Abstract
Background:
Patient support programs (PSP) help initiate and maintain disease-modifying treatment (DMT) for people with multiple sclerosis (PwMS). adveva® (worldwide), Alcura Healthcare (Netherlands), and MS LifeLines® (US) are nurse–pharmacy-led PSPs that collect clinical information, with consent, on PwMS receiving cladribine tablets (3.5 mg/kg over 2 years followed by a 2-year treatment-free period per product label).
Objective:
To evaluate initiation and completion of short courses of cladribine tablets treatment in Years 1 and 2 for PwMS and the frequency of subsequent treatment.
Design:
Retrospective, multinational, observational study using real-world data from PSPs.
Methods:
Clinical data from PSPs in Australia, Canada, Gulf, Latin America, Near East, Netherlands, UK, and US (December 2017–December 2023) included prior DMT use, cladribine tablets initiation and completion (Years 1 and 2), and subsequent treatment after Year 2 for those with ⩾48 months’ follow-up.
Results:
Across all PSPs, 22,082 PwMS initiated cladribine tablets (Australia, n = 799; Canada, n = 3665; Gulf, n = 258; Latin America, n = 1854; Near East, n = 86; Netherlands, n = 681; UK, n = 2320; US, n = 12,418), of whom 70.9%–81.3% were female (mean age 34.2–49.1 years). Most were DMT-experienced (Australia, no contribution to analysis; Canada, 100%; Gulf, 52.7%; Latin America, 57.7%; Near East, 86.0%; Netherlands, 81.4%; UK, 30.0%; US, 79.5%) with oral DMTs most commonly used (36.5%–69.3%). In PwMS with ⩾18 months’ follow-up, ⩾82.8% initiated Year 2 treatment at a mean of 12.4 (±3.8) to 13.9 (±3.5) months after initiating Year 1 treatment. Of those commencing Year 2 treatment, ⩾93.6% completed it. Additional treatment with cladribine tablets beyond 48 months was administered in a small proportion of PwMS: Canada, 5.6%; Latin America, 4.3%; Netherlands, 3.8%; and UK, 1.8%.
Conclusion:
Cladribine tablets adherence was high, with most PwMS completing short-course cladribine tablets treatment in Years 1 and 2. Of those with ⩾48 months’ follow-up, only a small proportion received additional cladribine tablets treatment.
Introduction
Multiple sclerosis (MS) is a chronic, inflammatory, demyelinating disease of the central nervous system that affects 2.9 million people worldwide,1,2 and it is one of the most common causes of neurological disability in young adults.3,4 Without appropriate treatment, MS can result in relentless neurological disability progression, however, in the past three decades, an increasing number of effective disease-modifying treatments (DMTs) have been approved, 5 resulting in improved clinical outcomes for people with MS (PwMS). 6 These DMTs vary substantially in efficacy, safety, dosing, and administration, and it is essential to understand their real-world use to inform treatment optimization. Among DMTs, cladribine tablets are the only oral therapy selectively targeting lymphocytes, resulting in sustained yet reversible immune modulation, enabling them to be administered in short courses that provide a durable benefit without the need for chronic administration.7,8
Cladribine tablets were first approved by the European Medicines Agency and Health Canada in 2017 and subsequently introduced to the European and Canadian markets in 2018, with many countries using the treatment since its launch date.9,10 In the United States (US), cladribine tablets became available in 2019 for the treatment of relapsing forms of MS. 11 As of June 2025, an estimated 131,017 PwMS have received cladribine tablets with 367,021 patient-years of exposure since final approval in 2017.
Cladribine tablets are indicated for the treatment of relapsing forms of MS in adults with active disease as defined by clinical or imaging features. 9 The recommended cumulative dose is 3.5 mg/kg over 2 years, administered as two short courses of 1.75 mg/kg at the start of each of Year 1 and Year 2. Each treatment course consists of two separate treatment weeks, one in the first month (Month 1) and another in the second month (Month 2) of each respective year. Following completion of the 2-year dosing regimen, no further treatment with cladribine tablets is required in Years 3 and 4.
The efficacy and safety of cladribine tablets have been demonstrated in several phase III clinical trials, including CLARITY and CLARITY Extension,12–16 ORACLE MS,17–19 and ONWARD. 20 Subsequently, phase IV trials, including the MAGNIFY-MS 21 and CLARIFY-MS extension trials, 22 as well as long-term, real-world data have confirmed the efficacy and safety profiles of cladribine tablets23–29 with no additional safety signals identified since marketing authorization was obtained in the European Union. Since their launch, more than 100,000 PwMS have been treated with cladribine tablets, with over 250,000 patient-years of experience being accumulated. 30
Cladribine tablets have been associated with improved health-related quality of life in PwMS, and a decreased need for health system support, which makes it an appealing DMT for PwMS and policymakers.31,32 Moreover, in the 6-month interim analysis of the CLARIFY-MS study, PwMS reported high treatment satisfaction in the Treatment Satisfaction Questionnaire for Medication following 6 months of treatment with cladribine tablets.33,34
Patient support programs (PSPs) play an important role in helping people with chronic conditions, including MS, with several studies indicating that PSPs have a positive impact on adherence to DMTs.35–37 PSPs are managed services provided by market authorization holders; they are an optional service beyond insured standards of care, with the goal of providing administrative support and educating people on their treatment journey and behavior to increase adherence.
The use of cladribine tablets is supported by PSPs in the US (MS LifeLines®), the Netherlands (Alcura Healthcare), and in the rest of the world (adveva®), and they provide administrative and educational support to PwMS initiating cladribine tablets. The goal of PSPs is to maximize adherence to cladribine tablets in the short and long term, to maximize the safety and effectiveness of the drug, and to improve health-related quality of life. With these core PSP objectives in mind, the purpose of this study was to evaluate the clinical characteristics of, and adherence to, cladribine tablet dosing in Year 2, and to assess whether additional treatment courses of cladribine were given beyond Year 2 among PwMS across many regions of the world, including Australia, North and Latin America, Europe, the Gulf region, and the Near East. We hypothesized that evaluating the characteristics of participants in PSPs would provide valuable insights into adherence to cladribine tablets in the real world.
Methods
Study design
Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines were followed for the conduct and reporting of this study (Supplemental Material). This cohort study is based on the secondary use of data from PSPs for cladribine tablets across eight distinct countries/regions: Australia, Canada, the Gulf region (Bahrain, Kuwait, Oman, and United Arab Emirates), Latin American (Argentina, Brazil, Chile, Colombia, Costa Rica, Dominican Republic, Ecuador, El Salvador, Honduras, Nicaragua, Peru, and Mexico), the Near East (Lebanon, Palestine), the Netherlands, the United Kingdom (UK), and the US. Dates of enrollment ranged from 2017 to 2023.
The study period for each country/region began with the initiation of cladribine tablets treatment (index date) for each participant and lasted until December 1, 2023 (data cut-off date). However, the study period for individual participants could end earlier due to loss to follow-up, consent withdrawal, or treatment discontinuation, whichever occurred first. Participants who completed the full 2-year regimen were considered to have completed treatment rather than discontinued. Follow-up extended up to 48 months, regardless of whether treatment was completed or discontinued early.
Data collection was integrated into the routine operations of the PSPs, with PSP nurses conducting telephone check-ins to gather information. Enrollment into the PSP occurred during the initial visit, when the HCP prescribed cladribine tablets. The HCP inquired with all patients initiating cladribine tablets whether they were willing to join the PSP and recorded consent. The aim of the first nurse-PSP contact was to set up clear expectations of cladribine tablets treatment (what is it, how does it work, how do I take it, what are the blood tests for, side effects, and frequently asked questions). The enrollment process varied among different countries and regions. All PSPs had scheduled visits during Year 1 and Year 2. The frequency and timing of check-ins were consistent across regions with slight variation in Canada. All PSPs had data collection points at: enrollment; day 0 (initiation of first treatment course); week 5 (initiation of second treatment course); week 9 (to provide information on treatment and a reminder blood test); month 6 (mid-year check in), month 10 or 11, and month 12 (to prepare for year 2 treatment). A similar pattern of visits occurred in Year 2. In Canada, there was follow-up at month 6 of Year 2, with no planned follow-up after this timepoint. In the Gulf Region and Latin America, proactive follow-up after Year 2 was conducted with patients contacted every 3 months and every 6 months, respectively.
In all other countries/regions, after Year 2, PwMS could be contacted for follow-up if needed, and PwMS in all countries could reach out at any time if needed. If additional treatment was initiated, a new set of follow-ups was triggered, and the PSP team was aware of any treatment discontinuation by patients or HCPs.
Study population
The study cohort consisted of PwMS enrolled in the PSP for cladribine tablets during the study period in one of the countries/regions of interest. Inclusion criteria consisted of PwMS newly initiating cladribine tablets, being ⩾18 years of age at the start of cladribine tablets treatment, and able to provide informed consent. There were no exclusion criteria. Due to the descriptive nature of the study, there was no pre-defined hypothesis tested in this study, so no formal sample size requirements were considered, and all eligible patients were enrolled.
Data collection/variables of interest
Variables of interest collected included: age at cladribine tablets initiation, biological sex, date of treatment initiation, and MS subtype at enrollment. The use of DMT prior to treatment with cladribine tablets was recorded, including whether individuals were DMT-naïve or DMT-experienced, the type of DMT, route of administration of the most recent DMT prior to enrollment, and the number of prior DMTs used. Treatment discontinuation (off therapy) and reasons for discontinuation were reported. We mapped the various terminologies used across countries to standardize definitions. For example, the terms “treatment stop” and “treatment discontinuation” were used interchangeably in different patient PSPs. To ensure clarity and accuracy in our definitions, we consulted with PSP personnel regarding data recording practices within each country’s PSP database. The operational definitions of each data element were harmonized across PSPs in a data specifications document by defining derived variables at the data cleaning and data analysis stages. A preliminary analysis was performed, and results of each data element were validated for each country/region PSP program.
Statistical methods
The descriptive statistics of participant characteristics, and variables of interest (relative proportion, mean and standard deviation (SD), median, interquartile range, and range (minimum and maximum)) were reported. A Kaplan–Meier analysis was used for time-to-event variables (e.g., time to treatment discontinuation).
The number of PwMS initiating Year 2 treatment (adherent with Year 2 dosing initiation) was evaluated for those PwMS who had at least 18 months of follow-up. The number of PwMS initiating additional treatments of cladribine and time to additional treatment initiation were also evaluated in PwMS with ⩾24 and ⩾48 months of follow-up.
Results
Across the eight countries/regions in this study, a total of 24,001 PwMS were enrolled, and 22,082 PwMS initiated treatment with cladribine tablets during the specified study period (Australia, n = 799; Canada, n = 3665; the Gulf region, n = 258; Latin America, n = 1854; the Near East, n = 86; the Netherlands, n = 681; the UK, n = 2320; and the US, n = 12,418). The country-specific total (22,081) differs by one PwMS because a PwMS who relocated to the Netherlands was enrolled in the PSP from Year 2; as the treatment start date was unknown, they were excluded from the country-specific denominator but retained in the overall total. Mean age (SD) at cladribine tablets initiation varied across countries/regions, ranging from 34.16 ± 9.63 in the Near East to 49.1 ± 12.10 in Australia. The majority of PwMS were female (range across regions: 70.9%–81.3%). All participants had relapsing–remitting MS. The patient characteristics are summarized in Table 1.
Patient characteristics and use of disease-modifying treatment prior to enrollment.

Dash indicates data not available or not collected.
DMT, disease-modifying therapy; MS, multiple sclerosis; Q1/Q3, first and third quartiles; RRMS, relapsing–remitting multiple sclerosis; SD, standard deviation; UK, United Kingdom; US, United States.
The proportion of PwMS with prior DMT use was 52.7% (136/258) in the Gulf region, 79.5% (9873/12,418) in the US, and 100% (3665/3665) in Canada. The UK was the only region where treatment-naïve PwMS constituted the majority (66.7% (1548/2320)) of participants. The number of prior DMTs used was reported in Australia, Canada, the Gulf region, and the Netherlands, and ranged from 1 to 8 prior DMTs. The proportion of PwMS who received at least one prior DMT was 52.7% (136/258) in the Gulf region, 62.2% (51/82) in Australia, 84.5% (554/656) in the Netherlands, and 100% (3665/3665) in Canada. The most common recent DMT prescribed before initiation of cladribine tablets varied by country/region and consisted of teriflunomide in the Netherlands and Latin America (56.5% (313/554) and 31.4% (316/1007), respectively); interferons in the Gulf region and Near East (39.3% (53/135) and 37.8% (28/74), respectively); dimethyl fumarate in Canada (21.0% (758/3618)); and ocrelizumab in the US (19.2% (1895/9873); Table 2).
Most recent DMT prescribed before initiation of cladribine tablets by country/region.

Shading in the table represents the proportion of PwMS. Darker shades indicate higher proportions of PwMS in the country/region taking the associated DMT, while lighter shades or unshaded cells represent lower proportions of PwMS in the country/region taking the associated DMT. Dash indicates data not available or not collected.
Further information on these specific DMTs is not provided in the current dataset.
DMT, disease-modifying therapy; PwMS, people with multiple sclerosis; US, United States.
Summary statistics on cladribine treatment discontinuation during follow-up, by country/region, are shown in Table 3. The total number of patients with complete data at 12, 24, and 48 months were 20,858, 4278, and 2309, respectively. Discontinuation rates were generally low: Australia, 3.6% (n = 29); Canada, 7.1% (n = 259); the Gulf region, 0.8% (n = 2); Latin America, 4.6% (n = 86); the Netherlands, 8.8% (n = 60); the Near East, not collected; the UK, 7.3% (n = 169); and the US, 12.9% (n = 1605). The timing of discontinuation varied by country/region and often occurred after the first 12 months of treatment (Table 3 and Figure 1). More granular data on the number of PwMS who discontinued treatment before or after the second dose of cladribine tablets were not available.
Summary statistics for discontinuation of cladribine tablets, by country/region.
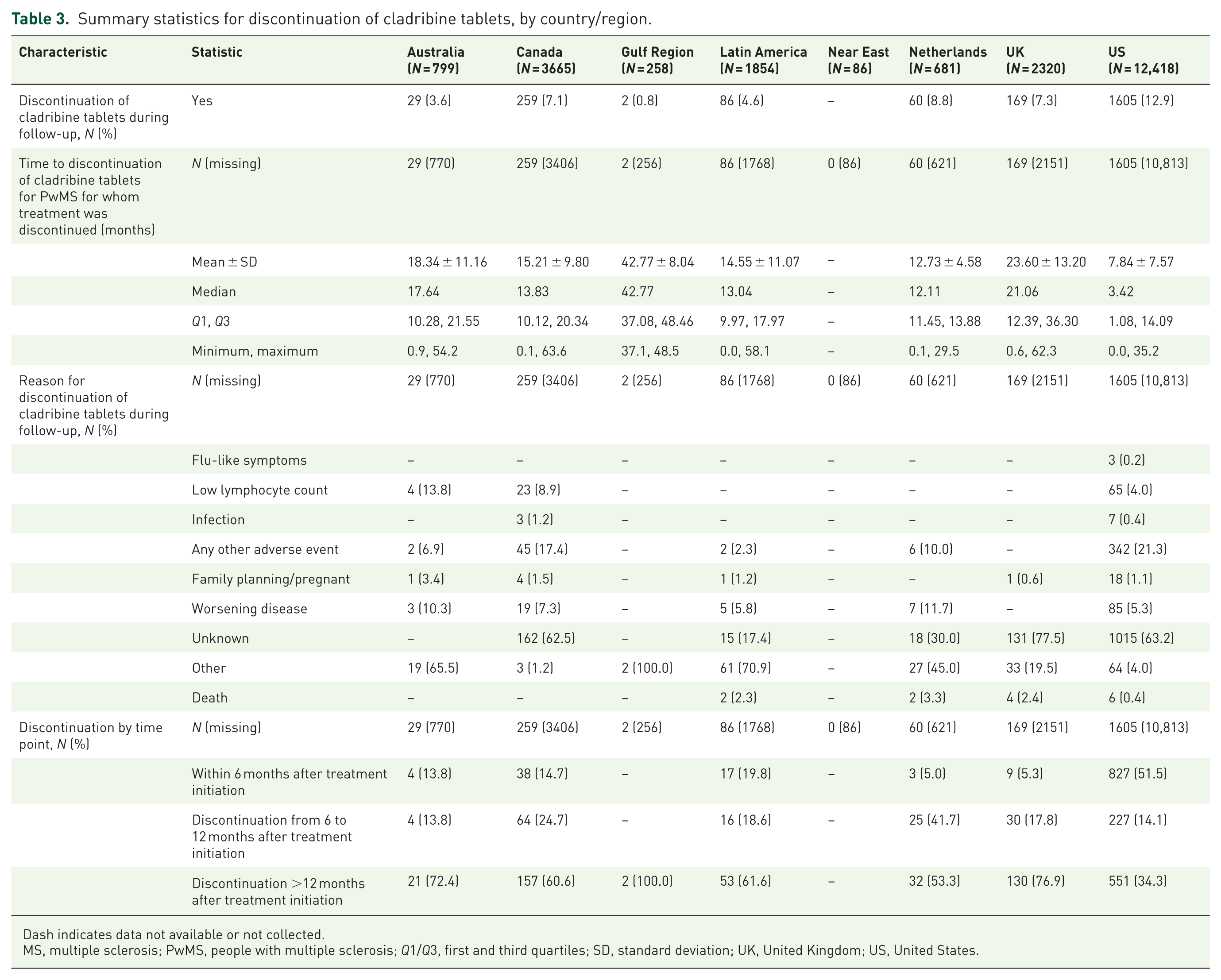
Dash indicates data not available or not collected.
MS, multiple sclerosis; PwMS, people with multiple sclerosis; Q1/Q3, first and third quartiles; SD, standard deviation; UK, United Kingdom; US, United States.

Time to discontinuation from the PSP by country. (a) Australia. (b) Canada. (c) Gulf Region. (d) Latin America. (e) Near East. (f) The Netherlands. (g) The UK. (h) The US.
In the majority of cases, the reason for discontinuation was either unspecified or was listed as “other” or “unknown”: Australia, not collected; Canada, 62.5% (162/259) unknown; the Gulf region, 100% (2/2) other; Latin America, not collected; the Near East, not collected; the Netherlands, 45.0% (27/60) other/30.0% (18/60) unknown; the UK, 77.5% (131/169) unknown; and the US, 63.2% (1015/1605) unknown. In cases where a reason for discontinuation was documented, adverse events were the most common reason in Canada (17.4% (45/259)) and the US (21.3% (342/1605)). In the Netherlands, however, disease worsening was the most frequently cited reason (11.7% (7/60)), followed by adverse events (10.0% (6/60)).
The first year of treatment was completed by the majority of PwMS across all countries/regions: Australia, 92.2% (n = 517); Canada, 99.3% (n = 2947); the Gulf region, 100% (n = 160); Latin America, 99.3% (n = 1077); the Near East, 100% (n = 76); the Netherlands, 99.6% (n = 479); the UK, 96.5% (n = 1848); and the US, 97.9% (n = 9155). Summary statistics of Year 2 treatment with cladribine tablets, by country/region in PwMS with at least 18 months of follow-up are shown in Table 4. Around 86.7% (14,398/16,601) initiated Year 2 of cladribine tablets treatment with a mean time (±SD) to initiation of 12.38 (±3.77) to 13.93 (±3.46) months. In seven of eight countries/regions, at least 93.6% of PwMS completed Year 2 treatment (Australia, NC).
Summary statistics for Year 2 treatment with cladribine tablets by country/region in PwMS with at least 18 months follow-up.

Dash indicates data not available or not collected.
Data did not contribute to the analysis due to database changes that prevented historic data transfer.
PwMS, people with multiple sclerosis; Q1/Q3: first and third quartiles; SD, standard deviation; UK, United Kingdom; US, United States.
In those with at least 24 months of follow-up (n = 4278; Table 5), the initiation of an additional course of cladribine tablets was uncommon, ranging from 1.6% (2/125) in the Gulf region to 13.8% (23/167) in Latin America. Mean (±SD) time to additional treatment initiation ranged from 32.57 (±12.48) months in Australia to 46.03 (±10.79) months in Canada.
Summary statistics for additional cladribine tablets treatment received after Year 2 treatment, by country/region.

PwMS who either did not require additional treatment or were lost to follow-up were excluded from the numerator.
Start of study period for each country/region—Australia: December 5, 2017; Canada: November 29, 2017; Gulf Region: March 15, 2018; Latin America: May 2, 2018; Near East: April 28, 2019; Netherlands: November 1, 2017; UK: September 7, 2017; US: March 29, 2019.
Among participants with ⩾24 months of follow-up since Year 1 initiation or participants stopping treatment with cladribine tablets.
Not estimated because data on only one PwMS was available.
Among participants with ⩾48 months of follow-up since Year 1 initiation.
PwMS, people with multiple sclerosis; SD, standard deviation; UK, United Kingdom; US, United States.
Among PwMS with at least 48 months of follow-up (n = 2309), data were available in Canada, Latin America, the Netherlands, and the UK. The proportion receiving additional treatment after 48 months was 5.6% (72/1294) in Canada, 4.3% (5/116) in Latin America, 3.8% (5/133) in the Netherlands, and 1.8% (14/766) in the UK (Table 5). Reasons for re-treatment and alternative subsequent DMT use were not captured.
Discussion
In this study, we investigated real-world adherence to treatment with cladribine tablets in PwMS by using data from PSPs in eight countries/regions worldwide and found that adherence was high, and the vast majority (⩾93.6%) of PwMS completed Year 2 of cladribine tablets treatment. Moreover, treatment discontinuation was uncommon, and in people with >24 and ⩾48 months of follow-up, additional courses of cladribine tablets beyond Year 2 were administered in only a small proportion of patients. Taken together, our results provide real-world evidence that adherence to cladribine tablets is high.
The observed high level of real-world adherence to cladribine tablets is likely related to the unique mechanism and short-term dosing/administration of cladribine that results in sustained benefits in many PwMS, in contrast to most other DMTs that require chronic administration to achieve sustained benefit.38–43 Unlike other DMTs, cladribine tablets achieve the full treatment effect after two short yearly courses, providing disease control for most PwMS without further treatment, 44 while a small percentage receive additional doses.38–43
This study was not designed to evaluate the effect of PSPs on medication adherence. As a result, it is purely descriptive, making it challenging to distinguish between adherence to the pharmacological effects of cladribine tablets and any impact of PSP-related support. However, the high rates of adherence of our data mirror findings from similar studies of PSPs, which report high rates of treatment completion. For example, Roche et al. 45 found that PwMS enrolled in a PSP had significantly higher adherence rates to subcutaneous interferon beta-1a compared to those receiving standard of care alone. At 12 months, 87.8% of PwMS remained on treatment versus 79.3% in the standard care group. The observed adherence gap widened at 24 months, with 76.2% adherence in the PSP group, compared to 61.8% in the standard of care group. Furthermore, high adherence has been observed in the CLARION non-interventional post-approval safety study, which included 3353 patients as of the second interim report (mean (SD) follow-up: 1.9 (1.2) years), 239 (6.7%) switched to another DMT during follow-up.
Lenz and Harms 37 examined adherence to DMTs (glatiramer acetate, interferon beta-1a, peg-interferon beta-1a, interferon beta-1b, dimethyl fumarate, and teriflunomide) over 24 weeks and found that PwMS using PSPs showed significantly higher adherence (92.9%) compared to those not using PSPs (61.8%, p = 0.02). Lapierre et al. 46 saw slightly lower, but still excellent, rates of adherence to fingolimod in their study of PwMS using PSPs, with 80.7% completing treatment at 12 months, 76.6% at 24 months, and 76.0% at 30 months, suggesting that PSPs potentially have a long-term beneficial effect on adherence. Notably, these studies primarily focused on continuously administered DMTs; this dosing regimen is quite different from the intermittent dosing regimen of cladribine tablets, given in two short courses 1 year apart. The unique dosing schedule of cladribine tablets likely facilitates adherence in comparison to chronically administered DMTs; however, it is probable that the PSP still plays an important role in adherence to cladribine tablets by educating patients on the intermittent dosing schedule and facilitating and reminding patients to start Year 2 of cladribine tablets at the appropriate time.
The findings from this study support these speculations, with all countries/regions reporting over 90% adherence to Year 1 treatment with cladribine tablets. Moreover, in PwMS with ⩾18 months’ follow-up, ⩾82.8% initiated Year 2 of cladribine tablets treatment with no relevant delay (mean of 12.4–13.9 months after Year 1 treatment), and at least 93.6% of PwMS completed Year 2 of cladribine tablets treatment. The high completion rate of Year 2 of cladribine treatment without a delay is likely related to the effectiveness of nurse–pharmacy-led PSP interventions in educating, motivating, and facilitating patients to complete their prescribed regimens, including regular follow-ups, education on the benefits of adherence, proactive management of concerns related to side effects, efficacy, and the provision of logistical support to complete relevant tests prior to Year 2 of treatment. 37
We observed relatively low rates of discontinuation in our study, ranging from 0.8% (the Gulf region) to 12.9% (the US). Discontinuations mainly occurred more than 12 months after Year 1 treatment initiation, except for in the US, where the majority of PwMS who discontinued treatment did so in the first 6 months following Year 1 treatment initiation. Reasons for discontinuation for PwMS who did not initiate Year 2 treatment varied across countries/regions. The most common reason for discontinuation was adverse events (variability across regions with 10% or less of PwMS in three of five countries/regions citing this reason, while the proportion was higher in Canada (17.4%) and the US (21.3%)). The next most common reason was worsening disease, ranging from 5.3% in the US to 11.7% in the Netherlands, across the follow-up period. Of note, discontinuation due to adverse events was still relatively low compared to previous studies, such as Lapierre et al., 46 where 59.8% discontinued fingolimod due to adverse events, and Tallantyre et al., 47 where 35% of discontinuations of DMTs were owing to adverse events. As MS nurses and PSPs are often the first point of contact when patients report side effects, these findings highlight the need for ongoing communication with PwMS to manage adverse events and explore options to mitigate these side effects, which could improve overall adherence.
Long-term follow-up (⩾48 months) was available for a small group of PwMS in Canada, Latin America, the UK, and the US. Among these, a small proportion initiated additional treatments beyond 48 months: 1.8% in the UK, 3.8% in the Netherlands, 4.3% in Latin America, and 5.6% in Canada. In the US and the Near East, the launch date for cladribine tablets precluded long-term follow-up, and no long-term data were available from Australia.
Although limited by the relatively small proportion of PwMS with 48 months or more of follow-up, these data suggest that in the real world, the efficacy of cladribine tablets may be sustained in many PwMS years after initiation of the first treatment course across many regions globally. However, given that information regarding the use of other DMTs during this long-term follow-up period was unavailable, this significantly restricts the ability to draw conclusions regarding the long-term efficacy of cladribine tablets, as well as long-term treatment strategies adopted by PwMS in these regions.
There is uncertainty around the longer-term management of MS with cladribine tablets since re-initiation of treatment after Year 4 has not been studied in clinical trials, and there is limited real-world evidence guiding treatment. A recent real-world study by Kowarik et al. 48 followed 187 participants with highly active relapsing MS for up to 5 years after initiating cladribine tablets treatment. Relatively low rates of treatment discontinuation were observed, with 63% of participants remaining off additional DMT in Year 5, supporting the durable efficacy of cladribine tablets. Expert opinions based on real-world cohort data suggest three treatment strategies for PwMS treated with cladribine tablets in Year 538–41,49: (1) extending the treatment-free period under structured monitoring, (2) initiating retreatment with cladribine tablets following a 2-year treatment-free interval, or (3) switching to another high-efficacy therapy with a different mode of action.38,40 Therefore, additional treatment courses may be administered based on clinical assessment at the discretion of the treating physician,42,49 and our data suggest that this is an approach that is utilized globally in a small proportion of PwMS. However, it is clear that additional evidence regarding the optimal approach to re-dosing of cladribine tablets is needed, and it should remain a subject of ongoing research. For the subset of PwMS who may require further treatment after the 2-year regimen, extended PSP support that can facilitate obtaining another course of cladribine tablets may be an important component of care.
Our study had limitations inherent to observational studies that use data from PSPs, and these need to be considered when interpreting the results. As data were not collected specifically for this study and were collected by nurses conducting telephone interviews not structured to classify data, it is likely that there was information bias due to under-reporting and misclassification (including inconsistent adverse event reporting); and some information (including reasons for discontinuation and re-dosing of additional treatment courses of cladribine) was under-recorded, which limits our ability to draw definitive conclusions for certain observations. Furthermore, the frequency and timing of telephone check-ins and data collection points varied across countries and regions. This heterogeneity may contribute to any regional variations in discontinuation and retreatment rates and represents a significant methodological limitation. In addition, in the majority of the PSPs, PwMS were only contacted if needed after Year 2, which might have resulted in underestimation of discontinuation rates.
Selection bias cannot be excluded in patients enrolled in the PSP, as those who choose to participate could have different baseline characteristics compared with non-participants. Enrolled patients might be more motivated and proactive in managing their health, which can positively influence adherence. However, it is unclear whether participants have less severe MS or higher disease activity, both of which could impact adherence. Another important potential limitation was the presence of immortal time bias; the latter may have arisen from the possible gap between completing the first course of therapy and initiating the second. To mitigate this bias, we calculated Year 2 dosing only for PwMS with ⩾18 months of follow-up, ensuring they had the opportunity for event occurrence. Although this may result in an overestimation of Year 2 treatment initiation, the observed rates are consistent with findings from other studies. In real-world studies, the proportion of Year 2 treatment initiation among patients prescribed cladribine tablets is notably high, ranging from approximately 91% to 97%.24,50 While these limitations are common in real-world studies, future efforts to minimize their impact in PSP-collected data studies include engaging academic researchers to provide input in the design of PSP data collection, 51 linking PSP data to other larger datasets,52,53 and utilizing predictive analytics. 54 Moreover, although a large number of PwMS from diverse regions of the world were included, our findings are not necessarily generalizable to all PwMS as there are regions of the world, including the Far East, that were not represented in this study. However, the large patient population that we included, and the fact that our study population closely approximates similar studies in age46,55,56 and sex/gender distribution, 46 suggests that our results are generalizable to a significant proportion of PwMS.
Conclusion
Real-world data from global PSPs collected from diverse regions of the world demonstrated that the vast majority of PwMS completed both short cladribine tablets courses in Year 1 and Year 2. In PwMS with long-term follow-up (⩾4 years), only a small portion received additional treatment with cladribine tablets, which suggests that at least a proportion of PwMS have durability of the treatment effect long after completion of the recommended two short treatment courses. Our results were consistent between countries/regions and demonstrate the utility of a PSP to not only support PwMS initiating and continuing cladribine tablets but also to facilitate real-world data collection that provides important insights into medication adherence in the real world.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261426842 – Supplemental material for Insights into adherence to cladribine tablets in the real world in multiple countries: results from patient support programs
Supplemental material, sj-docx-1-tan-10.1177_17562864261426842 for Insights into adherence to cladribine tablets in the real world in multiple countries: results from patient support programs by Jiwon Oh, Mavis Ayer, Raed Alroughani, Samar Farouk, Mounir Khoury, Sabrina de Souza, Michela Bossolasco, Kate Morgan, Laura Negrotto, Tracey Quinn, Amir Boshra, Joseph Youssef, Murad Al-Naqshbandi, Kelly MacDonald, Meritxell Sabidó, Jeffrey M. Muir and Berenice Silva in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
Natasza Klas, Alexander Jones, and Kathryn Ball of Springer Health+, Springer Healthcare Ltd., UK, provided medical writing support, which was funded and supported by Merck, in accordance with the Good Publication Practice 2022 Guidelines. Mariken Luca, PhD, of Merck B.V., Schiphol-Rijk, the Netherlands, an affiliate of Merck KGaA for critical review of the manuscript. All authors gave final approval of the version to be published; and agreed to be accountable for all aspects of the work.
Declarations
Supplemental material
Supplemental material for this article is available online.
