Abstract
T cells, genetically modified to express chimeric antigen receptors (CAR T), successfully used in hemato-oncologic malignancies, are showing promising and sustained benefits in refractory autoimmune neurological diseases, including Myasthenia Gravis, stiff-person syndrome, neuromyelitis optica, myositis, autoimmune neuropathies, and multiple sclerosis. Several reported patients with a steadily progressive disease and evolving disability unresponsive to available therapies, including rituximab and new biologics, after 2–3 months of treatment with CARs targeting CD19-positive, antibody-secreting, long-lived plasma cells, and plasmablasts exhibit impressive, long-lasting, and drug-free clinical improvements with the potential for immune reset shifting immunity to a healthy state without the need for continuing more immunotherapy cycles. The review discusses what the unmet needs are with the present neuroimmunotherapeutics pointing out the disease stage and patient subsets for which CAR T-therapy is most suitable highlighting that CAR T should be applied in the early stages of disability development when patients reach early-active/refractory status rather than waiting for very late disease progression when neurological deficits might be irreversible. The future trajectory of CAR T cells is also described as a promising means destined to change the present therapeutic algorithm in all neuro-autoimmunites, even offering a promising path toward a cure, pointing out that, in contrast to currently approved biologics that selectively target one immunoregulatory factor, CD19 CAR T cells exert effects even beyond B cells, cross the blood–brain barrier and lymphoid tissues, and expand as “living cells” to memory cells ensuring sustained long-term benefits. Key therapeutic uncertainties and practicalities are however highlighted, including the exact duration of CAR T-cell therapy-induced drug-free remissions, logistical challenges, economic limitations, and the need for extensive collaborative efforts with experts in specialized clinical centers.
Plain language summary
Because at least 20-25% of patients with common autoimmune neurological diseases, like Myasthenia Gravis, Stiff-Person Syndrome, Neuromyelitis, Myositis, Chronic Autoimmune neuropathies or Multiple Sclerosis, do not adequately respond or have stopped responding to current therapies exhibiting progressive or permanent disability, there is a need for novel therapeutic approaches. For such patients with refractory disease, CAR T cells can reverse the ongoing cumulative disability offering even a path toward a cure, without the need for continuing immunotherapy cycles; CAR T-therapies exert these actions because they can travel within the brain and the lymph nodes being able to modify the disease-specific lymphocytes responsible for the immune process and, most importantly, can expand as “living cells” ensuring their long-term benefits. The study discusses how these unique properties of CAR T cells can change the future treatment armamentarium in patients with autoimmune neurological diseases poorly responding to available therapies; describes the most suitable timing CAR T cells should be used after disease progression has begun; and highlights certain current uncertainties, such as the exact duration of CAR T-induced remissions, and economic or logistical challenges.
Keywords
Introduction
Based on controlled studies and clinical experience, patients with chronic autoimmune neurological diseases, such as Myasthenia Gravis (MG), Lambert–Eaton myasthenic syndrome (LEMS). Chronic inflammatory demyelinating polyneuropathy (CIDP), neuromyelitis optica spectrum disorder (NMOSD), Stiff person syndrome (SPS), progressive multiple sclerosis (MS), and inflammatory myopathies, respond to various immunotherapies for a period of time but in at least 20%–25% of them, the benefit is insufficient from the outset or declines over time due to disease progression. This applies not only to the currently used chemotherapeutic or immunomodulating agents and procedures such as IVIg, PLEX, or bone marrow transplantation, but also to the new biologics and monoclonal antibodies including those targeting B cells, complement, neonatal FcRn receptor (FcRn), or cytokines.1–3 As a result, despite the unprecedented progress we have been witnessing even with these novel immunotherapeutics, some patients not only require continuous cycling therapies, but they also remain poor responders with a steadily worsening symptomatology and cumulative disability. It is for these patient subsets that chimeric antigen receptors T cells (CAR T) therapies, by exerting global immune effects beyond targeting just one immunobiologic factor, as the current biologic agents do, appear highly promising not only in arresting disease progression but also in reversing disability even up to normalcy, as discussed for several autoimmune neurological diseases in the special series of TAND dedicated to CAR T.4–7
The present review is aimed to address the unmet treatment needs of autoimmune neurological diseases, based on currently applied immunotherapies, stressing for which patient subsets CAR T is most applicable and why CAR T is promising to change the therapeutic algorithm or even lead to a cure, by accomplishing an immune reset. It also highlights the present practical challenges, economic limitations, or uncertainties that impact the wide application of CAR T therapy in autoimmune neurology, while pointing out their very promising future trajectory.
Unmet needs of present neuroimmunotherapies and the concept of “active/refractory disease” status for which CAR T therapy is best applicable
Despite the revolutionary effects of the current or novel biologic therapeutics that offer undisputed benefits in all common autoimmune neurological diseases by successfully targeting key immunobiologic factors, there are several unmet needs that preclude a substantial number of patient subsets from feeling of normalcy most of the time. As highlighted below, the main unmet therapeutic needs relate to variable efficacy of these drugs, their cyclical administration pattern with “wearing off” effects after each cycle due to lack of long-term effectiveness, recurrent side effects, or poor tolerance repeatedly after each injection and, most importantly, their overall inadequacy in preventing disease progression in several patient subsets. These needs include the recognition of the following:
No efficacy or poor tolerance from the outset
Based on controlled trials with the present FDA-approved drugs, at least 20% of all patients may not exhibit substantial benefit from the outset or experience signs of intolerance, leading to early discontinuation.
Partial or declining efficacy
Although short- and long-term response to immunotherapies differs among various autoimmune neurologic diseases, like long-term remission achieved with B-cell-depleting therapies in several MS patients, compared to short-term or variable effects seen with many of the approved drugs in myasthenia or NMOSD, most patients across the autoimmune neurology spectrum initially respond fully or partially to approved immunotherapies, but in a number of them over the long course of the disease, usually 1–2 years after induction/maintenance therapy, their therapeutic efficacy starts to decline, as we have characteristically witnessed with IVIg maintenance therapy in a longitudinal study of patients with SPS. 8 Such partially effective agents, often given weekly, bi-weekly, monthly, or in 3–6 monthly patterns, not only cannot normalize the patient symptoms after several subsequent treatment cycles but cannot also fully halt disease progression; as a result, a number of patients exhibit an on/off therapeutic effect with some residual deficits after each immunotherapeutic cyclic period that gradually accumulate leading to early-onset, progressive disability. A typical therapeutic pattern witnessed in several currently treated patient subsets, is highlighted in Figure 1: after an initial improvement, during the first 6–24 months of treatment cycles either with IVIg, inhibitors of FcRn or complement, and with anti-B cells agents such as rituximab, ocrelizumab, or ofatumumab (A), there is a residual deficit during the off (maintenance) cycles (a); in the subsequent therapeutic cyclic periods (B → D) however, there is a partial and steadily declining improvement associated with declining residual benefits during the off (maintenance) dosing cycles (b → d), leading to increased disability over a 2–5-year period. 2 This therapeutic pattern that we refer to as “active/refractory” disease, is the most suitable for CAR T therapy, because the disease is “active,” as witnessed by a partial improvement, but also “refractory” because the deficit is not reversed but gradually is building up.
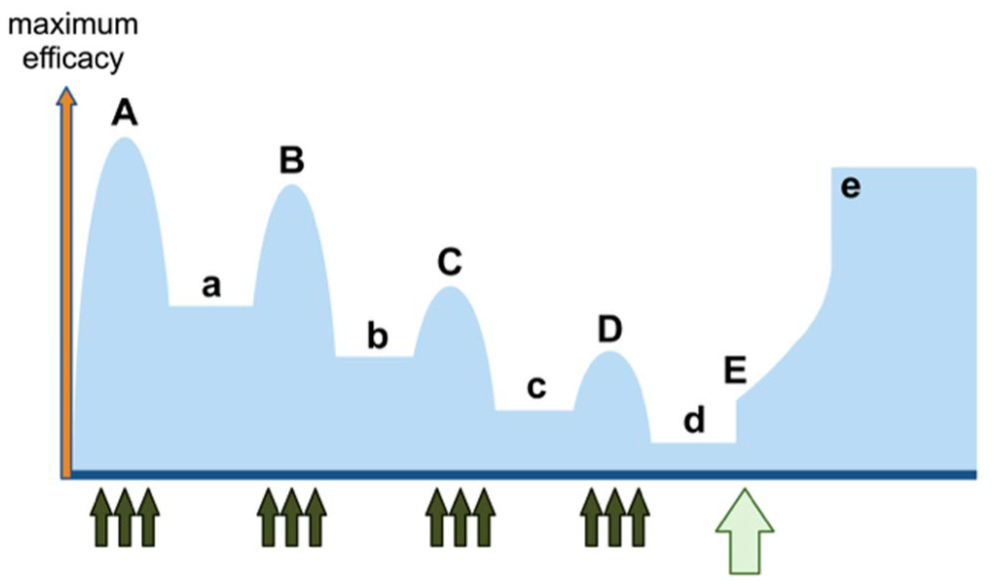
Therapeutic patterns of immunotherapy cycles in patients with late - “active/refractory” autoimmune neurological disease, relevant to anti-CD19 CAR T-therapy. (A) After the initial improvement with therapeutic cycling dosing (usually after the first 2–5 years), there is also partial residual benefit (a) during the off cycles. (B–D) Follow-up therapies continue with partial improvement but also steadily declining benefits with subsequent therapeutic dosing cycles with a steadily progressive decline of benefit between the maintenance cycling dosing (a => d). (E) Effects of initiating CAR T-cell therapy in patients with late-refractory but still active disease; even if late in disease progression, CAR T can reverse the decline and lead to steady improvement but not always reaching normalcy levels (e).
Early identification of the “active/refractory disease status”
To prevent the development of permanent disability and stop early the disease progression with expectations for a better therapeutic response, it is fundamental to recognize early in the disease course the time patients experience declining benefits with enhancing disability, while entering into an “active/refractory” status, for a timelier therapeutic intervention, as highlighted in Figure 2(A, B), rather than waiting longer as commonly practiced today due to lack of currently effective therapies, as depicted in Figure 1(A–D). 2 Although as we are learning from the ongoing studies, CAR T therapies can be effective in reversing disability even in some patients with late “active/refractory” stage (E, Figure 1), they have the potential to fully reverse disability progression, reaching even normal status, if applied earlier (C, Figure 2) because in the early “active/refractory” stage the CAR T-cells, by inducing an immune reset, provide the possibility not only of reversing disease progression but also leading fast toward a steady improvement without “wearing-off” effects or the need for repeated infusions, reaching even a normal status and achieving a sense of cure or normalcy with long-lasting benefits and patient satisfaction (c, Figure 2). If we do not intervene in the early stages of “active/refractory” status, some of the patients’ disability might have started to become irreversible as best highlighted from the effects of CAR T-therapy in a patient with SPS who impressively improved with reduced stiffness and spasms everywhere except of the stiffness in her left arm which had been immobilized in a stiff position for 10-years. 9 The example of this patient, along with our own experience of similar cases, supports the need for early (Figure 2), rather than late (Figure 1), treatment initiation during the active/refractory status, and before a potentially reversible neuronal dysfunction or axonal degeneration transition into neuronal loss and the clinical deficits enter into irreversible status, 10 as seen in long-term wheelchair-bound patients.

Therapeutic patterns of immunotherapy cycles in patients with early - “active/refractory” autoimmune neurological disease, best suitable for initiating anti-CD19 CAR T-therapy. (A) In these patient subsets, after the initial improvement with therapeutic cycling dosing (usually the first year) there is partial residual benefit during the off cycles (a). (B) Follow-up therapies continue with partial improvement but also declining benefits, even with subsequent therapeutic dosing cycles with two different immunotherapeutic agents, resulting in steadily progressive decline of benefit despite maintenance cycling dosing (a => b). (C) The most suitable time for initiation CAR T-cell therapy in patients with early-refractory but still active disease because one CAR T-cell infusion can reverse the disease decline and lead to fast improvement reaching even normalcy (c).
Patient satisfaction
At the patient’s level, the cyclic therapeutic patterns (weekly, monthly, or every 3–6 months), as with all current immunotherapies (B → D, Figure 1), translates into “wearing off” effects in-between each cyclic period affecting quality of life and preventing the induction of feeling of normalcy most of the time. In addition, the need for intravenous infusions at infusion centers or frequent self-subcutaneous injections, along with injection-related side effects, emotional stress, and tension associated with the need to continue receiving therapies at exact time points, and disturbing economic issues due to need for preapproval or periodic re-approval, are collectively further burdens, not seen after therapy has escalated to CAR and the B → D (a – d) has transitioned to E, e (Figure 1) or, even better, – A → B (a – b) has transitioned to C, c (Figure 2).
Why CAR T-cell-therapy can change the therapeutic algorithm in patients with active/refractory autoimmune neurological diseases
Although the experience with CD19 CAR T cells in neurology is still limited to a few case series in MG, SPS, myositis, MS, NMOSD, and encephalitis, their impressive success and expanding application in ongoing large-scale trials has been generating enthusiasm and hope as the only means of reversing the course of an active/refractory disease status with cumulative disability to a steadily increasing improvement, even approaching normalcy in just a few months. The unique superiority of CAR T therapies, and also their limitations, need to be explained not only to practicing neurologists but also to patients who must understand and appreciate the scientific reasoning of such promising success and the associated risks. Accordingly, even if redundant for the present TAND series,4–7 the following short explanation as to why CAR T cells can reverse the ongoing disability compared to the currently approved unsuccessful or partially successful immunotherapeutics that have led to the present refractory status, has become essential before practicing neurologists and their patients feel comfortable and appreciate the merits of this therapy, considering the many practical complexities and risks connected with the CAR T therapies:
What are the CAR-expressing T cells
As extensively discussed,1,4–7,10 CAR T-cells are genetically modified cells to effectively bind their targeted antigen, release cytotoxic molecules, like perforin or granzyme-B which kill targeted cells, and produce IL-2 via their integrated costimulatory domains CD28 or 4-1BB (CD137), that leads to their differentiation into effector memory and constantly active T-cells capable of exerting sustained cytotoxicity upon encountering their antigen, behaving as “living serial killer cells”2,10–14 or, simplistically, like they have been transformed into “super soldiers.”
How the CD19 CAR T cells differ from the monoclonal anti-B cell agents
The CD19 CAR T-cells penetrate fundamental disease-specific tissues, such as lymph nodes, thymic tissue, bone marrow, or blood–brain barrier (BBB), leading to B cell-depletion not only of the circulating B cells as the current drugs do but also within lymphoid or brain areas. 15 This is of importance to key neurological diseases, such as myasthenia gravis offering the prospects of eliminating thymic B-cells, or to MS and other CNS autoimmunitites because they can deplete meningeal ectopic lymphoid follicles or the resident B cells within the CNS compartments responsible for intrathecal antibody production. Such a direct effect within the CNS enhances their potential to arrest the neuroinflammatory process that drives the smoldering CNS inflammation and autoimmunity, currently unreachable with the present anti-B cell monoclonals that do not penetrate the CNS.1,10–15 Also, in contrast to anti-CD20 monoclonals currently used in MS or MG, the CD19 CAR T-cells eliminate a broad range of B cells, especially plasmablasts and long-lived plasma cells responsible for antibody production but without affecting the protective CD19-negative (but B-cell maturation antigen (BCMA), CD38, CD138-positive) bone marrow plasma cells and plasmablasts responsible for maintaining the reservoir of protective humoral immunity like the vaccine-induced IgG antibodies.1,10–15
CD19-CAR T induced sustained effects with B cell rebooting
In contrast to currently used therapeutic antibodies that have limited half-life requiring repeated infusions, the CAR T are “living cells” able to proliferate and constantly expand to memory cells that persist for years, ensuring sustained benefits without the need for re-treatments because of declining benefits or relapses as seen with the present drugs (Figure 1, A–D). It is on this basis that CAR T-cells have the potential to even eradicate the pathogenic B cell clones connected to the original disease and lead to nonclass-switched naïve B cells that could achieve an immune reset by rebooting the whole B-cell system.15,16
CAR T cells have broad immunotherapeutic actions
In contrast to current biologics that selectively target only specific immune players such as T cells, B cells, cytokines, or complement, collectively associated with antibody-dependent cellular phagocytosis or complement-dependent cytotoxicity, the CD19 CAR T cells have broad effects beyond B cells1,10 being effective not only in antibody-mediated neurological diseases but also in autoimmunities without pathogenic antibodies, like GAD-ab in SPS or myositis-associated-ab in inflammatory myopathies, or even in diseases without known antibodies like CIDP or MS; such global effects are also supported by their capacity to modify even regulatory or cytotoxic CD8 + T cells with the potential to shift the profiles of immune cell subsets toward a healthy state. 17
Have effects on two concurrent autoimmunities (“one stone targets two birds”)
CD19 CAR T-cells have been effective not only in one but in two concurrent autoimmune diseases seen in the same patient, each with different mechanism, like SPS and MG, 8 rheumatoid arthritis and MG, 18 or MG and Lambert–Eaton myasthenic syndrome, 19 enhancing further their prospects of changing the therapeutic algorithm because overlapping autoimmunitites occur in 25%–30% of autoimmune neurologic diseases. Witnessing such therapeutic effects in two different immunologically active but diverse diseases, each one contributing to the same patient’s overall disability, is a unique quality of CAR T-cells characterized as the phenomenon of “one stone targets two birds,” 10 that further expands their global efficacy in arresting disability progression caused by more than one disease as there is no need to treat these patients with two different disease-specific immunotherapies.
Status of CAR T therapies in neuroimmunological diseases: exciting potential in changing the current therapeutic algorithm
Based on the following case series in common autoimmune neurological diseases and the currently expanding large-scale studies, CAR T-cells seem destined to change the therapeutic armamentarium in patients with refractory autoimmune disease, being the only means of reversing disease progression, achieving long-lasting drug-free remission or even reaching normalcy.
Myasthenia gravis
In two MG patients with active/refractory disease, anti-CD19 CAR T cells led to long-term remission with stable IgG concentrations, reduced AChR-ab, and no increased susceptibility to infections. 20 In a phase Ib/IIa RNA-engineered CAR T cells targeting BCMA, five of the seven treated MG patients continued to show clinical improvement in MG-ADL (Activities of Daily Living), QMG (Quantitative MG), and MGC (MG Composite) scores after 12 months. 21 A large, phase III KYSA-6 (NCT06193889) multicenter international study with autologous CD19 CAR T cells is currently ongoing in refractory MG patients.
Lambert–Eaton myasthenic syndrome
A severely affected 54-year-old patient with LEMS and typical active/refractory disease with tetraparesis and severe gait disorder, showed clinical improvement after anti-CD19 CAR-T cell with an 8-fold increase in walking distance and reduction of the pathogenic VGCC (Voltage Gated Calcium Channel) antibody titers. 22
Stiff person syndrome
In two patients with active refractory disease,8,23 CD19 CAR T cell therapy reversed the patients’ progressive disability with signs of improvement starting a few weeks after infusion with steadily increasing benefits up to more than a year. An open-label study with the same CD19 CAR-T cells that has been now completed in 26 patients enrolled in 3 US centers, 10 we have seen very impressive and lasting benefits, as formally announced (KYSA-8 Topline results; Kyverna Therapeutics, December 15, 2025, Emerville, California USA).
Myositis
A patient with progressive myositis and interstitial lung disease due to antisynthetase syndrome refractory to available therapies including rituximab, impressively improved on muscle scores and pulmonary function tests 8 months after CD19-CAR T therapy without detectable myositis signs on the MRI; anti-Jo-1 antibody levels were reduced and serum muscle enzymes, CD8+ T-cell subsets, and cytokine secretion were normalized. 24 Three other myositis patients also had a major clinical response 3 months after infusion, with normalization of creatine kinase level, measured muscular functions, and extramuscular disease activity scores. 25
Chronic inflammatory demyelinating polyneuropathy
In two patients with refractory CIDP, anti-BCMA-chimeric antigen receptor T cell (BCMA-CAR-T) achieved drug-free remission within 6 months; one patient was stable for 24 months with reconstitution of circulating B cells, diminished autoantibodies, and sustained humoral immune suppression, while the other exhibited disease recurrence 12 months later when COVID triggered reactivation of pathogenic B cells and autoantibody recurrence.26,27 Two additional patients refractory to all immunotherapies, one with GM1-ab and the other with NF-155-positive paranodopathy being wheelchair-dependent, impressively improved 4–6 months after CD19 CAR T therapy becoming able to walk unassisted with improved electrophysiological findings and undetectable GM1 and NF-155 antibodies. 28
Neuromyelitis optica spectrum disorder
Anti-BCMA CAR T cells, induced drug-free and relapse-free remission after a median of 5.5 months in 11 of 12 adults with AQP4-IgG seropositive NMOSD, 29 but long-term data assessing the probability of relapses is lacking. In a 25-year-old patient with aggressive MOGAD and more than six relapses with severe visual impairment despite immunotherapies, CD19 CAR T induced disease stability without relapses for 1 year. 30 MOG-ab, being positive the seven previous episodes, became undetectable while vaccination-induced immunity remained unaffected. 30
Multiple sclerosis
CD19 CAR-T cells suppressed disease relapses in progressive MS unresponsive to ocrelizumab. Expansion of CAR T-cell was noted in the CSF with concurrent reduction of oligoclonal bands and intrathecal IgG 14 days after cell infusion, without signs of neurotoxicity, highlighting that CAR T-cells can safely penetrate the CNS compartments inaccessible to current B cell-depleting agents. 20 In a phase I trial (NCT06451159), CAR T-cells had a good safety profile in two patients with SPMS (Secondary Progressive MS) with elimination of oligoclonal bands after 43 days and easing disability 6 months after infusion. 31 A phase II clinical trial with anti-CD19 CAR T-cells in 12 patients with progressive MS (NCT06138132) is now ongoing hoping to also provide data on disability progression and value of specific biomarkers.
Autoimmune encephalitis
A 36-year-old man with rapidly progressive generalized myoclonus, head tremor, vertical binocular nystagmus, and tetraparesis due to anti-DAGLA-antibody-associated autoimmune encephalitis unresponsive to steroids, PLEX, and rituximab, improved after a single infusion of CD19 CAR T-cells 32 . CAR T-cells were present in the CSF leading to significant reduction of anti-DAGLA antibodies in serum and CSF, disappearance of CSF oligoclonal bands, and safe depletion of intrathecal B cells indicating that CD19-CAR T-induced deep B cell depletion can be a powerful treatment for antibody-associated encephalitis. 32
Therapeutic uncertainties and logistical challenges of CAR T-cell therapies
Although CAR T cell therapy is extremely promising and the only means for reversing an active/refractory autoimmune neurological disease, the following uncertainties, challenges, and concerns need to be highlighted:
What is the duration of CAR T-induced clinical remission and how are posttherapy relapses handled?
Despite the evidence that CAR-T cells persist and induce sustained B-cell aplasia with profound tissue-based B-cell depletion achieving an immune reset with persistent clinical effectiveness even after B-cell reconstitution, the exact duration of a CAR-T cell-induced drug-free clinical remission of treated autoimmune neurological disease remains unclear and probably unpredictable. Because CD19-CAR T-therapy cannot eliminate the patient’s pathologic T cells and some of the CD19-negative plasma cell-producing antibodies are expected to persist, the possibility of disease recurrence cannot be excluded after treatment, as highlighted with the following examples.
A patient with inflammatory myositis relapsed 9 months after CD19 CAR-T-induced remission but also failed to expand and improve when re-treated and reinfused with the same CD19 CAR-T cells. The patient achieved however stable drug-free remission after receiving BCMA CAR-T-cell therapy suggesting that BCMA-targeting has probably effectively addressed the CD19-resistant autoreactive plasma cells 33 . Whether switching CAR-T cell targets is an effective strategy to treat post-CAR T-therapy relapses, or need to explore other means to enhance the durability of CAR T cells, remains uncertain. 34 A seronegative rheumatoid arthritis patient relapsed after receiving CD19 CAR T-cell therapy but improved following hematopoietic stem cell transplantation 35 . In contrast, a patient with MG who relapsed after receiving CD19-CAR T-cells improved again after a second dose of CD19-targeting cells being in long-term remission afterward. 36 Of interest, a patient with autoimmune hemolytic anemia 37 who relapsed after CD19 CAR T-induced remission, regained remission for the second time after receiving a different therapeutic modality, a bispecific T cell engager.37,38
In cancer patients, CD19 or BCMA-negative B cells have re-emerged following CAR T therapy as a means of immune evasion of the malignant B cell population, 38 but whether a disease relapse in autoimmune patients has any similar analogy is unknown. The deep B-cell depletion induced by CD19 CAR T cells eradicates disease-associated B cell clones but whether it also causes secondary loss of autoreactive T cells, requiring their stochastic regeneration and activation with cognate T-cell help, has been speculated as a possibility. 39
At present, the clinical relevance of the magnitude of CD19+ B cell repopulation in reference to future disease activity or safety risks, like susceptibility to infections, remains uncertain. Importantly, the overall long-term safety of CAR T-cell therapies, risks for infections especially in patients without sufficient repopulation of B cells and prior series of immunotherapies or chemotherapeutic pretreatments, remain to be established with long-term follow-ups. Another uncertainty is the safety of CAR T-cell in certain age groups, currently excluded from ongoing trials, like children, fertility issues in women of reproductive age, and the very elderly patients with refractory neurological autoimmunities.
Collaborative needs for CAR T therapies in autoimmune neurological diseases and future prospects of CAR T-economics
All the CAR T clinical trials in neurological diseases are currently industry-sponsored with academic-led initiatives and close collaboration and interdisciplinary coordination of neurologists with hem/oncologists, cell therapy units, dedicated nursing staff, and clinical research administrators working closely with various regulatory agencies to successfully and timely complete therapy in case series ensuring safety standards and patient protection, while fostering efforts for innovative research to understand the mechanistics and consequences of deep B cell depletion. Since CAR T-therapies in neurology are currently conducted on experimental basis in academic university settings, such multidisciplinary CAR-T cell teams can perform clinical trials only in concert with industry. It is however hoped that when a CAR T therapy is approved for a specific neuroautoimmune disorder based on the ongoing FDA-approved clinical trials, the academic centers with experienced teams will be able to transition to offering dedicated CAR T-patient care with coordinated basic neuroimmunologic research to explore clinical assessments with the strategic timings of the immune reset.
Efforts on how best to improve cost-effectiveness remain however essential. Because autologous CAR T is a very costly multidisciplinary process requiring hospitalizations and collaboration with many experts beyond neurology, the allogeneic, “off-the-shelf” CAR T-cell therapy is making progress due to excellent therapeutic profile, albeit with inferior expansion and lesser duration of persistence compared to autologous CAR-T 40 . Whether the allogeneic CAR T-cells will be more widely utilized and more likely destined to change the neuroimmunologic therapeutic algorithm remains to be seen. The clinical rationale or the decision pathway as to when anti-CD19 CAR-T might be preferable to anti-BCMA CAR-T in initiating or follow-up therapy, remains also uncertain.
When discussing economics of CAR T therapies for patients with chronic neuro-autoimmune disorders, we need to take into consideration that these patients exhibit severe physical disability, poor quality of life, and mounting expenses for assistance in their daily living activities along with co-payments and frequent complexities in ensuring or gaining approval of their current, even if insufficiently-effective, biologic, and symptomatic therapies. Such cumulative expenses, combined with a psychologically stressful and steadily progressive disabling lifestyle, should be weighed against the prospects of CAR T-achieving a drug-free, normal quality of life fully able to perform physical activities with no disability and sustained benefits. An additional hopeful consideration favoring a CAR T option could be the optimistic expectation that the global cost of CAR T-cell therapy is expected to decrease over time.
Toxicities
The overall toxicity profile of CAR-T in treating autoimmune neurological diseases has been quite favorable compared to that of malignancies, as described in detail in this series.6,7 The most common, the cytokine release syndrome (CRS), is a systemic inflammatory reaction that presents with fever, chills, tachycardia, cellular edema, hypotension, and hypoxia associated with elevated inflammatory biomarkers such as CRP, ferritin, and IL-6, observed within the first week after the infusion.1,2,7,10 CRS occurs because activated CAR-T-cells release large amounts of cytokines, like IL-1β, IL-6, TNF-α, that cause cytopenia and cardiovascular or respiratory symptoms2,4,10; CRS in autoimmune diseases is not however as serious as in patients with malignancies and responds to antipyretics, corticosteroids, tocilizumab, or anakinra. The more serious immune effector cell-associated neurotoxicity syndrome (ICANS) that occurs in approximately 27% of malignancy-treated patients 10 has not been reported in the treatment of autoimmune diseases, or if it has rarely occured was not severe. ICANS is due to increased permeability of the BBB allowing pro-inflammatory cytokines to enter CNS triggering cerebral edema, requiring treatment with steroids and anakinra.2,4,7,10
The future trajectory of CAR T-cell therapy in changing the therapeutic landscape of autoimmune neurology
The future trajectory of CAR T-cell therapy for neuro-autoimmunities will be driven by establishing further its efficacy and safety, accessibility, cost, and expanding the multidisciplinary coordinated efforts between academia and industry. The first impressive success of CD19 CAR T-cell therapy in a large series of 26 patients with SPS, where 80% of previously disabled patients with refractory disease became able to walk independently or even run (as announced by Kyverna 12/2025), is the first example of a successful academia/industry coordinated effort serving as a stimulus for expanding further the clinical trial landscape to large-scale studies across the whole neuroautoimmunity spectrum. Despite the presently complex manufacturing procedures, costs, and inherent safety risks, it is expected based on the present enthusiasm and soon to be published results from FDA-approved successful trials, that future multidisciplinary efforts between industry and neuroimmunology groups will be substantially enhanced and become a strong driving force in establishing CAR T-University centers not only to serve patients and train clinicians but also to stimulate further the CAR T science to improve safety and explore in depth the basic effects of immune reset in inducing more sustained clinical benefits.
It is also expected that impressive results, as those announced for SPS, will become a strong impetus for the treating neurologists to identify in a timely fashion patients with active/refractory disease before their cumulative disability becomes irreversible and refer them promptly to appropriately established CAR T academic centers. Recognizing very early the timeline of the current treatment failures when the patients’ residual deficits are salvageable and reversible and not when long-standing disabilities have become irreversible due to chronic neuronal or synaptic degeneration and immunologically inactive process, will be essential in enhancing further the rate and magnitude of success when escalating to CAR T. On the patients’ side, the publicity of the success and safety profile, is expected to reduce possible hesitancies and inspire their willingness to seek CAR T-therapy early in their disease course when their disability starts building up. In parallel, as supported by the present compelling data, patient organizations, associations, foundations, and philanthropists is expected to provide stimulating support driven by the thought and expectation that an evolving neurological disability not only can be reversed but also can even normalize a disabling patient. Such collective coordinated efforts should slowly transform the CAR T-cells as a standard therapy for patients with early refractory autoimmune neurological disease.
Conclusion
Based on the unmet therapeutic needs of current patients with various neuroautoimmunities the study points out that CAR T-cell therapy is a very promising means of changing the therapeutic algorithm for patients with refractory autoimmune neurological diseases. It highlights the need to recognize the early stages of disability development when patients have reached an early-active/refractory disease status, where CAR T therapy can reverse disability and lead even to a cure, rather than waiting for very late disease progression when deficits might have become irreversible. Further, it summarizes the present status of CAR T in autoimmune neurology explaining at the level of general neurologist how CAR T cell therapy works in an effort to become comfortable in selecting suitable patients early, abort the ongoing failed treatments and start escalation to CAR T. The future trajectory of CAR T-cells in autoimmune neurology therapeutics is also discussed pointing out therapeutic uncertainties, duration of CAR T-induced remissions, logistical challenges, and economic limitations while highlighting the prospects of establishing multidisciplinary academic CAR T cell centers even as standard therapies.
