Abstract
Background:
In patients with posterior circulation stroke, the association between National Institutes of Health Stroke Scale (NIHSS) scores after thrombectomy and 90-day functional outcomes remains unclear.
Objectives:
We aimed to explore which factors among the 24-h NIHSS score, ΔNIHSS (baseline NIHSS minus 24-h NIHSS), and NIHSS score change rate (ΔNIHSS/baseline NIHSS × 100%) are associated with favorable functional outcomes at 90 days postoperatively in patients with posterior circulation stroke.
Design:
We performed a post hoc analysis of a prospective observational study utilizing key techniques of endovascular treatment and emergency workflow improvements from the acute ischemic stroke registry. The study included a cohort of 353 patients who underwent thrombectomy due to posterior circulation stroke. For all patients, we collected baseline characteristics, lesion locations, NIHSS scores, ΔNIHSS (baseline NIHSS minus 24-h NIHSS), NIHSS score change rate (ΔNIHSS/baseline NIHSS × 100), and 90-day postoperative modified Rankin Scale (mRS) score.
Methods:
A 90-day postoperative mRS score of 0–2 was defined as a favorable functional outcome, while a score of 3–6 was defined as an unfavorable functional outcome. The 24-h NIHSS score and ΔNIHSS score were converted into binary variables based on the Youden index to determine the optimal thresholds that best predict favorable functional outcomes at 90 days postoperatively. Adjusted logistic regression analysis was used to assess the predictive efficacy of the 24-h NIHSS score, ΔNIHSS (baseline NIHSS minus 24-h NIHSS), and NIHSS score change rate (ΔNIHSS/baseline NIHSS × 100) for the 90-day mRS. Subsequently, patients were categorized into cardioembolic embolism (CE) and large artery atherosclerosis (LAA) subgroups according to the Trial of Org 10172 in Acute Stroke Treatment classification, and the predictive efficacy of the optimal thresholds was examined within these subgroups.
Results:
Multivariate logistic regression analysis revealed that the 24-h NIHSS score was an independent predictor of 90-day functional outcomes (odds ratio (OR): 10.61, 95% confidence interval: 6.44–17.46, p < 0.001). The Youden index identified a 24-h NIHSS score of ⩽9 as the threshold for predicting an mRS score of 0–2, demonstrating good sensitivity (78.5%) and specificity (76.3%). The receiver operating characteristic curve indicated that the predictive model had good discriminative ability (area under the ROC curve = 0.8223). In subgroup analysis, a 24-h NIHSS score of ⩽9 also showed superior predictive efficacy in both the CE (sensitivity 67.8%, specificity 73.5%) and LAA (sensitivity 81.1%, specificity 74.4%) groups.
Conclusion:
The 24-h postoperative NIHSS score is a reliable predictor of 90-day functional outcomes in patients with posterior circulation stroke undergoing endovascular treatment. The predictive efficacy is optimal when the NIHSS score is ⩽9.
Introduction
Stroke remains a significant contributor to both disability and mortality worldwide. Revascularization therapies, including thrombolysis and thrombectomy, for acute ischemic stroke (AIS) have significantly improved stroke outcomes. 1 Endovascular thrombectomy is particularly effective for AIS of the posterior circulation, offering better outcomes than standard medical treatment.2–4 However, predicting the efficacy of thrombectomy at an early stage continues to be a challenge. Accurately assessing patient prognosis and predicting long-term functional outcomes after thrombectomy are areas that still necessitate further exploration.5–7 An accurate prognosis is essential for optimizing post-stroke care and rehabilitation efforts.8–10
The National Institutes of Health Stroke Scale (NIHSS) is a widely used clinical assessment tool that quantifies neurological deficits in patients with stroke.11,12 The early neurological state, as determined by the NIHSS score, exhibits a significant correlation with long-term functional outcomes. Although the NIHSS is a well-established tool for assessing stroke severity at baseline, 13 and the change in the NIHSS score indicates the deterioration or recovery of patients with stroke, its applicability in predicting the efficacy of thrombectomy has not been fully explored. Studies have shown its value in predicting long-term functional outcomes in patients with anterior circulation stroke14–16; however, its clinical utility in posterior circulation stroke requires further investigation.16–18 Therefore, this study aimed to explore the utility of the NIHSS score at 24 h post-thrombectomy as a predictor of 90-day functional outcomes in patients with posterior circulation stroke.
Methods
In this study, we employed a post hoc analysis of the endovascular treatment key technique and emergency workflow improvement of AIS (ANGEL-ACT) database, a prospective nationwide registry of 1793 continuous patients with AIS due to large vessel occlusion (LVO) undergoing endovascular therapy (EVT) in 111 hospitals across China from November 2017 and March 2019. 19 This subjects were enrolled according to the following criteria: (1) age ⩾18 years; (2) diagnosis of AIS confirmed by imaging, with intracranial LVO affecting the intracranial internal carotid artery (T/L), middle cerebral artery (M1/M2), anterior cerebral artery (A1/A2), basilar artery, vertebral artery (V4), and the posterior cerebral artery (P1); and (3) those who underwent any type of EVT, including intra-arterial thrombolysis, mechanical thrombectomy, angioplasty, or stenting.
The exclusion criteria were: (1) isolated cervical internal carotid artery or vertebral artery occlusion; and (2) no evidence of LVO on angiography. In our subgroups, we included patients with posterior circulation stroke who underwent thrombectomy. Of the 1793 patients initially enrolled in the study, 1440 were excluded owing to strokes affecting the anterior circulation or due to no thrombectomy being performed. The study received ethical approval from the Ethics Committees of Beijing Tiantan Hospital and all collaborating centers (KY 2017-048-01). All the procedures adhered to the principles outlined in the 1964 Declaration of Helsinki and its subsequent revisions. Furthermore, written informed consent was obtained from all patients or their legally appointed representatives (Figure 1).
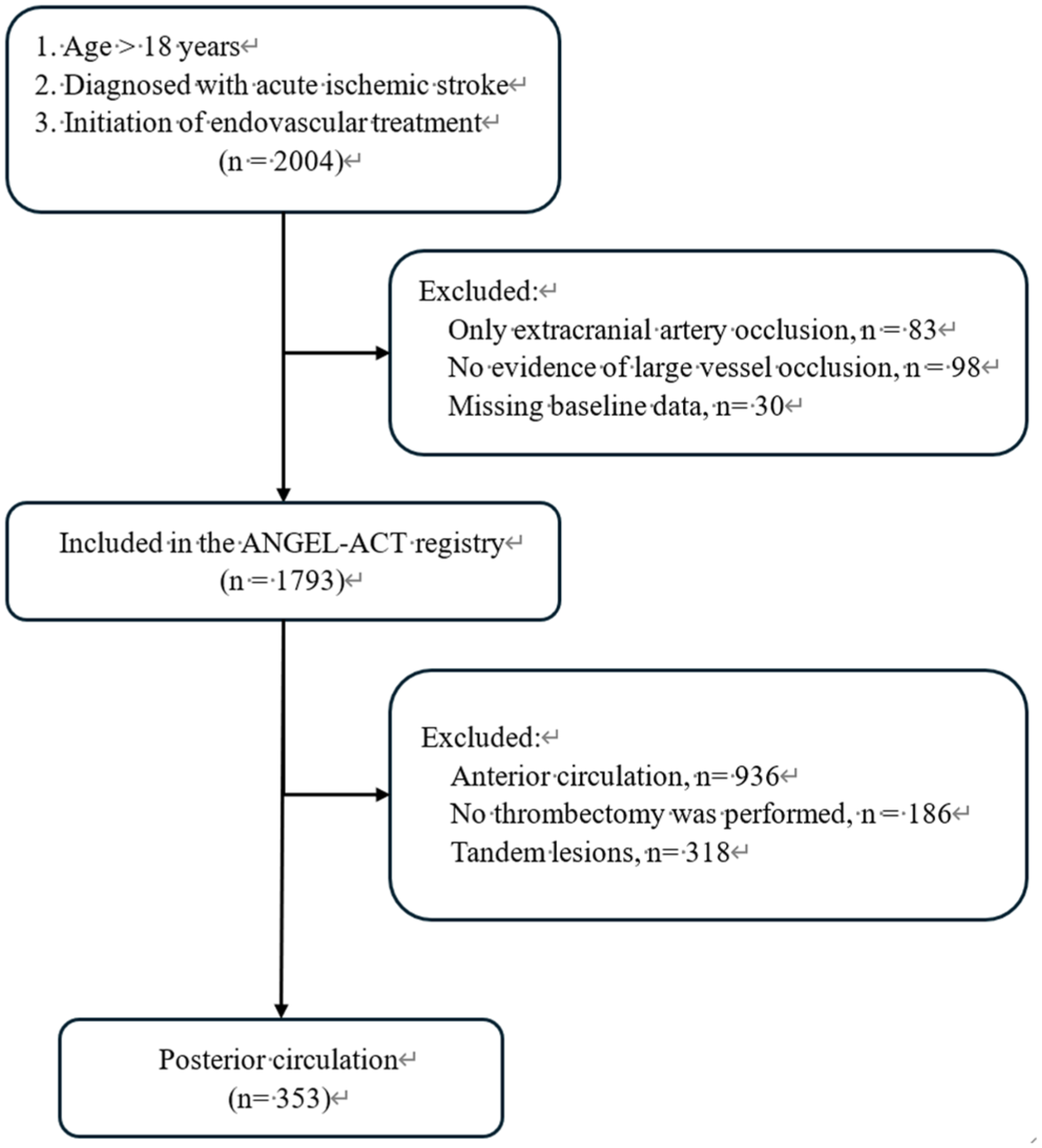
Flow chart of the patient selection process.
The following early clinical outcomes based on the NIHSS score were investigated: (1) a binary measure of the 24-h NIHSS threshold; (2) the threshold of change in NIHSS scores within 24 h after EVT (baseline minus 24-h NIHSS score (ΔNIHSS); binary measure); and (3) the percentage change from baseline (calculated as ΔNIHSS/baseline NIHSS × 100). The primary outcome measure was the 90-day modified Rankin Scale (mRS) score, categorized as favorable (mRS score: 0–2) or unfavorable (mRS score: 3–6).
The Youden index is a comprehensive measure of diagnostic test performance used to determine optimal thresholds for the 24-h NIHSS and ΔNIHSS, which best predict a favorable 90-day mRS score (0–2). This metric prioritizes maximizing the distinction between good and bad outcomes by balancing sensitivity and specificity, ultimately determining the optimal cutoff points. In addition, we dichotomized the 24-h NIHSS and ΔNIHSS at these optimal thresholds alongside commonly used alternatives. For continuous values (range 6–10), both measures were analyzed directly. The strength of the association between these NIHSS measures and 90-day mRS scores was assessed using two approaches: (1) multivariate logistic regression analysis (mRS score: 0–2), covariates including baseline NIHSS, age, hypertension, baseline glucose, ASPECTS, time to recanalization, successful recanalization, and tPA administration; and (2) an ordered regression model (7-level ordered mRS score). Furthermore, Spearman’s ρ coefficient was calculated to determine the potential predictive power of each covariate regarding 90-day mRS scores. This non-parametric measure indicates the strength and direction of the correlation between a set of predictors and the outcome, where higher values suggest a stronger relationship between them.
Data collection
Patient demographics and clinical variables were retrieved from electronic medical records. These included baseline NIHSS scores at admission and 24 h post-thrombectomy, the posterior circulation Alberta Stroke Program Early CT Score (pc-ASPECTS), 20 comorbidities (including atrial fibrillation, intravenous thrombolysis (IVT), hypertension, and diabetes), blood glucose levels, radiological findings (such as the site and extent of occlusion, presence of hemorrhage, and successful recanalization), and procedural details (i.e., time to recanalization).
Outcome measures
The primary outcome measure was the mRS score at 90 days post-thrombectomy, which categorizes functional outcomes as favorable (mRS: 0–2) or unfavorable (mRS: 3–6).
Statistical analysis
All statistical analyses were performed using SAS software (version 9.4; SAS Institute, Inc., Cary, NC, USA). The significance level was set at a two-sided p value of <0.05 for all tests. p-Values were reported, and all other effect sizes were reported with 95% confidence intervals (CIs).
Continuous and ordinal variables were reported as medians and interquartile ranges, while categorical variables were presented as frequencies and percentages. We assessed the normality of continuous variables before proceeding with further analyses.
The statistical analyses comprised descriptive statistics, univariate analyses to identify potential predictors of the primary outcome, and a multivariate logistic regression model to predict the primary outcome, with the 24-h NIHSS score serving as the key independent variable. Other potential predictors identified in the univariate analysis, such as age, baseline NIHSS score, radiological findings, and procedural details, were also included in the model. Receiver operating characteristic (ROC) curve analysis was performed to evaluate the discrimination (the model’s ability to distinguish between good and bad outcomes) and calibration (the agreement between predicted and observed probabilities) of the model.
Results
A total of 353 patients with posterior circulation stroke met the inclusion criteria. Patients with a favorable functional outcome at 90 days post-thrombectomy were 163, while those with an unfavorable outcome were 190. The corresponding numbers for each score segment are shown in Figure 2.

The corresponding numbers for each 90-day modified Rankin Score segment.
The mean age of the patients was 63 years and 77% were male. The median pc-ASPECTS was 9, and the median baseline NIHSS score was 21. Approximately 14% of patients (49/353) had atrial fibrillation, 27% (94/353) received prior IVT treatment, 15% (54/353) had coronary heart disease, 69% (242/353) had hypertension, and 21% (74/353) had diabetes. The median blood glucose level was 7.5 mg/dL. At 90 days post-thrombectomy, 46% of the patients achieved favorable functional outcomes (mRS: 0–2). The baseline patient characteristics are shown in Table 1.
Baseline characteristics of patients.
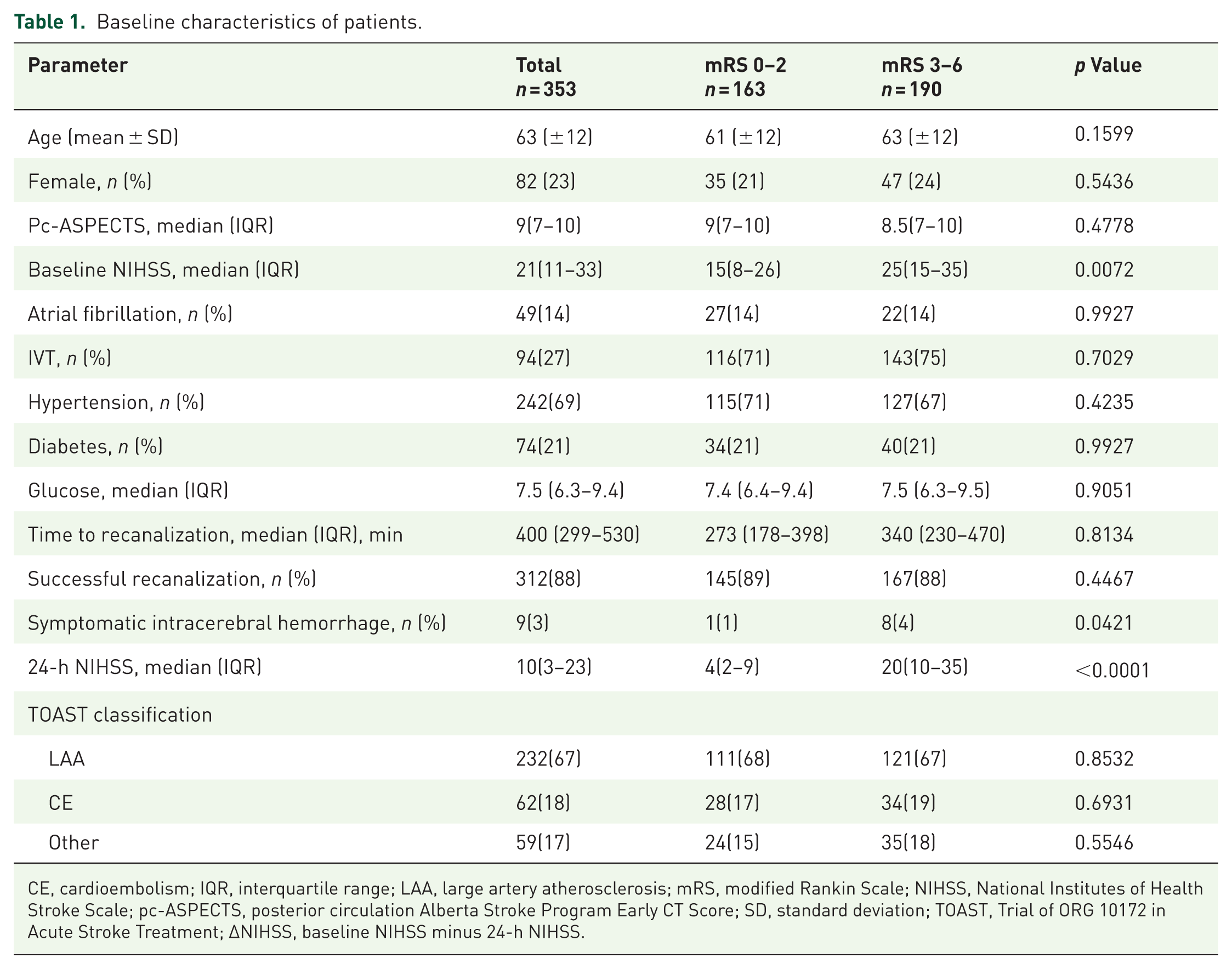
CE, cardioembolism; IQR, interquartile range; LAA, large artery atherosclerosis; mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale; pc-ASPECTS, posterior circulation Alberta Stroke Program Early CT Score; SD, standard deviation; TOAST, Trial of ORG 10172 in Acute Stroke Treatment; ΔNIHSS, baseline NIHSS minus 24-h NIHSS.
The NIHSS scores of patients’ 24-h post-thrombectomy demonstrated a strong association with their 90-day functional outcomes (p < 0.001). Multivariate logistic regression analysis revealed that the NIHSS score at 24 h was an independent predictor of 90-day functional outcomes (odds ratio (OR): 10.61, 95% confidence interval (CI): 6.44–17.46, p < 0.001).
The predictive model incorporating the 24-h NIHSS score showed good discrimination in predicting favorable outcomes, with an area under the ROC curve (AUC) of 0.8223.
NIHSS score at 24-h
Using the Youden index, we identified a 24-h NIHSS score of ⩽9 as the optimal threshold for predicting favorable functional outcomes (mRS: 0–2) at 90 days post-thrombectomy (sensitivity: 78.5%, specificity: 76.3%, AUC: 0.822, p < 0.001; Figure 3(a) and Table 2).

Receiver operating characteristic curves of NIHSS-based outcome measures predicting 90-day mRS scores (0–2). (a) 24-H NIHSS scores. (b) ΔNIHSS scores (baseline NIHSS minus 24-h NIHSS). (c) Percent change in NIHSS scores (ΔNIHSS/baseline NIHSS × 100). The sensitivity (y-axis), 100-specificity (x-axis), and AUC for 24-h NIHSS, ΔNIHSS, and percent change in NIHSS scores at differentiating 90-day mRS scores of 0–2 versus 3–6 are shown.
Association of various thresholds of the 24-h NIHSS and ΔNIHSS with 90-day mRS scores (0–2).

Adjusted for baseline NlHSS, age, hypertension, baseline glucose, ASPECTS, time to recanalization, successful recanalization, and tPA administration.
Adjusted for age, hypertension, ASPECTS, time to recanalization, successful recanalization, and tPA administration.
ΔNIHSS, baseline minus 24-h NIHSS; ASPECTS, Alberta Stroke Program Early CT Score; NIHSS, National Institutes of Health Stroke Scale; tPA,tissue plasminogen activator.
Among the 353 patients included in the study, 49% (173 patients) had a 24-h NIHSS score of ⩽9. This group had significantly higher odds of achieving a favorable mRS score (0–2) at 90 days compared with those with a higher score (OR: 11.78, 95% CI: 7.14–19.46, p < 0.001; Table 2). For reference, ROC data for a 24-h NIHSS cutoff value of 2 is also provided in Table 2.
ΔNIHSS score at 90 days post-thrombectomy
We identified a ΔNIHSS score of ⩾3 as the optimal threshold for predicting favorable functional outcomes (mRS score: 0–2) at 90 days post-thrombectomy (sensitivity: 59%, specificity: 65%, AUC: 0.65, p < 0.001). See Figure 3(b) and Table 2 for details. Among the 353 patients, 64% (225 patients) achieved a ΔNIHSS score of ⩾3. This group had significantly higher odds of achieving a favorable mRS score (0–2) at 90 days compared with those with a smaller improvement (OR: 3.51, 95% CI: 2.20–5.61, p < 0.001). Table 2 illustrates these details.
Percent change in NIHSS scores (continuous measure)
The analysis of the continuous percent change in NIHSS scores revealed an optimal cutoff point of >57.9% for predicting favorable functional outcomes (mRS score: 0–2) at 90 days post-thrombectomy (sensitivity: 59.9%, specificity: 81.0%, AUC: 0.734, p < 0.001; Figure 3(c)).
Among all potential indicators considered, the 24-h NIHSS score showed the highest predictive power, with a Spearman’s ρ 2 of 0.547 for an mRS score of 0–2 and 0.607 for ordinal mRS scores, indicating moderate to strong predictive power. ΔNIHSS scores ⩾3 exhibited the next best predictive power. Detailed Spearman ρ 2 values for these and other variables are outlined in Table 3.
Predictive power of various definitions of early neurological recovery in predefined multivariable logistic regression models.

IVT, intravenous thrombolysis; mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale; pc-ASPECTS, posterior circulation Alberta Stroke Program Early CT Score; ΔNIHSS, baseline minus 24-h NIHSS.
Predictive efficacy of 24 h NIHSS score in subgroups
The patients were categorized into the large artery atherosclerosis (LAA) group and the cardioembolic embolism (CE) group based on etiology. The two strongest predictive indicators
The differences between the two variables between the LAA group and the CE group.

Adjusted for baseline NlHSS, age, hypertension, baseline glucose, ASPECTS, time to recanalization, successful recanalization, and tPA administration.
Adjusted for age, hypertension, ASPECTS, time to recanalization, successful recanalization, and tPA administration.
ASPECTS, posterior circulation Alberta Stroke Program Early CT Score; CE, cardioembolic embolism; LAA, large artery atherosclerosis; NIHSS, National Institutes of Health Stroke Scale; OR, odds ratio; tPA, tissue plasminogen activator.
Discussion
This study identified the 24-h NIHSS score as the most valuable predictor of 90-day functional outcomes in patients with posterior circulation stroke who underwent thrombectomy. This finding is consistent with prior studies.3,21–23 Notably, our study is the first to demonstrate that the 24-h NIHSS score effectively predicts outcomes at 90 days post-thrombectomy. Our results indicate that a higher NIHSS score at 24 h is associated with an increased risk of unfavorable functional outcomes. Furthermore, we determined that a threshold of ⩽9 for the 24-h NIHSS score reliably predicted favorable functional recovery (mRS score: 0–2) at 90 days.
This information is crucial for clinicians to make informed decisions regarding post-stroke care, rehabilitation, and patient counseling. 10 Our findings align with emerging clinical studies demonstrating the effectiveness of thrombectomy in posterior circulation stroke, where accurate prognosis is crucial owing to the potentially wide range of outcomes. While good outcomes translate to well-preserved long-term prognosis, previous studies have reported mortality rates as high as 26.71% (95% CI: 19.35–34.71). Of all the predictors, we found that the 24-h NIHSS score was a more valuable factor than baseline NIHSS and ΔNIHSS scores.
This study suggested that the 24-h NIHSS score is the best predictor of functional outcomes in patients with posterior circulation stroke after thrombectomy. We speculate this may be as follows. First, the 24-h score assessed the state of the patient’s neurological function after surgery, thus reflecting the effect of the surgery and early recovery trajectory. Lastly, the 24-h score retains its value as a baseline indicator, critical for predicting prognosis. By encompassing both post-interventional effects and baseline information, the 24-h NIHSS emerges as the optimal predictor as it comprehensively considers both the post-surgery effect and the patient’s recovery status, providing an important reference for clinical decision-making.
Our study identified the 24-h NIHSS score as a more robust predictor of functional outcomes than ΔNIHSS scores in patients with posterior circulation stroke after thrombectomy. This preference arises from several limitations associated with the ΔNIHSS scores. First, the specificity of the indicator in predicting favorable outcomes was unacceptably low. Second, the ΔNIHSS does not fully explain the initial neurological state reflected by baseline NIHSS scores or the clinical assumptions surrounding the NIHSS. For example, for a patient with a baseline NIHSS score of 3, an NIHSS score of 4 has a very different meaning than for a patient with a baseline NIHSS score of 23. This highlights the critical limitation of ΔNIHSS in capturing the inherent biological significance of the baseline score. A low baseline NIHSS, as previous studies have shown,24,25 is strongly associated with smaller infarct core volumes and lesions in less critical locations, fundamentally altering the risk profile and prognostic implications of any subsequent change, compared to a high baseline NIHSS reflecting extensive or strategically located damage. ΔNIHSS fails to account for this baseline heterogeneity in underlying pathophysiology. In addition, a particular NIHSS score may have a different meaning depending on the hemisphere affected by stroke. Lastly, relying on the ΔNIHSS assumes a strong correlation between baseline NIHSS scores and their variance at 24 h, which might not always hold true in clinical practice. Owing to these drawbacks, the 24-h NIHSS provides higher predictive power and accuracy than the ΔNIHSS. Therefore, our study proves that the 24-h NIHSS regression model is a better method for analyzing relative changes in neurological function after thrombectomy than its counterpart, the ΔNIHSS.
In the subgroup analysis, we assessed the predictive efficacy of the 24-h NIHSS score ⩽9 and ΔNIHSS in the LAA group and the cardioembolic group. The results demonstrated that in both groups, the 24-h NIHSS score ⩽9 was a superior predictor of functional outcomes.
The limitations of this study include its retrospective nature and potential for selection bias. First, we included only patients with posterior circulation stroke who underwent thrombectomy, potentially excluding those with mild symptoms or high procedure risks who may not have been eligible for the procedure. Second, excluding patients without 90-day mRS scores might underestimate the rates of poor outcomes or serious events. Third, our study focused on patients treated between November 2017 and March 2019. Thrombectomy techniques and technology have likely evolved since then, potentially limiting the generalizability of our findings to current clinical practice. Fourth, the limited experience of the surgeons at the time might have influenced the procedural variables, such as the recanalization time, further impacting generalizability (399–530 min). This may limit the generalizability of the results. Lastly, the relatively low rate of IVT utilization (27%) observed in our cohort reflects the real-world practice patterns in the participating Chinese centers during the study period. This may be attributed to factors such as geographical disparities in access to comprehensive stroke centers equipped for rapid thrombolysis and variable levels of public awareness regarding acute stroke symptoms and treatment urgency, leading to delayed presentation. While this represents the actual clinical landscape in these centers, it could potentially introduce selection bias or outcome bias and warrants consideration when interpreting the generalizability of our findings.
Conclusion
The NIHSS score at 24 h post-thrombectomy is a promising tool for predicting the 90-day functional outcomes in patients with posterior circulation stroke. Early assessment of stroke severity following thrombectomy using the NIHSS score can aid clinicians in optimizing patient-care strategies and setting realistic expectations for patients and their families. Further research is required to validate its role in clinical practice.
