Abstract
Background:
Disease-modifying therapies (DMTs) have transformed multiple sclerosis (MS) care but are associated with substantial costs. Among high-efficacy DMTs, anti-CD20 antibodies are widely used, yet their economic value, particularly compared with different classes of DMTs, has not been comprehensively synthesized.
Objective:
To systematically review evidence on the cost-effectiveness of anti-CD20 antibodies compared with other DMTs or best supportive care (BSC) in MS.
Data sources and methods:
We searched PubMed, Embase, Web of Science, and International Network of Agencies for Health Technology Assessment (July 2025) for cost-effectiveness analyses comparing rituximab, ocrelizumab, ofatumumab, or ublituximab with other DMTs or BSC. Two reviewers independently screened studies, extracted data, and assessed reporting quality using the Consolidated Health Economic Evaluation Reporting Standards 2022 checklist. All costs were converted to 2024 US dollars and adjusted for inflation.
Results:
Of 92 records screened, 20 studies met inclusion criteria. Analyses were conducted in diverse but mostly high-income regions, most commonly using Markov models with long time horizons. Anti-CD20 antibodies were dominant (more effective, less costly) or cost-effective in the majority of studies, particularly when compared with platform therapies. In contrast, comparisons with other high-efficacy DMTs yielded more heterogeneous results, with immune reconstitution therapies more cost-effective in several studies. Reporting quality was generally high, although patient involvement, assessment of heterogeneity, and a health economic analysis plan were rarely addressed.
Conclusion:
These findings support the use of anti-CD20 antibodies as an economically reasonable option in many healthcare settings, particularly when compared with platform therapies, while underscoring that cost-effectiveness relative to other high-efficacy treatments is context-dependent. The lack of data from low- and middle-income countries and limited transparency in Health Technology Assessment reports represent major limitations. Future research should prioritize context-specific evaluations and promote full disclosure of economic data to strengthen the evidence base for clinically and economically informed reimbursement decisions in MS.
Trial registration:
The study was prospectively registered with PROSPERO (ID: CRD420251109958).
Keywords
Introduction
Multiple sclerosis (MS) is one of the leading causes of nontraumatic disability in young adults. 1 Its chronic course places a considerable burden not only on people with MS (PwMS) and their families but also on healthcare systems and society at large. In Europe, the average annual societal costs per PwMS have been estimated at approximately USD 46,000. 2 In the United States, annual medical costs have been estimated at USD 66,000 and annual indirect and nonmedical costs at USD 18,500 per PwMS. 3
Disease-modifying therapies (DMTs), which aim to reduce relapse rates and slow disability accumulation, have transformed MS management over the past decades. 4 DMTs are effective in preventing relapses and reducing disability accumulation over time, resulting in meaningful clinical benefits for patients by preserving neurological function and maintaining quality of life. 5 At the same time, they are associated with high drug acquisition costs: In theUnited States, more than half of direct medical costs for PwMS are attributable to DMTs. 3 Direct medical costs have risen substantially over the last years and are driven largely by escalating costs of DMTs. 6
Previous systematic reviews evaluating the cost-effectiveness of DMTs in MS have reported heterogeneous results.6–10 For certain drug classes, such as interferons, these reviews have even reached contradictory conclusions regarding cost-effectiveness. While one recent review described interferons as predominantly cost-effective, 9 another arrived at the opposite conclusion. 8 Overall, none of the existing reviews were able to draw consistent across the therapies examined. One potential explanation for this heterogeneity is that most reviews assess all available DMTs collectively, without accounting for substantial differences in therapeutic efficacy. By pooling therapies with distinct clinical benefits and cost profiles, the resulting estimates may mask the value of specific high-efficacy options. In this review, we therefore focus exclusively on B-cell-depleting anti-CD20 monoclonal antibodies—a subclass of highly effective DMTs with strong evidence for a higher clinical benefit compared to platform therapies. 5 We hypothesized that concentrating on this treatment class may reveal a more favorable cost-effectiveness profile compared with analyses that look at all available DMTs at the same time.
This work aims to provide a comprehensive overview of all currently available cost-effectiveness studies evaluating anti-CD20 antibodies in the treatment of MS. By synthesizing the existing evidence, the review seeks to inform healthcare practitioners as well as decision-makers, including payers and regulatory authorities, about the economic value of this class of therapies. In addition, identifying gaps and inconsistencies in the literature can help guide future research priorities, supporting researchers and health technology assessment (HTA) bodies in focusing on the most pressing unanswered questions in this field.
Methods
Registration and reporting standards
The study was prospectively registered with PROSPERO under the following ID: CRD420251109958 (https://www.crd.york.ac.uk/PROSPERO/view/CRD420251109958). 11 Registration was performed prior to study selection to ensure methodological transparency and to minimize the risk of selective reporting. The conduct and reporting of this review adhere to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. 12
Eligibility criteria
To address the research question, eligibility criteria were developed following the PICOS-scheme. 13 The relevant population, intervention, comparators, outcomes, and study types were defined as follows: (1) population: adults diagnosed with MS (regardless of disease course); (2) intervention: anti-CD20 antibodies European Medicines Agency and Food and Drug Administration approved or commonly used off-label for the treatment of MS (Rituximab, Ocrelizumab, Ofatumumab, Ublituximab); (3) comparator: other DMTs or best supportive care (BSC); (4) outcome: incremental cost-effectiveness ratio (ICER), incremental cost-utility ratio, or separately listed incremental costs and benefits; (5) study type: fully published economic analyses comparing the costs and benefits of two or more different therapies. Excluded were conference abstracts or documents in which relevant information (costs, effectiveness results) is redacted.
Search strategy and study selection
A systematic literature search was conducted on the following biomedical and economic databases: PubMed, Web of Science, Embase (via Ovid), and database of the International Network of Agencies for HTA on July 21, 2025. Results were limited to studies published in the year 2008 or later, the year in which the results of the first randomized controlled trials testing anti-CD20 antibodies in MS were published. The full search strategy for each database is found in the Supplemental Material.
After removal of duplicates, studies were independently screened by two reviewers (J.H., M.Z.). In the first step, title and abstract were screened and articles were categorized in exclusion or possible inclusion. In the second step, full articles were screened for definitive inclusion or exclusion. Disagreements between the two reviewers were dissolved by discussion with a senior reviewer (O.S.).
Data extraction and quality assessment
A Google Sheets form was created for data extraction and organization. The following variables were extracted from each included study: first author, year of publication, title, journal, country, type of analysis (cost-effectiveness analysis or cost-utility analysis), type of economic model, perspective, time horizon, intervention, comparators, discounting, base-case results, willingness-to-pay (WTP) thresholds used, results of deterministic sensitivity analysis with key drivers of results, and results of probabilistic sensitivity analyses (PSA). For studies that used more than one comparator, the base-case ICER values for each comparator were included separately. Data extraction was performed by one reviewer (J.H.) and validated by a second reviewer (M.Z.). Again, a senior reviewer (O.S.) was available for discussion in case of disagreement. All currency values not reported in US dollars were converted to US dollars using exchange rates from the respective year of the study. All currency values were then adjusted to inflation using the US consumer price index to 2024 US dollar values. The single HTA report included in this review contained two separate analyses: one submitted by the sponsor and one revised by the HTA agency. For this review, data from the agency’s reanalysis were extracted. The complete data extraction table is found in the Supplemental Material.
Quality of reporting was assessed using the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist. This checklist comprises 28 key criteria designed to ensure transparent, consistent, and comprehensible presentation of health economic evaluations. 14 Each item was rated as either fulfilled (1), not fulfilled (0), or not applicable (n/a).
Results
Study selection and characteristics
After removal of duplicates, 92 records were screened. Of these, 52 were excluded based on title or abstract. The remaining 40 records (published articles and HTA reports) underwent full-text review. Three of those records referred to HTA reports that were not publicly accessible, 17 articles were excluded after screening. A frequent reason for exclusion, particularly among HTA reports, was redacted cost information. Twenty studies met the eligibility criteria; one was a HTA report 15 and the remainder were published articles.16–34 The selection process is illustrated in Figure 1.
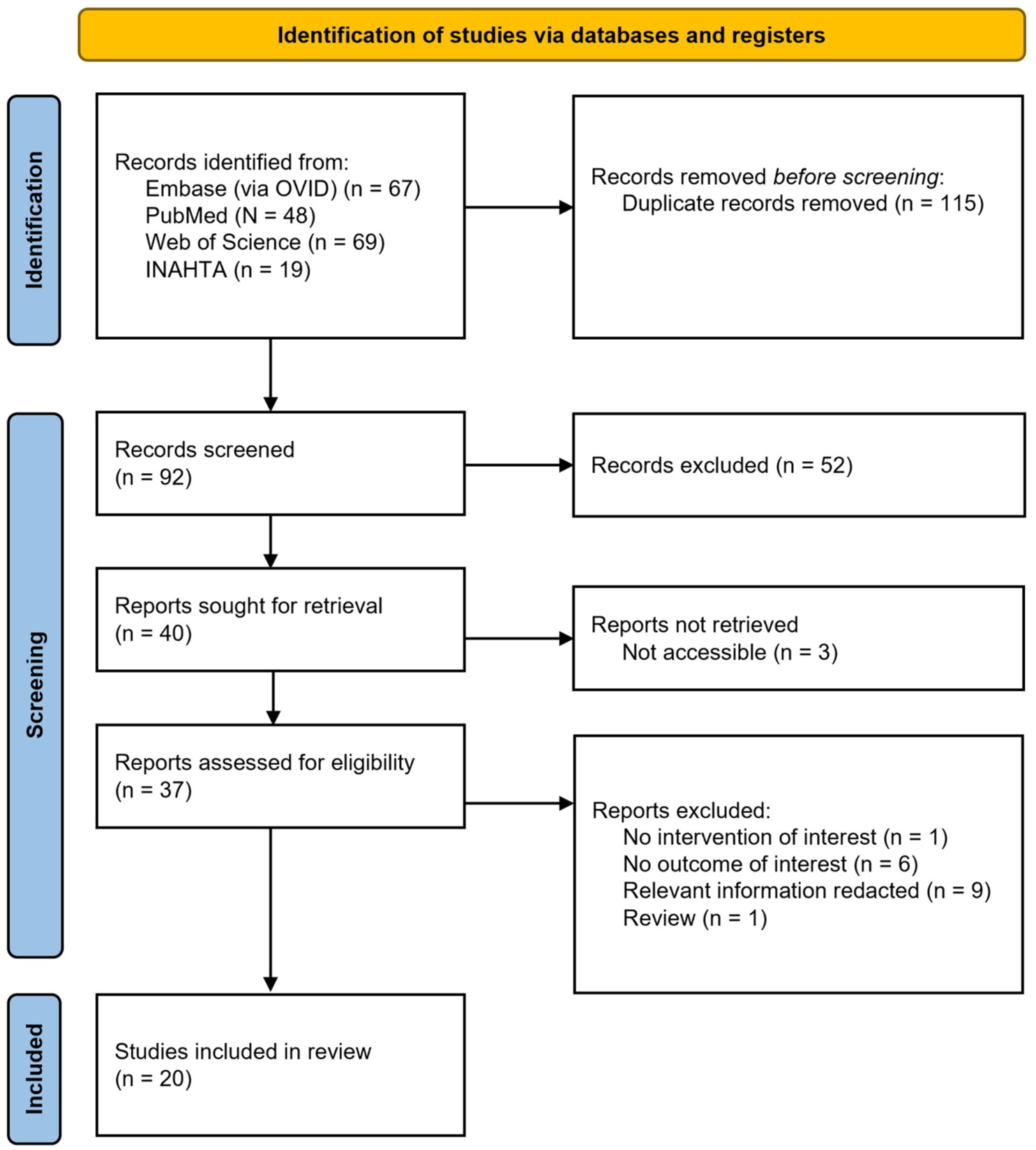
PRISMA diagram of the study selection process.
The included studies were published between 2017 and 2025. Study countries/regions included Canada (n = 2), Europe (n = 4), Middle East (n = 6), New Zealand (n = 1), South America (n = 2), Thailand (n = 1), and the United States (n = 4). Most studies relied on Markov models with a long time horizon of ⩾10 years, reflecting the chronic nature of MS and the ability of such simulations to project long-term outcomes. In contrast, the three real-world evidence studies adopted shorter time horizons (⩽5 years), as it is not feasible to follow actual patients over a longer time span within a single study. This inherent limitation of real-world observation may reduce the comparability of their results with simulated lifetime model projections.
A societal perspective including nonmedical costs and productivity loss was used in 8/20 (40%) studies; the remaining 12/20 (60%) were limited to a payer perspective. Anti-CD20 antibodies assessed as intervention or comparator included ocrelizumab (n = 13), ofatumumab (n = 5), and rituximab (n = 5). None of the studies analyzed the cost-effectiveness of the recently approved ublituximab. The study population consisted predominantly of adults with relapsing-remitting MS; one study each focused on adults with secondary progressive MS (SPMS) and primary progressive MS (PPMS). Study characteristics are summarized in Table 1.
Study characteristics.
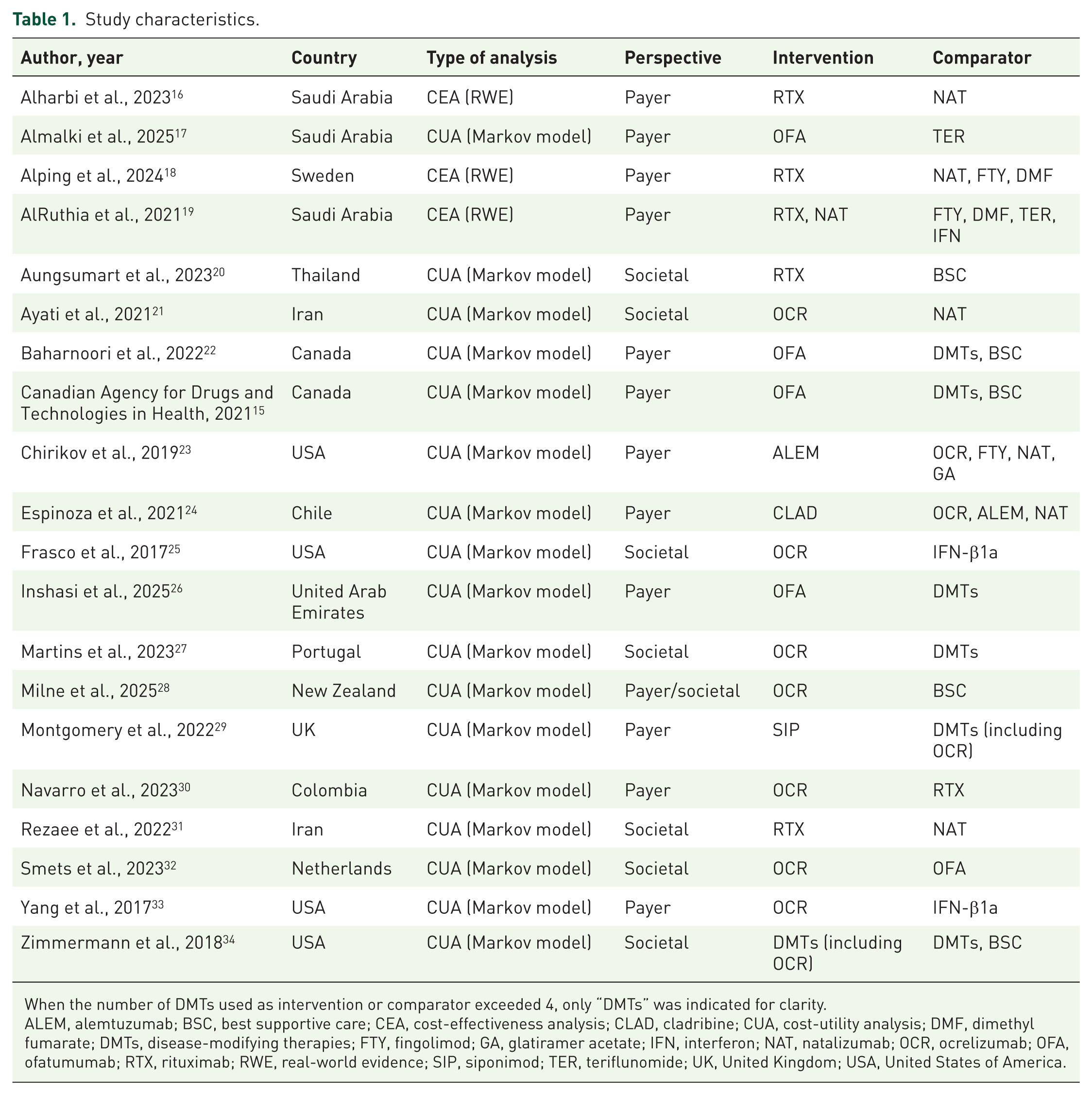
When the number of DMTs used as intervention or comparator exceeded 4, only “DMTs” was indicated for clarity.
ALEM, alemtuzumab; BSC, best supportive care; CEA, cost-effectiveness analysis; CLAD, cladribine; CUA, cost-utility analysis; DMF, dimethyl fumarate; DMTs, disease-modifying therapies; FTY, fingolimod; GA, glatiramer acetate; IFN, interferon; NAT, natalizumab; OCR, ocrelizumab; OFA, ofatumumab; RTX, rituximab; RWE, real-world evidence; SIP, siponimod; TER, teriflunomide; UK, United Kingdom; USA, United States of America.
Overall, the included studies show substantial methodological diversity in model type, perspective, time horizon, and comparator choice, factors likely to influence the reported cost-effectiveness of anti-CD20 antibodies.
Cost-effectiveness of anti-CD20 antibodies
The base-case results of the included studies are presented in Table 2. Because comparator choice can substantially influence cost-effectiveness outcomes, results are grouped according to comparator type: (1) Class-level comparison (e.g., monoclonal antibodies vs oral DMTs), (2) head-to-head comparisons between anti-CD20 antibodies, (3) comparisons between an anti-CD20 antibody and other DMTs or BSC.
Base-case cost-effectiveness results.

All currency values were converted to USD and adjusted for inflation as described in the “Methods” section.
ALEM, alemtuzumab; BSC, best supportive care; DMF, dimethyl fumarate; DMTs, disease-modifying therapies; FTY, fingolimod; GA, glatiramer acetate; GDP, ICER, incremental cost-effectiveness ratio; IFN, interferon; mAbs, monoclonal antibodies; NAT, natalizumab; OCR, ocrelizumab; OFA, ofatumumab; PPMS, primary progressive multiple sclerosis; QALY, quality-adjusted life year; RRMS, relapsing-remitting multiple sclerosis; RTX, rituximab; SIP, siponimod; SPMS, secondary progressive multiple sclerosis; TER, teriflunomide; UK, United Kingdom; USA, United States of America; WTP, willingness-to-pay.
One study conducted a class-level comparison. 19 Two studies conducted head-to-head comparisons of anti-CD20 antibodies.30,32 The remaining 17 compared at least one anti-CD20 antibody with a therapy from another drug class or with BSC.
Class-level comparison
In the one study conducting a class-level comparison, treatment with monoclonal antibodies (natalizumab or rituximab) was compared to oral DMTs (fingolimod, dimethyl fumarate, and teriflunomide) and interferons. 19 The outcome was a composite measure including clinical relapses, disability progression, and MRI activity.
Treatment with monoclonal antibodies was both more effective and more costly than the alternative regimens. Compared with oral medications, monoclonal antibodies reduced the odds of the composite outcome by 84.3%; compared with interferons, the reduction was 95%. Associated total costs amounted to USD 16,763 for monoclonal antibody therapy, compared with USD 15,381 for interferons and USD 11,045 for oral medications. Although costs can differ significantly between natalizumab and rituximab, the analysis did not disaggregate costs by individual therapy; therefore, total costs may have been largely driven by natalizumab treatment. The differences translated into ICERs of USD 3819 per composite event prevented for monoclonal antibodies (including rituximab) versus interferons and USD 19,473 versus oral medications.
Since the analysis did not use quality-adjusted life years (QALYs) as outcome measure, the ICERs cannot be compared against conventional WTP thresholds, limiting the ability to draw conclusions within standard cost-effectiveness frameworks.
Comparison of two anti-CD20 antibodies
There were two included studies that compared the cost-effectiveness of two different anti-CD20 antibodies. In one of these studies, ocrelizumab was both more effective and more expensive than rituximab. The ICER was USD 89,294/QALY, meaning that in the Colombian context of the study, ocrelizumab could not be considered cost-effective compared to rituximab. 30 In the second study, ocrelizumab was both more effective and more expensive than ofatumumab. With an ICER of USD 20,080/QALY, ocrelizumab could be considered cost-effective in the Dutch context of the study. However, the PSA of the study revealed a high degree of uncertainty in this result, so it could not be concluded with certainty that ocrelizumab is actually cost-effective compared to ofatumumab. 32
In three further studies comparing a variety of DMTs, two different anti-CD20 antibodies were among the therapies compared (ofatumumab and ocrelizumab).15,22,26 Here, the QALYs generated by the two drugs were very similar. In terms of cost, both antibody therapies were among the more expensive options within the DMTs examined.
Comparison of an anti-CD20 antibody with other DMTs
The majority (17/20) of the included studies compared the cost-effectiveness of at least one anti-CD20 antibody to at least one other DMT. Figure 2 illustrates the base-case ICER results of these analyses categorized by comparator therapy (platform therapy or high-efficacy DMT). In seven of those studies, the anti-CD20 antibody was dominant against the comparator, meaning it was less costly and more effective.16,18,20,21,25,31,33 In four other studies, the anti-CD20 antibody was considered cost-effective compared to the comparator therapies, taking into account the respective WTP threshold mentioned in the study. This means that the additional costs per QALY gained were below the WTP threshold, the costs of which are considered acceptable in the country where the study was conducted.17,22,27,34 In a study that used a number of comparators, the anti-CD20 antibody was dominant or cost-effective compared to all comparator therapies except cladribine. 26

Base-case ICER results of studies comparing an anti-CD20 antibody with other DMTs. Results are grouped by comparator type (platform therapies, top; high-efficacy DMTs, bottom). Most ICER values fall below commonly used WTP thresholds (50,000 and 100,000 USD per QALY). Green diamonds indicate dominance of the anti-CD20 antibody (more effective and less costly), whereas red diagonal crosses (saltires) indicate that the anti-CD20 antibody is dominated (less effective and more costly).
In three studies, the anti-CD20 antibody was dominated by another DMT, that is, the other therapy was both less costly and more effective. The dominant comparator was alemtuzumab in the first of these studies 23 and both alemtuzumab and cladribine in the second. 15 In the third study, a combined group of cladribine and natalizumab was both less costly and more effective, meaning that the anti-CD20 antibody was extendedly dominated. 24
In one study focusing on people with SPMS, an anti-CD20 antibody was used as the comparator, and the intervention therapy—siponimod—was deemed cost-effective. 29 Finally, one study conducted in people with PPMS reported divergent results depending on the chosen perspective: the anti-CD20 antibody was considered cost-effective from a societal perspective but not from a healthcare payer perspective. 28
The studies that compared an anti-CD20-antibody to different DMTs were further categorized by comparator type into those that used a platform therapy or BSC as comparator and those that used a high-efficacy therapy as comparator, as illustrated in Figure 2. For studies that used more than one comparator, the ICER values of every comparator versus the anti-CD20-antibody were separately included in the analysis. Of 23 comparisons against a platform therapy or BSC, the anti-CD20-antibody was dominant or the ICER below commonly used WTP-thresholds in 17/23 (74%) comparisons (WTP-threshold of USD 50,000) and 20/23 (87%) comparisons (WTP-threshold of USD 100,000).
In the 12 comparisons against another high-efficacy therapy, the anti-CD20-antibody was dominant or below a WTP-threshold of USD 100,000 in 7/12 (58%) comparisons and dominated or above the USD 100,000 WTP-threshold in 5/12 (42%) comparisons.
Quality of reporting
Quality of reporting evaluated by using the CHEERS checklist was generally high. Results are shown in Table 3. While most items were reported according to recommendations by all included studies, a few items were only reported by a minority of the studies. Areas recommended by the CHEERS statement but not reported routinely were a health economic analysis plan, characterizing heterogeneity and distributional effects and patient involvement.
Evaluation of report quality using the CHEERS checklist.
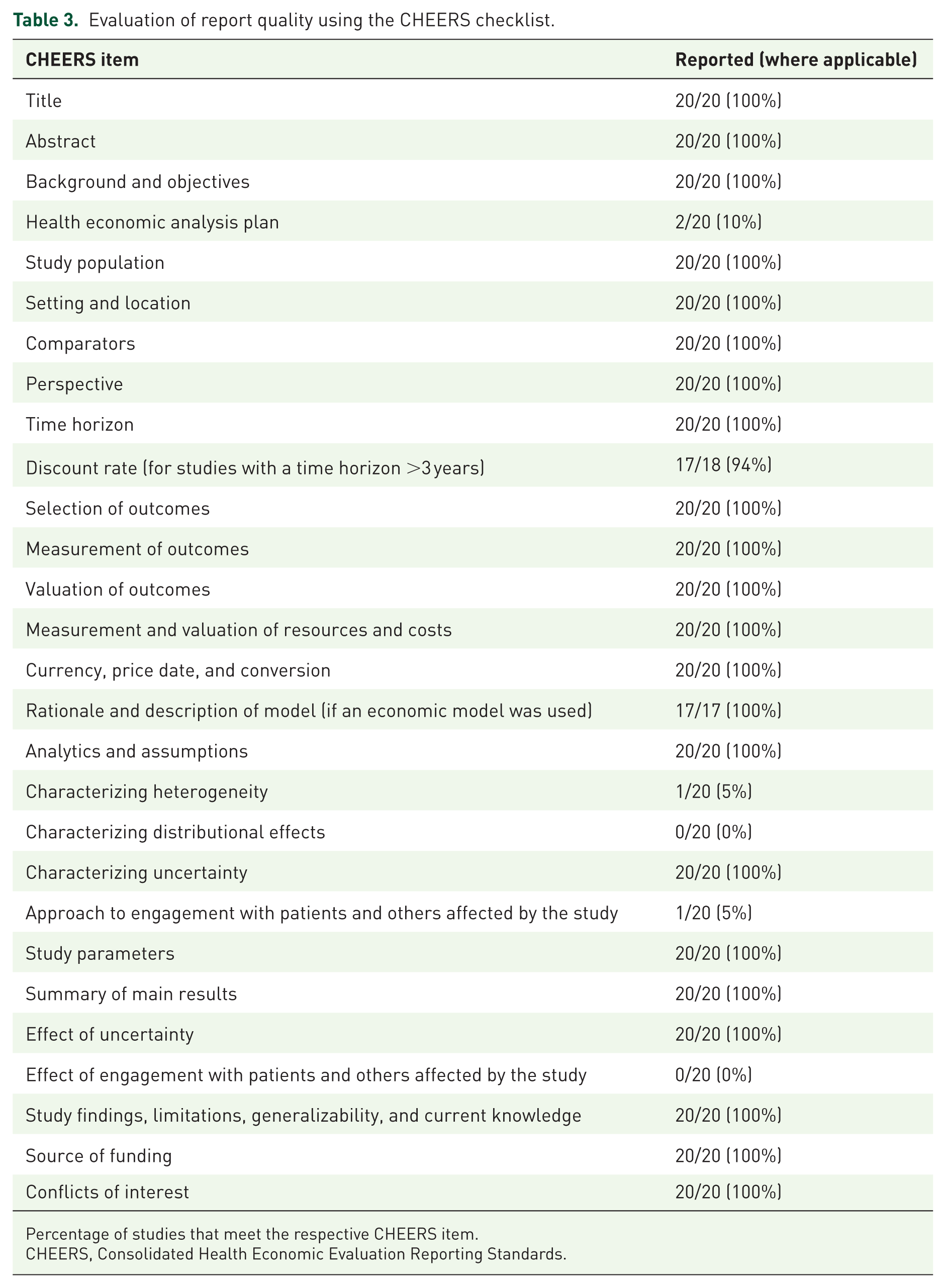
Percentage of studies that meet the respective CHEERS item.
CHEERS, Consolidated Health Economic Evaluation Reporting Standards.
Discussion
We systematically reviewed the literature regarding the cost-effectiveness of anti-CD20 antibodies in MS therapy. To our knowledge, this is the first review focusing exclusively on this clinically highly effective and commonly used drug class of DMTs. Despite substantial heterogeneity across studies in terms of healthcare setting, perspective, time horizon, and cost inputs, anti-CD20 antibodies were generally found to be cost-effective or dominant, particularly when compared with platform therapies. A quantitative meta-analysis was not feasible due to the context-specific nature of cost-effectiveness estimates and heterogeneity in model structures and assumptions. 35
The favorable economic profiles observed in many studies suggest that cost-effectiveness in MS is influenced not only by drug acquisition costs but also by long-term clinical outcomes. In chronic diseases such as MS, preventing disability progression may generate cumulative health gains and downstream cost savings. This concept aligns with the notion of “front-loading” costs, whereby higher early treatment costs may be offset by reduced long-term disability-related expenditures.36,37 Our findings are consistent with this paradigm, although the magnitude of economic benefit depends strongly on healthcare system characteristics and cost structures.
A potential economic disadvantage of anti-CD20 antibodies is the requirement for ongoing treatment over time. While extended dosing intervals or treatment discontinuation may be feasible in selected patients, most individuals require repeated administrations across the disease course.38,39 In contrast, immune reconstitution therapies (IRTs), which are administered in short, pulsed courses, may achieve sustained disease control with fewer treatment cycles. 40 Accordingly, cladribine and alemtuzumab were reported to be cost-effective compared with anti-CD20 therapies in several studies.15,23,24,26
However, economic advantages of IRTs must be interpreted in light of clinical applicability and safety. Alemtuzumab, despite high efficacy, is associated with substantial risks and is therefore reserved for selected patients.41,42 Cost-effectiveness results alone should not justify its preferential use over anti-CD20 antibodies. Cladribine, with a more favorable safety profile and limited treatment duration, may represent a more clinically realistic comparator and potential cost-efficient alternative in selected settings.
The analytical perspective can substantially influence study results. While most analyses reported favorable cost-effectiveness of anti-CD20 therapies, regardless of whether a healthcare or societal perspective was chosen, individual studies found fundamental differences depending on perspective choice. 28 Reporting quality was generally high, with good adherence to the CHEERS checklist, although patient involvement, assessment of heterogeneity, and prespecified economic analysis plans were infrequently reported. 7
Several limitations should be acknowledged. Transferability of cost-effectiveness results across countries is generally limited due to differences in healthcare systems, pricing, and reimbursement structures. 43 Since only a few analyses were available from individual countries, the overall assessment was not carried out separately for each country. In addition, evidence from low- and middle-income countries remains scarce. One notable exception demonstrated cost-effectiveness of biosimilar rituximab in a resource-limited setting with a low WTP threshold. 20 As patent protection for branded anti-CD20 antibodies expires, the introduction of biosimilars may further improve cost-effectiveness. The exclusion of HTA reports with redacted cost data and the limited evidence in progressive MS phenotypes further restrict generalizability.
Implications for clinical practice
Therapeutic decisions in MS are primarily guided by efficacy, safety, and patient-specific factors, although economic constraints are increasingly relevant in clinical practice. 44 We do not suggest that cost-effectiveness should override clinical judgment. Rather, our systematic review highlights that, in many healthcare settings, the use of anti-CD20 antibodies, particularly compared with platform therapies, appears economically reasonable and aligned with contemporary treatment strategies favoring early high-efficacy therapy. Consideration of cost-effectiveness evidence may therefore help bring together clinical and economic sustainability.
In contrast, comparisons between anti-CD20 antibodies and other high-efficacy DMTs yielded heterogeneous results, largely reflecting differences in pricing and national reimbursement structures. These findings underline that economic conclusions should not be generalized across countries and should not be used to justify preference for therapies with less favorable safety profiles solely on cost grounds.
Conclusion
From a health-economic perspective, anti-CD20 antibodies generally demonstrate favorable cost-effectiveness compared with platform therapies across a range of healthcare systems. When compared with other high-efficacy DMTs, results are more heterogeneous and strongly influenced by model assumptions like varying drug acquisition costs, uncertainty about long-term treatment effectiveness, and analytical perspective.
For clinical practice, these findings support the notion that use of anti-CD20 antibodies, when clinically indicated, does not appear to conflict with economic considerations in most high-income settings. However, cost-effectiveness evidence should be interpreted in a country-specific context and should complement, rather than replace, clinical decision-making based on efficacy, safety, and patient preference. Future research should prioritize transparent, context-specific analyses.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261426816 – Supplemental material for Economic value of anti-CD20 therapies in multiple sclerosis: a systematic review of cost-effectiveness studies
Supplemental material, sj-docx-1-tan-10.1177_17562864261426816 for Economic value of anti-CD20 therapies in multiple sclerosis: a systematic review of cost-effectiveness studies by Johannes Heinemann, Marco Zirkel, Oliver Schoeffski, Rick Dersch and Heinz Wiendl in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The idea for this review arose as part of a master’s thesis for the Master of Health Business Administration program at Friedrich-Alexander University Erlangen-Nuremberg.
Authors’ note
All authors have seen and approved the submission of this manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
