Abstract
Acute ischemic stroke (AIS) is a leading cause of long-term disability and mortality worldwide, necessitating the rapid implementation of time-sensitive reperfusion therapies to improve outcomes. The “drip and ship” (DS) model, in which intravenous thrombolysis (IVT) is initiated at a primary stroke center (PSC) followed by transfer for endovascular thrombectomy (EVT) at a comprehensive stroke center, is widely adopted, particularly in regions with limited immediate EVT access. This narrative review synthesizes evidence from randomized-controlled clinical trials, large-scale observational registries, meta-analyses, and expert-consensus statements to comprehensively analyze the DS model in AIS management, compare it with the mothership (MS) paradigm, and evaluate current evidence regarding workflow optimization, pharmacologic strategies, and system-level innovations. Evidence comparing DS and MS models highlights the complexity of balancing early IVT with minimizing delays to EVT, with regional factors influencing the optimal approach. Reducing door-in-door-out times is critical within DS pathways, as prolonged interhospital transfer is associated with worse outcomes, emphasizing the need for streamlined protocols, prehospital notification, and telemedicine integration. Bridging therapy with IVT, particularly using tenecteplase, is associated with improved rates of early recanalization, supporting its continued use within DS workflows. Emerging adjunctive therapies offer potential for enhancing arterial recanalization and microcirculatory reperfusion without delaying transfer. The “drive-the-doctor” paradigm, involving the transfer of neurointerventionalists to PSCs, may further reduce onset-to-reperfusion times in geographically challenging settings. Mobile stroke units, equipped with CT imaging and telemedicine capabilities, represent an additional strategy to initiate IVT in the field while expediting triage decisions for EVT. Collectively, these advancements support the continued refinement of the DS model, emphasizing the need for structured system-level improvements to optimize timely reperfusion and functional recovery in AIS patients. Continued research is necessary to further define optimal strategies within the DS framework to ensure equitable and effective stroke care across diverse healthcare environments.
Keywords
Introduction
Acute ischemic stroke (AIS) is a major global healthcare challenge, causing substantial morbidity, mortality, and healthcare costs.1,2 Advances in reperfusion therapy, particularly intravenous thrombolysis (IVT) and endovascular thrombectomy (EVT), have dramatically improved functional outcomes for AIS patients.3,4 These time-dependent interventions require coordinated systems to deliver care within narrow therapeutic windows.5–7
IVT, while standard for all AIS subtypes, achieves lower recanalization rates in large vessel occlusion (LVO). 3 The emergence of EVT has provided an effective intervention for LVO, demonstrating substantial improvements in functional outcomes.8–10 Despite these advances, logistical challenges related to the distribution of EVT-capable centers have resulted in the development of two primary care models: the mothership (MS) approach and the drip and ship (DS) approach. 11 The MS approach involves direct patient transport to a comprehensive stroke center (CSC) capable of delivering both IVT and EVT, whereas the DS-model emphasizes the early administration of IVT at a primary stroke center (PSC), followed by patient transfer for EVT when indicated.
The effectiveness of these models is influenced by various factors, including geographical location, hospital capabilities, prehospital triage, and system organization. Understanding the intricacies of the DS model and its potential advantages is essential for healthcare systems seeking to improve outcomes in AIS patients while managing resource constraints.
Methods
This narrative review synthesizes evidence from randomized-controlled clinical trials (RCTs), observational studies, meta-analyses, and expert recommendations to provide a comprehensive understanding of the DS model in AIS management.
A literature search was conducted in PubMed/MEDLINE, Scopus, and ClinicalTrials.gov up to 30 June 2025, using combinations of keywords such as “acute ischemic stroke,” “drip and ship,” “mothership,” “endovascular thrombectomy,” “intravenous thrombolysis,” “tenecteplase,” “door-in-door-out,” and “prehospital triage.” Reference lists of relevant articles were screened to identify additional studies. We included English-language publications reporting original clinical data or high-quality reviews relevant to DS workflows, comparisons with the MS paradigm, pharmacologic strategies, and system-level innovations. Studies exclusively focused on pediatric stroke, nonischemic stroke, or experimental animal models were excluded.
The review explores DS workflows, compares outcomes with the MS paradigm, and assesses workflow optimizations. Pharmacologic strategies, including the use of tenecteplase and alteplase, and emerging adjunctive therapies are analyzed for their potential to enhance recanalization and outcomes in the DS context. Additionally, innovative paradigms such as physician transfer models aimed at reducing door-in-door-out (DIDO) times are discussed.
Stroke systems of care: Definitions and organizational models
Stroke systems of care are structured to provide timely and effective reperfusion therapy to AIS patients. Hospitals are categorized based on their capabilities: acute stroke-ready hospitals, PSC, and CSC. 11 Acute stroke-ready hospitals provide basic stroke care and stabilization before transfer. PSCs can administer IVT but do not have the capacity for EVT, while CSCs are EVT-capable stroke centers that have the resources and expertise to perform EVT on a 24/7 basis.
The organizational approach to stroke care typically follows either the MS or the DS paradigm (Figure 1). The MS approach involves bypassing non-EVT centers to transport patients directly to CSCs, facilitating the prompt delivery of EVT without interhospital transfer delays. 12 This approach benefits urban areas with dense CSC coverage, allowing faster intervention.

Workflow and system pathways in AIS care. There are four primary AIS care pathways within modern stroke systems. Pathway (a): DS—The patient is transferred to a PSC, where diagnostic imaging and IVT are administered. If a LVO is identified, the patient is subsequently transferred to a CSC for EVT. Pathway (b): MS—The PSC is bypassed, and the patient is transported directly to a CSC for comprehensive imaging, IVT, and EVT without interhospital transfer delays. Pathway (c): Drive-the-Doctor—The patient is transferred to a PSC equipped with an angio-suite, where initial imaging and IVT are performed. If a LVO is identified, a neurointerventionalist travels from the CSC to the PSC to perform EVT onsite, avoiding patient transfer. Pathway (d): MSU—A MSU equipped with CT imaging, point-of-care laboratory testing, and telemedicine delivers prehospital diagnosis and IVT in the field. If no LVO is detected, the patient is transported to a PSC for further care; if a LVO is present, the patient is transported directly to a CSC for EVT.
In contrast, the DS-model emphasizes early treatment with IVT at PSCs, followed by rapid transfer to CSCs for EVT when necessary. 13 This model is particularly relevant in rural and semiurban settings where immediate access to CSCs may not be feasible due to geographical constraints. The DS approach leverages the widespread availability of PSCs to administer time-sensitive thrombolytic therapy, preserving brain tissue while preparing for potential EVT.
The organization of these systems requires clear protocols and effective prehospital triage to ensure that patients are routed to appropriate facilities based on stroke severity, suspected LVO, and geographical logistic considerations.
Prehospital stroke severity scales
An essential component of optimizing DS and MS routing strategies is the use of prehospital stroke severity scales to identify patients with a high probability of LVO. Several validated tools are employed in the field, including the Rapid Arterial oCclusion Evaluation scale, 14 Field Assessment Stroke Triage for Emergency Destination scale, 15 Los Angeles Motor scale, 16 Prehospital Acute Stroke Severity scale, 17 the Cincinnati Prehospital Stroke Severity scale, 18 and the Gaze–Face–Arm–Speech–Time scale. 19 These scales, often derived from subsets of the National Institutes of Health Stroke Scale (NIHSS), assess motor deficits, gaze deviation, and speech impairment to estimate LVO likelihood.
Integration of these tools into emergency medical services (EMS) protocols facilitates early CSC notification, informs bypass decisions, and reduces secondary transfers. From the perspective of the MS and DS paradigms, patients with high prehospital LVO scores in urban regions may benefit from direct transport to a CSC (MS model), minimizing time to EVT, while in rural or remote areas (where CSC access is delayed) a DS approach may still be favored to enable rapid IVT at the nearest PSC. Conversely, lower prehospital scores may support initial PSC routing in both settings, avoiding overtriage to CSCs.
The predictive accuracy of these scales varies, with sensitivity and specificity affected by symptom onset time, operator training, and patient presentation heterogeneity. Moreover, while clinical scales can guide routing, they may misclassify patients with atypical LVO presentations or stroke mimics. Emerging decision-support innovations aim to enhance accuracy and reduce delays. These include artificial intelligence-based algorithms integrated into EMS workflows, 20 geolocation systems calculating real-time transport times to PSCs and CSCs, and telemedicine-supported prehospital assessments. Interestingly, geospatial analysis has also been used to optimize Mobile Stroke Unit (MSU) distribution in TX, USA, demonstrating its potential to guide resource allocation. 21 Such an analysis could be successfully applied within DS models to inform individualized routing strategies across diverse regions, improving access and efficiency.
Evidence base: MS versus DS paradigms
The comparison between the MS and the DS models has been a focal point in the optimization of AIS care pathways. However, the majority of the AIS patients included in the phase III RCTs evaluating EVT had been treated in MS settings. 22 In the meta-analysis conducted by the HERMES collaboration, the MS group demonstrated significantly shorter onset-to-reperfusion times compared to the DS group, with median times of 251 versus 345 min, respectively (p < 0.001). 7 Additionally, the analysis showed that the likelihood of achieving functional independence at 3 months decreased as onset-to-reperfusion times increased, a possible disadvantage for DS. 7 Since then, observational studies and registry analyses suggest that patients directly admitted to CSCs may receive faster EVT, leading to improved outcomes compared to patients managed within DS models. A large-scale observational study of 1000 patients with severe stroke treated with EVT within 8 h found better clinical outcomes in the MS-model, with 60% achieving functional independence compared to 52% in the DS model (odds ratio (OR) = 1.38, 95% confidence interval (CI): 1.06–1.79; p = 0.02). 23 Hypothetical bypass modeling in this cohort suggested that although IVT would be delayed by 12 min, EVT could be performed 91 min earlier if patients were transported directly to EVT-capable centers. 23
However, modeling studies suggest that when PSCs achieve door-to-needle times of ⩽30 min and maintain efficient transfer workflows, the DS approach may still result in better outcomes in some regions. 24 The DS model allows for the earlier administration of IVT, which results in recanalization in a substantial number of cases, obviating the need for EVT. Seven observational studies25–31 and one RCT 32 evaluating EVT found no significant differences in functional outcomes between the MS and DS models. However, again, five of these studies reported significantly shorter onset-to-groin puncture times in the MS approach, with times ranging from 23 to 120 min faster (p < 0.001 in all studies).25–29 Additionally, one observational study documented a notably shorter onset-to-revascularization time in the MS model compared to the DS model (277 vs 420 min, p < 0.001). 31
A recent meta-analysis of 7824 patients compared the MS (n = 4639) and DS (n = 3185) models for AIS management. 33 The findings showed that the MS-model was associated with significantly shorter onset-to-puncture times and better functional outcomes at 90 days, with lower rates of symptomatic intracerebral hemorrhage compared to the DS model. 33 These findings align with an earlier meta-analysis conducted by our international group showing MS superiority for good functional outcome (OR = 1.34; 95% CI: 1.16–1.55; I2 = 30%). 34 Meta-regression analysis revealed association between onset-to-needle time and good functional outcome, with longer onset-to-needle time being detrimental. Similar rates of recanalization, symptomatic intracranial hemorrhage (sICH), and mortality at 90 days were documented between MS and DS. 34
The choice between MS and DS models often depends on regional infrastructure, transport logistics, CSC distribution, and prehospital triage capabilities. While the MS approach may be ideal in areas with accessible CSCs, the DS model remains a practical and effective strategy for extending reperfusion therapy to broader patient populations, particularly in remote regions with limited CSC access.
In the Unites States, 49% of LVO patients that were eligible for EVT received the treatment at the CSCs (MS approach). 35 These numbers were much higher for the urban patients compared to the rural patients: 70% for the urban population versus 28% for the rural population. Approximately 93% of AIS patients presented at the PSCs who were eligible for IVT received the systemic treatment and then followed the DS paradigm. However, one out of six LVO patients lacked access to EVT even despite the hospital transfer. 35
In Europe, only one-third of the countries had available CSCs that meet the needs for EVT. 36 Additionally, two-third of the European countries had less than one EVT-capable CSC per 1,000,000 inhabitants. 36 Therefore, the DS-model is used for the vast majority of AIS patients in Europe.
Up to now, there are only two completed RCTs that evaluated the two treatment paradigms in a real-world, nonurban context (Table 1). The Direct Transfer to an Endovascular Center Compared to Transfer to the Closest Stroke Center in Acute Stroke Patients With Suspected Large Vessel Occlusion (RACECAT) trial was a multicenter, cluster-randomized study conducted in nonurban areas of Catalonia, Spain, designed to evaluate the optimal prehospital transport strategy for patients with suspected LVO stroke. 37 Between March 2017 and June 2020, 1401 patients were randomized to direct transport to an EVT-capable center (MS; n = 688) or to the nearest local stroke center (DS; n = 713), which was not capable of performing EVT. In the target AIS population of 949 patients, there was no significant difference in 90-day functional outcomes between the two groups (adjusted common OR of 1.03; 95% CI: 0.82–1.29). Importantly, direct transport resulted in a higher likelihood of receiving EVT (48.8% vs 39.4%) but a lower rate of IVT (47.5% vs 60.4%), mortality rates at 90 days were similar between the groups. The trial was halted early for futility, and findings suggest that in nonurban settings, direct transport to EVT-capable centers (i.e., MS) does not significantly improve 90-day outcomes compared to initial care at local stroke centers (i.e., DS).
Randomized-controlled clinical trial directly comparing DS model versus MS model.

EVT, endovascular thrombectomy; IVT, intravenous thrombolysis; MS, mothership.
The TReatment Strategy In Acute Ischemic larGE Vessel STROKE (TRIAGE-STROKE) trial was a multicenter, randomized, assessor-blinded study in Denmark assessing whether bypassing a PSC for direct transport to a CSC improves outcomes in patients with suspected LVO who were eligible for IVT. 38 Between 2018 and 2022, 171 patients were randomized, of whom 104 had AIS. The trial found no statistically significant difference in 90-day functional outcomes, with an OR of 1.42 (95% CI: 0.72–2.82) favoring direct CSC transport. Direct CSC transport shortened onset-to-groin puncture times for EVT by 35 min but delayed IVT by 30 min compared to PSC-first routing. Post hoc analysis suggested that direct CSC transport may increase the likelihood of achieving ambulatory status at 90 days, with an OR of 2.64 (95% CI: 1.06–7.13, p = 0.034). Treatment rates for both IVT and EVT were high in both arms, and workflows were efficient, with a DIDO time of 67 min at PSCs. Although underpowered, the trial highlights the complexity of balancing timely IVT with expedited EVT and underscores the need for further randomized data to guide transport strategies for suspected LVO patients.
In the pooled analysis of these two RCTs, there is no difference in the adjusted common OR for reduced disability measured by modified Rankin Scale (mRS) at day 90 between the two paradigms (adjusted common OR 1.07 in favor of MS; 95% CI: 0.86–1.32; Figure 2).
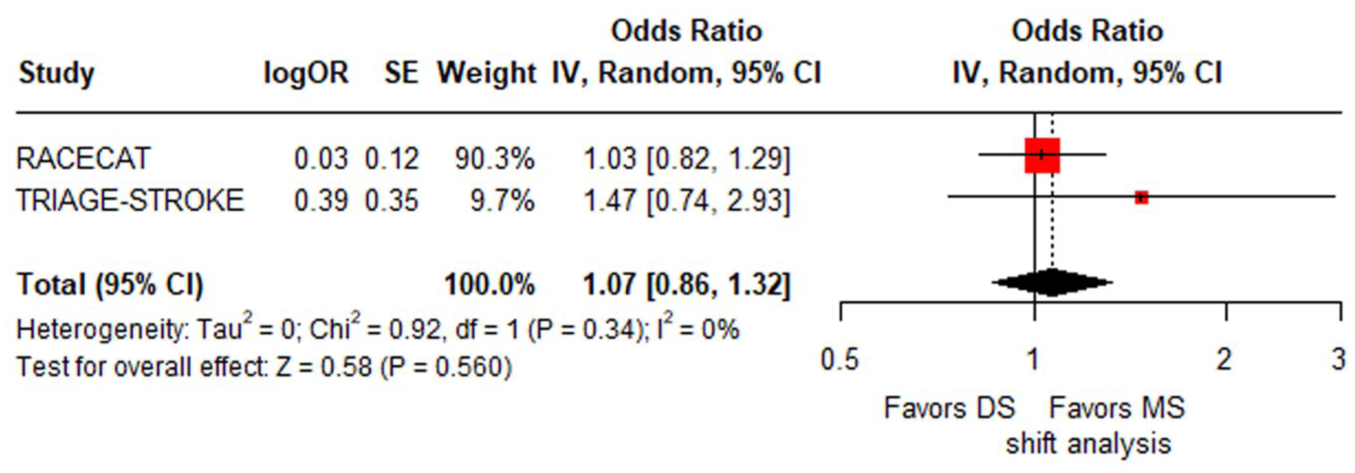
Forest plot presenting the adjusted common OR of reduced disability at 3 months among AIS patients managed in the DS versus the MS paradigm. Reduced disability at 3 months is defined as at least 1-point reduction across all mRS strata. For the evaluation of reduced disability at 3 months, the adjusted common OR with 95% CI was calculated using generic IV meta-analysis, using the R software version 3.5.0.
Despite the lack of strong evidence and based on consensus of expert opinion, current international guidelines recommend that if patients with suspected LVO are ineligible for IVT, they should be transported directly to a CSC to enable prompt access to EVT. 8 Conversely, for patients presenting within 4.5 h of symptom onset with anterior circulation LVO who are eligible for both IVT and EVT, the European Stroke Organisation/European Society for Minimally Invasive Neurological Therapy (ESO/ESMINT) guidelines advocate administering IVT promptly at the presenting nonthrombectomy-capable center followed by rapid transfer to a CSC, rather than omitting IVT while arranging transfer. 39 Importantly, the administration of IVT should not cause delays in initiating transfer to a CSC for thrombectomy. 39 Although based on low-quality evidence from a pairwise meta-analysis of observational studies, this recommendation is considered strong, reflecting expert consensus on the potential benefit of early IVT in preserving brain tissue while maintaining the urgency of achieving definitive reperfusion with EVT. 39
DIDO time: Critical metric in DS pathways
DIDO time is a crucial factor influencing outcomes in the DS-model, representing the period a patient spends at the transferring hospital before departure to the CSC. Reducing DIDO time is essential for minimizing delays in reperfusion and maximizing the benefits of EVT.
Studies have demonstrated significant variability in DIDO times, influenced by patient demographics, hospital protocols, and regional system organization. A US registry-based study evaluating interhospital transfers over 3 years, including both hemorrhagic and AIS patients, reported a median DIDO time of 174 min. 40 Longer median DIDO times were noticed for older patients (⩾80 years), female patients, non-Hispanic Black race, and Hispanic ethnicity. Importantly, shorter DIDO times were observed when EMS prenotification was utilized, in patients with higher baseline stroke severity (NIHSS >12), and among those eligible for EVT, with this subgroup having a median DIDO of 132 min.
Another study analyzing 130 patients across 15 PSCs with CTA-confirmed LVO found a median DIDO of 85 min, with a median door-to-puncture time of only 21 min upon arrival at the CSC. 41 While no clear association was found between DIDO and admission NIHSS or mRS, there was a nonsignificant trend toward higher NIHSS and mRS at discharge and at 90 days with increasing DIDO times. Critically, patients who failed to achieve complete reperfusion demonstrated worse outcomes associated with prolonged DIDO.
Workflow analyses indicate that the most prolonged components of DIDO are often related to the interval between imaging completion and the initiation of transfer requests. Addressing these delays requires targeted interventions, including streamlined imaging protocols, early communication with CSCs, and the use of prehospital notification systems to facilitate rapid decision-making. An Australian study demonstrated that DIDO comprises four key intervals: door-to-CT, CT-to-retrieval request, retrieval request-to-ambulance arrivals, and ambulance arrival-to-PSC departure times. 42 The longest segment was the CT-to-retrieval request phase, lasting nearly 1 h, encompassing imaging acquisition, reconstruction and interpretation, acute treatment decision-making, imaging transfer to the CSC, referral to the CSC, and the formal transfer request. Re-utilizing the initial ambulance crew for transfer and patient presentation during working hours significantly shortened DIDO times.
Supporting these findings, a Dutch study identified the major contributor to DIDO time as the CTA-to-ambulance notification interval, 43 while German data indicated the most significant delays occurred after EVT decisions had been made, with time consumed in organizing transfer logistics and scheduling EVT procedures. 44 Importantly, in these settings, IVT administration did not contribute to workflow delays, emphasizing its feasibility within efficient DS-models. 44
Recent data from the French multicenter Endovascular Treatment in Ischemic Stroke (ETIS) registry involving 3769 LVO patients with AIS transferred for EVT demonstrated that longer inter-hospital transfer times were significantly associated with lower rates of favorable 3-month outcomes. 45 Compared with transfer times under 120 min, transfer durations of 120–180 min and >180 min were associated with a 15% and 36% reduction in the odds of favorable outcomes, respectively, highlighting that faster interhospital transfer is critical for improving patient recovery within DS pathways. 45
Efforts to reduce DIDO times and enhance the DS efficiency have substantially benefited patient outcomes. 46 Successful strategies include (1) establishing standardized protocols for imaging and decision-making; (2) utilizing EMS prenotification to alert PSCs and CSCs of incoming stroke patients; (3) retaining the initial EMS crew until the transfer decision is finalized to expedite transport; (4) implementing telemedicine consultations between PSCs and CSCs to facilitate rapid assessment and EVT eligibility determination; (5) regular multidisciplinary meetings to review workflows, identify bottlenecks, and implement targeted improvements; (6) establishing direct communication channels between PSC and CSC specialists, ensuring that transfer decisions are made promptly; and (7) utilizing remote access systems for CSC interventionalists to review imaging directly, reducing delays associated with image transfer and interpretation. These interventions have been associated with significant reductions in DIDO times, improving onset-to-reperfusion intervals and outcomes for patients managed under the DS model. 47
Opportunities remain for further improvement. Retaining the ambulance crew until a transfer decision is made has proven effective in shortening DIDO without delaying other services. 48 In cardiology, DIDO benchmarks for acute myocardial infarction have been established at ⩽30 min, 49 while American Heart Association/Get With the Guidelines program advocates a target of ⩽90 min. 50 However, the optimal DIDO target for LVO stroke remains largely undefined, despite data suggesting that accelerating door-to-reperfusion time in AIS offer a five-fold greater benefit than in acute myocardial infarction. 51 Recent studies have attempted to identify an optimal DIDO cut-off for LVO patients but have not established a universally applicable threshold.
Bridging therapy versus direct EVT
Bridging therapy, involving the administration of IVT before EVT, remains a standard practice in the management of LVO in AIS patients within the DS model. 39 A previous systematic review and meta-analysis pooled observational evidence comparing both MS and DS approaches for bridging therapy. 52 This large meta-analysis of 38 observational studies evaluating bridging therapy versus direct EVT in patients with LVO demonstrated that bridging therapy is associated with improved outcomes, including higher rates of functional independence and lower mortality, without an increase in sICH. 52 These findings are relevant in the DS-model, where IVT is often administered at PSCs before EVT transfer. Despite concerns regarding the limited efficacy of IVT in proximal occlusions, this analysis suggests that administering IVT before EVT can enhance the likelihood of early neurological improvement, successful recanalization, and functional recovery, even in LVO patients.
Another recent meta-analysis evaluated the role of IVT administered at PSCs before transfer for EVT in AIS patients with LVO. Six observational studies comprising 1723 patients (mean age 71 years, 51% women) were included, with 53% receiving IVT before transfer. 53 The analysis found no significant difference in onset-to-groin puncture times between those receiving IVT and those undergoing EVT alone. However, patients treated with IVT prior to transfer (DS paradigm) demonstrated significantly higher odds of achieving excellent and good functional outcomes at 3 months and experienced reduced disability, without an increased risk of sICH or mortality.
Similar findings were confirmed by another systematic review and meta-analysis including 13 studies with 1561 patients (treated either within the DS- or the MS-model). 54 This study demonstrated that pretreatment with IVT resulted in successful reperfusion in approximately 11% of cases, effectively negating the need for EVT in about 1 out of every 10 patients. 54 This supports the continued use of IVT in eligible patients prior to transfer for EVT within the DS model, highlighting that early administration of IVT can lead to complete recanalization before the patient arrives at the CSC.
The rate of successful reperfusion was even higher in the DS arm of the RACECAT trial, where it was almost 16%, 37 while the PREDICT-RECANAL registry reported rates of 26% in DS patients compared to 10% in MS patients within the derivation cohort, and 22% versus 12% in the validation cohort. 55 Similarly, the Bridging Thrombolysis versus Direct Mechanical Thrombectomy in Acute Ischemic Stroke (SWIFT-DIRECT) trial demonstrated a threefold increase in pre-interventional reperfusion among patients pretreated with IVT, 56 while a German single-center study reported an 11-fold increase. 57 The Catalan Stroke Code and Reperfusion Consortium also noted a fourfold increase in preinterventional reperfusion in patients receiving IVT before EVT. 58
A large bi-center cohort study of 520 anterior circulation LVO patients transferred for EVT found that interhospital recanalization occurred in 21% of patients, which was strongly associated with IVT use (adjusted OR = 6.8; 95% CI: 4.0–11.6) and resulted in reduced infarct growth, greater NIHSS improvement during transfer, and significantly better 3-month outcomes. 59 A more recent prospective cohort analysis of 521 anterior circulation LVO patients transferred for EVT found that IVT before transfer was associated with significantly higher rates of good and excellent functional outcomes, as well as a fivefold increase in interfacility recanalization, without increasing the rates of sICH. 60 Critically, IVT was only used in 50% of patients, underscoring the need to explore expanding IVT criteria in the DS model to maximize its benefits. 60
Recent multicenter data in patients with basilar artery occlusion being transferred for EVT, showed that interhospital recanalization occurred in 15%, with prior IVT use at the referring PSC being the primary modifiable factor associated with recanalization. 61 Importantly, basilar artery recanalization during transfer was associated with favorable 3-month functional outcomes, underscoring the potential benefit of bridging therapy even in posterior circulation LVOs within the DS model. 61
Another observational study evaluating 504 LVO patients transferred for EVT demonstrated that substantial clinical changes during transfer are common, with 22% experiencing clinical improvement and 14% deterioration, each significantly impacting 3-month functional outcomes. 62 IVT use, more distal occlusions, and lower glucose were associated with improvement, while proximal occlusions and higher glucose predicted deterioration, highlighting the importance of pre-transfer treatment strategies to stabilize or improve clinical status during DS transfers. 62 Regarding safety outcomes, hemorrhagic transformation during interhospital transfer for EVT is uncommon (5.5%), but has been associated with subsequent hematoma expansion and poorer 3-month outcomes, particularly in patients with longer transfer times and higher pretransfer NIHSS scores. 63 Notably, hemorrhagic transformation has been strongly associated with interhospital arterial recanalization, emphasizing the need for strategies to mitigate hemorrhagic risk within DS workflows. 63
Only a limited number of observational studies have directly compared bridging therapy with direct EVT in patients treated under the DS-model. A subgroup analysis from the Optimizing Patient Selection for Endovascular Treatment in Acute Ischemic Stroke (SELECT) study, which evaluated LVO patients initially treated with IVT at PSCs before transfer, found that bridging therapy was associated with significantly higher rates of excellent functional outcomes (36% vs 10% with direct EVT). 64 This analysis also demonstrated an overall shift toward better functional outcomes with bridging therapy (adjusted common OR = 4.51; 95% CI: 1.44–14.15). 64 Additionally, analysis from the French ETIS registry showed that among 1507 anterior circulation LVO patients treated with EVT, those receiving bridging therapy in the DS subgroup had higher unadjusted rates of good outcomes (50.3% vs 39.7%, OR = 1.54; p = 0.005), although this association lost significance after propensity score matching. 65
The current ESO/ESMINT guidelines state that bridging therapy should be pursued in the DS scenario. 39 In the DS paradigm, direct EVT compared to bridging therapy was associated with lower odds of good functional outcome and a nonsignificant decrease in excellent functional outcome, with no difference in mortality. 39
Tenecteplase versus alteplase: Current evidence
The aforementioned ESO/ESMINT guidelines supporting the use of bridging therapy over direct EVT were based on the study-level meta-analysis of six RCTs, showing that direct EVT is not noninferior to bridging therapy in MS-patients with immediate EVT-access. 39 Those findings were later confirmed by the individual patient-data meta-analysis of the same six RCTs performed by the IRIS collaborators that again did not establish the noninferiority of EVT alone compared to bridging therapy in patients presenting directly at the CSCs. 66 However, in those RCTs, alteplase was the only thrombolytic agent used in the bridging therapy arm. More recently, the results of the Endovascular Treatment With Versus Without Intravenous rhTNK-tPA in Stroke (BRIDGE-TNK) were published. The BRIDGE-TNK trial, conducted in China, evaluated intravenous tenecteplase before EVT in AIS patients with LVO presenting within 4.5 h at CSCs. 67 Among 550 randomized patients, functional independence at 90 days was achieved in 52.9% receiving tenecteplase plus EVT, compared to 44.1% with EVT alone (risk ratio (RR) = 1.20; p = 0.04). Successful reperfusion before EVT was higher with tenecteplase (6.1% vs 1.1%), while post-EVT reperfusion rates were similar. These findings support the use of tenecteplase prior to EVT in eligible patients.
When the main analysis from the ESO/ESMINT guidelines 39 is updated to include the results of the BRIDGE-TNK trial, 67 the risk difference for achieving a good functional outcome at 3 months favors bridging therapy (risk difference: −3.05%; 95% CI: −6.98% to 0.89%; p = 0.13; I2 = 14%; Figure 3). This updated meta-analysis further reinforces that direct EVT fails to demonstrate noninferiority compared to bridging therapy, widening the margin of difference. In a network meta-analysis, only bridging therapy with tenecteplase was significantly associated with higher rates of good functional outcome compared to direct EVT (RR = 1.20; 95% CI: 1.01–1.43) and had the highest SUCRA value (89%), ranking first among the different thrombolytics (Figure 4).

Forest plot presenting the pooled risk difference (in percent) for good functional outcome (mRS 0–2 at 90 days) in patients with acute ischemic stroke treated with direct EVT versus bridging therapy (intravenous thrombolysis followed by EVT). The unadjusted pooled risk difference (in percent) with 95% confidence interval was calculated using random-effects meta-analysis, using the R software version 3.5.0. Different subgroups are presented according to the thrombolytic used in the bridging therapy: alteplase at a dose of 0.9 mg/kg, alteplase at a dose of 0.6 mg/kg, and tenecteplase at a dose of 0.25 mg/kg. The green dashed line indicates the prespecified noninferiority margin of −1.3%. 39
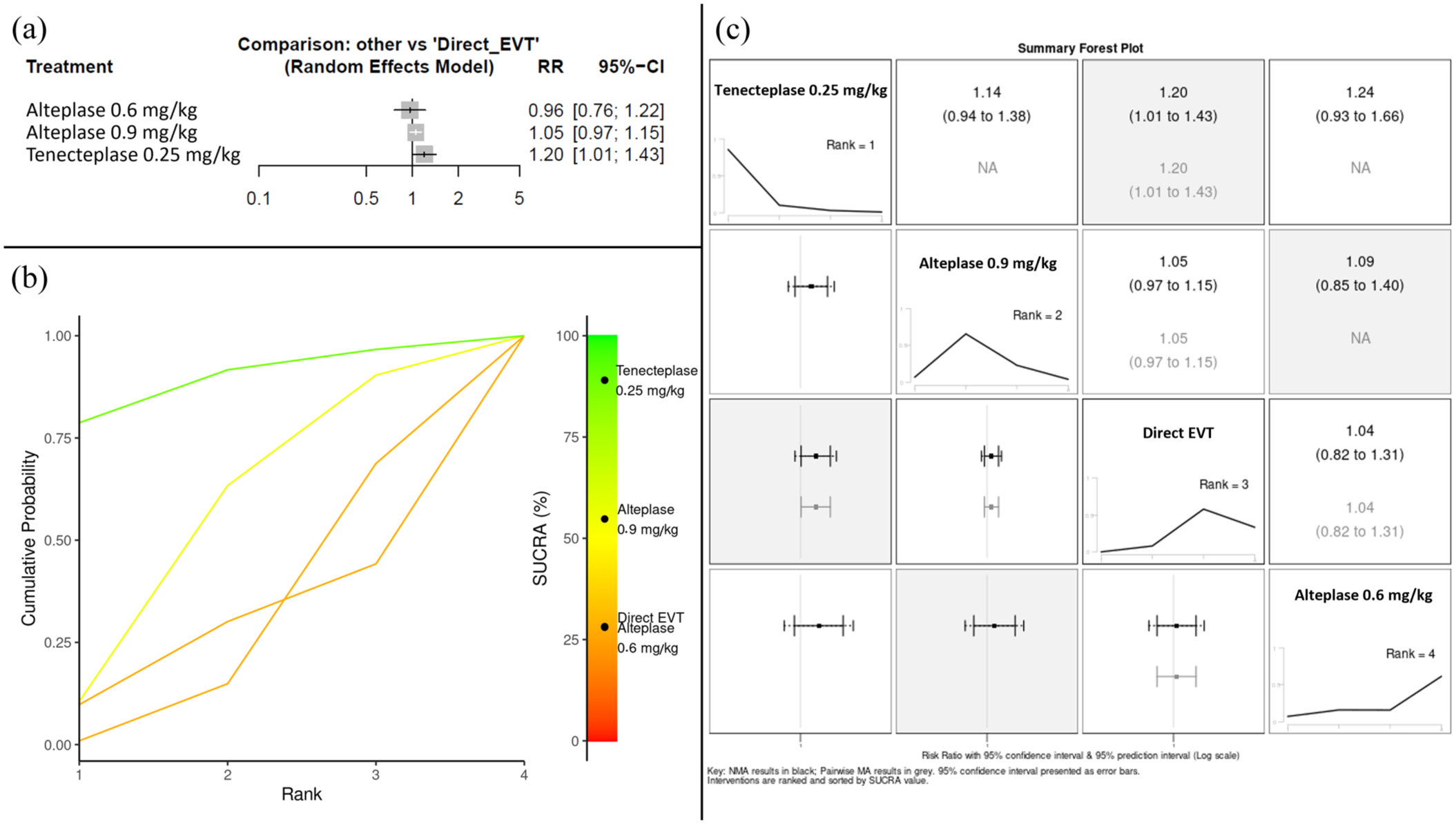
Network meta-analysis evaluating different thrombolytics during bridging therapy (intravenous thrombolysis followed by EVT) versus direct EVT. (a) Forest plot of the frequentist random-effects network meta-analysis presenting the RR of good functional outcome at 3 months according to the treatment group, using direct EVT as reference. (b) SUCRA plot for the good functional outcome at 3 months with higher SUCRA values and cumulative ranking curves nearer the top left indicating better performance. Tenecteplase 0.25 mg/kg had the highest SUCRA value (89%) and ranked first among the different treatment modalities, followed by alteplase 0.9 mg/kg (SUCRA value = 55%), and then by direct EVT (SUCRA value = 28%) and alteplase 0.6 mg/kg (SUCRA value = 28%). (c) Summary forest plot for the RR of good functional outcome at 3 months: network meta-analysis results in black; pairwise meta-analysis results in gray; 95% confidence interval presented as error bars; interventions are ranked and sorted by SUCRA value.
Tenecteplase has emerged as a promising alternative to alteplase in the treatment of AIS. 68 Studies have demonstrated that tenecteplase is associated with higher rates of pre-interventional reperfusion and may facilitate faster workflows due to its single-bolus administration in contrast to the infusion-based delivery required for alteplase. 69
According to the expedited ESO recommendations, tenecteplase is favored over alteplase in patients with LVO AIS, as its use was independently associated with a statistically significant 16% increase in the rate of good functional outcomes. 70 Based on these recommendations, for patients with LVO AIS within 4.5 h of onset who are eligible for IVT and present directly to a CSC, there is an expert consensus statement advocating for administering tenecteplase rather than withholding IVT. 70 Similarly, for eligible patients presenting to nonthrombectomy-capable centers (PSCs) within the same timeframe, tenecteplase at 0.25 mg/kg is recommended, followed by urgent transfer to an EVT-capable center. 70
The Tenecteplase versus Alteplase before Endovascular Therapy for Ischemic Stroke (EXTEND-IA TNK) trial has previously demonstrated that the rate of pre-interventional reperfusion increased from 10% with alteplase to 22% with tenecteplase. 71 Comparable results were reported in a French cohort, which was observed as a pre-interventional reperfusion rate of 21% with tenecteplase. 72 Additionally, a well-designed study from Texas, conducted within a 10-hospital regional network transitioning from alteplase to tenecteplase, compared workflow metrics between the two thrombolytics. 73 In the DS scenario, the DIDO times were significantly shorter for patients treated with tenecteplase compared to alteplase. 73 This finding was also reproduced in the French TETRIS registry, where IVT-to-puncture times were reduced by 11 min with tenecteplase relative to alteplase, reflecting faster interhospital transfer for EVT. 74 Critically, tenecteplase was associated with improved clinical outcomes across both MS and DS cohorts. 74
A recent analysis from the Get with the Guidelines—Stroke registry, representing the largest observational study to date comparing tenecteplase and alteplase in AIS, evaluated over 79,000 patients treated within 4.5 h of symptom onset. 75 The study found no significant differences in good functional outcome at discharge, sICH, or in-hospital mortality between tenecteplase and alteplase in the overall cohort. Importantly, among patients who were eligible for but did not undergo EVT, tenecteplase was associated with higher rates of discharge home, reduced in-hospital mortality, and lower composite rates of mortality or hospice discharge, demonstrating superiority in this subgroup. These findings underscore tenecteplase as a safe and effective alternative to alteplase and support its preferential use in patients presenting to PSCs, even when subsequent transfer for EVT is not feasible within the DS paradigm, maximizing the potential for favorable outcomes in routine stroke care. 76
Specifically for patients with LVO, the ESO expedited recommendations strongly advocate the preferential use of tenecteplase over alteplase, reflecting its emerging role as the thrombolytic of choice in this context. 70 This aligns with growing evidence that tenecteplase not only facilitates workflow efficiency due to its single-bolus administration but may also enhance early reperfusion rates, offering a practical and effective approach for improving outcomes in AIS patients within both MS and DS care pathways.
Adjunctive thrombolytic therapies and innovations
Emerging adjunctive therapies aim to enhance the effectiveness of reperfusion strategies in AIS. Although most have been evaluated primarily in MS settings, they may have potential applications within the DS-model, particularly in enhancing recanalization or improving microcirculatory reperfusion without delaying interhospital transfer. 3 These adjuncts include sono-thrombolysis, direct thrombin inhibitors, glycoprotein IIb/IIIa inhibitors, and therapeutic antibodies targeting glycoprotein VI.
Sono-thrombolysis, which uses ultrasound to augment thrombolytic activity, has shown promise in improving recanalization rates without increasing adverse events. An individual patient data meta-analysis of 272 LVO patients across 7 RCTs demonstrated that sono-thrombolysis was associated with higher rates of complete recanalization and a nonsignificant numerical increase in excellent and good functional outcomes, with no identified safety concerns. 77 As anticipated, earlier treatment initiation was critical, as each 10-min delay in sono-thrombolysis reduced the odds of recanalization and improved outcomes. These findings are now being evaluated in the phase III Aureva Transcranial Ultrasound Device With tPA in Patients With Acute Ischemic Stroke (TRUST; NCT03519737) trial, in the United States, which is investigating the Aureva transcranial ultrasound device in AIS patients with CTA-confirmed anterior LVO within the DS setting.
Other adjunctive therapies, including direct thrombin inhibitors and glycoprotein IIb/IIIa inhibitors, are under investigation for their potential to improve recanalization rates and prevent re-occlusion. Argatroban, a direct thrombin inhibitor, has been investigated as adjunctive therapy with IVT or EVT to improve microcirculatory reperfusion. The Randomized Controlled Trial of Argatroban With Tissue Plasminogen Activator (tPA) for Acute Stroke (ARTSS-2) trial suggested safety and a potential signal for improved recanalization, 78 but the recent Multiarm Optimization of Stroke Thrombolysis (MOST) phase III trial found that adjunctive argatroban did not improve outcomes and was associated with increased mortality (24% vs 8% with placebo), indicating it cannot currently be recommended in AIS care pathways. 79
Eptifibatide, a glycoprotein IIb/IIIa inhibitor, has been studied with IVT as adjunctive therapy to prevent re-occlusion.80–82 While feasibility and safety have been demonstrated, no clear efficacy signal has emerged. The MOST trial further confirmed the lack of benefit and showed increased mortality with adjunctive eptifibatide (12% vs 8% with placebo). 79 Robust data are still lacking to support its use in AIS.
Tirofiban, another glycoprotein IIb/IIIa inhibitor, has been evaluated in registries and small trials during EVT, suggesting potential for improved recanalization and prevention of re-occlusion without increasing hemorrhagic complications, though evidence has been inconclusive. Recently, a phase III multicenter RCT in China evaluated tirofiban post-IVT in noncardioembolic AIS patients within 4.5 h not eligible for EVT. 83 Among 832 patients, tirofiban significantly increased excellent outcomes (mRS 0–1 in 65.9% vs 54.9%, p = 0.001) with low but higher sICH rates (1.7% vs 0%) and similar mortality (4.1% vs 3.8%), indicating tirofiban may improve outcomes post-IVT in selected patients with modest bleeding risk.
Glenzocimab is a monoclonal antibody targeting glycoprotein VI, preventing platelet aggregation while minimizing bleeding risk. The Acute Ischemic Stroke Interventional Study (ACTIMIS) phase II trial demonstrated favorable safety with potential reduction in early re-occlusion post-IVT. 84 The ACuTe Ischemic Stroke Study Evaluating Glenzocimab Used as Add-on Therapy Versus placEbo (ACTISAVE; NCT05070260) phase III trial has since evaluated glenzocimab as an adjunct to reperfusion therapies, aiming to improve recanalization and microcirculation. Although no mRS differences were observed in the overall cohort, a potential signal of benefit was noted in patients receiving EVT with complete recanalization, suggesting a possible role for glenzocimab in optimizing microvascular reperfusion postrecanalization.
While data for these agents in DS workflows are limited, their ability to act without delaying transfer makes them potential candidates for future integration if efficacy and safety are confirmed in larger trials.
The drive-the-doctor paradigm: Physician transfer
The drive-the-doctor paradigm (Figure 1(c)), involving the transfer of neurointerventionalists to PSCs to perform EVT rather than transferring patients to CSCs, has been proposed as an alternative strategy to reduce onset-to-reperfusion times. 85 This approach may be particularly beneficial in regions where patient transfer times are prolonged due to geographical barriers.
Studies have demonstrated that the drive-the-doctor approach can significantly reduce time to recanalization and may improve functional outcomes in AIS patients requiring EVT. 86 Additionally, this model may offer cost-effectiveness advantages by reducing the need for patient transfers and optimizing resource utilization within stroke systems of care. 87
MSUs: Expanding prehospital capabilities
MSUs have emerged as a transformative innovation within stroke systems of care, designed to bring advanced diagnostics and treatment capabilities directly to the patient in the prehospital setting (Figure 1(d)). 88 Equipped with onboard CT imaging, point-of-care laboratory testing, and telemedicine connectivity to stroke specialists, MSUs enable early diagnosis of AIS and facilitate the administration of IVT in the field, significantly reducing onset-to-treatment times.
Studies have demonstrated that the use of MSUs leads to faster initiation of IVT compared to standard EMS, with time savings ranging from 20 to 40 min.89,90 Additionally, MSUs can perform prehospital imaging to identify LVO, aiding in triage decisions for direct transport to EVT-capable centers or early activation of EVT teams, thereby supporting both MS and DS workflows. 89 The ESO guidelines suggest that MSU management in AIS patients improves 90-day functional outcomes, reduces onset-to-treatment times, and increases rates of IVT within 60 min of onset without safety concerns, supporting MSUs as an effective tool to enhance prehospital AIS care. 91
While the integration of MSUs into stroke systems poses logistical and financial challenges, including staffing, operational costs, and regional coverage considerations, 92 their potential to enhance timely access to reperfusion therapies is significant, particularly in urban and suburban areas.93,94 Further research is needed to evaluate their cost-effectiveness across different healthcare settings and to explore their integration within the DS-model, where MSUs could initiate IVT while coordinating rapid transfer to CSCs, thereby optimizing outcomes in AIS care pathways.
Future directions and research priorities
Further research is needed to optimize the DS-model in AIS management, with a focus on conducting RCTs specifically within the DS context to evaluate interventions and workflow optimizations. Broadening IVT indications before interhospital transfer for EVT also represents a critical research priority, particularly for patients presenting beyond the 4.5-h time window or those receiving anticoagulation, since current IVT utilization before transfer remains low (~50%) despite its potential to enhance early recanalization and outcomes. Expanding the use of telemedicine and artificial intelligence for prehospital triage and LVO detection will be important for improving patient routing decisions and ensuring timely care. Investigating the integration of MSUs into prehospital care could enable the initiation of IVT and imaging in the field, potentially reducing time to reperfusion. Evaluating the long-term outcomes and cost-effectiveness of innovative models, such as the drive-the-doctor approach and adjunctive therapies, will further guide system improvements. Finally, given its strong association with functional outcomes in transferred patients, optimizing DIDO times should be considered a high-priority research target within DS-model workflow optimization. Future studies should aim to identify universally applicable DIDO benchmarks and evaluate interventions to achieve them in diverse healthcare settings. These initiatives will refine the DS-model, improve outcomes, and expand access to advanced reperfusion therapies.
Conclusion
The DS-model remains a vital strategy in delivering timely reperfusion therapy to AIS patients, particularly in regions without immediate CSC access. Bridging therapy with IVT, preferably using tenecteplase, should be promptly administered at PSCs to facilitate early reperfusion. Workflow optimization is essential for preserving the benefits of EVT in transferred patients, with DIDO time reduction being a central priority. Shortening DIDO times has a direct and substantial impact on onset-to-reperfusion intervals and functional outcomes and should therefore be a key performance metric in DS systems. Emerging adjunctive therapies and innovative models represent promising avenues for enhancing AIS care within the DS framework. Continued research and system-level investments are necessary to further refine these pathways and improve outcomes for patients with AIS.
