leads to functional results comparable with those observed in patients with lower risk cancers.
Extended lymph node dissection during radical prostatectomy in high-risk patients
The European Association of urology guidelines [Heidenreich et al. 2011] recommend to perform an extended pelvic lymph node dissection (EPLND) in all high-risk cases, as the estimated risk for positive lymph nodes based on nomograms will range from 15% to 40% [Briganti et al. 2006]. According to the North American NCCN guidelines [National Comprehensive Cancer Network, 2010], a lymphadenectomy can be excluded in patients with <2% predicated probability of nodal metastases by a nomogram [Cagiannos et al. 2003]. EPLND is preferred when lymphadenectomy is performed, irrespective of risk strata.
An EPLND includes the obturator, external iliac, and hypogastric with or without the presacral and common iliac nodes. In comparison to a limited template, an EPLND has been reported to improve detection of lymph node invasion [Briganti et al. 2009]. Complication rates as high as 20% attributable to an EPLND has been described in reports published more than 30 years ago [Dillioglugil et al. 1997]. In contemporary series, the frequency and severity of these complications did not differ significantly between limited and extended lymphadenectomy [Allaf et al. 2004; Bader et al. 2003; Heidenreich et al. 2002]. Some authors report that EPLND performed by an experienced surgeon is associated with a complication rate of 7% and a symptomatic lymphocele rate of 2–4% [Heidenreich et al. 2007]. Moreover, low rates of venous thromboembolic events and intraoperative laceration of the ureter or obturator nerve have been described in patients undergoing EPLND. Despite the controversy, EPLND does not seem to be an entirely innocuous procedure, even in the hands of the most experienced surgeon. Therefore, Heidenreich and colleagues suggest that treatment-associated morbidity can be reduced significantly when (1) all lymphatics lateral to the external artery are saved, (2) the distal ends of the lymphatics are either ligated or clipped with small clips exerting a higher pressure to the lymphatic vessels than large clips, (3) two drains are placed in each side of the pelvis, (4) drains are left in place until <50 ml/day is drained, and (5) low-molecular heparin is injected into the upper arm [Heidenreich et al. 2007]. It remains to be seen whether EPLND has an impact on potency and continence recovery rates. The few studies addressing this relationship could not corroborate this hypothesis [Burkhard et al. 2006; Michl et al. 2006].
Nonetheless, the therapeutic role of an EPLND is still debated [Briganti et al. 2009]. Unfortunately, prospective randomized trials are lacking and the results of retrospective series are controversial. Some investigators suggest that EPLND might be beneficial with regard to PCa-specific survival and symptomatic progression [Joslyn and Konety, 2006; Masterson et al. 2006]. Prospective randomized trials examining the effect of lymph node dissection in high-risk patients can further define its effect on PCa outcomes.
Morbidity after radical prostatectomy for high-risk cancers
Most urologists recommend RP in low- and intermediate-risk patients. However, they do not recommend RP in patients harboring high-risk disease. This is surprising given that the latter are those who may benefit the most with regards to the risk of local recurrence and may even be cured. The perception of increased technical difficulty and morbidity rate has been a major source of concern for RP in locally advanced PCa. However, realistic expectations regarding the success of RP alone or in the context of a multimodal approach should be provided during patient counseling [Yossepowitch and Eastham, 2008].
Berglund and colleagues examined a cohort of 281 patients with locally advanced disease, of which 31% received neo-adjuvant androgen-deprivation therapy and/or chemotherapy [Berglund et al. 2006]. Mean hospitalization was 2 days and mean catheter duration was 9 days. The return of continence was described as qualitatively and quantitatively similar to that observed in the low-risk population (90%). The complication rate in a subset of 134 patients was 9.7% compared with 6.9% in patients with less-advanced disease. However, this study was descriptive and the control characteristics were not revealed. Ward and colleagues examined 5652 men who underwent RP at the Mayo Clinic [Ward et al. 2005]. Within a subgroup of 842 having cT3 cancer, morbidity was similar to patients with cT2 disease, but direct statistical comparisons were not made. Specifically, a deep vein thrombosis rate of 8% in the cT3 subgroup appears to be elevated compared with generally cited rates after RP (0.4–1.3%) [Rabbani et al. 2010], raising doubts that the rates reported in that specific study truly mirror morbidity rates in lower risk diseases. Novara and colleagues also reported that preoperative high-risk constellation does not influence complication outcomes [Novara et al. 2010]. In multivariable analyses, D’Amico risk groups were not significantly associated with any grade complications in 415 patients after robotic-assisted surgery. Conversely, a recent report by Agarwal and colleague using a standardized methodology of reporting complication rates, showed that in 3317 patients undergoing robotic-assisted RP, higher preoperative PSA levels represented an independent risk factor for any class of medical complications (p = 0.002) [Agarwal et al. 2011]. Similarly, patients with biopsy Gleason score 8 and higher had a 64% higher risk of experiencing any surgical complications than patients with Gleason score 6 and lower (p = 0.02). Interestingly, data derived from the CaPSURE database, a longitudinal, observational disease registry of men with PCa, revealed that patients belonging to the high-clinical-risk group had a twofold higher non-cancer-specific mortality rate than those in the low-clinical-risk group [Simone et al. 2008]. However, the examined study cohort consisted of patients treated with either RP or radiotherapy. The authors did not comment specifically on this finding and state that this result warrants further analyses in prospective trials. Another issue in high-risk patients is recovery of urinary incontinence. Reports focusing on continence rates uniformly do not show differences in urinary continence after RP when preoperative risk strata are examined [Novara et al. 2010; Pierorazio et al. 2007; Gontero et al. 2007; Kessler et al. 2007, Loeb et al. 2007; Nandipati et al. 2007; Bastide et al. 2006; Sacco et al. 2006; Kundu et al. 2004; Catalona et al. 1999; Eastham et al. 1996]. Moreover, a recent publication suggests that RP for high-risk localized PCa is associated with a reduction in mortality relative to radiation therapy and androgen-deprivation monotherapy [Cooperberg et al. 2010]. Another publication demonstrated a lower risk of metastatic progression as well as PCa-specific death in RP patients than patients after radiotherapy [Zelefsky et al. 2010]. However, these findings have to be corroborated by future, well-designed studies.
Taken together, large-scaled studies assessing complication rates after RP in patients with high-risk clinically localized PCa in a standardized fashion are lacking. Based on the contemporary evidence presented here, it is not possible to conclude that high-risk disease patients are at a higher risk of experiencing perioperative complications, whereas recovery of urinary incontinence seems to be unaffected.
Oncological outcomes after radical prostatectomy for high-risk cancers
In the majority of cases, BCR is used as a surrogate for more clinically meaningful endpoints such as metastatic progression or cancer-specific mortality [Freedland et al. 2005; Pound et al. 1999]. The 5-year BCR-free survival of these high-risk patients can vary substantially and may range from 24% to 68%, depending on the type of definition (Table 1). For example, Walz and colleagues showed that the rate of BCR-free survival within a large pooled study cohort of patients undergoing RP in Europe and North America varied substantially, depending on the definition used [Walz et al. 2011]. Specifically, the 5-year BCR-free rates after RP were 47.4% if D’Amico criteria were used to define high-risk PCa. The corresponding 5-year BCR-free rates if clinical stage, PSA ≥20 ng/ml and Gleason sum 8–10 were used separately were 51.6, 39.8 and 35.4%, respectively (Figure 1). Moreover, patients with cT3 PCa had the highest 5-year BCR-free rates (51.6%) but the lowest rate of pathologically favorable disease (13.7%), which means that favorable pathology is not directly related with BCR-free status and adverse pathology is not synonymous with BCR. In addition, patients with only one risk factor had the most favorable 5-year BCR-free survival (50.3%), relative to patients with two or more risk factors (27.5%). Yossepowitch and colleagues also examined pathological and clinical outcomes among high-risk patients treated with RP [Yossepowitch et al. 2008, 2007]. The authors applied eight definitions of high-risk cancers and showed that the proportion of high-risk patients was between 3% and 38% depending on the criteria. One third of them (22–63%) had organ-confined cancers on pathology and nearly half of their study population (41–74%) remained progression-free 10 years after surgery as monotherapy. Furthermore, up to 76% of the high-risk patients were alive and free of additional cancer therapy 10 years after RP.
The probability of biochemical recurrence (BCR)-free survival rates in a large European and North American cohort of patients with high-risk prostate cancer (A). The effect of clinical stage (B), biopsy Gleason sum (C), stratified pretreatment serum prostate-specific antigen (PSA) levels (D), D’Amico risk grouping (E) on BCR-free survival rates are depicted. Graphical comparison of the BCR-free survival rates according to the four high-risk definitions (F) and the effect of the cumulative number of risk factors (G) are illustrated. (Reprinted with permission from Walz et al. [2011] Copyright © [2011][Wiley-Blackwell] [BJUI]. Access at: http://onlinelibrary.wiley.com/doi/10.1111/j.1464-410X.2010.09594.x/abstract;jsessionid=33AE69ED23E0C02B29DB9B14EB3A444E.d02t04.).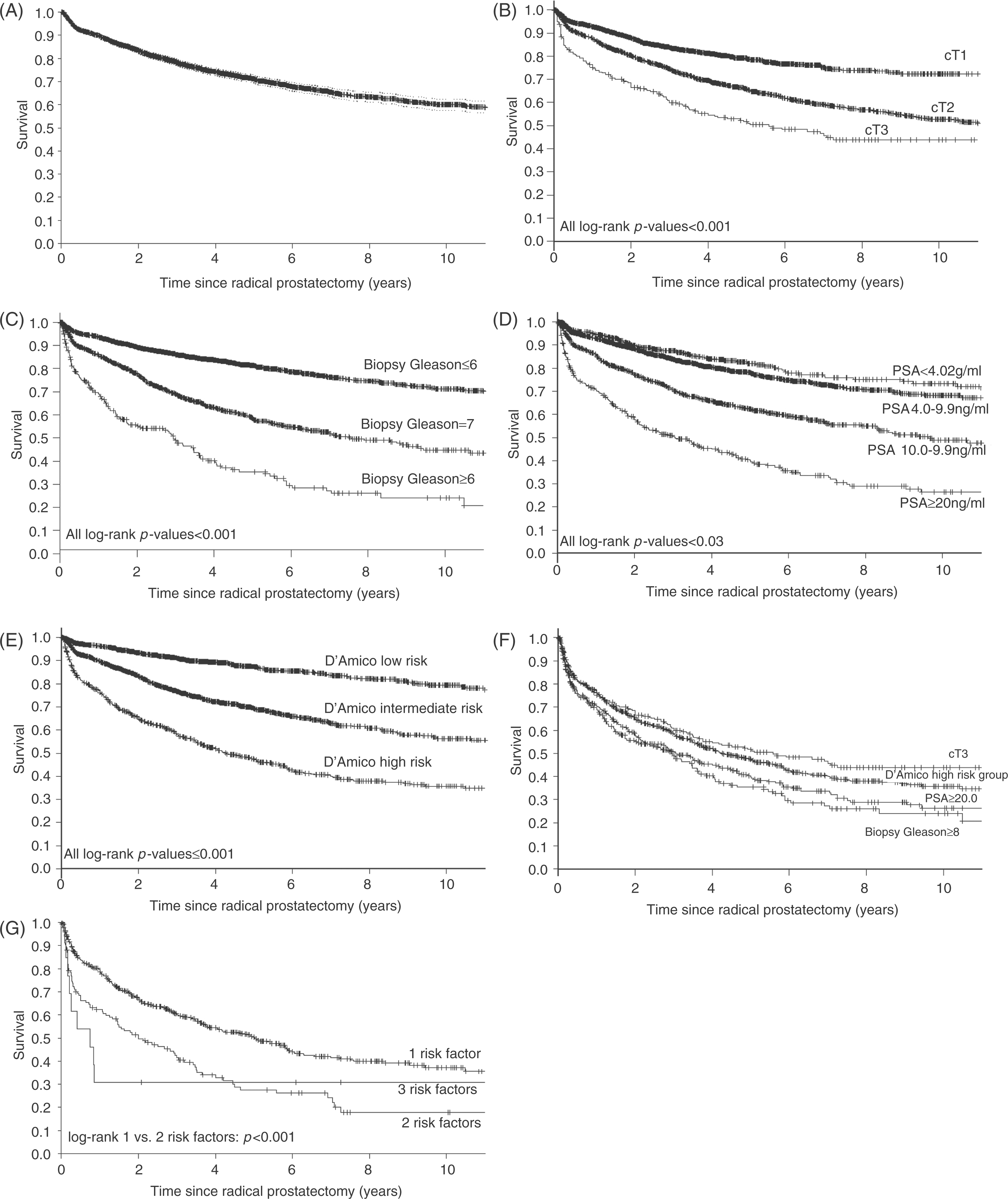
These findings clearly illustrate the complex natural history of high-risk PCa and emphasize the need for more specific and accurate definitions for high-risk disease. In addition, in comparison with high-risk PCa patients harboring two or more primary risk factors, those with only one primary risk factor have a better oncological outcome. In consequence, it might be postulated that patients with one primary risk factor are more likely to benefit from RP alone, whereas patients with multiple of risk factors may warrant a multimodal treatment approach.
Conclusions
The complex natural history of high-risk PCa, as well as a lack of specific and accurate definitions for high-risk disease impede treatment decision-making for patients with high-risk PCa and emphasize the need for more specific and accurate high-risk definitions. The historical perception of higher rates of urinary incontinence, erectile dysfunction and perioperative complications after RP in this patient group is not supported by the current literature. Performing a nerve-sparing technique in the vast majority of these patients seems to be oncologically safe, if properly selected. A concomitant EPLND contributes to improve pathological staging, while marginally affecting the morbidity of RP. Moreover, patients harboring only one primary risk factor may benefit from RP alone, whereas patients with multiple risk factors are more likely to warrant a multimodal treatment approach. In consequence, RP in high-risk PCa patients is a valuable therapeutic possibility.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
