Abstract
Background:
Higher blood pressure variability (BPV) in patients suffering from acute ischemic stroke were shown to be associated with worse functional outcome and hemorrhagic transformation.
Objectives:
To assess the influence of blood pressure variability during endovascular stroke treatment on functional outcome and parenchymal hemorrhage.
Design:
We performed a post-hoc exploratory analysis of the individualized blood pressure management during endovascular thrombectomy under procedural sedation in acute ischemic stroke (INDIVIDUATE) study, which was a randomized clinical trial investigating an individualized blood pressure management strategy in comparison to a standardized treatment strategy during endovascular stroke treatment.
Methods:
Several BPV parameters, such as procedure time of systolic blood pressure (SBP) in a range of a preprocedural baseline SBP ± 10 mmHg and ±20 mmHg, maximal and minimal SBP, variance and average real variability of intraprocedural SBP values were tested for association with functional outcome and PH1/PH2 hemorrhages in the current post-hoc analysis.
Results:
Regression analyses were performed in 250 patients and revealed an association of variance of intraprocedural SBP (aOR, 1.002 (95% CI, 1.0004–1.004); p = 0.016) and average real variability of SBP (aOR, 1.105 (95% CI, 1.019–1.199); p = 0.016) with favorable outcome (modified Rankin Scale 0–2).
Conclusion:
In our study, intraprocedural BPV parameters during endovascular stroke treatment were positively associated with favorable clinical outcomes. Potential underlying mechanisms should be further explored to better understand the effects of hemodynamics during the hyperacute time frame of endovascular stroke therapy.
Trial registration:
Clinicaltrials.gov; NCT04578288.
Introduction
Many aspects of blood pressure management for stroke patients in the acute setting remain to be elucidated. One topic of interest is blood pressure variability (BPV). Studies have shown that increased variability during the acute and subacute phase in stroke patients is associated with worse functional outcome and higher mortality. Most of the recent studies were focused on the acute or subacute phase after endovascular treatment of patients with acute ischemic stroke and showed that in the early 24–72 h after intervention, higher BPV was associated with poor functional outcome,1–4 mortality, 2 and intracranial hemorrhage.5,6 Some studies suggest that BPV is associated with functional outcome in the subacute phase rather the acute phase.7,8 Studies evaluating BPV with a focus on the hyperacute intraprocedural time frame during endovascular stroke treatment are scarce but gaining further insights in this vulnerable phase of stroke treatment is important to guide intraprocedural blood pressure management. Here, we analyzed different parameters of systolic blood pressure (SBP) variability from the “Individualized blood pressure management during endovascular treatment of acute ischemic stroke under procedural sedation” (INDIVIDUATE) trial for its association with functional outcome and intracerebral hemorrhage as well as for interaction effects of different blood pressure management strategies.
Methods
Study design
This post-hoc secondary exploratory analysis was performed on data of the single-center, randomized, controlled trial INDIVIDUATE with blinded endpoint evaluation. Patients with acute ischemic stroke of the anterior circulation with a National Institutes of Stroke Scale (NIHSS) score of at least 8 and who underwent endovascular stroke treatment were recruited between 1 October 2020 and 7 July 2022 at the Heidelberg University Hospital, Department of Neurology, Germany. Participants had to meet all the following inclusion criteria: Decision for thrombectomy according to local protocol for acute recanalizing stroke treatment, age of 18 years or older, NIHSS ⩾ 8, acute ischemic stroke in the anterior circulation with isolated or combined occlusion of internal carotid artery (ICA) and/or middle cerebral artery (MCA). The informed consent by the patient him-/herself or his/her legal representative was obtained within 72 h of treatment. Patients with following criteria were excluded: Intracerebral hemorrhage, coma on admission (Glasgow Coma Scale ⩽ 8), severe respiratory instability, loss of airway protective reflexes or vomiting on admission, where primary intubation and general anesthesia is deemed necessary, and an intubated state before randomization, as well as severe hemodynamic instability. Patients were randomized in a 1:1 ratio to either keeping the intraprocedural systolic blood pressure within the target range of a baseline SBP (bSBP) ±10 mmHg (intervention) or to a standard blood pressure management (control group) where intraprocedural systolic blood pressure was to be maintained between 140 and 180 mmHg. The intraprocedural time frame was defined as the time from groin puncture to the last thrombectomy maneuver before the final reperfusion result. Baseline systolic blood pressure as defined as the first value measured at the emergency department or, if that value was not available, at the angiography suite. The lowest target range was prespecified as 110 ± 10 mmHg and the highest depended on whether the patient received intravenous thrombolysis: In patients who had received prior systemic thrombolysis therapy with recombinant tissue plasminogen activator (rtPA), the highest target range was predefined as 170 ± 10 mmHg, while the highest possible target range for patients without rtPA therapy was 190 ± 10 mmHg. Hemodynamic targets were monitored noninvasively in the patients at least every 5 min. Norepinephrine was used as the primary vasopressor medication to increase blood pressure, and urapidil was used as the primary antihypertensive medication. After successful reperfusion, patients were primarily transferred to our stroke unit; only in cases of emergent conversion to general anesthesia, they were admitted to our neurological intensive care unit. An SBP target of 100–160 mmHg was maintained for 24 h after intervention. The study results were neutral for the individualized versus standardized BP management approaches and have been published elsewhere. 9 For this post-hoc analysis we included all patients enrolled in the INDIVIDUATE trial.
Outcome and exposures
We investigated favorable functional outcome measured as modified Rankin Scale (mRS) score 0–2 at day 90 (±14 days) after the stroke and the occurrence of parenchymal hematoma type 1 and 2 (PH1 and PH2) in the postinterventional imaging studies as outcomes in this post-hoc analysis.
BPV is defined inconsistently in the existing literature. 10
We used different exposure values as BPV parameters for association analyses with the outcomes, namely the intraprocedural time spent in bSBP ± 10 mmHg and bSBP ± 20 mmHg in minutes, their respective proportion of intraprocedural time spent in the range of bSBP ± 10 mmHg and bSBP ± 20 mmHg in percent, the variance of per-patient intraprocedural SBP, the average real variability of intraprocedural SBP, and the minimal and maximal intraprocedural SBP. We also chose to investigate interaction effects for the different blood pressure management strategy (individualized vs standard).
Statistical analysis
BPV parameters, clinical covariates, and outcomes of the study cohort were described using appropriate summary statistics for the respective empirical distributions (mean, standard deviation, median, quartiles, minima, and maxima for continuous variables; absolute and relative frequencies for categorical variables). The dichotomous endpoints (favorable/nonfavorable mRS (0–2 vs 3–6) and the nonoccurrence/occurrence of PH1 and PH2) were analyzed by fitting a multivariable logistic regression model. The main model was adjusted for the covariates age, premorbid mRS score, NIHSS score at admission, preintervention Alberta Stroke Program Early CT Score (ASPECTS), and time from groin puncture to final reperfusion. We have performed additional adjusted analyses. First, we added the duration of intraprocedural infusion of urapidil and norepinephrine as the main primary vasoactive drugs as a covariate to the main model (which is designated as model 2 in Supplemental Tables 1 and 2). Second, we added intravenous thrombolysis (IVT) and onset to reperfusion time as covariates to the main model (which is designated as model 3 in Supplemental Tables 1 and 2). The BPV parameters were included as independent covariates and as interactions with the INDIVIDUATE trial’s treatment group. Covariate effects estimated for the regression models were reported as adjusted odds ratios (aORs) with 95%-confidence intervals and descriptive p values. Due to the exploratory nature of our analysis, we refrained from formal multiplicity correction of statistical tests. Therefore, all reported p-values are to be interpreted in a descriptive sense. Model fit was checked using residual plots and deemed acceptable. The data were analyzed using R version 4.4.2. 11
One of the subjects had a missing value in the ASPECTS score. We conducted 150-fold imputation using predictive mean matching and pooling according to Rubin’s rules for imputing the missing value. Twenty-six missing intraprocedural SBP values were imputed in 12 patients by the mean of the intraprocedural values before and after the missing value. Use of dichotomized mTICI (0–2a vs 2b–3) as an adjustment covariate was considered but omitted as the imbalanced distribution of modified Thrombolysis in Cerebral Infarction (mTICI) scores resulted in model instability. Analysis of occurrence of symptomatic intracerebral hemorrhage as an outcome was also considered but omitted due to very few events.
Furthermore, we performed a propensity score matching of the patients with and without favorable outcomes based on their propensity for favorable outcome modeled by the INDIVIDUATE trial’s treatment group, age, premorbid mRS, NIHSS at admission, preintervention ASPECTS, time from groin puncture to reperfusion, IVT, and time from onset to reperfusion. In order to integrate both multiple imputation and matching, we first performed matching separately on each imputed data set, subsequently calculated descriptive effect measures separately and finally pooled the effect measures. We used robust linear models for effect estimation which accounted for the cluster effects of the matched pairs as well as for the varying sample sizes of the matched sets.
Results
Of the 250 patients enrolled in the INDIVIDUATE trial, 123 were randomly assigned to the individualized treatment group and 127 to the standard treatment group. One hundred and eight (43.2%) patients were male. Mean (SD) age was 77 (12.2) years, and the mean (SD) stroke severity measured as baseline NIHSS score was 16.6 (5.2). 182 (72.8%) patients of the cohort had a prestroke mRS score of 0–2 and 186 (74.4%) patients had a history of hypertension. The number (%) of patients achieving an mRS score of 0–2 at 3 months were similar between the individualized and the standard treatment group (31 (25) vs 30 (24)) (see Table 1). Additional baseline demographics and clinical characteristics were published previously. 9
Outcome data.
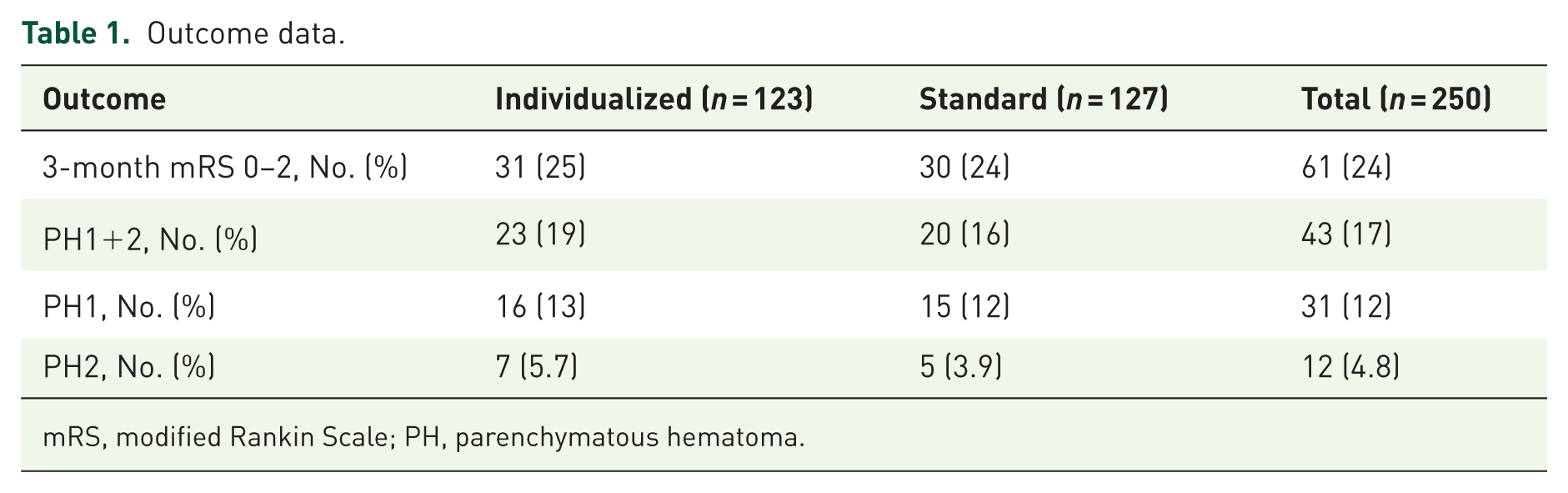
mRS, modified Rankin Scale; PH, parenchymatous hematoma.
Blood pressure profiles
Mean (SD) baseline SBP was 171 (25) mmHg in the individualized group and 171 (30) mmHg in the standard group. Mean (SD) of the per-patient mean intraprocedural SBP was 157 (19) mmHg in the individualized group and 154 (18) mmHg in the standard group. Vasopressors were used in 98 (80%) patients in the individualized group and in 82 (65%) patients in the standard group. Intraprocedural blood pressure drops of 20% occurred in 53% versus 57% of the patients in the individualized versus standard group. Further blood pressure profiles and descriptive summaries are depicted in Table 2.
Blood pressure and procedural vasoactive medication data.
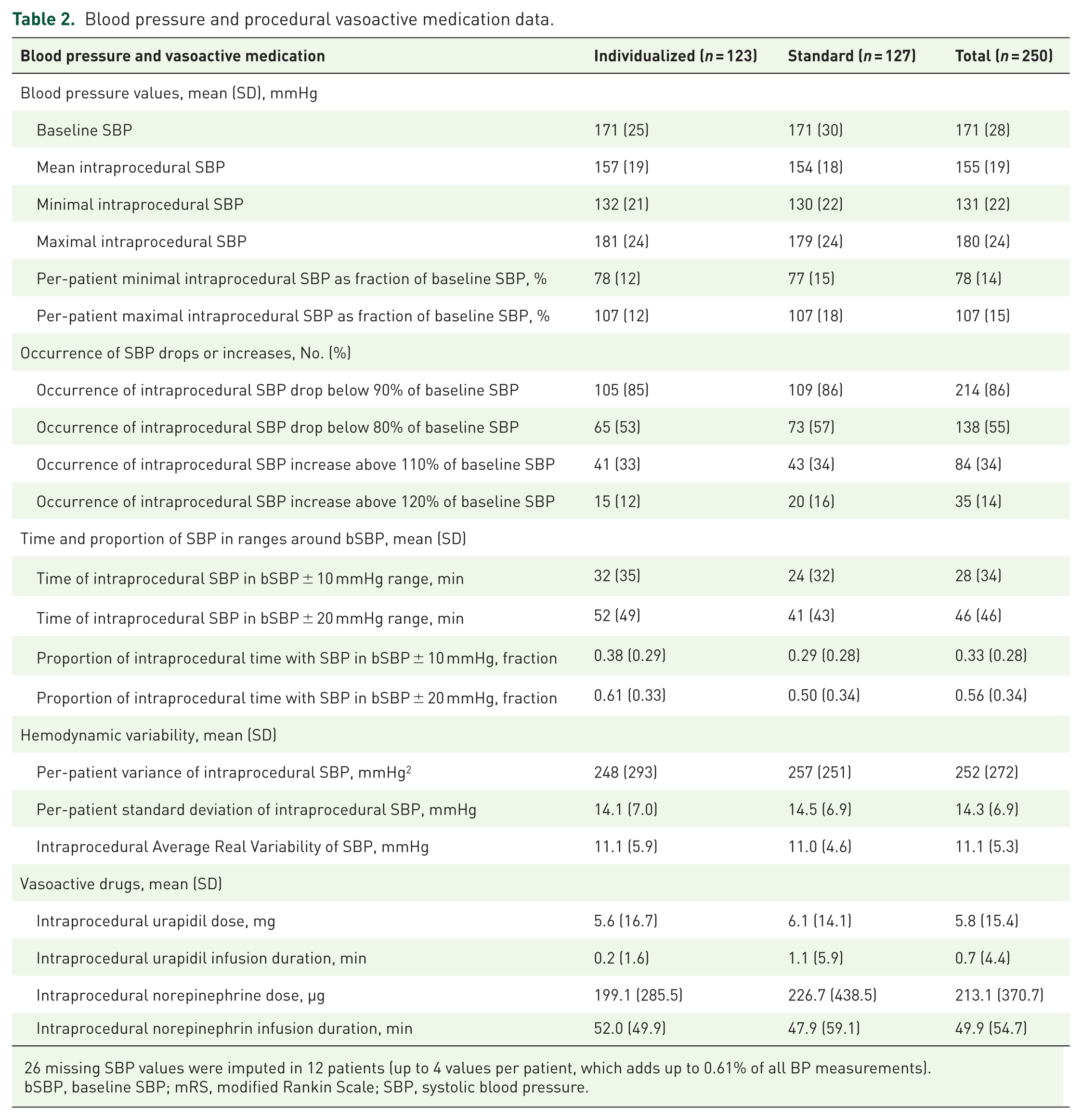
missing SBP values were imputed in 12 patients (up to 4 values per patient, which adds up to 0.61% of all BP measurements).
bSBP, baseline SBP; mRS, modified Rankin Scale; SBP, systolic blood pressure.
Association analyses of different parameters of blood pressure variability with functional outcome
Time and proportion of intraprocedural SBP spent in a range of 10 or 20 mmHg around the baseline SBP and maximal and minimal intraprocedural SBP were not associated with functional outcome. However, higher variance (aOR, 1.002 (95% CI, 1.0004–1.004); p = 0.016) and higher average real variability of SBP (aOR, 1.105 (95% CI, 1.019–1.199); p = 0.016) during thrombectomy was associated with higher odds to achieve a favorable outcome (see Table 3). In complementary analyses with duration of the infusion of urapidil and norepinephrine as additional covariates (model 2, see Supplemental Tables 1) or IVT and onset to reperfusion time as additional covariates (model 3, see Supplemental Table 1) the association of variance and average real variability remained significant with similar aORs. Furthermore, model 2 revealed significant associations of higher minimal intraprocedural SBP with lower odds of achieving favorable functional outcome (aOR 0.973 (95% CI, 0.948–0.999); p = 0.044). The duration of urapidil and norepinephrine application was not associated with functional outcome (see Supplemental Table 3 and 4).
Association of different blood pressure characteristics with favorable outcome (=mRS 0–2).

aOR > 1 depict higher odds to achieve a favorable outcome. Results were adjusted for INDIVIDUATE trial’s treatment group and its interaction with the respective exposure, age, prestroke mRS, baseline NIHSS score, procedure time and ASPECTS. The ASPECTS score was imputed in one patient.
ASPECTS, Alberta Stroke Program Early CT Score; bSBP, baseline SBP; mRS, modified Rankin Scale; NIHSS, National Institutes of Stroke Scale, aOR, adjusted odds ratio; SBP, systolic blood pressure.
Association analyses of different parameters of blood pressure variability with PH1 or PH2 hemorrhages
No significant associations were found for different parameters of blood pressure variability with PH1 and PH2 events in the main model (see Table 4). Higher maximal intraprocedural SBP was associated with lower odds of occurrence of PH1/2 events (aOR 0.978 (95% CI, 0.957–0.9999) p = 0.049), when infusion duration of urapidil and norepinephrine was used as additional covariates (model 2, see Supplemental Table 2).
Association of different blood pressure characteristics with PH1 or PH2.

aOR > 1 depict higher odds for occurrence of PH1 or PH2 events. Results were adjusted for INDIVIDUATE trial’s treatment group and its interaction with the respective exposure, age, prestroke mRS, baseline NIHSS score, procedure time and ASPECTS. The ASPECTS score was imputed in one patient.
ASPECTS, Aberta Stroke Program Early CT Score; bSBP, baseline SBP; mRS, modified Rankin Scale; NIHSS, National Institutes of Stroke Scale, aOR, adjusted odds ratio; SBP, systolic blood pressure.
Group interaction effects of blood pressure management strategy
There was no significant interaction effect with respect to blood pressure management strategy (individualized vs standardized) in the analyses of the different blood pressure variability exposures (see Supplemental Table 5).
Propensity-score-matched comparison of exposures
Propensity scores for the propensity to favorable outcome were calculated based on INDIVIDUATE trial’s treatment group, age, premorbid mRS, NIHSS at admission, pre-intervention ASPECTS, time from groin puncture to reperfusion, IVT and time from onset to reperfusion. On average, this resulted in samples of about 35 patient pairs, with a mediocre balance in the NIHSS and good balance in all other covariates. None of the effects showed a p-value < 0.05, with the smallest p-values (<0.15) being found for ARV and variance of intraprocedural SBP. These effects hinted at slightly higher blood pressure variation in patients with favorable functional outcome. The results of the propensity-score matched analyses are qualitatively in line with the main analysis based on logistic regression models but appear to have lower power due the reduced sample size (see Table 5).
Comparison of exposures in propensity-score-matched data set.

Propensities for favorable outcome were calculated based on the INDIVIDUATE trial’s treatment group, age, premorbid mRS, NIHSS at admission, preintervention ASPECTS, time from groin puncture to reperfusion, IVT and time from onset to reperfusion. p Values for the mean difference in exposure for mRS 0–2 versus 3–6 were calculated using robust linear models and are presented together with the pooled regression coefficients Beta.
ASPECTS, Alberta Stroke Program Early CT Score; bSBP, baseline SBP; mRS, modified Rankin Scale; NIHSS, National Institutes of Stroke Scale, SBP, systolic blood pressure.
Discussion
This post-hoc analysis provides insights regarding aspects of blood pressure variability during the hyperacute interventional treatment phase of acute ischemic stroke and their possible impact on functional outcome and postinterventional hemorrhage. Our analysis revealed the principal finding that higher variance and average real variability of SBP during the intraprocedural time frame of EST were associated with a favorable outcome, which stands in contrast to several other retrospective studies.1–4,7,8 However, most of those studies have focused on the subacute phase following EST and not on the time frame during EST. BPV during the postprocedural phase might lead to detrimental effects in recanalized patients due to the increased direct exposure of the vulnerable recanalized post-ischemic brain tissue to perfusion fluctuations; however, this explanation has not been clearly verified. 2 The intraprocedural phase differs from the postprocedural subacute phase, as monitoring and pharmacologic interventions are more intensive and tightly controlled by the attending anesthesiologist (or neurointensivist in our setting). Moreover, the endovascular intervention itself, particularly the use of intraluminal devices, may alter collateral blood flow and affect perfusion in vulnerable brain regions. The occluded vessel and inserted intraluminal device could reduce direct exposure of blood pressure and perfusion fluctuations in the oligemic or ischemic brain downstream and thus prevent harmful effects of BPV.12,13 This explanation remains speculative, and further research is required to replicate this finding and to uncover any underlying mechanisms for this association.
On the other hand, some studies have found an association of higher BPV with worse outcome during the intraprocedural time frame.14,15 The study population of those studies differed from the current study: In the ASTER trial, the occlusion site was mainly localized at large vessels, the patients had a better prestroke functional status, had more futile recanalization and were younger compared to the current study cohort. Xu et al. also showed a worse outcome in patients who had higher BPV parameters during thrombectomy of large vessel occlusion stroke, but the mean baseline and intraprocedural SBP were lower than in the current study (baseline mean SBP of 144–158 vs 171 mmHg and mean intraprocedural SBP of 124–126 vs 154–157 mmHg).
Several analyses of the intraprocedural time frame and blood pressure focused on blood pressure drops and showed a positive association with worse outcome.16–21 An association between time-dependent blood pressure deviations and unfavorable clinical outcomes was demonstrated in a post-hoc individual patient data meta-analysis of randomized controlled trials evaluating sedation strategies during endovascular stroke therapy. 22
If a causal relationship between BPV and clinical outcome exists, different mechanisms are likely involved at various time points in stroke patients. Higher BPV may not always be associated with worse outcomes and could depend on factors such as occlusion site, collateral status, baseline blood pressure values, and other individual patient characteristics. Its role in modulating clinical outcome needs to be further elucidated before any clinical recommendations can be established.
One limitation for the interpretation of the INDIVIDUATE trial was, that the actual treatment-specific differences in SBP between the different treatment groups were not significant, so no inference on the actual effect of blood pressure on functional outcome could be made. Thus, additional analysis of time and proportion of SBP around the baseline SBP as an additional BPV parameter was performed to investigate whether a higher adherence in terms of time and proportion of intraprocedural time to baseline values was associated with functional outcome; however, no significant association was found. This contrasts with another retrospective analysis from observational data, where an association of higher proportion of intraprocedural time of SBP in a 20% range with favorable functional outcome was found. 23 Mean SBP and mean arterial pressure (MAP) was lower in the current study, and the relationship between baseline SBP adherence and clinical outcomes may vary depending on the baseline SBP level itself.
Regarding postprocedural hemorrhages, we could not find any association of BPV parameters with PH1+2 occurrence except an association of higher maximal SBP with lower occurrence of PH1+2 hemorrhages in the complementary regression model 2 of the current study. This needs to be interpreted cautiously, as the finding reached only marginal statistical significance and could not be reproduced in other models. While a lower hemorrhage rate in the context of higher maximal SBP would contradict the pathophysiological plausibility that elevated SBP contributes to hemorrhagic events due to higher chance of vessel rupture, one study also showed an association of lower odds of hemorrhage with higher intraprocedural MAP and SBP, which might be mediated by the smaller infarction volumes observed in patients with higher blood pressures in that study. 24 The existing retrospective evidence is conflicting. Some studies showed no association of BPV with hemorrhagic events: One study analyzed a cohort of 2640 acute ischemic stroke patients and could not find an association of 24 h post-EST BPV with symptomatic intracerebral hemorrhage. 1 In another study with 141 patients, BPV during the intraprocedural time frame was not associated with parenchymal hematoma. 15
On the other hand, BPV in the first 48 h was associated with parenchymal hematoma in patients with atrial fibrillation and acute stroke in a study by Wu et al. 25 especially when the major vessel responsible for the infarcted area was recanalized. This was speculated to be attributed to reperfusion injury, where BPV may lead to blood–brain barrier dysfunction, as increasing pulse pressure could lead to stretching of arteries and endothelial dysregulation. Other studies showed that BPV within 24 h of stroke onset 6 and during the first 24 h after mechanical thrombectomy was associated with sICH.3,5 These positive studies investigated cohorts with Asian ethnicity, where there is evidence for a higher risk of hemorrhagic transformation after reperfusion therapy26,27 and these findings require validation in other ethnic populations.
Lastly, different blood pressure strategies (standardized or individualized to preprocedural baseline blood pressure levels) showed no interaction effect on the results of association analyses of different BPV with functional outcome or occurrence of parenchymal hematoma. As the actual blood pressure differences and BPV parameters (see Table 2) between the two strategies were negligible, this was an expected finding. Another small single-center RCT investigating an individualized blood pressure management strategy aimed at keeping the intraprocedural MAP during EST at the baseline MAP level showed difficulties in achieving blood pressure targets but could produce a difference in mean MAP and SBP during procedures. 28 Maximum and minimum blood pressure values were similar, and clinical outcomes did not differ significantly, although the small sample size (n = 60) may have precluded detection of meaningful effects. Publication from another multicenter RCT is expected shortly. 29 Furthermore, RCTs investigating postinterventional blood pressure interventions yielded a harmful effect of intensive blood pressure lowering (<140 mmHg SBP) compared to standard BP management (<180 mmHg SBP) after thrombectomy. 30
One strength of this study is the investigation of a prospective trial cohort with broad inclusion criteria for stroke patients and protocolized blood pressure management approaches. The current study has some limitations. Given that any modulatory effect of blood pressure on outcome is likely to be small, the limited statistical power may have precluded the detection of clinically relevant associations. The patient cohort is derived from a single-center trial and generalizability of conclusions is limited. Another limitation is that we could not incorporate TICI scores as covariate, as the models yielded unstable aORs for it. Although the analyses were adjusted for known confounders, residual confounding by unmeasured variables cannot be ruled out.
Conclusion
In this study, the intraprocedural BPV parameters variance and average real variability observed during endovascular stroke treatment have demonstrated a positive association with improved clinical outcomes. Although causal inferences cannot be drawn from these analyses, the results may nonetheless provide valuable insights into intraprocedural blood pressure fluctuations and their potential impact on clinical outcomes.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251391837 – Supplemental material for Association of hemodynamic variability during endovascular stroke treatment with functional outcome and parenchymal hemorrhage
Supplemental material, sj-docx-1-tan-10.1177_17562864251391837 for Association of hemodynamic variability during endovascular stroke treatment with functional outcome and parenchymal hemorrhage by Min Chen, Lukas D. Sauer, Arne Potreck, Meinhard Kieser, Martin Bendszus, Peter Ringleb, Markus Möhlenbruch and Silvia Schönenberger in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
