Abstract
Background:
Perampanel (PER) is effective in treating focal and generalised seizures. The PERaMpanel pooled analysIs of effecTiveness and tolerability (PERMIT) Extension study was a large, pooled analysis of PER clinical practice studies that assessed the effectiveness and safety/tolerability of PER in over 6800 people with epilepsy.
Objectives:
To determine clinical factors associated with PER retention, response, seizure freedom and tolerability when used in clinical practice.
Design:
An exploratory post hoc analysis of data from all individuals included in PERMIT Extension.
Methods:
Univariate and multivariable logistic regression analyses were performed to identify baseline factors associated with retention rate, responder rate (⩾50% seizure frequency reduction), seizure freedom rate (no seizures since at least the previous visit) and incidence of adverse events (AEs).
Results:
A total of 6822 people with epilepsy treated with PER were included. Baseline factors associated with retention were absence of psychiatric comorbidity (odds ratio [95% confidence interval], 1.99 [1.403–2.825]; p < 0.001), fewer focal seizures (1.01 [1.004–1.014]; p < 0.001) and fewer previous antiseizure medications (ASMs; 1.06 [1.013–1.119]; p = 0.013). Factors associated with response were absence of focal seizures (2.12 [1.532–2.924]; p < 0.001), fewer previous ASMs (1.19 [1.136–1.250]; p < 0.001) and absence of concomitant sodium channel blocker (SCB) ASM(s) (1.96 [1.455–2.628]; p < 0.001). Factors associated with seizure freedom were fewer total seizures (1.04 [1.020–1.061]; p < 0.001), absence of focal seizures (3.45 [2.387–4.979]; p < 0.001), fewer previous ASMs (1.11 [1.028–1.205]; p = 0.008), absence of concomitant SCB ASM(s) (1.46 [1.051–2.028]; p = 0.024) and absence of concomitant gamma-aminobutyric acid (GABA)-ergic ASM(s) (2.08 [1.266–3.412]; p = 0.004). Factors associated with occurrence of AEs were older age (1.01 [1.006–1.015]; p < 0.001), longer epilepsy duration (1.01 [1.010–1.012]; p = 0.044), presence of psychiatric comorbidity (1.74 [1.469–2.062]; p < 0.001) and greater number of previous ASMs (1.09 [1.069–1.119]; p < 0.001).
Conclusion:
This study identified clinical factors associated with PER’s real-world effectiveness and tolerability, which may help inform treatment decisions in clinical practice.
Keywords
Introduction
Epilepsies are a group of brain disorders with a high burden due to substantial morbidity and mortality, 1 often requiring life-long treatment with antiseizure medications (ASMs).2–6 The management of epilepsy aims to achieve seizure freedom while minimising adverse events (AEs).7–9 Treatment must be individualised based on seizure type or epilepsy syndrome, clinical characteristics (e.g. comorbidities) and personal preferences. 10 There are currently approximately 30 ASMs available,11,12 and it can be difficult to select the most appropriate treatment option. Perampanel (PER) is an ASM with a unique mechanism of action offering a new approach in the management of epilepsy.13,14 As a highly selective, noncompetitive α-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate (AMPA) receptor antagonist, PER has shown potential as a broad-spectrum ASM that may be useful for individuals with various seizure types15,16 and can target different aspects of the mechanisms underlying seizure generation. Its efficacy in treating both focal and generalised epilepsy has been demonstrated in numerous clinical trials and clinical practice studies.17–23
The PERaMpanel pooled analysIs of effecTiveness and tolerability (PERMIT) Extension study was a pooled analysis of data from the PERMIT and Perampanel Real-wOrld eVidEnce (PROVE) studies, in which people with focal and generalised epilepsy were treated with PER under real-world clinical practice conditions. 24 PERMIT was a multinational pooled analysis of prospective, retrospective and cross-sectional studies and work groups, 22 and PROVE was a retrospective, noninterventional, observational phase IV study from medical and pharmacy records at sites across the United States. 23 PERMIT Extension assessed the effectiveness and safety/tolerability of PER when used in everyday clinical practice and is the largest pooled analysis of PER clinical practice data conducted to date, including data from over 6800 individuals with epilepsy. 24 Retention rates at 3, 6 and 12 months were 88.0%, 77.6% and 61.4%, respectively; responder rates at 12 months were 58.5% for total seizures, 54.6% for focal seizures and 77.7% for generalised seizures, and corresponding seizure freedom rates were 23.6%, 19.0% and 51.3%, respectively. 24 Several subanalyses of PERMIT Extension have been previously conducted in subgroups of individuals with different seizure types, 24 epilepsy aetiologies,25,26 ages, 24 ethnicities 27 and stages of treatment. 28 PER was effective across all subgroups, but some variability in response was identified. Here, we present the results of a further analysis of PERMIT Extension that was conducted to determine clinical factors associated with PER retention, response, seizure freedom and tolerability when used in clinical practice.
Methods
Study design
The design of the PERMIT Extension study and details of the foundational PERMIT and PROVE studies have been previously reported.22–24 Both PERMIT and PROVE assessed retention on PER treatment and its effectiveness, safety and tolerability over at least 12 months.22,23 In PERMIT Extension, deidentified individual participant data from PERMIT and PROVE were pooled together for baseline number of seizures, type of epilepsy/seizures, prior ASMs, dosage, effectiveness at various timepoints and AEs. 24 As in PERMIT and PROVE, PERMIT Extension assessed retention on PER treatment and its effectiveness and safety/tolerability over at least 12 months. 24 The studies included in PERMIT used broad inclusion/exclusion criteria to be representative of people with epilepsy treated in clinical practice. 22 To be eligible for inclusion in PROVE, participants had to have a diagnosis of epilepsy, have been prescribed PER after 1 January 2014 (based on their treating clinician’s recommendation) and have attended their usual epilepsy clinic. 23 The current analysis included all patients from PERMIT Extension.
Study assessments
Retention on PER was assessed after 12 months. Effectiveness was assessed at the last observation of each participant (defined as the ‘last visit’; i.e. last observation carried forward) by seizure type (total, focal, generalised) in terms of responder rate and seizure freedom rate. Response was defined as ⩾50% seizure frequency reduction from baseline (i.e. prior to PER initiation), including those achieving seizure freedom. Seizure freedom was defined as no seizures since at least the prior visit (either 3 or 6 months, depending on the time point at which seizure freedom was assessed). The mean (95% confidence interval (CI)) retention time in PERMIT Extension was 10.5 (10.4, 10.7) months, 24 giving an approximation of the time at which final response was measured. Tolerability was assessed for the duration of PER treatment by evaluating the occurrence of AEs.
Statistical analysis
A descriptive analysis of recorded quantitative and qualitative variables was performed. Quantitative variables were summarised using mean, standard deviation (SD), median, minimum and maximum values, CI or interquartile range, and number of valid cases. Qualitative variables were assessed using absolute frequencies and percentages. Dependent variables were defined as retention, response, seizure freedom and occurrence of AEs. Univariate logistic regression analyses were performed to determine the individual factors (independent variables) associated with each of the dependent variables. These independent variables included demographic and baseline characteristics (including the presence of psychiatric comorbidities, as defined by the participating clinicians), as well as treatment variables (e.g. PER dose at initiation, type of concomitant ASM, mechanism of action of concomitant ASMs (Table 1)). Once potential relationships between the independent variables and each of the dependent variables were determined, those with p < 0.10 were pre-selected to build a multivariable binary logistic regression model. In this way, the probabilities of retention, response, seizure freedom and AEs were estimated as a function of the different independent variables. Odds ratios, 95% CIs and p values were calculated for the univariate and multivariable analyses. The number of previous ASMs used, and PER doses at initiation and the last visit, were compared between the subgroups of individuals with focal and generalised seizures using the Mann–Whitney test. The significance level was set at 5% and the statistical package SPSS 28.0, IBM was used for all analyses.
Categorisation of concomitant ASMs by mechanism of action.

ASM, antiseizure medication; GABA, gamma-aminobutyric acid; SCB, sodium channel blocker; SV2A, synaptic vesicle glycoprotein 2A.
Results
Sample characteristics: independent and dependent variables
A total of 6822 people with epilepsy were included in the current analysis, representing the entire PERMIT Extension full analysis set. 24 The independent variables included in the analysis are summarised in Table 2. These included the number and types of previous ASMs (i.e. ASMs tried before the study) and concomitant ASMs (i.e. ASMs being used when PER treatment was initiated), as well as the mechanism of action of concomitant ASMs (Table 1). The mean (SD) number of previous ASMs was significantly higher in the subgroup of participants with focal versus generalised seizures (4.9 (3.8) vs 3.3 (3.1); p < 0.001). The mean (SD) PER dose at initiation was the same in those with focal and generalised seizures (both 2.7 (1.7) mg/day) but significantly higher in those with focal versus generalised seizures at the last visit (6.3 (2.8) vs 5.7 (2.8) mg/day; p < 0.001).
Summary of independent variables.

International League Against Epilepsy 2017 classification. 29
⩾1% of participants.
Mean (SD; median) number of previous ASMs was significantly higher in individuals with focal versus generalised seizures (4.9 (3.8; 4) vs 3.3 (3.1; 3); p < 0.001).
⩾5% of participants.
Carbamazepine, eslicarbazepine acetate, oxcarbazepine, phenobarbital, phenytoin, primidone.
See Table 1.
Mean (SD) PER dose at initiation was the same in individuals with focal versus generalised seizures (both 2.7 (1.7) mg/day).
Mean (SD) PER dose at the last visit was significantly higher in individuals with focal versus generalised seizures (6.3 (2.8) mg/day vs 5.7 (2.8) mg/day; p < 0.001).
ASM, antiseizure medication; GABA, gamma-aminobutyric acid; IQR, interquartile range; max, maximum; min, minimum; PER, perampanel; SCB, sodium channel blocker; SD, standard deviation; SV2A, synaptic vesicle glycoprotein 2A.
With regards to the dependent variables, the retention rate at 12 months was 61.4% (3614/5888 participants); the responder rate at the last visit was 50.1% (2109/4209); the seizure freedom rate at last visit was 20.8% (953/4571); and the proportion of individuals experiencing AEs during the study was 49.2% (3065/3168).
Factors associated with retention at 12 months
The independent variables with p < 0.10 in the univariate logistic regression analysis of factors associated with retention at 12 months, which were therefore selected for the multivariable logistic regression analysis, are highlighted in grey in Table 3. Multivariable analysis identified that psychiatric comorbidity, baseline focal seizure count and the number of previous ASMs were significantly associated with retention at 12 months: individuals without psychiatric comorbidity at baseline were more likely to be retained on PER at 12 months than those with psychiatric comorbidity at baseline (p < 0.001), as were those with a lower versus higher number of focal seizures at baseline (p < 0.001), and those with a lower versus higher number of previous ASMs at baseline (p = 0.013; Table 3; Figure 1(A)).
Summary of univariate and multivariable logistic regression analyses for factors associated with retention at 12 months.

Grey cells indicate independent variables with p < 0.10 in the univariate logistic regression model that were entered a priori into the multivariable logistic regression model. For the multivariable logistic regression analysis, results are only presented for those independent variables with p < 0.05.
↑, Higher number; ↓, lower number; ASM, antiseizure medication; CI, confidence interval; GABA, gamma-aminobutyric acid; IQR, interquartile range; max, maximum; min, minimum; OR, odds ratio; PER, perampanel; SCB, sodium channel blocker; SE, standard error; SV2A, synaptic vesicle glycoprotein 2A.

Multivariable logistic regression analysis: forest plots of ORs (95% CIs) for independent variables associated with (A) retention at 12 months, (B) response at the last visit, (C) seizure freedom at the last visit and (D) occurrence of AEs during the study.
Factors associated with response at the last visit
The independent variables with p < 0.10 in the univariate logistic regression analysis of factors associated with response at the last visit are highlighted in grey in Table 4. Multivariable analysis identified that the presence of focal seizures at baseline, number of previous ASMs and use of a sodium channel-blocking concomitant ASM at baseline were significantly associated with response at the last visit: participants without focal seizures at baseline were more likely to respond to treatment than those with focal seizures at baseline (p < 0.001), as were those with a lower versus higher number of previous ASMs at baseline (p < 0.001), and those not using versus using a sodium channel-blocking concomitant ASM at baseline (p < 0.001; Table 4; Figure 1(B)).
Summary of univariate and multivariable logistic regression analyses for factors associated with response at the last visit.

Grey cells indicate independent variables with p < 0.10 in the univariate logistic regression model that were entered a priori into the multivariable logistic regression model. For the multivariable logistic regression analysis, results are only presented for those independent variables with p < 0.05.
↑, Higher number; ↓, lower number; ASM, antiseizure medication; CI, confidence interval; GABA, gamma-aminobutyric acid; IQR, interquartile range; max, maximum; min, minimum; OR, odds ratio; PER, perampanel; SCB, sodium channel blocker; SE, standard error; SV2A, synaptic vesicle glycoprotein 2A.
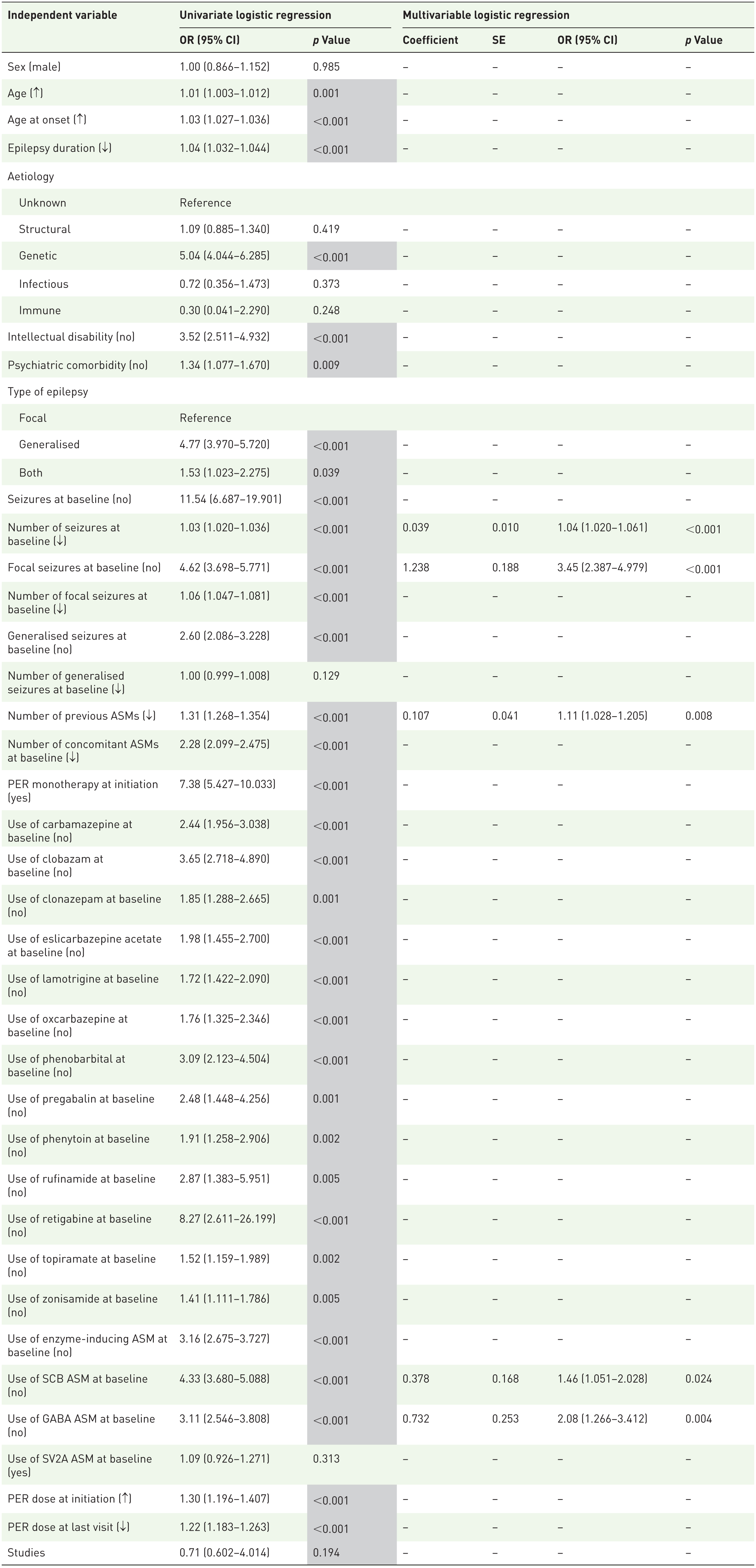
Factors associated with seizure freedom at the last visit
The independent variables with p < 0.10 in the univariate logistic regression analysis of factors associated with seizure freedom at the last visit are highlighted in grey in Table 5. Multivariable analysis identified that the number of seizures at baseline, presence of focal seizures at baseline, number of previous ASMs, use of a sodium channel-blocking concomitant ASM at baseline and use of a concomitant ASM targeting the gamma-aminobutyric acid (GABA) system at baseline were significantly associated with seizure freedom at the last visit: individuals with a lower baseline monthly seizure frequency were more likely to be seizure free at the last visit than those with a higher baseline monthly seizure frequency (p < 0.001), as were those without versus with focal seizures at baseline (p < 0.001), those with a lower versus higher number of previous ASMs at baseline (p = 0.008), those not using versus using a sodium channel-blocking ASM at baseline (p = 0.024) and those not using versus using a GABAergic concomitant ASM at baseline (p = 0.004; Table 5; Figure 1(C)).
Summary of univariate and multivariable logistic regression analyses for factors associated with seizure freedom at the last visit.

Grey cells indicate independent variables with p < 0.10 in the univariate logistic regression model that were entered a priori into the multivariable logistic regression model. For the multivariable logistic regression analysis, results are only presented for those independent variables with p < 0.05.
↑, Higher number; ↓, lower number; ASM, antiseizure medication; CI, confidence interval; GABA, gamma-aminobutyric acid; IQR, interquartile range; max, maximum; min, minimum; OR, odds ratio; PER, perampanel; SCB, sodium channel blocker; SE, standard error; SV2A, synaptic vesicle glycoprotein 2A.
Factors associated with the occurrence of AEs during the study
The independent variables with p < 0.10 in the univariate logistic regression analysis of factors associated with AE occurrence during the study are highlighted in grey in Table 6. Multivariable analysis identified that age, duration of epilepsy, presence of psychiatric comorbidity at baseline and number of previous ASMs at baseline were significantly associated with the occurrence of AEs during the study: older participants were more likely to experience AEs than younger participants (p < 0.001), as were those with longer versus shorter duration of epilepsy (p = 0.044), those with versus without psychiatric comorbidity at baseline (p < 0.001) and those with a higher versus lower number of previous ASMs at baseline (p < 0.001; Table 6; Figure 1(D)).
Summary of univariate and multivariable logistic regression analyses for factors associated with the occurrence of AEs during the study.
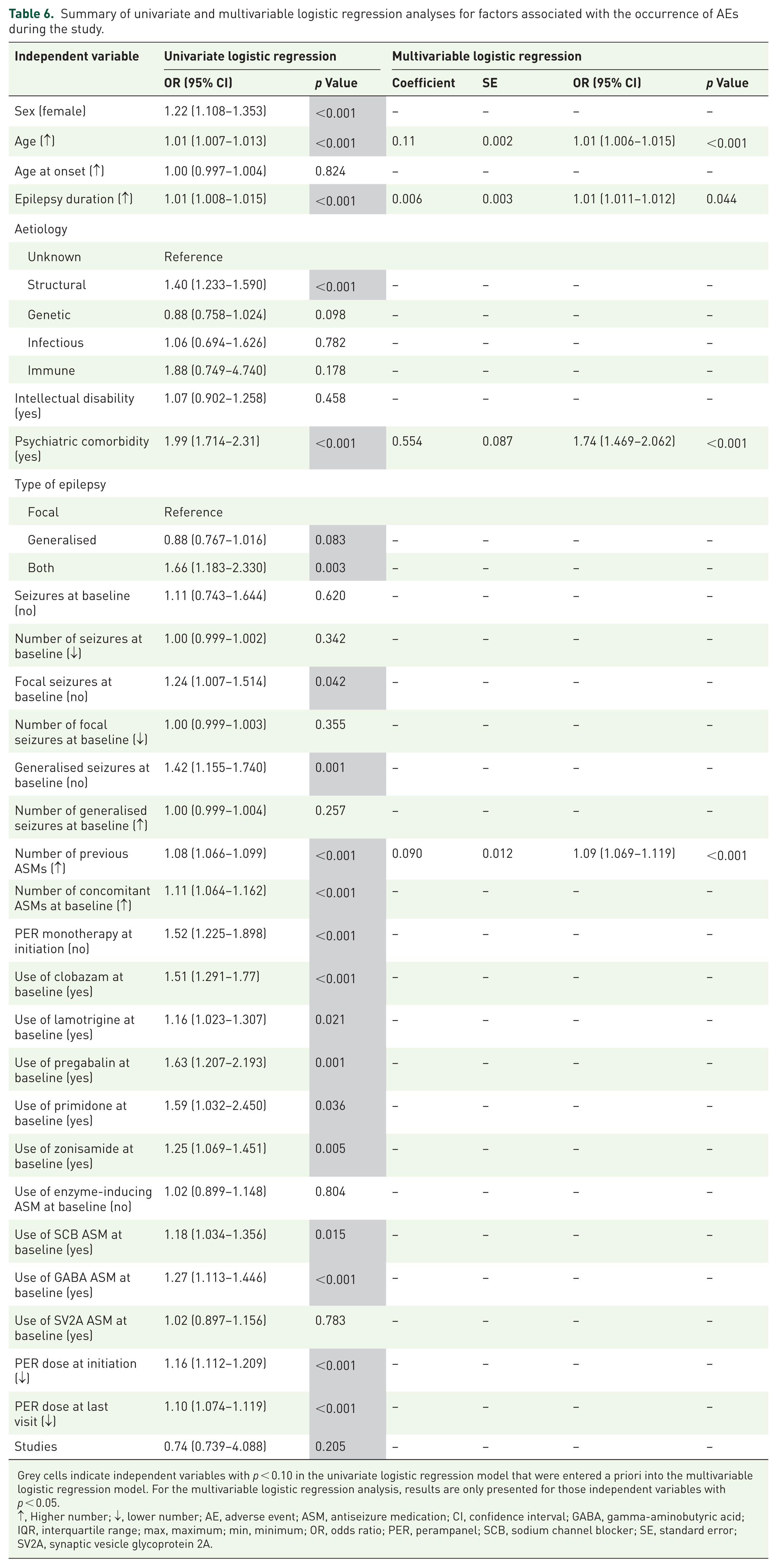
Grey cells indicate independent variables with p < 0.10 in the univariate logistic regression model that were entered a priori into the multivariable logistic regression model. For the multivariable logistic regression analysis, results are only presented for those independent variables with p < 0.05.
↑, Higher number; ↓, lower number; AE, adverse event; ASM, antiseizure medication; CI, confidence interval; GABA, gamma-aminobutyric acid; IQR, interquartile range; max, maximum; min, minimum; OR, odds ratio; PER, perampanel; SCB, sodium channel blocker; SE, standard error; SV2A, synaptic vesicle glycoprotein 2A.
Discussion
Our study identified key clinical factors associated with retention, response, seizure freedom and AEs during treatment with PER, including psychiatric comorbidity, seizure type and prior ASM use. The number of previous ASMs individuals were treated with prior to starting PER was inversely and significantly associated with retention at 12 months, as well as response and seizure freedom at the last visit, consistent with previous clinical practice studies demonstrating PER’s effectiveness when used as an early add-on therapy28,30–36 or monotherapy.37–42 These findings are also consistent with previous reports demonstrating that most individuals with epilepsy are likely to achieve effective seizure control early in their disease course with their first or second ASM, the likelihood of seizure freedom diminishing with each subsequent ASM regimen. 43 In the current study, response and seizure freedom at the last visit were also significantly associated with the absence of focal seizures at baseline, while retention at 12 months was inversely and significantly associated with the frequency of focal seizures at baseline. The inverse association between the presence of focal seizures and PER’s effectiveness may reflect the fact that responder and seizure freedom rates in PERMIT Extension were higher in individuals with generalised seizures than in those with focal seizures, 24 in line with findings on other ASMs, such as levetiracetam and brivaracetam.44,45 Moreover, individuals with focal seizures had been treated with significantly more previous ASMs than those with generalised seizures and were therefore likely to be more refractory to treatment. This is also evidenced by the fact that although PER doses at initiation were the same for the subgroups of participants with focal and generalised seizures, the doses at the last visit were significantly higher in those with focal versus generalised seizures. Participants with focal seizures may have been more treatment refractory and/or at a later disease stage than those with generalised seizures because there are currently more treatment options available for focal versus generalised seizures. Nevertheless, the reason for the observed inverse association is not clear and might also reflect the heterogeneity of the population included in the PERMIT Extension study. Response and seizure freedom were shown to be significantly less likely in individuals being treated with concomitant sodium channel-blocking ASMs at baseline than in those who were not being treated with sodium channel blockers. This may in part be because some sodium channel-blocking ASMs, such as carbamazepine and phenytoin, are strong enzyme inducers and may therefore reduce exposure to PER. 46 In addition, since sodium channel-blocking ASMs have historically been the most efficacious in treating seizures, treatment failure (i.e. the need for adding PER to the treatment regimen) may indicate that those being treated with sodium channel blockers at baseline had more drug-resistant epilepsy. Although it might be thought that sodium channel-blocking ASMs are likely to be used mainly to treat focal seizures, no significant interaction between the presence of focal seizures and the use of sodium channel blockers was identified, and such an interaction was therefore not included in the logistic regression model. Moreover, the model reflects the fact that there were individuals with generalised epilepsies (who only had generalised tonic-clonic seizures) who were treated with sodium channel blockers; however, it should be borne in mind that the diagnosis of generalised epilepsy was made by the treating clinician. Seizure freedom was also shown to be significantly less likely in those being treated with GABAergic concomitant ASMs at baseline than in those not being treated with such agents. This might be related to the fact that the GABAergic ASM group includes enzyme inducers, such as phenobarbital, which could have affected the effectiveness of PER. The use of GABAergic ASMs, such as phenobarbital, may also be associated with a longer duration of epilepsy and/or treatment refractoriness, resulting in a poorer response to PER in this study.
Participants with psychiatric comorbidity at baseline were significantly less likely to be retained on PER treatment and significantly more likely to experience AEs than those without psychiatric comorbidity. PER treatment is commonly associated with psychiatric AEs,47,48 particularly aggressive behaviours, 49 and it may aggravate pre-existing psychiatric symptoms in some people with epilepsy. 50 Although increased levels of glutamate are thought to be associated with increased aggressive behaviour,49,51 glutamate’s effects on behaviour are complex, and evidence suggests that blocking of AMPA receptors may both decrease and increase aggressive behaviour,51,52 which may in part account for the findings observed in the current study (since PER is an AMPA receptor antagonist47,48). However, a post hoc analysis of PER clinical trials demonstrated that a history of psychiatric comorbidity appeared to increase the risk of psychiatric AEs in individuals randomised to PER at doses of 8 and 12 mg/day, as well as in those randomised to placebo. 53 Moreover, prospective audits of a range of ASMs have demonstrated that people with epilepsy who have a pre-existing psychiatric history are significantly more likely to discontinue a new ASM due to psychiatric issues than those with no prior psychiatric diagnosis. 54 These audits have also demonstrated that people with focal seizures are more likely to have psychiatric diagnoses than those with generalised genetic epilepsies, 54 consistent with our observation that 12-month retention on PER was inversely and significantly associated with baseline focal seizure frequency. Our study also demonstrated that AEs were significantly more likely to occur in older versus younger individuals, those treated with more versus less previous ASMs, and those with longer versus shorter duration of epilepsy. These observations may reflect a greater likelihood of drug–drug interactions and associated AEs in these subgroups, since older individuals are more likely to have comorbidities requiring treatment than younger individuals, and those with longstanding and/or refractory epilepsy are more likely to be treated with multiple ASMs than those in earlier disease stages. 55
Other studies have attempted to identify clinical factors associated with response to PER treatment in clinical trial and clinical practice settings (Table 7).22,56–62 The results of the current study are broadly consistent with those of these previous studies and, taken together, the findings provide insights that can potentially help guide clinicians when making treatment decisions regarding the use of PER. For example, the findings support the use of PER as an early intervention (i.e. in individuals not treated with many previous ASMs or concomitant ASMs) but perhaps indicate that PER should be used with caution in those with pre-existing psychiatric conditions, and that therapeutic drug monitoring and/or dose adjustment may be required when using PER in combination with strong enzyme inducers. It is also important to point out that the dose of PER used (median 6 mg/day at the last visit 24 ) was lower than that used in clinical trials (up to 12 mg/day 13 ). This is likely related to the fact that a higher dosage is generally used in clinical trials in order to show efficacy, whereas, in clinical practice, treatment is tailored to the patient’s needs and characteristics to balance efficacy and tolerability, resulting in a lower dosage. In addition, the PERMIT Extension included individuals with less refractory epilepsy (including those with idiopathic generalised epilepsy) than in clinical trials.
Summary of other studies of clinical factors associated with response to PER treatment.

Carbamazepine, oxcarbazepine or phenytoin.
Included in PERMIT Extension.
ASM, antiseizure medication; CNS, central nervous system; FBTCS, focal to bilateral tonic-clonic seizures; OLE, open-label extension; OR, odds ratio; PER, perampanel; PERMIT, PERaMpanel pooled analysIs of effecTiveness and tolerability.
Studies have also investigated clinical factors associated with response to other ASMs, including eslicarbazepine acetate, cenobamate, levetiracetam and lacosamide (Table 8).64–69 In addition, the EXPERIENCE study assessed the overall effectiveness and tolerability of brivaracetam in routine practice in a large population (N = 1644), and subgroup analyses in individuals treated with different numbers of prior ASMs showed that those treated with fewer prior ASMs experienced numerically higher responder rates (⩾50% seizure reduction from baseline), seizure freedom rates and retention rates than those who received a higher number of prior ASMs, 70 in line with the current findings. Other studies have attempted to identify clinical factors associated with treatment response in populations treated with a range of different ASMs. For example, PRO-LONG – an Italian multicentre, retrospective cohort study – employed multinomial logistic regression models to identify factors associated with remission among 1006 children and adults with newly diagnosed epilepsy, who were followed for approximately 18,000 person-years. 71 The types of epilepsy were focal (61.8%), generalised (35.0%), combined focal and generalised (2.7%) and unknown (0.5%) and the most frequently prescribed ASMs at diagnosis (⩾10% of the cohort) were carbamazepine (33.9%), valproate (30.7%) and phenobarbital (15.6%). 71 Prognostic factors for 5-year remission were ⩽2 seizures at diagnosis (vs >10; adjusted hazard ratio (aHR) = 1.03; p = 0.0495), generalised epilepsy (vs focal epilepsy; aHR = 1.29; p = 0.0458), psychiatric comorbidity absent (vs present; aHR = 1.36; p = 0.0263), treatment with one ASM during follow-up (vs 3+ ASMs; aHR = 2.71; p < 0.001) and treatment with two ASMs during follow-up (vs 3+ ASMs; aHR = 1.89; p < 0.001). 71 Overall, our results are broadly in line with similar studies of other ASMs, but they also suggest that a better response to PER is expected in individuals with generalised seizures than in those with focal seizures.
Summary of studies of clinical factors associated with response to other ASM treatments.

ASM, antiseizure medication; ESL, eslicarbazepine acetate; FBTCS, focal to bilateral tonic-clonic seizures; LCM, lacosamide; LEV, levetiracetam; OR, odds ratio; PER, perampanel; RRR, relative risk ratio; SCB, sodium channel blocker; TLE, temporal lobe epilepsy.
A strength of this study is the size of the PERMIT Extension cohort, which allowed meaningful statistical analyses to be conducted. However, as previously reported, 24 the study is limited in being a pooled analysis of two previous studies (PERMIT and PROVE), one of which (PERMIT) was itself a pooled analysis of previous studies and work groups. 22 Moreover, pooled analyses of real-world studies can overestimate treatment benefit due to factors such as regression to the mean and bias caused by discontinuation of individuals who do not respond to treatment, and most of the studies included in PERMIT and PROVE were retrospective in nature and might therefore additionally be subject to selection bias. 24 The study was also limited by the lack of a control group and the fact that the observation timepoints of some studies included in PERMIT Extension did not match those used for evaluation, which might have affected some of the results. 24 It should also be noted that specific seizure types were not considered in the current study; thus, results might differ across the subgroups of individuals experiencing different types of focal and generalised seizures; for example, previous studies have shown that PER demonstrated similar efficacy in generalised and focal to bilateral tonic-clonic seizures.72,73 Finally, individual subject data were not reviewed systematically but were reviewed by the investigators of the original studies that were then pooled in PERMIT Extension. 24
Conclusion
This study has identified clinical factors associated with PER’s real-world effectiveness and tolerability in treating individuals with focal and generalised epilepsy, which may help inform treatment decisions in clinical practice.