Abstract
Background:
Despite the availability of several disease-modifying therapies for multiple sclerosis (MS) patients, the optimal strategy remains debated. High-efficacy therapies may better prevent subclinical disease activity and long-term disability; however, escalation remains frequent in real-world practice. Ofatumumab, an anti-CD20 monoclonal antibody, demonstrated robust efficacy and safety in clinical trials; however, real-world data are also essential.
Objectives:
To evaluate the effectiveness and safety of ofatumumab in relapsing MS (RMS), comparing outcomes between treatment-naïve and previously treated patients, and identify predictors of suboptimal response.
Design:
Prospective longitudinal observational study of RMS patients followed at the Verona MS Center.
Methods:
Clinical assessments, annual 3.0T brain MRI, and comprehensive neuropsychological testing were performed throughout follow-up. Treatment effectiveness was evaluated using no evidence of disease activity (NEDA), progression independent of relapse activity (PIRA), and cognitive PIRA. Safety was assessed by recording adverse events and treatment discontinuations.
Results:
Eighty-nine RMS patients (68.5% female, mean age 38.0 ± 10.4 years) were followed for a mean of 3 years. Overall, 91% achieved NEDA-3, with one relapse and one case of MRI activity. Seven patients (7.9%) developed PIRA, six of whom also fulfilled criteria for cognitive PIRA. Among 46 NEDA-3 patients who underwent longitudinal cognitive assessment, 37 (80.4%) did not exhibit cognitive worsening during follow-up and were classified as NEDA-4, indicating stability across clinical, radiological, and cognitive domains. In general, among patients with longitudinal cognitive data, 37 of 54 (68.5%) were NEDA-4. Naïve and previously treated patients showed comparable outcomes. Patients who failed to maintain NEDA-3 were older and had higher baseline disability, whereas in multivariable analyses, baseline Expanded Disability Status Scale (EDSS) was the only factor independently associated with NEDA-3 loss. When cognitive outcomes were included, associations between baseline clinical variables and NEDA-4 loss were attenuated. Ofatumumab was well-tolerated, with mostly mild transient injection-related reactions and 2 (2.2%) treatment discontinuations due to adverse events.
Conclusion:
Ofatumumab provided sustained multidimensional disease control, high NEDA-4 rates, and excellent tolerability. Baseline EDSS was the primary predictor of suboptimal response, underscoring the importance of early intervention. These findings support early anti-CD20 therapy as an effective strategy to preserve neurological function and limit long-term progression.
Introduction
Multiple sclerosis (MS) is a chronic, immune-mediated disorder of the central nervous system (CNS) characterized by inflammation, demyelination, and neurodegeneration, leading to progressive disability in young adults. 1 Although disease-modifying therapies (DMTs) have significantly improved disease control, the optimal treatment strategy remains debated. 2 Mounting evidence suggests that neurodegeneration in MS begins early, 3 even before overt clinical progression, and is driven by chronic compartmentalized inflammation and silent lesion activity.4,5 Delayed escalation therapy may, therefore, allow irreversible injury, whereas early, aggressive intervention may help preserve neurological reserve. Several studies have shown that early high-efficacy therapy (HET) is associated with lower relapse rates, reduced MRI activity, and slower disability accumulation compared to delayed treatment.6,7 However, despite this growing evidence, real-world practice, especially in Italy, still largely relies on an escalation paradigm, heavily conditioned by safety considerations and the current lack of long-term comparative evidence directly comparing these strategies. 8
Among the immunopathogenic mechanisms of MS, B lymphocytes play a central role in CNS inflammation and disease progression.9,10 The presence of oligoclonal bands at diagnosis highlights early intrathecal B-cell activation.9,11 B cells contribute to disease activity through antibody production, antigen presentation, and the release of pro-inflammatory mediators that amplify T-cell-driven immune responses.12 –14 Moreover, B cells represent a key component of meningeal tertiary lymphoid-like structures associated with cortical pathology and more aggressive disease courses.15,16 The persistent activity of B cells throughout the disease course may contribute to the accumulation of both anti-inflammatory mediators (such as IL-10 and TGFβ) and pro-inflammatory mediators (including). 17 The balance between these mediators is crucial in influencing neuroaxonal damage, either directly or indirectly by mediating alterations and activation of glial cells.
Ofatumumab is a fully human anti-CD20 monoclonal antibody that binds a distinct CD20 epitope, inducing a strong B-cell depletion through both complement-dependent and antibody-dependent cytotoxicity. 18 However, although randomized controlled trials provide essential efficacy and safety data, their selected populations may not fully reflect real-world clinical heterogeneity.19 –21 Moreover, clinical trials often overlook key dimensions of the MS framework, for example, progression independent of relapse activity (PIRA) and cognitive outcomes.22,23 Real-world evidence is, therefore, crucial to assess effectiveness and tolerability across broader patient groups, including individuals switching from other DMTs and patients at higher risk of reactivation after discontinuation of HETs such as natalizumab.
In this context, only a few recent observational studies provided evidence from real-world data on patients with ofatumumab: these findings highlight the need for additional observational studies to define whether an escalation strategy remains acceptable or whether early initiation of HET, such as ofatumumab, should be considered the new therapeutic standard.24,25 The present study aims to contribute to this discussion by providing real-world data from a single-center Italian MS cohort, focusing on treatment outcomes in naïve versus previously treated patients and evaluating the impact of ofatumumab on MRI activity, disability progression, and safety in everyday clinical practice.
The primary objective of this study was to evaluate the real-world effectiveness and safety of ofatumumab in patients with relapsing multiple sclerosis (RMS). Specifically, the study aimed to: (i) compare treatment outcomes between treatment-naïve and previously treated patients, focusing on disease activity and disability progression; (ii) assess the impact of ofatumumab on clinical outcomes (including relapses and disability progression) and radiological measures (MRI activity); and (iii) describe the frequency and severity of adverse events (AEs) in a real-world setting. Additionally, a secondary objective was to explore potential predictors of suboptimal response to better identify patient profiles that may benefit most from the early initiation of HET.
Methods
This prospective, longitudinal, observational study included patients with RMS treated with ofatumumab and followed at the Multiple Sclerosis Center of the Azienda Ospedaliera Universitaria Integrata in Verona, Italy. All consecutive patients with a diagnosis of RMS who initiated ofatumumab at our center between January and November 2022, were aged 18–65 years, and had a minimum clinical and radiological follow-up of 2 years, were included. Dropouts were registered in case of legitimate study abandonment or transfer to another MS center. Patients who discontinued ofatumumab for any clinical reason were included in the analyses throughout the study.
According to Italian Medicines Agency (AIFA) guidelines, treatment initiation was allowed in adult patients with RMS who met at least one of the following conditions: (i) evidence of high disease activity despite prior treatment with at least one DMT, defined by ongoing clinical relapses and/or MRI activity; or (ii) a severe, rapidly evolving disease course, defined by the occurrence of two or more disabling relapses within 1 year, together with the presence of one or more gadolinium-enhancing lesions on brain MRI or a significant increase in T2 lesion burden compared with a recent previous MRI examination. These criteria reflect current Italian real-world prescribing regulations for HETs (see Table 1).
Baseline demographic and clinical characteristics of the study population.

Treatment-naïve patients fulfilled the Italian MEdicines Agency (AIFA) reimbursement criteria for rapidly evolving severe relapsing multiple sclerosis, while previously treated patients met AIFA criteria for high disease activity despite prior disease-modifying therapy. All patients were therefore eligible for ofatumumab according to national prescribing regulations at the time of treatment initiation (see Methods section). Continuous variables are expressed as mean ± standard deviation (range), except for EDSS, which is presented as median (range). p-values < 0.05 are highlighted in bold.
EDSS, Expanded Disability Status Scale; NEDA, No Evidence of Disease Activity; RMS, relapsing multiple sclerosis.
Patients were evaluated at 6-month intervals through standardized neurological assessments, including determination of the Expanded Disability Status Scale (EDSS) by a certified rater. Brain MRI scans were acquired annually on a 3.0 Tesla scanner using a standardized protocol that included T1-weighted, T2-weighted/FLAIR, and post-contrast T1 sequences. All MRI scans were systematically reviewed to assess the presence of new or enlarging T2 lesions and contrast-enhancing lesions, which were used to define radiological disease activity. For a subsample of patients, comprehensive neuropsychological assessments were also performed throughout the entire follow-up period.
STROBE guidelines were consulted when preparing the manuscript, and a complete checklist has been submitted as Supplemental Material.
Data collection and variables
Baseline demographic and clinical data, including age, sex, disease duration, prior exposure to DMTs, and EDSS score, have been collected. Treatment history was categorized as treatment-naïve or previously treated based on prior DMT use.
Clinical and MRI outcomes were monitored throughout follow-up to evaluate treatment effectiveness, defined using the No Evidence of Disease Activity (NEDA-3) composite endpoint. NEDA-3 status was defined as the absence of clinical relapses, MRI activity (including new white matter or cortical lesions, CLs), and confirmed disability progression during the observation period.
A relapse was defined as the onset of new or recurrent neurological symptoms lasting ⩾ 24 hours, associated with new objective neurological findings and/or new radiological activity, and not attributable to fever or infection.
MRI activity was defined as the presence of new or enlarging T2-hyperintense lesions or gadolinium-enhancing lesions on brain or spinal MRI.
Disability progression was defined as a clinically meaningful increase in EDSS score, confirmed after at least 6 months, according to the following thresholds:
⩾1.5 points from a baseline EDSS = 0
⩾1.0 point from a baseline EDSS between 1.0 and 5.0
⩾0.5 points from a baseline EDSS ⩾ 5.5
The reference EDSS score was updated in case an increase in EDSS (or in any component of the composite measure) was observed and confirmed at the subsequent visit, or when a relapse resulted in residual disability.
Patients who fulfilled all three conditions (no relapses, no disability progression, and no MRI activity) were classified as NEDA-3 positive.
According to the standardized criteria proposed, PIRA was defined as a confirmed increase in disability occurring in the absence of any evidence of disease activity, either clinical or radiological. 26 Specifically, patients were classified as having PIRA when disability progression occurred without a relapse within the interval from 30 days before to 90 days after the onset of progression and without MRI evidence of new or enlarging T2 lesions or gadolinium-enhancing lesions within 180 days before or after the progression event.
Neuropsychological assessment
Cognitive functioning was assessed at ofatumumab initiation and throughout the entire follow-up period using the Brief Repeatable Battery of Neuropsychological Tests and the Stroop Test. 27 Neuropsychological assessments were performed when feasible as part of routine clinical care. Cognitive testing is not systematically conducted in all patients at our center due to limited personnel resources and organizational constraints, and patient willingness to undergo formal evaluation also contributed to missing data.
Patients were classified into three groups based on their performance across all neuropsychological tests: either cognitively normal (CN, 0 failed subtests) or cognitively impaired (CI), subdivided into mildly cognitively impaired (mCI, one or 2 failed subtests) and severely cognitively impaired (sCI, 3 or more failed subtests). 28
The occurrence of cognitive PIRA was also evaluated using a conservative approach, considering a significant cognitive decline (as determined by a reliable change index methodology) in the Symbol Digit Modalities Test (SDMT) that occurred in the absence of relapse activity 9 months before/after the cognitive decline. 29 To ensure identification of cognitive progression independent of acute inflammatory activity, we focused on those patients without evidence of MRI activity in the same period of time, in addition to no evidence of clinical relapses. 30 Cognitive decline was defined using a sliding window approach, in which cognitive performance at each observation was re-baselined relative to the function measured at the preceding assessment.
We, therefore, arbitrarily decided to consider all patients who fulfilled NEDA-3 criteria (absence of relapses, disability progression, and MRI activity) and did not exhibit evidence of cognitive PIRA as achieving NEDA-4, indicating no disease worsening across clinical, radiological, and cognitive domains.
MRI protocol
The following image sets were acquired:
(a) 3D T1weighted magnetization prepared rapid gradient echo with Repetition Time (TR) = 8.4 ms, Echo Time (TE) = 3.7 ms, voxel size = 1 × 1 × 1 mm3, field of view (FOV) = 240 × 240 × 180, number of excitations (NEX) = 1;
(b) 3D Fluid Attenuated Inversion Recovery (FLAIR) with TR/TE = 8000/292 ms, Inversion Times (TI) = 2350 ms, voxel size = 1 × 1 × 1 mm3, FOV = 240 × 240 × 180, NEX = 1;
(c) 3D Double Inversion Recovery with TR/TE = 5500/292 ms, TI = 2550 ms, delay 450 ms, voxel size = 1 × 1 × 1 mm3, FOV = 240 × 240 × 180, NEX = 3;
(d) 3D-T1 weighted TFE post-contrast with the same parameters as the pre-contrast sequence (acquisition 5:51 min).
All images were quality-checked for scanner inhomogeneities and artifacts.
MRI examinations were reviewed for evidence of acute inflammatory activity, including new T2 or CLs, Gd+ lesions, and enlarging T2 lesions.
Safety assessment
AEs were recorded at each visit and categorized by type (injection-related reactions, infections, and malignancies), severity, and relationship to treatment. The need for treatment discontinuation was also documented, specifying whether it was due to AEs, MRI activity, or clinical progression.
Statistical analysis
Given the observational and real-world nature of the study, no formal sample size calculation or a priori power analysis was performed. The study population comprised all consecutive patients treated with ofatumumab at our center during the study period. Patients who discontinued treatment contributed available data up to the time of discontinuation and were not excluded from outcome analyses. No imputation methods were applied.
Continuous variables are reported as mean ± standard deviation (SD) for normally distributed variables and as median (range) for ordinal or non-normally distributed variables. Normality was assessed using the Shapiro–Wilk test and visual inspection of histograms/Q–Q plots. Group comparisons were performed using Welch’s t-test for normally distributed continuous variables, the Mann–Whitney U test for non-normal or ordinal variables (including EDSS), and Fisher’s exact test for categorical variables.
Exploratory logistic regression analyses were performed to evaluate baseline factors associated with loss of NEDA status. Separate models were constructed for NEDA-3 loss and NEDA-4 loss. Covariates considered included age, sex, disease duration, baseline EDSS, and previous treatment status. Given the limited number of outcome events, multivariable regression analyses were considered exploratory and interpreted accordingly.
Because NEDA-4 loss is a time-dependent outcome, time-to-event analyses were also performed. Kaplan–Meier curves were used to illustrate time to NEDA-3 and NEDA-4 loss, with time zero defined as the date of ofatumumab initiation and censoring set at 48 months for patients without events.
In addition, Cox proportional hazards models were applied to assess the association between baseline factors and time to NEDA-4 loss. A parsimonious multivariable Cox model including baseline EDSS, age, and previous treatment status was performed. More complex multivariable Cox models were not fitted due to the limited number of NEDA-4 loss events.
NEDA-4 analyses were restricted to patients with available longitudinal cognitive assessments; patients without cognitive follow-up were considered not evaluable for NEDA-4 and were excluded from NEDA-4 denominators.
Statistical significance was defined as a two-sided p-value < 0.05. All analyses were conducted using SPSS (version 30.0; IBM Corp., Armonk, NY, USA).
Results
Demographic and baseline characteristics
A total of 89 patients with RMS were included in the analysis (68.5% females), with a mean baseline age of 38.0 ± 10.4 years (range, 20 – 62 years) and a mean baseline disease duration of 9.4 ± 7.4 years (range, 1 – 21 years). The median baseline EDSS was 2.0 (range 0.0 – 6.5). The mean follow-up duration after initiation of ofatumumab was 3.0 ± 0.8 years (range, 2.0 – 4.0 years). No dropouts were registered. Details are reported in Table 1.
Naïve versus previously treated patients
Of the total cohort, 24 (27.0%) were treatment-naïve, 51 (57.3%) had received one prior DMT, and 14 (15.7%) two or more DMTs. All patients had evidence of disease activity within the year preceding treatment initiation. Specifically, according to national guidelines, all naïve patients had experienced at least two markers of disease activity (clinical relapse or new MRI lesions), while previously treated patients had shown at least one sign of disease reactivation.
When stratified by treatment history, naïve patients (n = 24) and previously treated patients (n = 65) showed comparable distribution of age, sex, baseline EDSS, and short-term disability change (all p > 0.4; see Table 1). As expected, previously treated patients had a significantly longer disease duration than naïve patients (10.7 ± 7.0 vs 5.6 ± 7.2 years; p = 0.003). The proportion achieving NEDA-3 was high in both groups (92.3% vs 87.5%; p = 0.68), with comparable relapse occurrence and MRI activity (both 1/89, 1.1%) (Table 1).
Clinical and radiological outcomes—NEDA-3
Across the entire cohort, 81 patients (91.0%) achieved NEDA-3 during follow-up (Figure 1(a)): according to the definition, they did not show any clinical (relapses or EDSS progression) or radiological activity (new WM or CLs). Only one patient (1.1%) experienced a clinical relapse accompanied by MRI activity, characterized by the development of new T2 lesions. Additionally, seven patients (7.9%) demonstrated progression independent of relapse and of MRI activity. No new CLs were detected. Group-level disability remained globally stable (ΔEDSS = 0; range −3.0 to +2.0). Five patients (5.6%) discontinued ofatumumab: two due to AEs (see below), one for the occurrence of new MRI lesions, and two due to PIRA.

Kaplan–Meier estimates for NEDA maintenance. (Panel a): NEDA-3: Kaplan–Meier curve showing the proportion of patients maintaining NEDA-3 over time. Time zero corresponds to ofatumumab initiation. Loss of NEDA-3 was defined as the event, and patients without events were censored at 48 months. Numbers at risk are shown below the curve at 0, 12, 24, 36, and 48 months. (Panel b): NEDA-4: Kaplan–Meier curve showing the proportion of patients maintaining NEDA-4 over time among patients with available longitudinal cognitive assessment. Time zero corresponds to ofatumumab initiation. Loss of NEDA-4 was defined as the event, and patients without events were censored at 48 months. Numbers at risk are shown below the curve at 0, 12, 24, 36, and 48 months.
Clinical and radiological outcomes—NEDA-4
At treatment initiation, 74 patients underwent a comprehensive neuropsychological assessment. Fifty-five patients (74.3%) were classified as CN, while 19 (25.7%) showed cognitive impairment (CI), including 16 with mild and 3 with severe impairment. During follow-up, 54 patients were re-evaluated from a cognitive perspective. Among them, 44 were classified as CN and 10 as CI (8 mild and 2 severe). When comparing individual trajectories, 45 patients (83.3%) remained cognitively stable, 8 (14.8%) changed from CI to CN, and only 1 (1.9%) worsened from CN to CI.
Overall, seven patients (13.0%) exhibited a meaningful cognitive decline during follow-up. Of these, one patient showed cognitive deterioration associated with both a clinical relapse and new MRI activity, while six fulfilled criteria for cognitive PIRA. Notably, these six patients with cognitive PIRA were also included in the overall PIRA group, as their “mental” functional system score increased by at least one point, thereby meeting the established criteria for PIRA.
Importantly, among the 54 patients with available longitudinal cognitive data, 46 (85.2%) were NEDA-3, while 8 (14.8%) showed clinical and radiological disease activity. Among 46 NEDA-3, 37 (80.4%) maintained NEDA-3 status and also did not exhibit cognitive worsening during follow-up and were, therefore, classified as achieving NEDA-4, encompassing stability across clinical, radiological, and cognitive domains. In general, among patients with available longitudinal cognitive assessment, 37 of 54 patients (68.5%) maintained NEDA-4 (Figure 1(a) and (b)).
Predictors of suboptimal response
Demographic and clinical characteristics according to NEDA status are reported in Table 2.
Demographic and clinical characteristics according to NEDA-4 status.

Continuous variables are expressed as mean ± standard deviation (range), except for EDSS, which is presented as median (range). p-values < 0.05 are highlighted in bold.
EDSS, Expanded Disability Status Scale; NEDA, No Evidence of Disease Activity.
When considering NEDA-3 as the outcome, patients who failed to maintain NEDA-3 were older than those who maintained NEDA-3 (47.7 ± 5.2 vs 39.3 ± 10.4 years, p = 0.002) and showed higher baseline disability, as reflected by a higher median EDSS score (4.5 (range 3.0–6.0) vs 2.0 (0–6.5), p < 0.001). No significant differences were observed between NEDA-3 groups with respect to sex distribution, disease duration, or prior exposure to DMTs.
When NEDA-4 was used as the outcome, age was no longer significantly different between groups (41.4 ± 8.6 vs 38.7 ± 10.3 years, p = 0.36). Patients who failed to maintain NEDA-4 exhibited higher baseline disability compared with those who maintained NEDA-4 (median EDSS 3.0 (0–6.5) vs 2.0 (0–6.5), p = 0.018). No significant differences were observed in sex distribution, disease duration, or prior treatment exposure, although a qualitatively higher proportion of previously treated patients maintained NEDA4.
Baseline EDSS values according to NEDA status are shown in Figure 2. Patients who lost NEDA-3 had higher baseline EDSS scores than those who maintained NEDA-3 (Figure 2(a)). Exploratory logistic regression illustrated an increasing estimated probability of NEDA-3 loss with increasing baseline EDSS (Figure 2(b)).
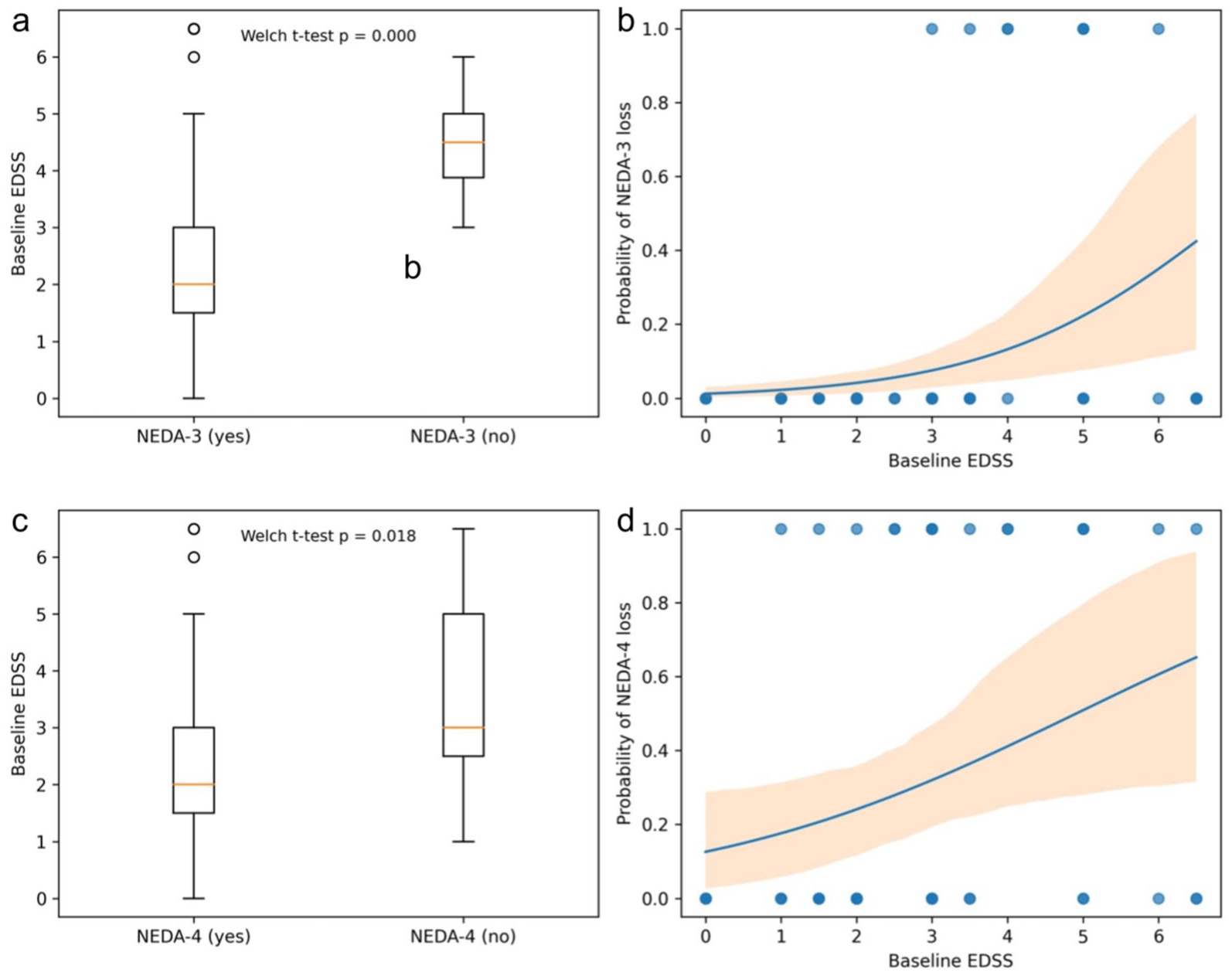
Baseline EDSS and probability of NEDA loss. Relationship between baseline EDSS and NEDA-3 status (figure above) and between baseline EDSS and NEDA-4 status (figure below). (Panel a): Distribution of baseline EDSS values according to NEDA-3 status (Yes vs No). Group comparison is shown for descriptive purposes. Welch’s t-test is reported within the plot. (Panel b): Exploratory logistic regression analysis showing the relationship between baseline EDSS and the probability of NEDA-3 loss (NEDA-3 No = 1). Each dot represents an individual patient. The solid line indicates the fitted logistic model, and the shaded area represents the 95% confidence interval. Higher baseline EDSS values were associated with an increased probability of NEDA-3 loss, suggesting that patients with greater baseline disability are less likely to maintain complete disease control under treatment. Given the limited number of NEDA-3 loss events, this analysis should be interpreted as exploratory. (Panel c): Distribution of baseline EDSS values according to NEDA-4 status (Yes vs No). Group comparison is shown for descriptive purposes. Welch’s t-test is reported within the plot. (Panel d): Exploratory logistic regression analysis showing the relationship between baseline EDSS and the probability of NEDA-4 loss (NEDA-4 No = 1). Each dot represents an individual patient. The solid line indicates the fitted logistic model, and the shaded area represents the 95% confidence interval. Higher baseline EDSS values were associated with an increased probability of NEDA-4 loss, suggesting that patients with greater baseline disability are less likely to maintain complete disease control under treatment.
Similarly, patients who lost NEDA-4 had higher baseline EDSS values than those who maintained NEDA-4 (Figure 2(c)). Exploratory logistic regression showed an increasing estimated probability of NEDA-4 loss with higher baseline EDSS (Figure 2(d)).
Results of exploratory multivariable logistic regression analyses are shown in Figure 3. In the multivariable model for NEDA-3 loss, baseline EDSS was associated with increased odds of NEDA-3 loss (OR = 1.85 per EDSS point, p = 0.027), whereas age, sex, disease duration, and prior treatment were not associated (Figure 3(a)). The overall model showed acceptable goodness-of-fit (AIC = 53.9; Nagelkerke R2 = 0.28).

Multivariable analysis of factors associated with NEDA loss. Forest plot of odds ratios (ORs) and 95% confidence intervals derived from an exploratory multivariable logistic regression model including baseline EDSS, age, sex, disease duration, and previous treatment as predictors of NEDA-3 loss (NEDA-3 No = 1; Panel a) and NEDA-4 loss (NEDA-4 No = 1; Panel b). The dashed vertical red line represents the null value (OR = 1). Among all variables, baseline EDSS was the only independent predictor of NEDA-3 loss (p = 0.027).
In the multivariable model for NEDA-4 loss, none of the variables included were statistically significant (AIC = 70.3; Nagelkerke R2 = 0.19) (Figure 3(b)).
In an exploratory multivariable Cox proportional hazards model including baseline EDSS, age, and previous treatment, higher baseline EDSS was associated with a higher hazard of NEDA-4 loss (HR 1.35 per EDSS point, 95% CI 1.00–1.83; p = 0.047), whereas age (HR 0.99, 95% CI 0.94–1.05; p = 0.756) and previous treatment (HR 0.44, 95% CI 0.16–1.21; p = 0.114) were not statistically significant.
AEs and treatment tolerability
Ofatumumab was generally well-tolerated. The most common AE was a transient febrile reaction after the first injection, occurring in 58 patients (65.2%), and after the second administration in 11 patients (12.4%); no further episodes occurred thereafter. All were mild and managed with antipyretics. One patient experienced nausea and vomiting after the first injection. Another patient reported transient vertigo and headache, both of which resolved within 24 h.
Furthermore, treatment was discontinued in two patients, in the first case due to a flare-up of psoriasis, while in the second case due to an atypical nevus with suspected melanoma in situ. Additionally, one patient developed mild injection-site erythema only after the first injection. No hospitalizations or severe systemic reactions occurred, confirming a favorable safety profile over the longitudinal follow-up.
Discussion
This prospective longitudinal study with a 3-year follow-up provides real-world evidence of the sustained efficacy of ofatumumab in patients with relapsing MS. Our findings demonstrate robust treatment effectiveness across clinical, radiological, and cognitive domains, extending the results of pivotal randomized controlled trials into routine clinical practice. After 3 years of continuous treatment, most patients were stable, with 91% achieving NEDA-3 and nearly all showing stable disability and minimal MRI activity. When cognitive outcomes were incorporated, most patients also met the criteria for NEDA-4, indicating preservation of cognitive function alongside clinical and radiological control. Together, these findings support the sustained effectiveness and favorable tolerability profile of continuous B-cell depletion with ofatumumab over long-term follow-up in a heterogeneous real-world population including both treatment-naïve and previously treated patients. 31
The observed NEDA-3 rate of approximately 91% over a 3-year follow-up is higher than that reported in extension phases of randomized controlled trials and in most previously published real-world cohorts. This finding should be interpreted in light of several factors. First, patient selection was driven by AIFA reimbursement criteria, which restrict the use of ofatumumab to patients with either rapidly evolving severe disease or high disease activity despite prior treatment. As a result, both treatment-naïve and previously treated patients were characterized by a high inflammatory burden at baseline but were treated early with an HET. Second, the consistent and early use of ofatumumab in a single tertiary MS center, combined with standardized MRI monitoring and close clinical follow-up, may have contributed to optimized disease control. Third, although treatment discontinuations occurred, patients were not excluded from the analyses and contributed data up to the time of discontinuation, partially mitigating survivorship bias.
Finally, NEDA-4 was assessed only in patients with longitudinal cognitive follow-up, without imputing missing data. This methodological aspect should be taken into account when interpreting NEDA-4 rates and comparing results across studies.
Our results are broadly consistent with previously published real-world outcomes from large multicenter cohorts. In an Italian multicenter study, ofatumumab showed high effectiveness and good tolerability in both treatment-naïve patients and switchers, including those transitioning from natalizumab, with relapse-free and MRI activity-free rates exceeding 85% at 12 months. 24 Similarly, a recent Polish multicenter real-world study reported favorable short-term outcomes, with NEDA-3 achieved in approximately 76% of patients at 12 months. 25 Together, these studies confirmed the robust short-term efficacy and safety of ofatumumab in routine clinical practice, as well as the convenience in maintaining long-term disease control.
Compared with these multicentric analyses, our study provides complementary evidence from a highly homogeneous single-center cohort. All patients were evaluated by the same neurologist, underwent MRI examinations using a standardized protocol on the same 3-Tesla scanner, with images consistently interpreted by the same neuroradiologist, and neuropsychological assessments performed by the same neuropsychologist. This high level of methodological uniformity minimizes inter-observer and inter-instrument variability and allows a more reliable assessment of longitudinal clinical, radiological, and cognitive changes over a longer follow-up period.
Beyond confirming the clinical effectiveness of ofatumumab, our findings support its role within current therapeutic strategies in MS, where early use of HETs is increasingly recognized as a key determinant of long-term outcomes. 7 Recent advances in MS diagnostics, aimed at enabling earlier disease recognition and treatment initiation, together with the central role of B-cell-driven inflammation, highlighted by the early presence of oligoclonal bands, further support the rationale for prompt intervention with highly effective targeted therapies. 32 In this context, our real-world data add supportive evidence that early and sustained B-cell depletion with ofatumumab can achieve durable disease stability in routine clinical practice.
Within this framework, our data provide exploratory insights into which patients may be at higher risk of a suboptimal response despite treatment with an HET. In an exploratory multivariable logistic regression analysis, higher baseline EDSS was the only variable associated with failure to achieve NEDA after adjustment for age, disease duration, sex, MRI activity, and prior treatment exposure. However, given the low number of outcome events, this finding should be interpreted with caution and considered hypothesis-generating rather than confirmatory. Nevertheless, they are consistent with previous large-scale studies indicating that baseline disability, together with age, represents a major determinant of subsequent disease activity and relapse risk over long-term follow-up. 33
Notably, a qualitative review of clinical trajectories revealed that the main driver of suboptimal response in this subgroup was PIRA, characterized by a gradual worsening of disability unrelated to overt relapses or new MRI lesions, 22 despite HET showing a lower rate of PIRA than low-to-moderate efficacy treatments. 34 This observation aligns with the growing understanding that PIRA reflects ongoing, compartmentalized inflammation and neurodegeneration, processes that are less responsive to peripheral immunosuppression once established. Consequently, the reduced likelihood of achieving NEDA-4 in patients with higher EDSS at baseline likely reflects the presence of this smoldering, relapse-independent disease component. These results reinforce the concept that timing of treatment initiation is crucial, as delaying the introduction of HET may allow PIRA mechanisms to emerge and limit the reversibility of tissue damage. 35 Early initiation of B-cell-depleting therapy, by contrast, may not only suppress focal inflammatory activity but also indirectly mitigate chronic intrathecal inflammation, including possibly B-cell accumulation in meningeal tertiary lymphoid structures, and reduce the long-term risk of progression.31,36 It is worth mentioning that early anti-CD20 treatments may also contribute to the elimination of CD20+ T cells, 37 even though their role in MS remains unclear.
Importantly, the strong NEDA rates among younger patients and those with lower baseline disability reinforce the idea that early B-cell depletion can maximize clinical benefit. 38 Initiating anti-CD 20 treatment at an earlier disease stage may not only suppress focal inflammatory activity but also delay the emergence of PIRA, by preventing the establishment of compartmentalized inflammation and irreversible neuroaxonal loss. 20
It is also important to consider the cognitive outcomes observed in our cohort. Overall, the majority of patients with available longitudinal neuropsychological assessments showed no significant change in cognitive status over the observational period, indicating a general stability of cognitive performance during ofatumumab treatment. A limited number of patients showed a change in dichotomous cognitive classification from “impaired” to “normal”: however, such changes should be interpreted with caution, as they may reflect learning/practice effects, regression to the mean, or fluctuations in inflammatory activity rather than real cognitive improvement.39 –41 Moreover, the use of a dichotomous classification approach inherently limits the ability to capture subtle longitudinal changes in cognitive functioning. 42
This is true not only for cognitive status but also for cognitive decline: in particular, only a limited number of patients experienced cognitive progression independent of relapse and MRI activity, with a lower prevalence than in previous studies.29,30,43,44 Therefore, ofatumumab could not only play a role in the prevention of clinical PIRA but also of cognitive PIRA. This is noteworthy considering that a cognitive PIRA is highly frequent in MS patients and represents the major cause of cognitive decline in MS patients.29,30,43,44 Moreover, cognitive PIRA often occurs also independently from MRI activity and from physical disability worsening:29,30,43,44 the results of the present study demonstrating no significant differences between NEDA-4 results and those adding cognitive progression advocate in favor of the overall efficacy of ofatumumab considering multiple domains of patients.
These findings prompt further consideration of the NEDA concept, particularly when cognitive outcomes are considered. In our cohort, a subset of patients classified as having disease activity according to the NEDA-4 definition did not exhibit overt clinical relapses or new MRI lesions but did show cognitive worsening detectable only through neuropsychological assessment. Conversely, a proportion of patients who met NEDA-3 criteria demonstrated evidence of disease activity when cognitive outcomes were considered, underscoring the added sensitivity of incorporating cognition into disease activity assessment. 45
These observations highlight that disease activity may remain clinically silent on neurological and radiological grounds while still affecting cognitive functioning, which can only be identified through a structured neuropsychological evaluation. This finding underscores the importance of a multidisciplinary clinical approach integrating neurological examination, MRI, and cognitive assessment to more comprehensively capture disease activity in MS. 45
The relationship between baseline disability and disease control varied depending on the NEDA definition applied. While a higher baseline EDSS was associated with loss of NEDA-3 also in multivariable analyses, this association was attenuated when the more stringent NEDA-4 definition was considered. This difference likely reflects variations in endpoint composition and event rates, as NEDA-4 incorporates cognitive stability in addition to clinical and radiological measures.
Exploratory analyses further showed that baseline EDSS was associated with an increased estimated probability of NEDA-4 loss in univariable models, although this association did not consistently retain statistical significance after multivariable adjustment. This attenuation is consistent with the smaller evaluable sample size for NEDA-4 and the limited number of outcome events, which reduces the power of multivariable modeling.
Taken together, these findings suggest that baseline disability more consistently captures vulnerability to loss of clinical–radiological disease control than to failure of the broader NEDA-4 construct. They also underscore the importance of endpoint definition when evaluating predictors of treatment response. Given the observational design and exploratory nature of these analyses, confirmation in larger cohorts with systematic cognitive follow-up is warranted.
The high overall tolerability and very low discontinuation rate observed in our cohort support the use of ofatumumab as a front-line option in appropriate patients, balancing efficacy, safety, and patient convenience. These findings are consistent with the most recent 7-year extension data presented at the ECTRIMS 2025 congress, which confirmed sustained efficacy and a stable safety profile of continuous ofatumumab treatment over long-term follow-up.46,47 Although in these studies using data from clinical trials, the injection-related reactions occurred more rarely compared to what we found in the present study (26% compared to 65%), our prevalence of these AE is in line with previous real-word conducted which we discussed above: in the Italian multicentric cohort, the prevalence of fever after the first administration was 72%, and in the Polish study, the prevalence of injection-related reactions was 63%.24,25 Together, these real-world and extension data further consolidate the role of ofatumumab as a well-tolerated, durable, and patient-friendly HET for RMS.
This single-center, observational study has several limitations. While the single-center design represents a methodological strength, ensuring uniform neurological, neuroradiological, and neuropsychological assessments without inter-rater variability, it also introduces potential sources of bias that should be acknowledged.
The study was conducted in a tertiary MS center and included a population with relevant disease activity; however, the early initiation of HET and the availability of home-administered ofatumumab may have favored the inclusion of patients who were clinically stable enough to adhere to self-administration. As a result, the generalizability of our findings to other clinical settings and to patients with more advanced disability or different care pathways may be limited.
In addition, the sample size and the low number of NEDA-3 loss events limit the statistical power of multivariable analyses and increase the risk of model overfitting. Accordingly, results from regression analyses should be interpreted as exploratory and hypothesis-generating and require confirmation in larger, independent cohorts. Given the limited number of events, the interpretative values of Kaplan–Meier analyses are also constrained, and survival curves should, therefore, be considered descriptive rather than inferential.
Third, some missing data in the cognitive cohort may introduce selection bias. Finally, MRI assessments, although performed on the same 3T scanner by experienced readers, would benefit from external validation in multicenter datasets. A future multidisciplinary and longitudinal research approach may help clarify whether early anti-CD20 therapy compared to late treatment facilitates the fastest and deepest B-cell depletion from the CSF and CNS in MS.
Conclusion
In this real-world, single-center cohort, treatment with ofatumumab was associated with sustained disease control over a 3-year follow-up, with high rates of NEDA-3, low relapse activity, and overall stability of disability. When cognitive outcomes were considered, a substantial proportion of patients with available longitudinal assessments also maintained NEDA-4, highlighting the relevance of comprehensive outcome evaluation in routine clinical practice.
Exploratory analyses suggested an association between baseline disability and loss of disease control, particularly when using clinical–radiological endpoints, while this relationship was attenuated when a more stringent definition incorporating cognitive stability was applied. These findings underscore the importance of early and effective disease control and support the role of continuous anti-CD20 therapy as a valuable option in the management of RMS.
Together with accumulating real-world evidence, our results support the feasibility and long-term effectiveness of sustained B-cell depletion with ofatumumab in routine clinical practice. Larger studies with systematic and comprehensive follow-up are warranted to further clarify predictors of long-term disease control and progression.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261434351 – Supplemental material for Real-world effectiveness and safety of ofatumumab in multiple sclerosis: a longitudinal study integrating clinical, cognitive, and MRI outcomes
Supplemental material, sj-docx-1-tan-10.1177_17562864261434351 for Real-world effectiveness and safety of ofatumumab in multiple sclerosis: a longitudinal study integrating clinical, cognitive, and MRI outcomes by Stefano Ziccardi, Damiano Marastoni, Pietro Biasi, Agnese Tamanti, Francesca Benedetta Pizzini, Milena Calderone, Valentina Camera, Maddalena Bertolazzo, Alice Daldosso, Bianca Milocco, Roberta Magliozzi and Massimiliano Calabrese in Therapeutic Advances in Neurological Disorders
