Abstract
Background:
Motor fluctuations and OFF episodes are common complications of long-term levodopa therapy in Parkinson’s disease (PD) and significantly impair quality of life. Apomorphine, a short-acting dopamine agonist, is available in multiple formulations for on-demand symptom relief; however, comparative evidence across administration routes remains limited.
Objectives:
To evaluate the efficacy and safety of apomorphine across various routes of administration in patients with PD.
Design:
Systematic review and meta-analysis of randomized controlled trials and randomized crossover trials.
Data sources and methods:
A comprehensive search of PubMed, Scopus, Web of Science, CENTRAL, and Embase was conducted from inception to December 28, 2025. Eligible studies included randomized controlled or crossover trials assessing apomorphine versus placebo in PD. Primary outcomes were motor improvement measured by Unified Parkinson’s Disease Rating Scale III (UPDRS-III) or Movement Disorder Society-UPDRS-III (MDS-UPDRS-III) and OFF time. Safety outcomes included treatment-related adverse events. Risk of bias was assessed using the Cochrane RoB 2 tool. Pooled analyses were conducted using standardized mean differences (SMD), mean differences (MD), and risk ratios (RR) with 95% confidence intervals (CIs).
Results:
Thirteen studies involving 557 patients were included. Apomorphine significantly improved motor symptoms across multiple administration routes. Intermittent subcutaneous (SC) injection showed the greatest efficacy (SMD: −2.19, 95% CI: (−3.32, −1.05), p < 0.0001). The inhalation and sublingual routes also showed significant improvement (SMD: −1.11, 95% CI: (−1.52, −0.7), p < 0.0001, I2 = 0%), (SMD: −1.69, 95% CI: (−1.99, −1.38), p < 0.0001, I2 = 0%) respectively. Intermittent SC injections also significantly reduced OFF time (MD = −1.62 h; 95% CI = (−2.59, −0.65); p < 0.00001). Adverse events were more frequent with apomorphine (RR 1.50, 95% CI = 1.09–2.06), particularly nausea, vomiting, dyskinesia, somnolence, yawning, and rhinorrhea.
Conclusion:
Apomorphine is an effective on-demand therapy for reducing motor symptoms and OFF time in PD, particularly when administered as intermittent SC injection. Although associated with increased mild-to-moderate adverse events, treatment choice should be individualized.
Trial registration:
PROSPERO (CRD42024548330).
Introduction
Parkinson’s disease (PD) is the second most common neurodegenerative disorder after Alzheimer’s disease. It is estimated that PD has exhibited the fastest-growing prevalence and disability disorder among neurological diseases worldwide, with a particularly pronounced increase over the past two decades.1,2 The hallmark of PD is the progressive degeneration of dopaminergic neurons in the substantia nigra, which leads to a reduction in dopamine levels in the striatum, a key component in the basal ganglia responsible for smooth and coordinated purposeful movements.3,4 The exact etiology of dopaminergic neuron degeneration in PD remains unknown. However, a complicated interplay between genetic and environmental factors likely plays a role. 4 Despite the advances in management strategies, PD treatment remains challenging, as there are no approved pharmacologic treatments that can stop or slow its progression, and all the current therapies mainly focus on relieving its motor and non-motor symptoms. 5
Oral levodopa remains the cornerstone of symptomatic therapy for PD; however, prolonged use is associated with motor complications such as motor fluctuations and dyskinesia.6–8 Within 4–6 years of treatment, approximately 40% of patients develop these complications, with the figure rising to 70% after 9 or more years. 9 These motor fluctuations can lead to multiple OFF-episodes per day, cumulatively accounting for up to 50% of a patient’s waking day,10,11 significantly impacting their quality of life.12,13 To manage levodopa-related motor complications, three main approaches are commonly employed: the addition of oral adjuncts, continuous drug delivery (e.g., infusion), and the use of “on-demand” medications designed to abort OFF episodes rapidly. 14 In addition, in advanced PD, device-aided therapies such as deep brain stimulation (DBS) are also established options for selected patients with disabling motor fluctuations. 15 Apomorphine, a short-acting dopamine (D1 and D2) receptor agonist, is notable for its efficacy in reducing motor symptoms, with a more rapid onset of action than levodopa, albeit with a shorter duration.16,17
Apomorphine, a drug chemically synthesized from morphine in the mid-1800s that was initially used to treat psychiatric conditions as a sedative, is one of the emerging medications. 18 In the 1950s, it was discovered that apomorphine has a structure similar to dopamine and was then considered a possible treatment for PD. However, oral apomorphine was not found to be very effective at that time due to its short duration of action, poor bioavailability, and high “first-pass” metabolism in the liver. 19 Researchers then started to explore other novel preparations of apomorphine, including subcutaneous (SC) injection, SC infusion, and sublingual and nasal forms that have been proven to be effective in multiple clinical trials.10,18–20 To date, there has been no comprehensive systematic review of the efficacy of all apomorphine protocols in PD. Therefore, we conducted this systematic review and meta-analysis to assess the safety and effectiveness of various apomorphine modalities in PD.
Methods
This systematic review and meta-analysis were conducted following PRISMA guidelines 21 and the Cochrane Handbook for Systematic Reviews of Interventions. 22 The protocol for this study was registered in PROSPERO (CRD42024548330).
Data source and search strategy
A systematic literature search starting from inception until May 22, 2024, and was later updated up to December 28, 2025 to include studies measuring the efficacy of apomorphine modalities in PD. We searched PubMed, Scopus, Web of Science, CENTRAL, and Embase with relevant keywords. The PubMed search strategy was: ((apomorphine OR AZ-009 OR Apo-morphine OR Apokyn OR Kynmobi OR APO-Go OR C17H17NO2 OR SL-APO OR sc-apmo) AND (Parkinson’s OR Parkinsonism OR Parkinsonian OR Parkinsonism-plus OR Parkinson) AND (randomized controlled trial OR RCT OR cohort study OR case-control study OR registr* OR clinical trial OR prospective study OR retrospective study OR controlled OR PLACEBO)). The screening was done by four independent authors (O.Y., A.Z.A., H.S., and M.A.Z.) using the Rayyan software. 23 The first author (M.E.A.) resolved any conflicts.
Eligibility criteria
Study design
Randomized controlled trials (RCTs) and randomized crossover trials that investigate the use of apomorphine in PD patients were included. Crossover RCTs were included if washout periods were clearly defined and data could be extracted from the first treatment phase, as recommended by the Cochrane Handbook, to expand generalizability. 22 Population: Patients diagnosed with PD. Intervention: Apomorphine in its different modalities (e.g., SC, Intravenous, inhalation, etc.). Comparator: Placebo or standard care. Outcomes: efficacy measured by the Unified Parkinson’s Disease Rating Scale III (UPDRS-III) 24 or the Movement Disorder Society version (MDS-UPDRS-III) 25 and/or OFF-time, and safety was measured by adverse events. Exclusions: Observational studies, single-arm trials, reviews, case series, animal studies, and no extractable quantitative outcomes.
Data extraction and risk of bias assessment
Relevant data from selected studies were independently extracted by four review authors into Google Sheets. The extracted information included the study design, study population characteristics, and outcome measures. The primary outcomes of this study were improvement in motor symptoms, measured by UPDRS III and MDS-UPDRS III, off-time, and adverse events.
The risk of bias was evaluated using the Cochrane risk of bias tool RoB-2 for RCTs and modified RoB-2 for crossover trials, 26 and studies were assessed as low, with some concerns, and a high risk of bias. The RoB-2 tool consists of five domains: (1) bias arising from the randomization process, (2) bias due to deviations from intended interventions, (3) bias due to missing outcome data, (4) bias in the measurement of the outcome, and (5) bias in the selection of the reported result. At the same time, a sixth domain is added for crossover trials: period and carryover effect. Studies were assessed as having low, some concerns, or high risk of bias across these domains.
Data synthesis and analysis
R version 4.4.1 with meta and metafor packages.27,28 For Windows, the packages were used for analysis with meta-analysis packages. Continuous pooled outcomes were reported as Standardized mean differences (SMD) and 95% confidence interval (CI) for pooling MDS-UPRS III and UPRS III results together, while mean difference (MD) with 95% CI for was used for off-time outcome and risk ratios (RR) and 95% CI for an adverse event. Peto methods with odds ratios (ORs) were used for analyses in which all events in one group across studies were zero. To assess heterogeneity, we visually inspected the forest plot and tested the Chi-squared distribution of the Cochrane Q statistic (a Chi-square p-value <0.1 indicating significant heterogeneity) and the I2 statistic (>50% indicating considerable heterogeneity). In cases of significant heterogeneity, a random-effects model was used; otherwise, a fixed-effects model was used. For the meta-analytic effect sizes, a p-value <0.05 was considered statistically significant. We could not assess publication bias using the funnel plot and Egger’s test because the number of studies for each analysis model was less than 10. 29 Outcomes of RCTs and randomized crossover trials were pooled after proper data management following Cochrane guidelines. 22 First MD and their standard error (SE) for change scores of RCTs were transformed to SMD and SE through the metafor package. While SMD and SE of crossover trials were calculated using the equations below, with a correlation coefficient of 0.7 calculated from included studies, after that, pooling of these results was conducted as SMD and SE through the metafor package.
Results
Study selection
After preliminary database and reference searches, 2088 results were found; out of them, only 13 were included in our systematic review and meta-analysis.20,30–41 The PRISMA flow diagram for the identification and selection of research is shown in Figure 1.

PRISMA flowchart summarizing the study selection process.
Characteristics of the included studies
We included 13 studies conducted across multiple countries, including the United States, the United Kingdom, France, Germany, Italy, Japan, Sweden, Spain, and the Netherlands. Of these, seven were RCTs, and six were randomized crossover studies. Various administration routes were used in these studies, including SC infusion, SC injection, sublingual, intranasal spray, and inhalation. Detailed characteristics of the included studies are presented in Table 1. The total number of patients across all studies was 557, with an overall mean age of 62.84 years, and 61.78% of participants were male (seeTable 2 for baseline characteristics).
Summary of included studies.
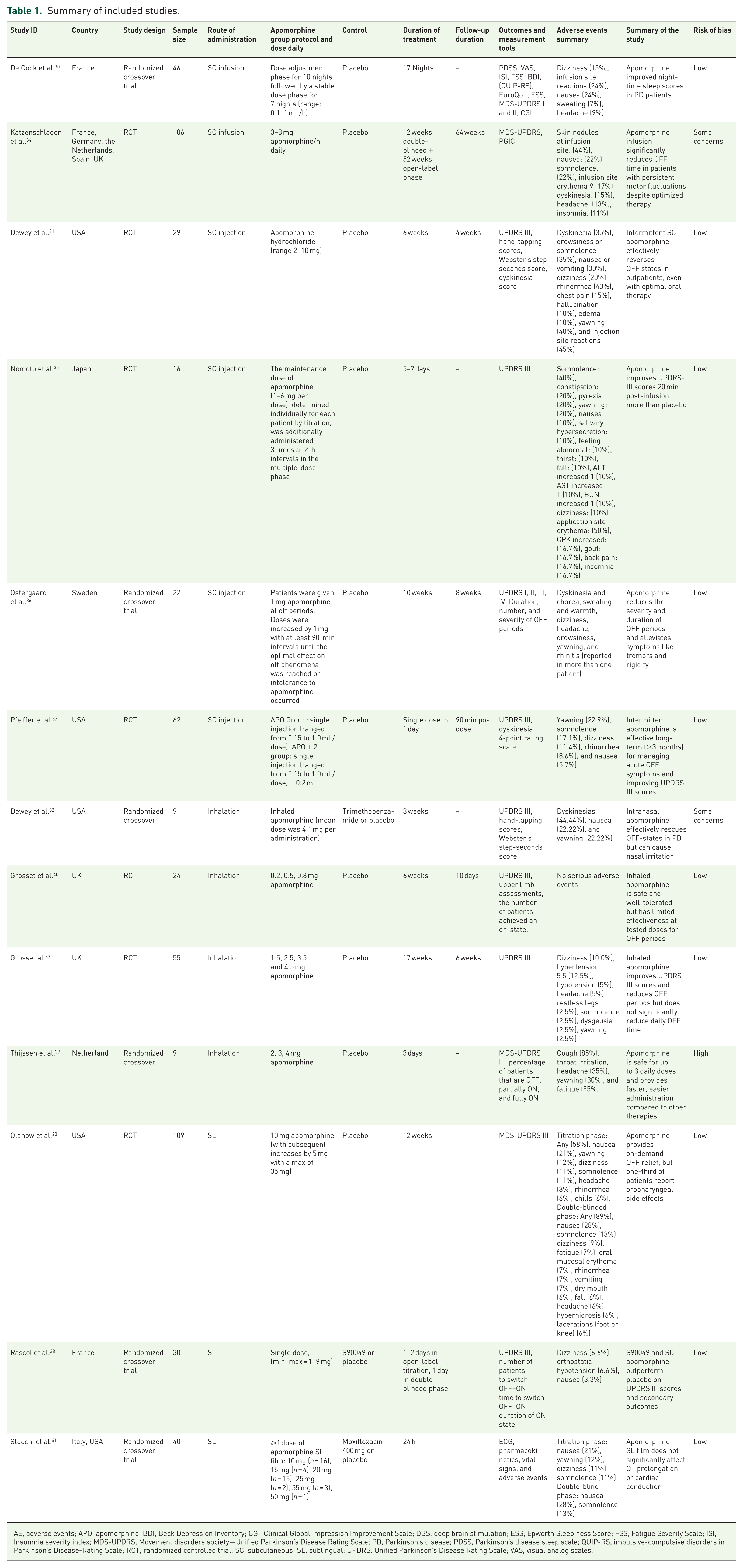
AE, adverse events; APO, apomorphine; BDI, Beck Depression Inventory; CGI, Clinical Global Impression Improvement Scale; DBS, deep brain stimulation; ESS, Epworth Sleepiness Score; FSS, Fatigue Severity Scale; ISI, Insomnia severity index; MDS-UPDRS, Movement disorders society—Unified Parkinson’s Disease Rating Scale; PD, Parkinson’s disease; PDSS, Parkinson’s disease sleep scale; QUIP-RS, impulsive-compulsive disorders in Parkinson’s Disease-Rating Scale; RCT, randomized controlled trial; SC, subcutaneous; SL, sublingual; UPDRS, Unified Parkinson’s Disease Rating Scale; VAS, visual analog scales.
Baseline characteristics of included studies.

APO, apomorphine; COMT, catechol-O-methyltransferase; DBS, deep brain stimulation; LEED, levodopa equivalent daily dose; MAOB, monoamine oxidase type B; MDS-UPDRS, movement disorders society—unified Parkinson’s Disease Rating Scale; SC, subcutaneous; SL, sublingual; NA, not available.
Risk of bias in studies
Ten studies were assessed using the RoB 2 tool. Among these, seven studies19,30,32–34,36,39 demonstrated an overall low risk of bias. In addition, six crossover studies were assessed using the RoB 2 tool for crossover trials, with two studies showing some concerns while the others showed a low risk of bias (see Figure 2).

RoB2 results for randomized controlled trials and crossover trials.
Impact of apomorphine administration route on MDS-UPDRS-III and UPRS-III
The SC injection subgroup, comprising 3 studies with 107 participants, showed a statistically significant difference favoring apomorphine (SMD = −2.19, 95% CI: (−3.32, −1.05), p < 0.0001, I2 = 76%; Figure 3). This significant heterogeneity was resolved (I2 = 0%) after the exclusion of the Dewey et al.’s 31 study, and results remained significant (SMD: −1.68, 95% CI: (−2.20, −1.16), p < 0.0001; Figure 4). The sublingual subgroup, including 2 studies with 139 participants, also demonstrated a significant effect favoring apomorphine (SMD = −1.69, 95% CI: (−1.99, −1.38), p < 0.0001, I2 = 0%, p < 0.0001). Similarly, the inhalation subgroup, with 3 studies totaling 59 participants, showed a significant benefit for apomorphine (SMD = −1.11, 95% CI: (−1.52, −0.70), p < 0.0001, I2 = 0%). By contrast, the subcutaneous infusion subgroup, which included 1 study with 59 participants, found no significant difference between apomorphine and placebo (SMD = −0.24, 95% CI: (−0.75, 0.27), p = 0.37). Overall, the pooled analysis showed a statistically significant improvement for apomorphine across most subgroups, with substantial heterogeneity resolved through subgrouping (see Figure 3). A statistically significant difference in MDS-UPDRS III and UPDRS-III was observed between the four groups. After excluding the outlier by the subcutaneous infusion study, the test of subgroup differences remained significant, where SC injections were better than sublingual and inhalation routes (p < 0.0001; Figure 5).

Forest plot of the efficacy of apomorphine on UPDRS-III with subgroups according to the route of administration.
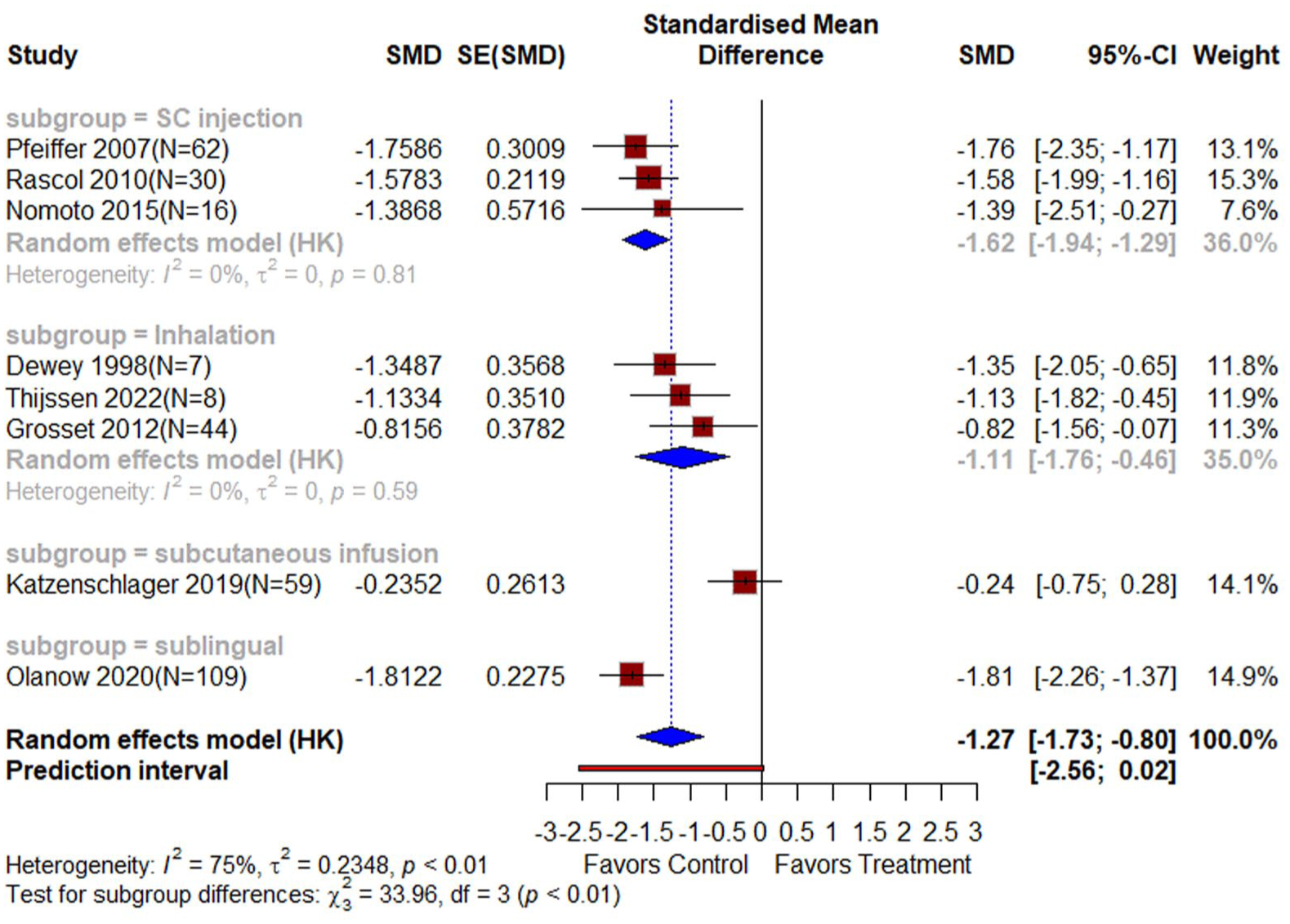
Sensitivity analysis of SC injection after exclusion of Dewey et al. 31

Sub-group differences test between inhalation and subcutaneous injection routes.
Impact of apomorphine on off-time
Two studies33,34 with a total of 82 patients in the apomorphine group and 63 patients in the placebo group were analyzed for off-time hours’ outcomes. The analysis revealed a statistically significant decrease in off-time for patients receiving apomorphine (MD: −1.62, 95% CI: (−2.59, −0.65), p < 0.00001, I2 = 0%), compared to those receiving placebo (see Figure 6).

Forest plot of the efficacy of the apomorphine on off-time hours.
Adverse events
Eight studies reported treatment-related adverse effects, involving 263 patients in the apomorphine group and 238 in the placebo group. The analysis revealed a statistically significant increase in adverse effects in the apomorphine group compared to placebo (RR = 1.50, 95% CI: (1.09, 2.06), I2 = 74%). Subgroup analysis by route of administration showed that subcutaneous infusion (2 studies, RR = 1.60, 95% CI: (1.27, 2.02)) significantly increased adverse effects in the apomorphine group, while sublingual administration (3 studies, RR = 2.10, 95% CI: (0.86, 5.12)), inhalation (1 study, RR = 1.18, 95% CI: (0.85, 1.64)), subcutaneous injection (2 studies, RR = 1.05, 95% CI: (0.61, 1.80)) showed no significant difference. The test for subgroup differences indicated no significant interaction between route of administration and adverse effects (p = 0.24; Figure 7).
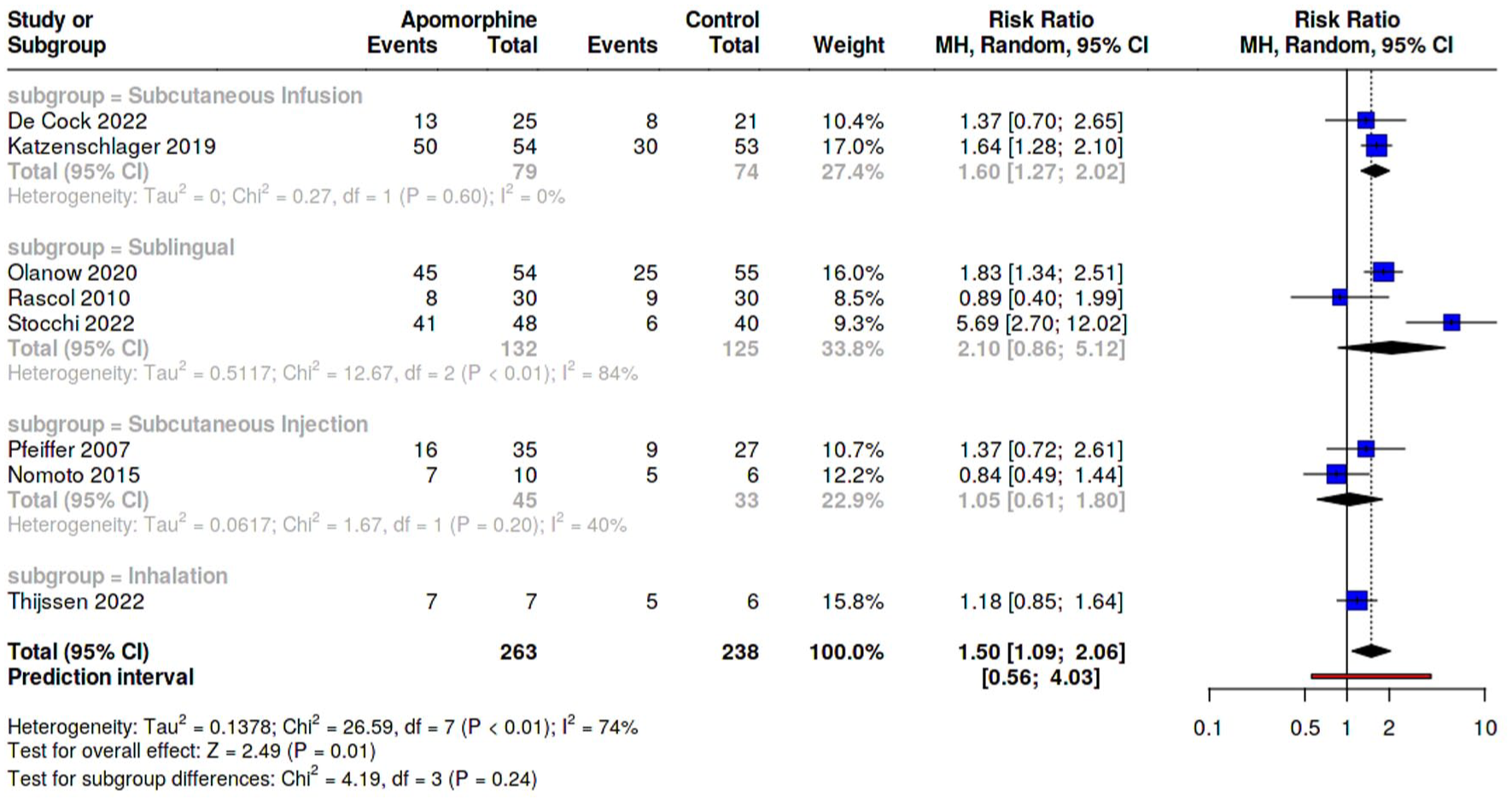
Forest plot of risk ratios of meta-analysis for treatment-related adverse events.
Five studies with 169 patients in the apomorphine group and 160 in the placebo group reported headaches, showing a significant increase in headache risk with apomorphine (RR = 3.37; 95% CI: (1.20, 9.45); p = 0.02; I2 = 0%). Subgroup analysis by route of administration revealed no significant differences between subgroups (p = 0.89, Figure 8(a)), and none of the routes were significant. Similarly, 6 studies involving 221 patients in the apomorphine group and 190 in the placebo group reported increased somnolence risk (RR = 6.07; 95% CI: (2.74, 13.43); I2 = 0%; Figure 8(b)). Subgroup analysis showed significant increases for subcutaneous infusion (RR = 5.89; 95% CI: (1.38, 25.06)) and sublingual administration (RR = 5.62; 95% CI: (1.73, 18.25)), with no significant differences between subgroups (p = 1.00). This was further confirmed by yawning analysis where for yawning, 3 studies with 120 patients in the apomorphine group and 88 in the placebo group reported a pooled significant increase in risk with apomorphine (RR = 3.81; 95% CI: (1.37, 10.60); p = 0.01; I2 = 0%, Figure 8(c)). Subgroup analysis showed increased risks for subcutaneous injection (RR = 3.67; 95% CI: (1.13, 11.94)), but no significant differences for inhalation or sublingual administration. No significant differences were observed between subgroups (p = 0.99).

Forest plot of risk ratios of meta-analysis for headache (a), somnolence (b), and yawning (c).
Six studies, including 204 patients in the apomorphine group and 173 in the placebo group, reported dizziness. The analysis found no significant difference between the groups (RR = 2.57; 95% CI: (0.82, 8.08); I2 = 20%), with no significant subgroup differences based on the route of administration (p = 0.69, Figure 9(a)) or significant effects for any of the subgroups. However, analysis of five studies demonstrated a significant increase in risk of nausea with apomorphine (RR = 3.82; 95% CI: (1.52, 9.59); p < 0.01; I2 = 34%, Figure 9(b)), with none of the individual routes showing significance. This was further supported by two sublingual apomorphine studies reporting a significant increase in risk of vomiting with apomorphine (OR = 7.67; 95% CI: (2.47, 23.79); p < 0.01; I2 = 0%, Figure 9(c)). Furthermore, an analysis of three studies revealed increased overall dyskinesia risk (RR = 5.08; 95% CI: (1.24, 20.89); I2 = 0%), although none of the individual routes were significant. The risk of both fatigue and orthostatic hypotension was not significant across studies (see Figure 10(b) and (c)). As for the risk of rhinorrhea, it was significant for both subcutaneous injections and sublingual routes with an overall effect size of (RR = 6.45; 95% CI: (1.84, 22.56); I2 = 0%,Figure 11(a)). Paying additional interest for injection site reactions accompanied by apomorphine administration, it was not significant for either injection (RR = 0.81; 95% CI: (0.43, 1.51); I2 = 0%) or perfusion (RR = 1.34; 95% CI: (0.35, 5.09); I2 = 88%) or both of them (RR = 1.07; 95% CI: (0.53, 2.15); I2 = 70%; see, Figure 11(b)).

Forest plot of risk ratios of meta-analysis for dizziness (a), nausea (b), and vomiting (c).

Forest plot of risk ratios of meta-analysis for dyskinesia (a), fatigue (b), and orthostatic hypotension (c).

Forest plot of risk ratios of meta-analysis for rhinorrhea (a) and injection site reactions (b).
Discussion
This systematic review and meta-analysis evaluated the efficacy of apomorphine in treating PD, primarily measuring the improvement in motor symptoms using the MDS-UPDRS-III and UPDRS-III. The findings indicated that apomorphine significantly improved motor symptoms compared to placebo. Among the administration routes, apomorphine inhalation, intermittent SC injection, and sublingual significantly reduced motor scores compared to placebo, with intermittent SC injection being the most effective modality, reducing UPDRS-III by a large effect. By contrast, continuous subcutaneous infusion did not demonstrate a significant benefit over placebo. Apomorphine also significantly decreased OFF time by an average of 1.62 h, indicating a clinically meaningful improvement in motor fluctuations. In terms of safety, apomorphine did not significantly differ from placebo in causing fatigue, dizziness, orthostatic hypotension, and injection site reactions. However, it was associated with a higher incidence of headaches, dyskinesia, somnolence, yawning, rhinorrhea, dyskinesia, vomiting, nausea, and treatment-related adverse events.
Apomorphine is a dopamine agonist that binds to postsynaptic dopamine receptors and exerts a therapeutic effect by direct stimulation of postsynaptic striatal dopamine receptors. 42 The motor response of apomorphine is similar to that of levodopa, but with a faster onset, making it one of the rescue medications in cases of unpredictable off periods. 42 However, the oral route of apomorphine is subject to extensive first-pass metabolism, resulting in a bioavailability of less than 4%, so it is considered unfeasible as it requires very high doses to achieve a therapeutic effect. 43 Therefore, alternative administration routes, including subcutaneous, sublingual, and inhalation, were explored to identify the most suitable options for patients with PD.
The most common route of administration for apomorphine is subcutaneous, either as an intermittent injection or via continuous infusion. Intermittent SC injections were first investigated as acute treatment for off-medication state, but were later investigated as repeated doses or SC infusion for preventing off states and as a treatment for PD. The intermittent SC injection route was the most effective in improving motor symptoms in our meta-analysis. Dewey et al.’s 31 study was the main source of heterogeneity in the SC injection subgroup with a very large effect. This was the earliest RCT and had some concerns in one of its domains. Regarding safety, among pooled adverse events, SC injection was significantly associated with yawning and rhinorrhea. However, intermittent SC apomorphine may cause injection site reactions such as bruising, granulomas, or itching, but these typically do not lead to discontinuation. The included studies showed that intermittent SC injection of apomorphine provided rapid relief from off periods. However, intermittent SC injection is unsuitable when off periods occur frequently, as it would require multiple injections. In addition, severe off periods may limit a patient’s ability to self-administer the injection, and the absence of a caregiver to assist further could limit its feasibility. 44 Continuous subcutaneous apomorphine infusion provides constant drug delivery. It is usually considered for patients with severe PD who cannot maintain adequate ON-time or tolerate levodopa or DBS. Katzenschlager et al. examined the efficacy of CASI on MDS-UPDRS III during and found no significant difference compared to placebo. This study measured only the MDS-UPDRS III in the ON state, and motor symptoms may already have been optimized by baseline levodopa treatment, which could explain the insignificant results. The dilemma of apomorphine SC infusion is still present with younger patients showing better results with DBS, while older patients eventually switch to oral therapies due to the inconvenience of the Pump, and can be at most a “transition” treatment between PD therapies. 45 However, a recent RCT published in The Lancet examined SC night-time-only apomorphine infusion to treat insomnia in patients with PD and found SC night-time apomorphine infusion to enhance sleep quality and improve self-reported motor conditions. 30 Katzenschlager et al. found a significant increase in infusion-site reactions, while De cock et al. did not, which can be explained by the longer duration of the Katzenschlager et al. study.
To avoid the inconvenience of invasive SC injections and infusions, alternative forms of apomorphine delivery have been developed. Inhaled (intranasal) apomorphine provides rapid symptom relief and significant improvements in UPDRS-III scores, making it beneficial for the acute management of OFF periods.33,40 The inhalation route improved motor symptoms and was not significantly associated with any of the adverse effects in our meta-analysis. The primary advantage of inhalation is patients’ acceptance and convenience, as intermittent SC injection may be problematic for some patients. However, commonly reported symptoms may include mild throat irritation and cough after inhalation that resolved within minutes.33,39,40 Despite the improvement in UPDRS III reported in studies, daily off time was not significantly affected in any of the included studies. However, inhaled apomorphine showed a similar onset of action (8.1 min) to subcutaneous apomorphine (7.5–14 min). It was faster than other rescue treatments, such as sublingual apomorphine (28 min), as Grosset et al. 33 noted.
The sublingual route demonstrated a significant benefit over placebo for motor symptoms. However, SL apomorphine may irritate, including tissue swelling, pain, ulceration, or erythema, which can lead to discontinuation in a significant proportion of patients. While SL films may offer rapid conversion of OFF episodes to ON states, though with somewhat less robust efficacy compared to SC routes.20,35,38 The efficacy and safety of SL apomorphine have been further supported by a recent non-randomized, open-label study that reported significant clinical improvement in more than 60% of patients with PD with off episodes over 48 weeks. However, a high rate of discontinuation due to adverse events was still reported (28%). 46
Regarding the pharmacokinetics of “on demand” apomorphine formulations, SC apomorphine has 100% bioavailability, 47 whereas SL apomorphine has a bioavailability of only 17%–18%, partly due to absorption through the gastrointestinal tract.43,48 These differences can affect the time course and intensity of the drug’s effect, suggesting that SC apomorphine provides more reliable and rapid symptom relief compared to SL administration. 49 However, a previous randomized crossover study compared SL-APO with subcutaneous apomorphine in patients with PD and OFF episodes, showed no significant difference in motor improvement (MDS-UPDRS part III) and adverse events but found 72.2% of patients preferred SL-APO with greater convenience and satisfaction rates. 50 These findings are supported by a choice experiment that revealed that PD patients theoretically preferred SL films over inhaled SC modes of medicine administration. Even with adverse events as possible risks, participants preferred oral adverse events over inhaled-related (cough) or SC-related (injection-site reaction) events. 51 These findings shall be taken into consideration when interpreting our results.
Our results suggest that apomorphine provides clinically meaningful benefits in PD, with subcutaneous injection showing the largest improvement in motor symptoms compared with other routes, while also reducing OFF-time, which may translate into better daily functioning. The observed heterogeneity across routes indicates that effectiveness varies by formulation and delivery method, so treatment choice should be individualized and informed by patient preference and available evidence, particularly for less-studied modalities. Future studies should focus on long-term safety and efficacy, especially for night-time infusion, inhaled, and intranasal formulations, including effects on disease course, sleep, and overall tolerability. Research is also needed to define optimal dosing, identify which patient subgroups benefit most from each route, improve objective measurement of OFF periods (given diary–clinician discrepancies), and evaluate how different delivery methods affect adherence and quality of life over time.
Regarding the strengths of this study, it is the first meta-analysis to assess the effects of various apomorphine modalities on motor symptoms in PD. We performed a subgroup analysis to determine the efficacy of each route of administration and identified SC injection as the most effective modality. Variations in study designs limit the generalizability of these findings. However, the study has limitations. Significant heterogeneity was detected and addressed using a random-effects model, leave-one-out analyses, and subgroup analyses. The follow-up periods were insufficient to assess the long-term safety and efficacy of apomorphine. In addition, only MDS-UPDRS III, UPDRS III, and off times were analyzed, as these were the consistently available outcomes. The relatively small sample size may lead to spurious effects, and the power of the subgroup analysis was limited by the small number of included studies, potentially reducing the precision of the results. The inclusion of crossover trials may introduce some heterogeneity. However, we handled them in accordance with the Cochrane guidelines. We were unable to conduct subgroup analyses to assess the effects of variations in doses, study designs, and measurement tools due to the limited number of studies. In addition, most studies did not use a uniform dose protocol but based on doses for each participant individually.
Conclusion
According to our study, apomorphine has the potential to be a transformative treatment for PD patients who are unsuitable for surgery or have not had a sufficient response to medications such as levodopa. The treatment markedly improves motor complications in PD patients, as evidenced by a significant reduction in UPDRS-III and MDS-UPDRS-III scores, indicating enhanced motor functions. In addition, apomorphine decreases off-time by an average of 1.62 h, thereby further improving the quality of life for these patients. The safety profile of apomorphine is acceptable, with no significant difference in adverse events such as dizziness, orthostatic hypotension, injection site reactions, and fatigue compared to placebo. However, some side effects, including headache, vomiting, somnolence, yawning, rhinorrhea, nausea, and treatment-related events, were more common but generally manageable. Despite these promising results, further studies with larger sample sizes and longer follow-up periods are necessary to confirm the long-term safety and efficacy of apomorphine. This will ensure a comprehensive understanding of its potential benefits and limitations, guiding its optimal use in clinical practice.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261436541 – Supplemental material for Efficacy and safety of apomorphine in the treatment of Parkinson’s disease: a systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-tan-10.1177_17562864261436541 for Efficacy and safety of apomorphine in the treatment of Parkinson’s disease: a systematic review and meta-analysis of randomized controlled trials by Moaz Elsayed Abouelmagd, Abdallah Abbas, Obai Yousef, Atef Abdelrahman Hassan, Omar Kassar, Khalid Sarhan, Asmaa Zakria Alnajjar, Haneen Sabet, Mohamed Ahmed Zanaty, Hoda Awad, Muataz Kashbour, Fakhruddin Almuzghi, Matthew J. Barrett, Brian Berman and Ahmed Negida in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

