Abstract
Background:
The quality and clinical outcomes of mechanical thrombectomy (MT) performed at thrombectomy-capable stroke centers (TSCs) versus comprehensive stroke centers (CSCs) remain insufficiently characterized.
Objective:
To compare MT outcomes between TSCs and CSCs and to develop and externally validate an online tool for individualized prognosis and decision support.
Design:
Retrospective cohort study including derivation and external validation cohorts from multiple stroke centers.
Method:
Patients with anterior circulation large vessel occlusion who underwent MT within 24 h were analyzed. Inverse probability of treatment weighting (IPTW) and multivariable logistic regression estimated the effects of stroke center certification. Sensitivity analyses using alternative model specifications, patient subsets, and predefined subgroups assessed robustness and heterogeneity. A prognostic model was developed using least absolute shrinkage and selection operator regression after IPTW, externally validated using 2023–2024 data from different centers, and deployed as a Shiny-based online tool predicting 90-day modified Rankin Scale outcomes (0–2 for independence, 0–5 for survival).
Results:
The median age was 69 years (interquartile range (IQR) 60–77) in the derivation cohort (n = 975) and 72 years (IQR 64–80) in the validation cohort (n = 484). Functional outcomes and survival probabilities were similar between cohorts. After IPTW and adjustment, logistic regression showed CSCs were associated with higher 3-month survival (OR, 1.70 (95% CI: 1.31–2.22)). Sensitivity and subgroup analyses validated findings. The online prediction model, incorporating eight variables, demonstrated strong discriminative ability for functional outcomes (C-statistic 0.77 (95% CI: 0.73–0.81)) and survival (C-statistic 0.77 (95% CI: 0.71–0.82).
Conclusion:
CSCs were significantly associated with a higher probability of survival compared to TSCs, while no significant difference was observed in favorable functional outcomes. An online multivariable model could predict clinical outcomes and guide decision-making between TSCs and CSCs in routine clinical practice.
Plain language summary
A stroke happens when a blood clot blocks blood flow to the brain. One treatment, called mechanical thrombectomy (MT), removes the clot with a special device. Not all hospitals provide the same level of stroke care. Some are thrombectomy-capable stroke centers (TSCs), while others are comprehensive stroke centers (CSCs), which usually have more resources and specialists. We studied nearly 1,000 stroke patients who received MT within 24 hours of stroke onset. About half were treated at TSCs and half at CSCs. We also checked our findings in another group of almost 500 patients. We found that patients treated at CSCs were more likely to survive three months after stroke compared with those treated at TSCs. However, the chances of recovering independence—being able to live without major disability—were about the same at both types of centers. To help doctors and patients, we created a simple online tool. By entering information such as age and stroke severity, it can estimate an individual’s likelihood of survival and recovery. In short, survival after thrombectomy is higher in comprehensive stroke centers, but functional recovery is similar. Our tool may help guide patients, families, and clinicians when considering treatment options.
Introduction
Stroke centers are critical hubs for time-sensitive stroke interventions. Robust evidence demonstrates a volume–outcome relationship in endovascular therapy, with high-volume centers achieving superior procedural results.1–4 Institutional certification has been introduced as a quality assurance mechanism, shown to reduce mortality and mitigate resource disparities in community hospitals.5,6 This credentialing framework stratifies mechanical thrombectomy (MT)-capable facilities into two tiers: thrombectomy-capable stroke centers (TSCs) focusing on acute interventions, and comprehensive stroke centers (CSCs) providing full-spectrum cerebrovascular care. 7 While certification has been associated with increased thrombolysis utilization, 8 its specific impact on clinical outcomes following MT for large vessel occlusion (LVO) remains poorly characterized—a critical knowledge gap given MT’s expanding role in stroke management.
Despite approximately 50% of MT patients attaining functional independence, substantial interindividual outcome variability persists, influenced by complex interactions between procedural factors, biological responses, and post-interventional care.9–11 Previous research has demonstrated that well-validated prognostic models outperform clinicians in accurately predicting patient prognosis.10,12–14 However, these studies predominantly focus on CSCs, with limited data from TSCs. Notably, existing validation studies predominantly derive from CSC cohorts, leaving TSC-specific predictive accuracy unverified. This oversight is clinically consequential given the distinct operational profiles: TSCs excel in rapid thrombectomy delivery but require patient transfers for advanced neurosurgical/neurocritical care, whereas CSCs provide integrated management from acute intervention through rehabilitation. 15 Current comparative analyses remain superficial, focusing on service availability rather than quantifying outcome differentials attributable to center-level capabilities.
The tiered certification system thus creates divergent care pathways: TSCs deliver time-critical MT with specialized stroke teams and basic rehabilitation, while CSCs provide comprehensive multidisciplinary services, including advanced surgeries and neurocritical care. Inter-center transfers from TSCs may introduce delays that offset MT’s initial benefits. Crucially, beyond quantifying center-level differences, practical tools that translate comparative prognostic models into bedside decision support are lacking. To address this gap, we used a population-based prospective registry from 37 stroke centers across 20 provinces (11 CSCs and 26 TSCs) to develop and validate the first comparative prognostic models for functional outcomes and survival in LVO patients undergoing MT. To implement these models in a validated, user-friendly Shiny-based online tool to aid clinicians in triage and transfer decisions.
Subjects and methods
This study was designed, conducted, and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline. It is a nationwide, prospective, observational multicenter study evaluating outcomes of mechanical thrombectomy and the performance of a prediction model across thrombectomy-capable and comprehensive stroke centers. The study was registered at ClinicalTrials.gov (NCT05988125, registered on October 18, 2021).
Derivation and validation cohorts
We formed a derivation cohort using the Endovascular Treatment for Acute Ischemic Stroke in the database. ETERNITY is a national prospective registry conducted from November 2021 to December 2022 in 37 hospitals across 20 provinces, including 11 CSCs and 26 TSCs. The Supplemental Methods outline the key standards and distinctions between TSCs and CSCs.
The study population was consecutive adult patients with AIS due to LVO in the anterior circulation treated with MT. The external validation cohort from four medical centers located in different provinces of China (two primary and two comprehensive) includes 484 patients from January 2023 to December 2023. The external validation cohort was derived from a prospective study across four geographically diverse centers in China using identical inclusion criteria. It comprised patients from two participating centers (sampled from a subsequent period) and two independent centers, with strict verification to ensure no overlap with the derivation cohort.
Patient selection criteria for MT in TSC and CSC: 1. Diagnosis of AIS caused by imaging-confirmed intracranial LVO; 2. Patients whose onset-to-presentation time surpasses 6 h should have CTP and meet the DAWN 16 or DEFUSE’s 17 criteria; 3. ASPECT score ⩽ 6, NIHSS score ⩽ 6, or occlusion of the anterior cerebral artery should have multimodal imaging and cranial perfusion imaging with a clear salvageable hemidiaphragm greater than 50 ml or a ratio greater than 1.8.
Study population
All enrolled residents with LVO undergoing MT in stroke centers were certified by the Ministry of Health China Stroke Prevention Project Committee (CSPPC) (Figure 1).18,19 Eligible participants required according to the inclusion and exclusion criteria (eMethods 1).
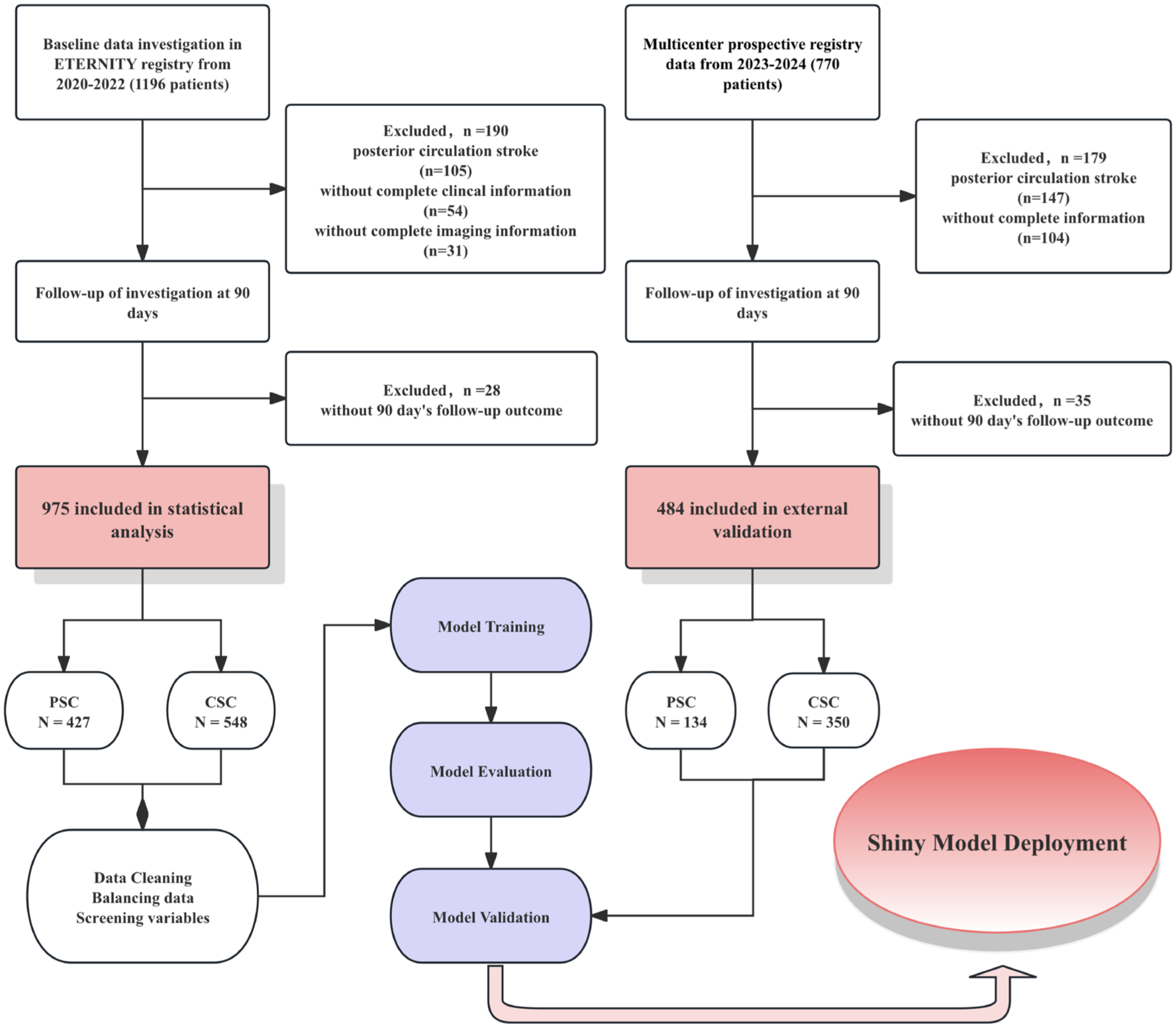
Flow chart.
Study outcome
The primary clinical outcome involved mRS assessments at 90 days, 20 which were evaluated by telephone calls or scheduled follow-up visits. Favorable functional outcome was defined as an mRS score ⩽ 2 at 90 days. Survival was defined as mRS 0–5 (alive at 90 days), which complements the functional outcome endpoint by capturing survival irrespective of disability level. Successful reperfusion was defined as eTICI 21 2b or higher. Safety outcomes included symptomatic intracranial hemorrhage (sICH) and mortality at 90 days. sICH was defined as an increase of ⩾4 points in the NIHSS score, according to the Heidelberg criteria. 22
Candidate predictor variables
Candidate predictor variables for our models were identified based on recent literature, expert opinion, clinical relevance, and potential predictor variables identified prior to and during MT with LVO.9,14,23 All sites received standardized training on an electronic data capture system for consistent data entry and electronic signatures, with blinded statisticians conducting central quality checks to ensure data consistency and completeness.
Baseline patient characteristics, including age, sex, smoking, and relevant clinical findings, were comprehensively analyzed. Presenting factors included admission NIHSS score, primary occlusion site, and imaging techniques independently evaluated by neurologists, neuroradiologists, and neuro-interventionalists following established protocols. Treatment variables encompassed symptom onset-to-treatment time (OTT), door-to-puncture time (DPT), puncture-to-reperfusion time (PTR), frontline techniques for mechanical thrombectomy based on neuro-interventionist discretion, use of stent retriever, angioplasty, and intra-arterial (IA) tissue plasminogen activator (tPA) administration. Baseline CT/magnetic resonance and CT angiography/magnetic resonance angiography, digital subtraction angiography, and postprocedural CT were assessed by an imaging core laboratory, which was blinded to clinical data and outcomes. All imaging was independently evaluated by two neuro-radiologists, with a third available for adjudication if necessary. The variables with less than 20% missing values are retained, and the missing value is filled by the method of multiple interpolation. The specific missing data rates for all included variables are detailed in Supplemental eTable 1. Processing of missing values of data was supplemented in the eMethods 2. Multi-collinearity was assessed using the variance inflation factor (VIF; eTable 2).
Model derivation
The multivariable models were adjusted for age, baseline NIHSS, Acute Stroke Prognosis Early Computed Tomography Score (ASPECTS), smoking, hypertension, diabetes, stroke history, location of occlusion, intravenous thrombolysis, atrial fibrillation, OTT, DPT, PRT, tandem occlusion, stroke causative mechanism, and stroke certification for centers.
Baseline characteristics of the two groups were balanced using inverse probability treatment weighting (IPTW) based on propensity scores. In the IPTW cohort, the treatment effect was estimated using the Inverse Probability Weighted Regression Adjustment model. The balance of all covariates between groups was assessed using the standardized mean difference (SMD). 24 A smaller SMD indicates a smaller difference between covariates, leading to a better balance, particularly when SMD is less than 0.1. 25
In the IPTW-adjusted and multivariable logistic regression analysis adjusted for confounders, effect sizes were estimated using weighted logistic regression models for binary outcomes and weighted ordinal logistic regression models for shifts in the mRS score, with TSC as the reference. From these models, odds ratios (ORs) along with their 95% CIs for favorable functional outcome and survival probability were derived with PCSs as the reference.
To prevent overfitting and address collinearity, we used the least absolute shrinkage and selection operator (LASSO) regression to select the best predictors from highly correlated candidate variables. Ten-fold cross-validation determined the optimal lambda (λ), with lambda.1se chosen for simplicity and accuracy. Models were built for a favorable functional outcome and survival at 90 days.
Prediction model performance
For external validation, we used a nationwide prospective observational study from three stroke centers located in different provinces of China, all of which were not involved in the derivation cohort, ensuring no geographic or institutional overlap. The participation centers and geographical distribution are provided in eTable 3. Consecutive patients with ischemic stroke who underwent MT between January and December 2024 were screened. The same inclusion and exclusion criteria used for the derivation cohort were applied to ensure consistency in patient selection. Data from the external validation cohort were analyzed in April 2025. Model performance was assessed using discrimination and calibration metrics. 26 The updated final model was implemented in a web application that predicts functional outcomes at 90 days for individual ischemic stroke patients treated at TSCs and CSCs using routinely available information. The application displays the predicted probabilities of a favorable functional outcome and mortality based on the ETERNITY Registry.
Statistical analysis
Multiple interpolation was used for the dropouts or missing data. Continuous variables were presented as medians with interquartile ranges and compared using the Mann-Whitney test after assessing normality. Categorical variables were presented as numbers and percentages and compared using the Pearson χ² test or Fisher’s exact test, depending on theoretical numbers. IPTW was used to balance the data, as the traditional IPTW may lead to over-weighting of the sample size, so we used stabilized weights to improve the IPTW. 27 The process was performed in eMethods 3.
Sensitivity analyses
To address potential reverse causality where CSC may treat MT for patients with lower NIHSS scores, we analyzed the association, excluding those patients. We also excluded those with anterior cerebral artery occlusion to evaluate its impact. In addition, we reanalyzed the stroke center-prognosis link, excluding patients with other etiologies to test the robustness. We further performed a sensitivity analysis using 90-day mortality (mRS = 6) as the alternative survival endpoint to evaluate whether the choice of definition affected the observed center-level differences.
Finally, we examined treatment effect heterogeneity for favorable functional outcome and 90-day survival across subgroups (age, gender, NIHSS, ASPECT, OTT, and IV-tPA) and adjusted for multiple comparisons using the Bonferroni correction. The ORs and 95% CIs for the effect size of each group were computed. Calibration plot was used to verify the level of agreement between observed outcomes and predicted probabilities. Statistical analyses were performed with R statistical software, version 4.4.1 (R Foundation for Statistical Computing, Vienna, Austria). All hypothesis tests were two sided, with p < 0.05 considered statistically significant.
Sample estimation
The development of a binary outcome multivariable prediction model with 18 candidate predictor parameters requires a minimum sample size. 28 Based on prior evidence and current sample statistics, with incidence rates of 16% and 46%, we could not obtain the Cox-Snell R-squared estimate, but the reported C-statistic is 0.80. A sample size of 530 is needed to predict an mRS score of 0–2, and 938 for survival probability.
Results
Patient characteristics
From November 2021 to December 2022, a total of 975 patients with LVO who underwent MT were enrolled in the ETERNITY registry. External validation on data from 484 cases from multiple centers in different provinces in China, treated in clinical practice from Jan 2023 to Dec 2024 (Figure 1). We adjusted the scatter plots to better visualize the data distribution and linear trends. The linear relationship between continuous variables and outcomes is presented in Figure S1.
Table 1 shows the baseline characteristics of patients by stroke center after multiple imputations for missing values before and after PSM. After IPTW adjustment based on PSM, all baseline variables had a SMD of less than 10%. Probability distribution of propensity score stratified by stroke centers was presented in eFigure 2(A). SMD values at baseline variables before and after IPTV were shown in eFigure 2(B). External validation on data from 484 cases from multiple centers in different provinces in China, treated in clinical practice from Jan to Dec 2024. Median age was 72 years (IQR, 64–80), and 51.5% were male. Supplemental eTable 4 shows baseline characteristics after missing value adjustment by derivation and external validation cohort.
Clinical profile of patients with MT according to thrombectomy-capable and comprehensive stroke center before and after IPTW based on PSM.

Data are presented as median (IQR) or n (%).
ACA, anterior cerebral artery; ASPECTS: Alberta Stroke Program Early CT Score; CE, cardioembolism.; DTP, door-to-puncture; ICA, intracranial carotid artery; IPTW, inverse probability treatment weighting; LAA, large-artery atherosclerosis; MCA, middle cerebral artery; MT, mechanical thrombectomy; NIHSS, National Institutes of Health Stroke Scale; OTT, onset-to-treatment time; PRT, puncture-to-reperfusion time; PSM, propensity score matching; SMD, standardized mean difference; IQR, interquartile range.
Outcomes according to the study group
CSCs were not associated with a higher degree of favorable functional outcome compared to TSCs, both before (OR, 1.24 (95% CI: 0.96, 1.6)) and after IPTW analysis (OR, 0.876 (95% CI: 0.679, 1.13)). The shift in 90-day mRS is shown in Figure 2. In the unadjusted analysis, CSCs were associated with a higher rate of survival (86.7% vs 73.1%) and successful reperfusion (92.9% vs 79.2%) compared to TSCs. In the IPTW analysis set, CSCs remained associated with a survival benefit (OR, 1.64 (95% CI: 1.19, 2.26)), whereas the difference in successful reperfusion was no longer statistically significant (OR, 1.01 (95% CI: 0.693, 1.46)). No significant difference was observed in the rates of sICH (OR, 0.868 (95% CI: 0.553, 1.36)).
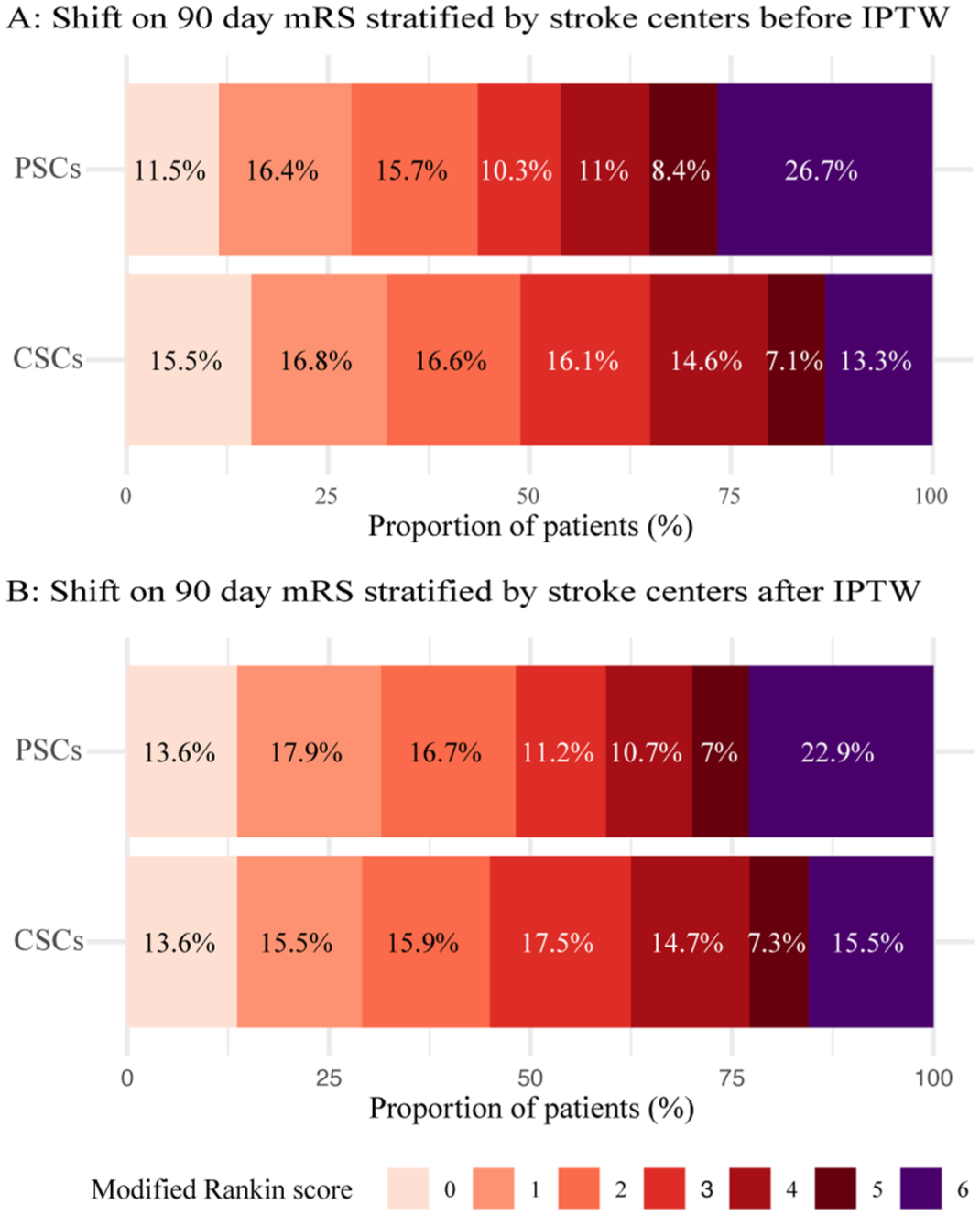
mRS scores distribution at 90 days stratified by stroke centers before and after IPTW at ETERNITY. Modified Rankin score (0: no disability to 5: severe disability and 6: death) at discharge comparing TSCs and CSCs before IPTW (a) and after IPTW (b).
After adjustment for age, baseline NIHSS, ASPECT, puncture-to-reperfusion time (PRT), intravenous thrombolysis, stroke causative mechanism, and location of occlusion, CSCs remained associated with a survival benefit (OR, 1.71 (95% CI: 1.18, 2.48)). However, there remained no significant differences between CSCs and TSCs in favorable functional outcome (mRS 0–2) (OR, 0.838 (95% CI: 0.625, 1.13)), successful reperfusion (OR, 0.998; (95% CI: 0.679, 1.46)) or sICH (OR, 0.926; (95% CI: 0.580, 1.48)) between the study groups (Table 2).
Adjusted association of efficacy and safety outcomes between different stroke centers in ETERNITY registry before and after IPTW.

The propensity score was calculated with all parameters in Table 1. IPTV-adjusted analysis: adjusted for age, baseline NIHSS, ASPECT, PRT, intravenous thrombolysis, Location of occlusion, and stroke causative mechanism.
Data are numbers of patients, with percentages in parentheses. ‡Data in parentheses are 95% CIs.
aOR, adjusted rate ratio; CSCs, comprehensive stroke centers; IPTW, inverse probability treatment weighting; mRS, modified rankle scale; OR, odds ratio; sICH, symptomatic intracranial hemorrhage; TSCs, thrombectomy-capable stroke center.
Sensitivity analyses confirmed the robustness of this result across different datasets with different subsets (eTable 5). The forest plot (eFigure 3) compares CSC and TSC in terms of favorable functional outcome (mRS 0–2) and survival (mRS 0–5). CSC significantly outperforms TSC with an overall aOR of 2.40 (95% CI: 1.73–3.32, p < 0.001), indicating a higher survival rate for patients treated at CSCs. The difference in favorable functional outcome is not statistically significant, with an aOR of 1.24 (95% CI: 0.96–1.60, p = 0.097). Subgroup Analysis: Patients with lower ASPECT scores, lower baseline NIHSS, and those without intravenous thrombolysis are more likely to benefit from CSC care (p for interaction < 0.05). Interaction tests show no significant differences between subgroups.
Model development and external validation
The results of the LASSO model are shown in eFigure 4. For the model predicting a favorable functional outcome, the number of variables associated with predicting a complete response was reduced to eight using LASSO regression. Subsequently, eight variables were included in the final model: age, baseline NIHSS, ASPECT, puncture-to-reperfusion time, intravenous thrombolysis, occlusion location, stroke causative mechanism, and stroke center. After optimism correction through internal validation, the final model demonstrated discriminative ability with C-statistics of 0.79 (95%CI 0.76–0.82) for predicting favorable functional outcomes and 0.82 (95%CI 0.79–0.86) for survival. External validation yielded comparable performance, showing C-statistics of 0.77 (95%CI 0.73–0.81) for functional outcomes and 0.77 (95%CI 0.71–0.82) for survival prediction (Table 3). Calibration analysis revealed close agreement between model predictions and actual observations, with no significant deviations in either functional outcome attainment or survival rates. Observed favorable functional outcome was similar (intercept 0.12, slope 1.04 (95% CI: 0.82–1.26)), whereas observed survival was slightly higher than predicted by the model (intercept 0.16, slope 0.77 (95% CI: 0.71–0.82)) (Figure 3).
Model performance metrics (95% CI) are presented for both derivation (n = 975) and external validation (n = 266) cohorts, with statistical comparisons using DeLong’s test for AUC equivalence.

The AUC quantifies discriminatory power across outcome probability strata, with an interpretation scale of 0.5 (random chance) to 1.0 (perfect separation). Calibration intercept quantifies systematic prediction bias (ideal = 0), with bootstrap-corrected estimates validated through the Hosmer-Lemeshow test. Calibration slope assesses predictor effect magnitude (target = 1.0), where deviations indicate overfitting (<1) or underfitting (>1) patterns.
AUC, area under the curve; C-statistic, concordance statistic; mRS, modified Rankin Scale.
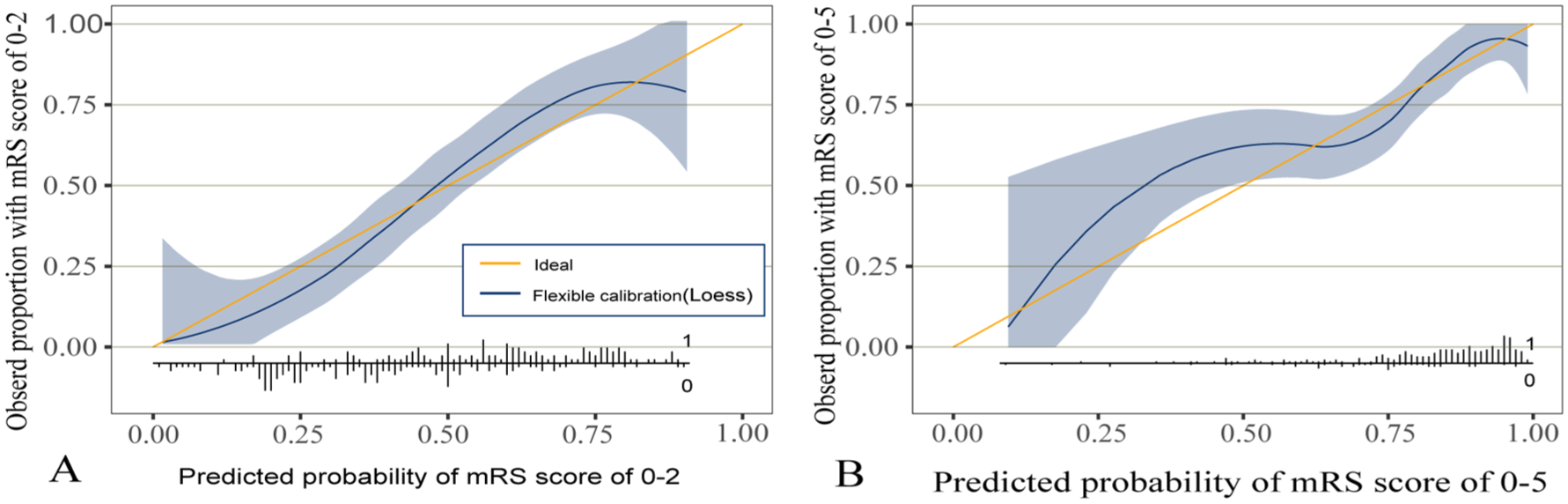
Calibration plot for (a). Favorable functional outcome (mRS 0–2) and (b). Survival status (mRS 0–5) in the external validation cohort.
Final model
The online tool, PREDICTION@ETERNITY, is available for clinical use at https://eternity123.shinyapps.io/shiny/ (temporary link) and will be maintained on a stable server to ensure long-term availability (eFigure 5). The application is intended primarily for bedside clinical support, while also serving research and quality-improvement purposes. Clinicians can enter patient-specific data such as age, NIHSS score, ASPECT score, puncture-to-reperfusion time (PRT), location of occlusion, use of intravenous thrombolysis, and stroke causative mechanism. The system predicts probabilities for different clinical outcomes between primary and comprehensive stroke centers.
Discussion
TSCs and CSCs differ significantly in their ability and performance in MT. Evaluating the prognostic impact of MT for ischemic stroke between TSCs and CSCs is crucial for patients, their families, and physicians. CSCs are equipped with advanced imaging technologies (such as multimodal MRI and CTA) and have 24/7 availability of specialized staff, including vascular neurologists, neurosurgeons, and endovascular specialists.29–32 They also have intensive care units (ICU) and neuroscience ICU to manage complex and severe stroke cases. In acute stroke treatment, CSCs administer intravenous tissue plasminogen activator (IV tPA) more frequently and promptly than TSCs, and they perform intra-arterial thrombectomy (IA therapy) more frequently. 33
Our study revealed that CSCs were associated with a higher survival, but not with favorable functional outcome at 3 months, sICH, or successful reperfusion. CSCs have more stringent enrollment criteria with equipment requirements and organizational structure than TSCs to provide more advanced life support. This apparent discrepancy—improved survival without corresponding gains in functional independence—may reflect several factors. Potential explanations include differences in neurocritical care quality, heterogeneity in post-acute rehabilitation pathways, delayed transfers from primary hospitals, and variations in patient selection, such that CSCs may treat more clinically complex patients who survive the acute event but retain significant long-term disability. In addition, residual confounding may also contribute, as unmeasured differences in premorbid status, frailty, secondary prevention adherence, socioeconomic rehabilitation access or admission glucose levels and stress hyperglycemia could bias estimates toward a survival advantage without proportional functional benefit. 34 A significant proportion of patients with acute ischemic stroke due to LVO fail to receive timely follow-up or emergency transfer to stroke centers, creating critical intervention gaps that may adversely affect long-term prognosis.35,36 Conversely, universal admission of LVO survivors to CSCs risks overutilizing specialized care capacity without clinical justification.
A potential limitation is our use of mRS 0–5 to denote survival rather than mortality (mRS = 6). We chose this definition to reflect survival irrespective of functional status and to increase power for center-level comparisons. Nevertheless, we performed sensitivity analyses using mortality (mRS = 6) as the endpoint; those results were directionally consistent with the primary analysis but less precise because of fewer events.
Several studies have shown an association between hospital MT volume and outcomes1,2,37–39; however, limitations in sample size, methodology, and representativeness have prevented these analyses from influencing treatment guidelines and policies. CSC had optimized workflows, high procedural volumes, and extensively trained and experienced operators, which may result in improved discharge outcomes and lower in-hospital mortality rates. 4 Overall, the survival advantage of CSC care is clear across most subgroups, while a favorable functional outcome shows more variability, favoring certain groups such as females and those with worse baseline stroke characteristics. This suggests that patients treated in CSCs are significantly more likely to survive with less severe disability. The most notable benefits are seen in patients with lower ASPECT scores (more severe strokes), those receiving thrombolysis, and patients with lower baseline NIHSS. Both young and old, male and female patients benefit from CSC care for survival. This risk-tiered approach enables dynamic outcome prediction and care coordination throughout post-acute transitions while providing actionable data for precision resource distribution. Elevated mortality correlates with three clinical imperatives: complication-prone profiles necessitating neurospecialist evaluation, and care pathways demanding substantial resource investment.
A multivariable model comprising seven variables demonstrated excellent discriminative ability in both internal and external validation cohorts from multiple clinical trials and routine patient practice. The model showed good discriminative performance, with C-statistics ranging from 0.77 to 0.82. Confidence intervals indicate stable performance and minimal overfitting. Although the discriminative ability is similar to existing prognostic tools, the model’s practical features enhance its clinical value. It uses routinely collected clinical variables, has been externally validated, and is available as a user-friendly online tool. These characteristics allow clinicians to estimate individualized 90-day outcomes after MT and support resource planning, discharge decisions, and post-acute care coordination. This work integrates these variables into a multivariate risk model shiny presentation, allowing for personalized estimation of endovascular treatment of acute ischemic stroke encountered in clinical practice. The predictor variables used in this model can be readily obtained after MT, making it feasible for implementation within discharge planning processes to identify patients at high risk of favorable prognosis and mortality. The model has been made available online for clinical use and demonstrates clinical outcomes between TSCs and CSCs.
Validating clinical prediction models is crucial for their development. The necessity of external validation for predictive models lies in ensuring their generalizability and reliability across diverse populations. External validation helps confirm that the model performs well not just in the original cohort but also in different settings and demographics, which is crucial for clinical applicability. Internal validation demonstrated preserved calibration fidelity, suggesting minimal overfitting risk in the derivation cohort. A critical prerequisite for clinical deployment involves multisite external validation across geographically distinct populations with varying care protocols. Notably, the model derivation leveraged China’s National Stroke Registry data, ensuring population-level generalizability through deliberate sampling stratification. The parsimonious variable set comprises routine clinical parameters (age, NIHSS, ASPECTS) and daily assessments, facilitating seamless electronic medical record integration via prebuilt clinical decision support modules. The models were validated in a distinct, external cohort and significantly extend the literature on the association between stroke center accreditation status and clinical outcomes. This approach enhances the model’s robustness and supports its implementation in various clinical settings.
Although the external validation cohort was expanded to 484 patients from multiple Chinese stroke centers, the model has not yet been tested in non-Chinese healthcare systems. Differences in MT workflow organization, operator experience, anesthesia practices, and postprocedural care across countries may limit generalizability. Future studies should validate the model in multicontinental cohorts to ensure applicability across diverse clinical environments.
Finally, limited external validation may contribute to the underestimation of the model’s performance. Clinical prediction models can also be undervalued due to factors such as overfitting, advancements in surgeons’ skills and cognitive abilities, as well as innovations in technology and equipment upgrades. To enhance the value of these models in the future, efforts should focus on continuous external validation, updating models with new data, and improving their interpretability.
Limitations
Four key limitations merit discussion. First, the observational registry-based design inherently carries selection bias risks in center allocation patterns, despite the application of IPTW to address indication bias. Second, the exclusion of thrombolysis responders (defined as NIHSS improvement ⩾4 within 2hr post-IVT without requiring MT) may attenuate thrombolytic treatment effect estimates in comparative analyses. Thirdly, although diabetes history was recorded, admission glucose and stress hyperglycemia were not, and residual confounding from unmeasured factors such as premorbid status, frailty, secondary prevention adherence, or rehabilitation access may bias survival estimates without proportional functional benefit. Finally, there is a lack of external validation for people outside China, and the validation population needs to be further expanded in the future to increase the universality of the model.
Conclusion
Patients treated at CSCs were associated with higher survival following mechanical thrombectomy, although the degree of post-stroke functional recovery was similar across centers. A multivariable model comprising eight variables demonstrated excellent discriminatory ability in both internal and external validation cohorts. The model is available online as a tool to support the prediction of clinical outcomes after MT across different stroke centers.
Supplemental Material
sj-doc-1-tan-10.1177_17562864251411020 – Supplemental material for Comprehensive versus thrombectomy-capable stroke centers: a web-based model to predict outcomes after mechanical thrombectomy
Supplemental material, sj-doc-1-tan-10.1177_17562864251411020 for Comprehensive versus thrombectomy-capable stroke centers: a web-based model to predict outcomes after mechanical thrombectomy by Shujuan Gan, Weifeng Huang, Tingyu Yi, Wenli Zhang, Xiongwei Lu, Zhiting Chen, Jinfeng Miao, Yanmin Wu, Meihua Wu, Caixia Li, Yining Yang, Jinhua Ye, Huanghuang Chen, Ying Wu, Xiaona Zhuang, Yuxin Xu, Liqun Jiao and Wenhuo Chen in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251411020 – Supplemental material for Comprehensive versus thrombectomy-capable stroke centers: a web-based model to predict outcomes after mechanical thrombectomy
Supplemental material, sj-docx-2-tan-10.1177_17562864251411020 for Comprehensive versus thrombectomy-capable stroke centers: a web-based model to predict outcomes after mechanical thrombectomy by Shujuan Gan, Weifeng Huang, Tingyu Yi, Wenli Zhang, Xiongwei Lu, Zhiting Chen, Jinfeng Miao, Yanmin Wu, Meihua Wu, Caixia Li, Yining Yang, Jinhua Ye, Huanghuang Chen, Ying Wu, Xiaona Zhuang, Yuxin Xu, Liqun Jiao and Wenhuo Chen in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
We would like to thank the sponsor from Medtronic Medical and all individuals and organizations participating in the 37 Stroke Centers.
Declarations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
